Abstract
Aging is associated with specific physiological changes. One change that influences the functionality of an individual is the decrease in skeletal muscle. Research efforts to maintain skeletal muscle through dietary modifications have primarily focused on protein intake, including composition, quantity, and timing. Evidence demonstrates that adequacy, quality, and balance of protein intake are important; however, it should not replace the other dietary needs of the individual. The purpose of this article is to summarize the vast area of research regarding protein consumption in the aging population and place it in context of other nutritional concerns that affect this demographic. Efforts to educate older adults without providing overcomplicated guidelines will reduce the likelihood of individuals feeling overwhelmed, increasing dietary adherence.
Current research demonstrates that routine physical activity and adequate protein consumption may lessen consequences of aging.
While the process of aging is inevitable, proper nutrition, in conjunction with regular physical activity, may help maintain skeletal muscle, which may increase quality of life. Many researchers have noted the influential role skeletal muscle mass plays in functional performance and an individual’s ability to maintain a healthy, active lifestyle. Age-related losses can threaten that independence. Lean muscle mass decreases approximately 50% from young adulthood to the age of 75 to 80 years. Increased fat mass can mask the dramatic change in muscle mass, leading to negative body composition changes.1,2 Age-associated alterations in body composition reduce resting energy expenditure. 3 The impact of these changes can lead to increased frailty, sarcopenia, or sarcopenic obesity, which all have the potential to become debilitating and life threating. Current research demonstrates that routine physical activity and adequate protein consumption may lessen consequences of aging. The purpose of this article is to summarize the vast area of research regarding protein consumption in the aging population and place it in context of other nutritional concerns that affect this demographic. Lastly addressed is the role nutrition professionals have in assisting older adults in applying necessary dietary recommendations.
Protein: What Matters Most?
Advanced aging contributes to increased protein turnover from skeletal muscle and decreased type II muscle fibers, resulting in diminished strength. Ultimately, consequences associated with decreased mobility and functioning leads to more sarcopenia-related falls.3,4 In addition to the age-related morphology, some researchers have discussed the concept of “anabolic resistance,” contributing to the difficulties in maintaining skeletal muscle. In a recent review, Breen and Phillips 5 placed greater importance on the decreased responsiveness to protein and amino acids consumption in synthesizing protein, rather than on overall muscle protein metabolism. Therefore, they concluded that more rapid protein turnover is the body’s resistance to anabolic stimuli. Exact mechanisms of protein metabolism in the older adult have been detailed elsewhere 6 and consequently will not be expanded on here. This seemingly correctable or reducible phenomenon has caught the attention of hundreds of researchers, all looking for the best approach to decrease the aging burden on skeletal muscle.
With evidence of altered protein metabolism, increased skeletal muscle loss, and a decrease in anabolic responsiveness, various researchers have concluded that older adults would benefit from greater protein consumption. Currently, the Recommended Dietary Allowance (RDA) for protein set by the Institute of Medicine is 0.8 g/kg/day (for males and females ≥ 19 years). 7 The RDA is set to meet the needs of almost all (97% to 98%) individuals in a Life Stage group, determined by sex and age. 7 Despite differences between age groups and sex in terms of lean body mass, overall body weight differences are expected to compensate. 8 The RDAs are set to provide adequacy, and for protein this translates into a state of nitrogen equilibrium. The goal and focus of current research strives to develop an optimal level of protein to combat aging, which has produced results consistently higher than the RDA. Much of the literature has concluded that intakes between 0.9 and 1.5 g/kg/day are necessary for both men 9 and women 10 aged 65 years and older. The majority of the extensive reviews of literature and epidemiology research reserves the upper end of this range (1.2-1.5 g/kg) for older adults with acute or chronic diseases (including sarcopenia) and the lower end (1.0-1.2 g/kg) for otherwise healthy older adults.3,11,12 Observed benefits in both walking and balance tests, 13 as well as the prevention of unintentional weight loss independent of other nutritional factors, have been reported. 14 Despite the amount of evidence supporting protein consumption of at least 1.0 g/kg/day, data from the National Health and Nutrition Examination Survey 2005-2006 discovered 12% of men and 24% of women older than 70 years consumed significantly less than the RDA. 15
Quality and Timing
