Abstract
The passage of Title IX in 1972 resulted in a tremendous increase in the number of females participating in high school and collegiate athletics. This rise in female athletes sparked new focus areas of research in sports medicine related to woman with an important area emerging in 1980, the female athlete triad. This triad consisting of low energy availability, menstrual irregularities, and bone health disruption spans a spectrum of severity and has evolved both in diagnosis and in management throughout the years. Many health questions arise for female athletes and their health care providers, often concerning the most effective management and treatment strategies for this triad. This review examines the research and latest advancements in recognizing and understanding the female athlete triad and explores the most current recommendations for treatment and prevention.
‘The triad of low energy availability, menstrual dysfunction, and low BMD has been well researched and established. . . . there may be a fourth component of the triad, endothelial dysfunction, making the triad a tetrad.’
Organized athletic participation in the United States has seen a 1000% rise in the volume of female athletes at the high school level and a 140% rise at the collegiate level since the passage of Title IX in 1972.1,2 The increase in participating female athletes has further increased the recognition and understanding of the female athlete triad. The first published recognition of the effects of exercise on the female reproductive function was in 1980 with ballet dancers. 3 Yeager et al formally defined the triad of disordered eating, amenorrhea, and osteoporosis as the female athlete triad in 1993. 4 In 2007, the American College of Sports Medicine (ACSM) updated the 1997 statement on the triad, placing emphasis on decreased energy availability, with or without disordered eating, as the initiating insult in female athletes, which in turn leads to menstrual irregularities and negative effects on overall bone health.5,6
Numerous studies have made an effort to document the prevalence of the female athlete triad; however, varying methodology and definitions of disordered eating, menstrual dysfunction, and low bone mineral density (BMD) precludes true comparisons. In high school athletes, disordered eating and low energy availability has been documented at 35.4% among 331 female athletes as studied by Thein-Nissenbaum and Carr in 2011. 7 Previous studies found prevalence values of 20.9% in 423 female athletes and 18.2% in 170 athletes. These percentages were found by using the Eating Disorders Examination Questionnaire, a self-reported questionnaire consisting of 28 food-related questions. These authors used a standard mean score to determine the presence of disordered eating. The variation among prevalence estimations is thought to be due to the specific sports included and underreporting of disordered eating. 7 In college athletes, Beals and Hill, in 2006, studied division II female athletes from 7 different sports and found 25% with disordered eating, 26% with menstrual dysfunction, 10% with low BMD, and 2.6% with all 3 components of the triad. 8
The triad of low energy availability, menstrual dysfunction, and low BMD has been well researched and established. However, there is increasing evidence that there may be a fourth component of the triad, endothelial dysfunction, making the triad a tetrad.9-12 This becomes quite important, as endothelial dysfunction is the first stage in the pathogenesis of cardiovascular disease. The ACSM stresses the importance of viewing each component of the triad on a spectrum from healthy to disease. 6
Definitions and Epidemiology
Energy availability is the dietary energy that remains after exercise training is completed. The 2007 ACSM position stand refocused the definition of disordered eating as a spectrum of disease encompassing low energy availability. That spectrum of low energy availability ranges from inadequate intake without associated abnormal eating behaviors to diagnosed eating disorders, such as bulimia and anorexia nervosa. Some athletes may have low energy availability due to purposeful calorie restriction or increased expenditure, whereas others may fall into that category because they are simply unaware of their true caloric needs. When energy availability falls below a threshold, normal physiological maintenance, growth, and reproduction mechanisms are disrupted. 6
Some studies have defined decreased energy availability as less than 45 kcal/kg of lean body mass per day, whereas others use less than 30 kcal/kg of lean body mass. In 2009, Hoch et al looked at 80 varsity high school athletes and 80 sedentary control students. Using a definition of low energy availability as less than 45 kcal/kg of lean body mass, 36% of the athletes and 39% of the sedentary controls were found to have decreased energy availability. 13 This study suggests that the complications of low energy availability may not be unique to the adolescent female athlete.
Menstrual function ranges from eumenorrhea to amenorrhea. Primary amenorrhea is defined as failure to menstruate by the age of 15. Secondary amenorrhea is defined as absence of menses for more than 3 months in the setting of a previous monthly cycle. 14 Functional hypothalamic amenorrhea is a condition characterized by the absence of menses due to the suppression of the hypothalamic–pituitary–ovarian axis in the setting of no identifiable anatomical or organic disease and is the most likely cause of secondary amenorrhea in female athletes. 15 Within the spectrum is also oligomenorrhea, which involves having fewer than 9 cycles per year. Menstrual irregularities are a cause of concern as they are directly related to BMD.