As previously mentioned, a potential explanation for decreases in skeletal muscle mass in older adult is “anabolic resistance.” Some suggest that “resistance” places greater importance on both the quantity of protein consumed and the timing of protein ingestion. Breen and Phillips 5 hypothesized that it is the decreased utilization of amino acids (especially leucine) that prevents effective preservation of lean body mass. Concerns related to aging, intensified by the increased prevalence of sarcopenia, have encouraged researchers to look beyond total quantity of daily protein intake and look closer at the quality of protein consumed. Current findings regarding protein intake for the older adult place importance on high biological value (BV) sources to assist in both overall protein and nitrogen balance.6,16 Nitrogen retained by the body for maintenance, repair, and growth determines the BV of a protein source. Other important components of a high-quality protein source include digestibility and, above all, composition of essential amino acids, in both quantity and proportion. 17 Sources of complete proteins (foods containing all 9 essential amino acids in quantities needed by the human body) are mainly of animal origin including milk, yogurt, cheese, eggs, meat, fish, and poultry, all having a digestibility between 90% and 99%. Being of plant origin, soy protein is one exception, which supplies sufficient amounts of essential amino acids. 17 However, when comparing the muscle protein synthesis (MPS) stimulating effects of whey and soy, it is the higher leucine content of whey, not the total amino acid profile, that contribute to the greater MPS potential seen in whey. 5 Leucine’s role in triggering anabolic pathways in muscle tissue, including during periods of food restriction, is well established.18,19 As a result, a recommendation of a minimum of 2.5 grams of leucine per meal helps promote muscle synthesis throughout the day. 20 While leucine is present in all foods that contain protein, consuming complete protein sources, especially beef, poultry, fish, milk, and eggs, likely supply more than enough leucine (chicken [3 oz cooked] contains 2.0 grams). 21
In efforts to combat anabolic resistance, distributing protein consumption throughout the day, including an amount ranging from 25 to 40 grams of high-quality protein (containing 2.5-3 grams of leucine), can encourage anabolism through stimulating MPS regardless of age-related changes in otherwise healthy, older adults.5,11,22 Consideration for this approach in hospitalized older adults is justified. 23 Drastic variability in protein consumption throughout the day is thought to attribute to increased protein breakdown. Current dietary patterns existing in the United States skew most of the day’s protein toward the evening meal. Recent research has called attention to this trend, intuitively asking if a more evenly distributed approach would decrease protein breakdown. A recent study in middle-aged adults demonstrated that an equal protein distribution (in grams) between 3 meals (30:30:30) resulted in greater MPS over a 24-hour period compared with a more typical approach (10:15:60). 24 Conversely, Kim et al 9 demonstrated that in the context of a mixed meal, MPS differences were insignificant among older adults when accounting for whole body net protein balance. Exceeding the RDA for protein intake was more influential on whole protein balance and MPS than protein intake distribution.
Conclusion: Protein
The existing evidence is clear that to slow the loss of skeletal muscle caused by aging, dietary interventions through increased protein intake is necessary. Dietary protein of high quality/biological value, specifically with significant amounts of leucine, may be the most effective in stimulating protein synthesis and slowing protein breakdown. More research is needed to determine if equalizing protein distribution throughout the day is necessary to encourage optimal skeletal muscle maintenance among this population, or if overall daily consumption is more important. Given these 2 dietary approaches, the former may present more logistical problems.
Putting This Into Practice
Recommendations to simply increase one specific macronutrient may be shortsighted and simplistic for this population with diverse age-related physiological changes. While many suffer from malnutrition, classic sarcopenia, and increased frailty, age-associated shifts in body composition and decreased energy expenditure contribute to the rate of obesity in this demographic. More than 30% of adults age 65 and older are classified as obese. 25 In older adult men and women, low muscle mass and high fat infiltration into the muscle are associated with decreased strength, increased risk of losing mobility, and decreased functionality. 26 With older adults losing muscle mass and increasing adipose tissue, including the amount found intramuscularly, the term sarcopenic obesity has become more common. Primary management of sarcopenic obesity aims to sustain muscle strength and function, while secondarily decreasing body fat through a combination of moderate calorie restriction, increased protein intake, and physical activity.12,27 More detailed protein guidelines, appropriately addressing the broader implications on the individual’s overall health, deserve further consideration.