In the general population, the prevalence of primary amenorrhea is less than 1% and up to 4% for secondary amenorrhea.16,17 Studies have reported the presence of menstrual irregularities in the high school athletic population with estimates ranging from 20% to 54%, whereas others have reported rates among the collegiate athletic population ranging from 26% to 69%.13,18-20 Thein-Nissenbaum et al found a prevalence of menstrual irregularity among 249 female high school athletes to be 19.7%. They defined menstrual irregularity as athletes who had 9 or fewer cycles in the past 12 months, or those athletes who were older than 15 years who had not begun menstruation. Among those with menstrual irregularities, the prevalence of musculoskeletal injury was 63.1%. The athletes with menstrual irregularities were 3 times more likely to sustain an injury resulting in 7 or more days of lost time from sport. 21 Beals and Manore demonstrated similar prevalence of musculoskeletal injury among collegiate athletes with menstrual irregularities, at 65.9%. 22 Injury prevalence and menstrual irregularities is an important correlation to make as these injuries may be attributed from low BMD.
Much like energy availability and menstrual irregularity, bone strength also lies along a disease spectrum. Since the 1980s, bone strength has been assessed with the use of dual-energy x-ray absorptiometry (DXA). The spectrum of bone disease consists of osteopenia and osteoporosis, which both refer to a reduction in BMD. DXA measurements offer both a T-score and a Z-score. The T-score is a measurement that compares BMD to a reference of the young adult population, whereas the Z-score is a comparison of BMD to an age-matched population. The Z-score is of primary interest in the young-athlete population. Currently, there is no consensus on the terminology or cutoffs for classification of BMD DXA scores and disease in premenopausal women, between the World Health Organization, the International Society of Clinical Densitometry (ISCD), and the ACSM 6 (see Table 1).
Classification of BMD DXA Scores and Disease.
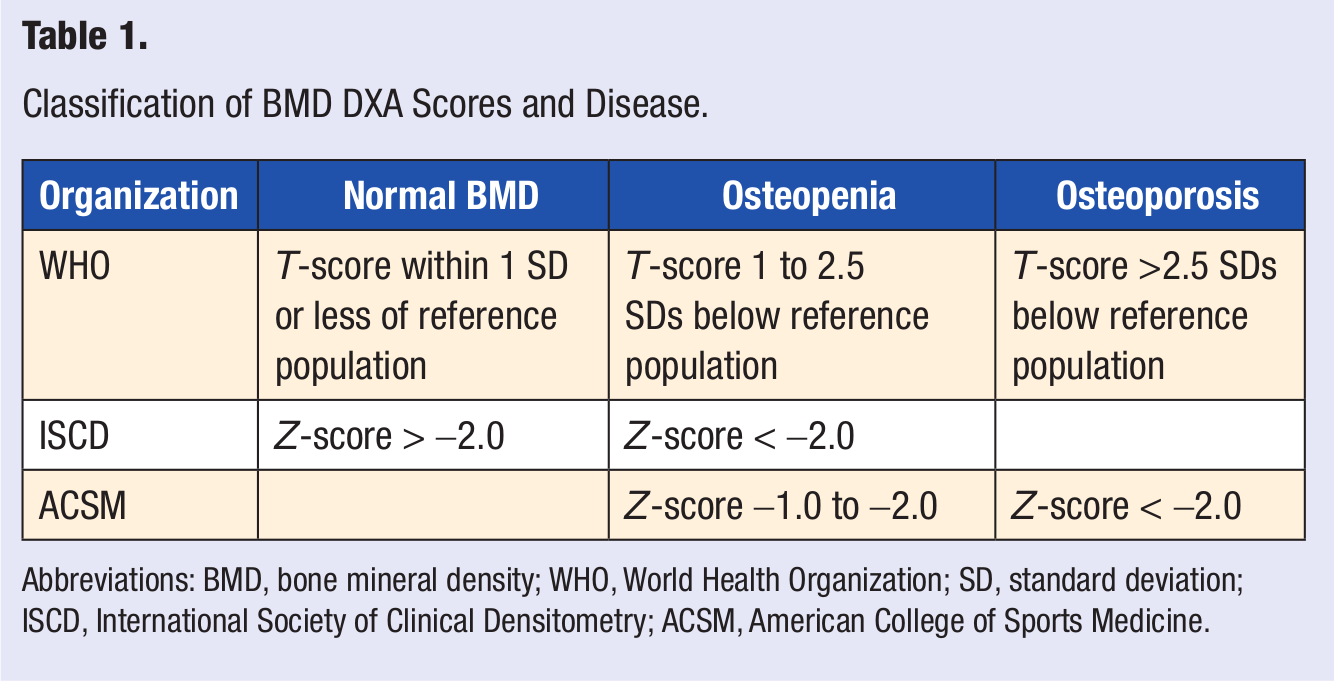
Abbreviations: BMD, bone mineral density; WHO, World Health Organization; SD, standard deviation; ISCD, International Society of Clinical Densitometry; ACSM, American College of Sports Medicine.
The ISCD recommends scanning the lumbar spine and hip in adults. The hip is an unreliable measurement in the growing adolescent; thus, the spine is suggested for this population. 23 Although BMD at the hip may be well within the normal range in athletes engaged in repetitive weight bearing on the lower limbs, those same athletes with menstrual disturbances may display low BMD at the lumbar spine and distal forearm. 24 The lumbar spine and distal forearm should be monitored carefully as they are relatively non-weight-bearing and rich in trabecular bone. Site-specific variation among sport participation was demonstrated by Mudd et al, in 2007, when they found that runners as well as swimmers and divers were more likely to have lower BMD values in the lower leg in comparison to gymnasts, rowers, soccer, field hockey, and softball players. 25 Axial quantitative computed tomography and peripheral quantitative computed tomography have the capacity to measure bone mass, volume, and provide further BMD data; however, these are mainly used in the research setting, thus having limited availability. 26
BMD scores can also provide a false sense of reassurance. Studies have demonstrated that many athletes in weight-bearing sports have a BMD 5% to 30% higher than sedentary controls. Thus, in this population, a decrease in BMD using standard testing may suggest much more advanced disease compared to their controls.27-29 With this known, prevalence studies of low BMD in the athletic population are more difficult.
The 3 components of the female athlete triad, low energy availability, menstrual irregularities, and low BMD, have been thoroughly studied and are well established; however, there is now increasing evidence pointing toward a fourth component, endothelial dysfunction. It is known that coronary and peripheral vessels have estrogen receptors that allow estrogen to bind, stimulating the production of nitric oxide, which in turn leads to vasodilation. 30 We also know that in amenorrheic females, they have hormone profiles similar to women in menopause, with low levels of estrogen, suggesting a loss of normal endothelial function.
The gold standard for the evaluation of endothelial dysfunction is by noninvasive high-resolution ultrasound of the brachial artery diameter in response to hyperemic flow. 31 Measurements of flow velocity are taken at baseline and then after forearm occlusion with a blood pressure cuff. Using this measuring device, studies have shown a positive correlation between brachial artery endothelial dysfunction and coronary artery endothelial dysfunction. 32 This may be important, since coronary artery endothelial dysfunction is a precursor to cardiovascular events, such as unstable angina, myocardial infarction, or ischemic stroke.
Several studies have demonstrated a correlation between amenorrheic athletes and endothelial dysfunction. In 2003, Hoch et al found a significantly decreased brachial artery vasodilator response to hyperemic flow in amenorrheic runners (1.08 ± 0.91%) compared to a control group (6.38 ± 1.38%) and oligomenorrheic runners (6.44 ± 1.28%). 9 Rickenlund et al suggested similar findings in 2005. 10 In 2011, Hoch et al investigated a cohort of professional ballet dancers, finding 64% with decreased brachial artery vasodilatory response, and of those 72% reported menstrual dysfunction. 12 These were relatively small studies that consistently demonstrate an association between amenorrhea and endothelial dysfunction. In the future, larger sample sizes are needed to investigate this potential correlation further, as the clinical implications are great.