Past lifestyle interventions using a combination of diet and exercise, while advocating for adequate high-quality protein intake, have been successful in producing a weight loss of 10%, leading to increased physical function, decreased cardiovascular risk, and metabolic outcomes.28,29
Even distribution of protein throughout the day, consuming 25 to 40 grams of protein (2.5-3.0 grams of leucine) at each meal, compared to consuming 1.2 g of protein per kilogram of bodyweight daily, may offer this population a way to maintain lean body mass, while increasing the possibility of creating an overall calorie deficit. Table 1 depicts the energy needs of an obese (body mass index = 40) 75-year-old male and compares the two different protein intake methods described previously. Assuming that both protein intake methods are effective in maintaining muscle mass, calorie consumption from fat and carbohydrates remain constant, and physical activity is minimal, even distribution (30:30:30) could result in a 200 kcal difference (equivalent of 0.4 lbs per week).
Common Protein Recommendations and the Impact on Total Calories.
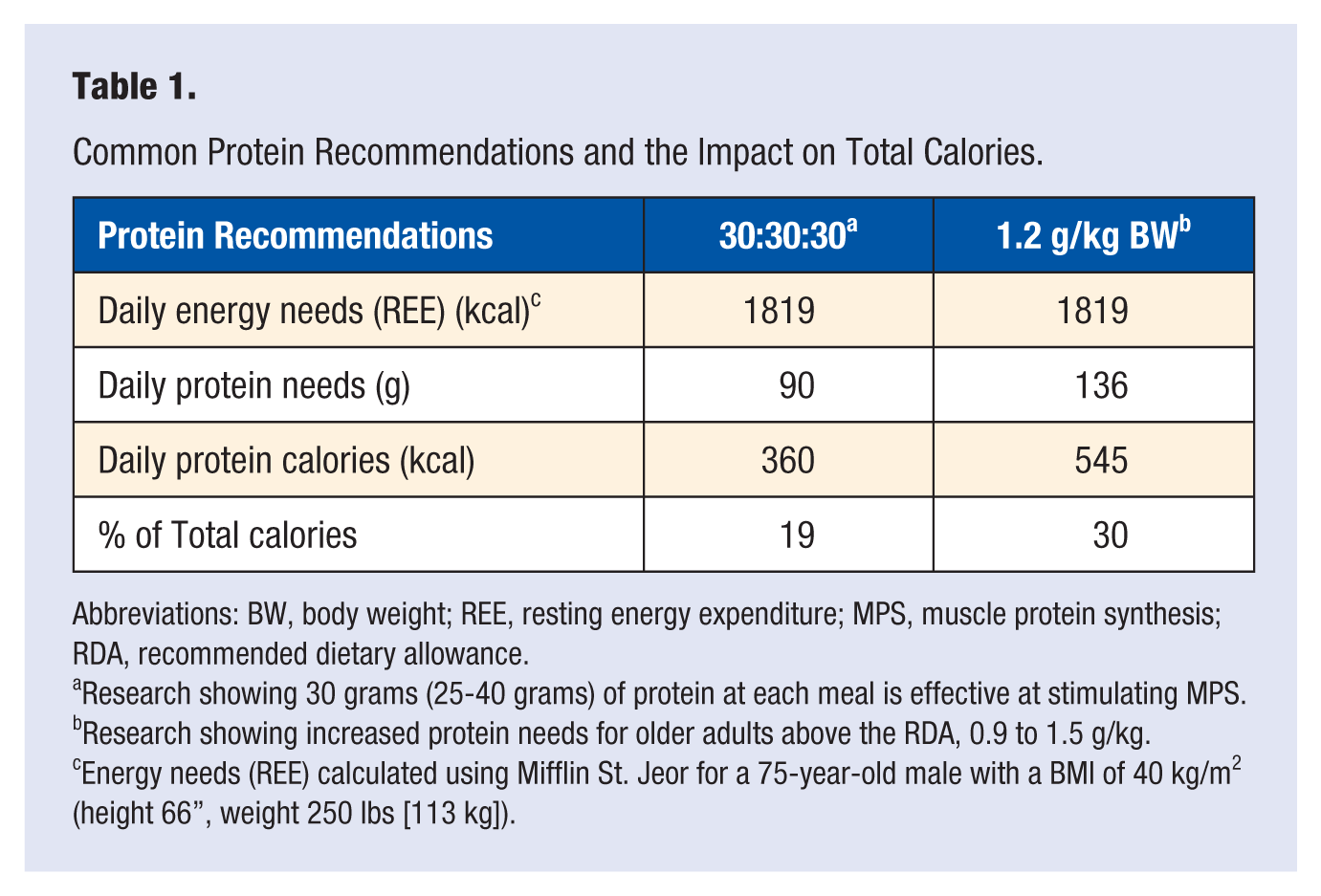
Abbreviations: BW, body weight; REE, resting energy expenditure; MPS, muscle protein synthesis; RDA, recommended dietary allowance.
Research showing 30 grams (25-40 grams) of protein at each meal is effective at stimulating MPS.
Research showing increased protein needs for older adults above the RDA, 0.9 to 1.5 g/kg.
Energy needs (REE) calculated using Mifflin St. Jeor for a 75-year-old male with a BMI of 40 kg/m2 (height 66”, weight 250 lbs [113 kg]).
Other Dietary Recommendations to Consider
In addition to sarcopenic obesity, the older adult population experiences many diseases that benefit from medical nutrition therapy. Addressing dietary guidelines that accompany these diseases, while implementing the increased protein needs, is a necessary part of diet planning. It is estimated that more than 70% of adults older than age 60 years and more than 83% of adults age 80 years and older have cardiovascular disease (CVD). 30 Of adults older than age 65 in the United States, 86% have at least one nutrition-related risk factor for CVD. 31 The American Heart Association recommends a dietary pattern that contributes less than 7% of all calories from saturated fat, and contains no more than 1500 milligrams of sodium. The Dietary Guidelines for Americans (DGA) 2010 congruently recommend that individuals who are 51 and older, of African American decent, and individuals diagnosed with hypertension, diabetes, or chronic kidney disease reduce their sodium intake to 1500 mg. Additionally, DGA also advise lowering consumption of saturated fat to no more than 7% of total calories, in an effort to further reduce the risk of CVD. 32 Unfortunately, many of the high-quality protein sources also contain saturated fat, and oftentimes sodium, further complicating the diet planning process. In addition to heart disease, 25.9% of individuals 65 years and older have diabetes. 33 Formulation of specific carbohydrate recommendations typically include the assistance of a certified diabetes educator. Generally, consistency among meals in terms of grams of carbohydrates is foundational. Furthermore, these two disease states share the recommendation to consume adequate fiber in accordance with the Institute of Medicine’s DRIs (30 grams and 21 grams for men and women, respectively, 51 years and older). 7 Table 2 demonstrates the complexities of meal planning, including nutrients of concern, while accounting for the three common disease states of sarcopenic obesity, CVD, and diabetes.
One-Day Sample Menu Addressing Common Dietary Guidelines for Older Adults a .
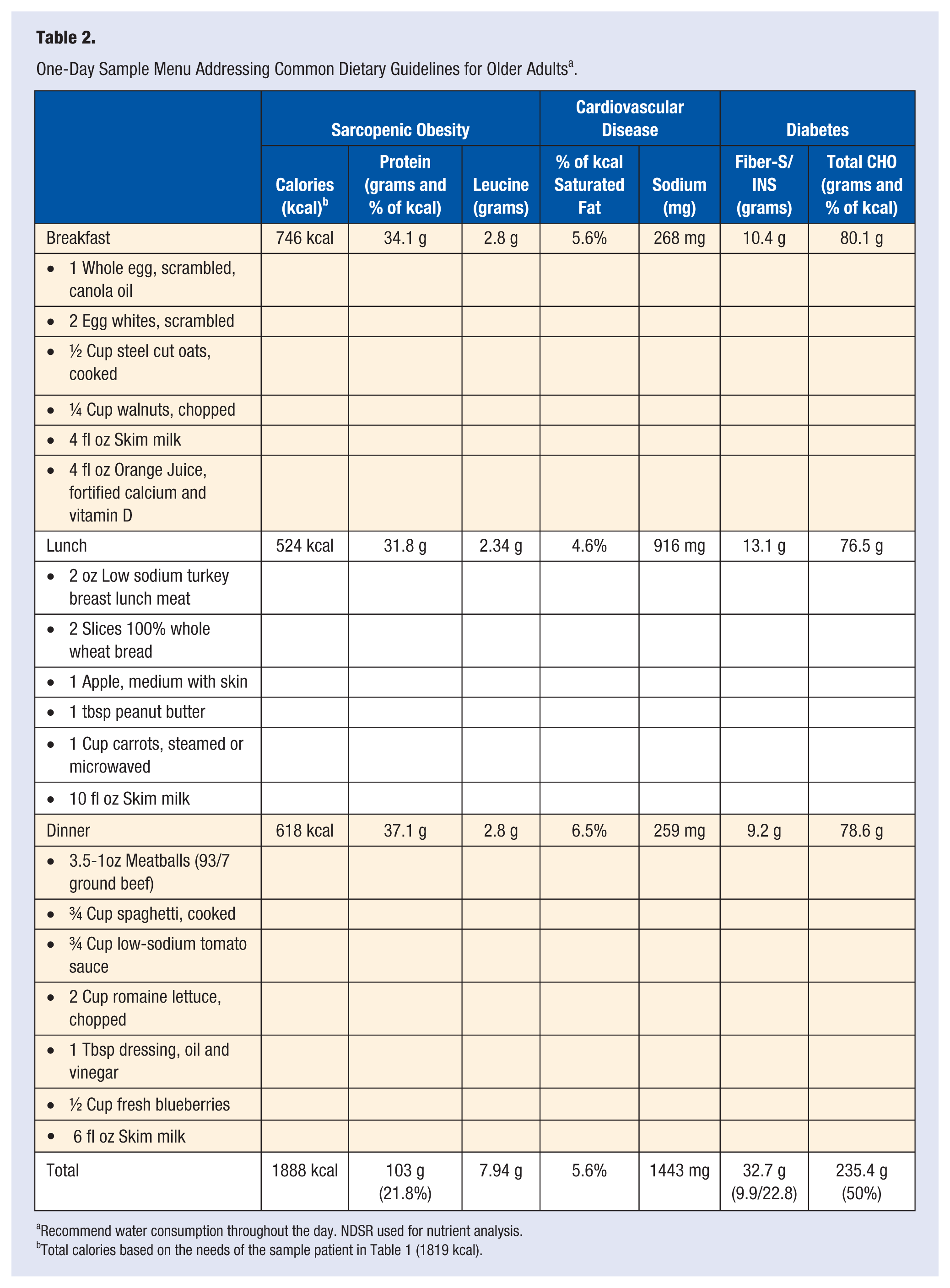
Recommend water consumption throughout the day. NDSR used for nutrient analysis.
Total calories based on the needs of the sample patient in Table 1 (1819 kcal).
Consuming sufficient, high-quality protein to meet the discussed recommendations, while attempting to stay within the various disease-specific recommendations, is difficult. To emphasize the importance of protein, maximize anabolic potential, and supply sufficient leucine, even distribution of protein intake across all three meals is advised. Additionally, protein intake with adequate leucine has been shown to stimulate postprandial insulin release, as well as slow the rise in blood glucose following a meal. 33 Certain allergies and intolerances, such as lactose intolerance, something that often develops due to gradual decline of lactase activity in older adults, 34 limits some high-quality, leucine-rich foods from consideration. Last, Table 2 addresses the nutritional concerns of sarcopenic obesity, CVD, and diabetes due to their prevalence, but other disease such as chronic kidney disease and osteoporosis introduce further challenges when developing an appropriate diet plan. The one-day sample menu in Table 2 does supply the RDA for calcium, but only half (10.4 µg) of the RDA for vitamin D.
Role of the Nutrition Professional
Efforts to educate older adults without providing overcomplicated guidelines will reduce the likelihood of individuals feeling overwhelmed, increasing dietary adherence. Skilled nutrition professionals with advanced knowledge of food, medical nutrition therapy, and the needs of the aging population are crucial. Adequate and quality protein intake is important; however, it should not replace the other dietary needs of the individual. Further considerations should seamlessly account for other nutritionally influenced conditions, the living environment of the individual, and food preferences. While food should be the primary avenue of nutrition, the trained nutrition professional can also support the older adult in evaluating the appropriateness and efficacy of supplementation given their unique and changing health needs.
Footnotes
Acknowledgements
Bianca Gerardi and Dr Jesse Morrell assisted in editing the article.