Pathophysiology
Clinical eating disorders include anorexia nervosa, bulimia nervosa, and disordered eating not otherwise specified. Several predisposing factors for clinical eating disorders have been identified; dieting is the most common factor. Dieting has been associated with a 5-fold to 18-fold increased risk of developing a clinical eating disorder 6 months later. 33 Other factors include low self-esteem and self-control. It is also important to recognize clinical eating disorders are often accompanied by comorbid psychological conditions, including depression, anxiety disorder, and obsessive–compulsive disorder. 34 And finally, there is also mounting evidence for a genetic component to eating disorders.35,36 With these associations known, antidepressants have been suggested as a treatment modality for anorexia or bulimia.
The etiology of low energy availability in athletes is less clear. Athletes may attempt to lose weight or body fat for the benefit of their athletic performance. This may consist of knowingly restricting caloric intake or an unintentional lack of adjusting caloric intake to correspond with increased energy expenditure. Nutritional guidance helps show athletes how to eat accordingly to replace energy expenditure as opposed to in response to hunger; studies have shown that although decreased dietary intake does increase hunger, exercise that results in equal energy deficit does not increase hunger.6,36
When net energy availability is insufficient, the body’s natural response to promote survival is to reduce the amount of energy used for growth and reproduction. In females with insufficient net energy, there is a disruption in the pulsatile secretion of gonadotropic-releasing hormone (GnRH) from the hypothalamus, thus causing a disruption in pulsatile secretion of luteinizing hormone (LH) from the pituitary. The result is a functional hypogonadotropic hypothalamic amenorrhea or dysmenorrhea in athletes, and the specific pathway of this disruption is yet to be identified. 6 This can occur even after just 5 days of low energy availability. 37 LH pulsatility is abruptly disrupted when energy availability drops below 30 kcal/kg of lean body mass per day, as opposed to a linear relationship between LH pulsatility and energy availability. 37 Furthermore, neither low body weight nor the act of exercising itself can explain the disruption of LH pulsatility in athletes, suggesting that LH pulsatility depends more importantly on energy availability. 38
Functional hypothalamic amenorrhea appears to also have a genetic component, and several genes may add to the susceptibility of certain females to the functional changes in GnRH secretion. 39 There has also been some evidence that the hormone leptin may serve as a signal to the hypothalamus regarding energy availability. In one study, treatment of amenorrheic athletes with recombinant human leptin restored menstrual cycles. 40 The roles of several hormones, including insulin, insulin-like growth factor-I, cortisol, and growth hormone functioning as possible signaling molecules remains unclear. 41
In postmenopausal women, the most important contributor to low bone mass and osteoporosis is estrogen deficiency, which is related to estrogen’s suppression of osteoclast activity. 42 The relative estrogen deficiency in athletes with amenorrhea contributes to the increased osteoclast activity, although this is only partially responsible for low bone mass in amenorrheic athletes. This is demonstrated best by a lack of normalization of BMD even after years of estrogen replacement.43,44 Undernutrition has also been shown to reduce the rate of bone formation. 45 One randomized trial placed women into either a dieting group or exercising group with the goal energy availability to be below 30 kcal/kg lean body mass per day. Within 5 days blood markers of bone formation were noted to be decreased, and bone resorption increased. 46 Other factors that affect bone acquisition include leptin, growth hormone, and insulin-like growth factor, which have been discussed in the literature as being affected by low energy availability, further explaining a mechanism by which low energy availability can translate into low BMD.
The statement “Senile osteoporosis is a pediatric disease” alludes to the concept that failure to achieve peak bone mass during adolescence and young adulthood often leads to osteoporosis and increased fracture risk later in life. 47 Most women achieve peak bone mass during their 20s, and while the majority of variability in peak bone mass is determined by genetics, the remaining 30% to 40% of variability can be attributed to environmental factors including diet and exercise. 48 This important understanding in the pathophysiology of osteoporosis in postmenopausal women again underscores the need to prevent or treat low energy availability, amenorrhea, and low bone density that can occur in young women.
Screening and Diagnosis
History
The greatest opportunity to screen for 1 or more components of the female athlete triad occurs most often at the preparticipation physical exam (PPE) or annual physical exams. The Female Athlete Triad Coalition, a group of representatives from the ACSM, International Olympic Committee, American Medical Society for Sports Medicine, American Academy of Orthopaedic Surgeons, American Academy of Pediatrics, NCAA, American Dietetic Association, and National Athletic Trainers Association worked to generate a list of 12 items to screen for the triad on PPEs (see Table 2).
Female Athlete Triad Coalition Screen for the Triad on PPEs.
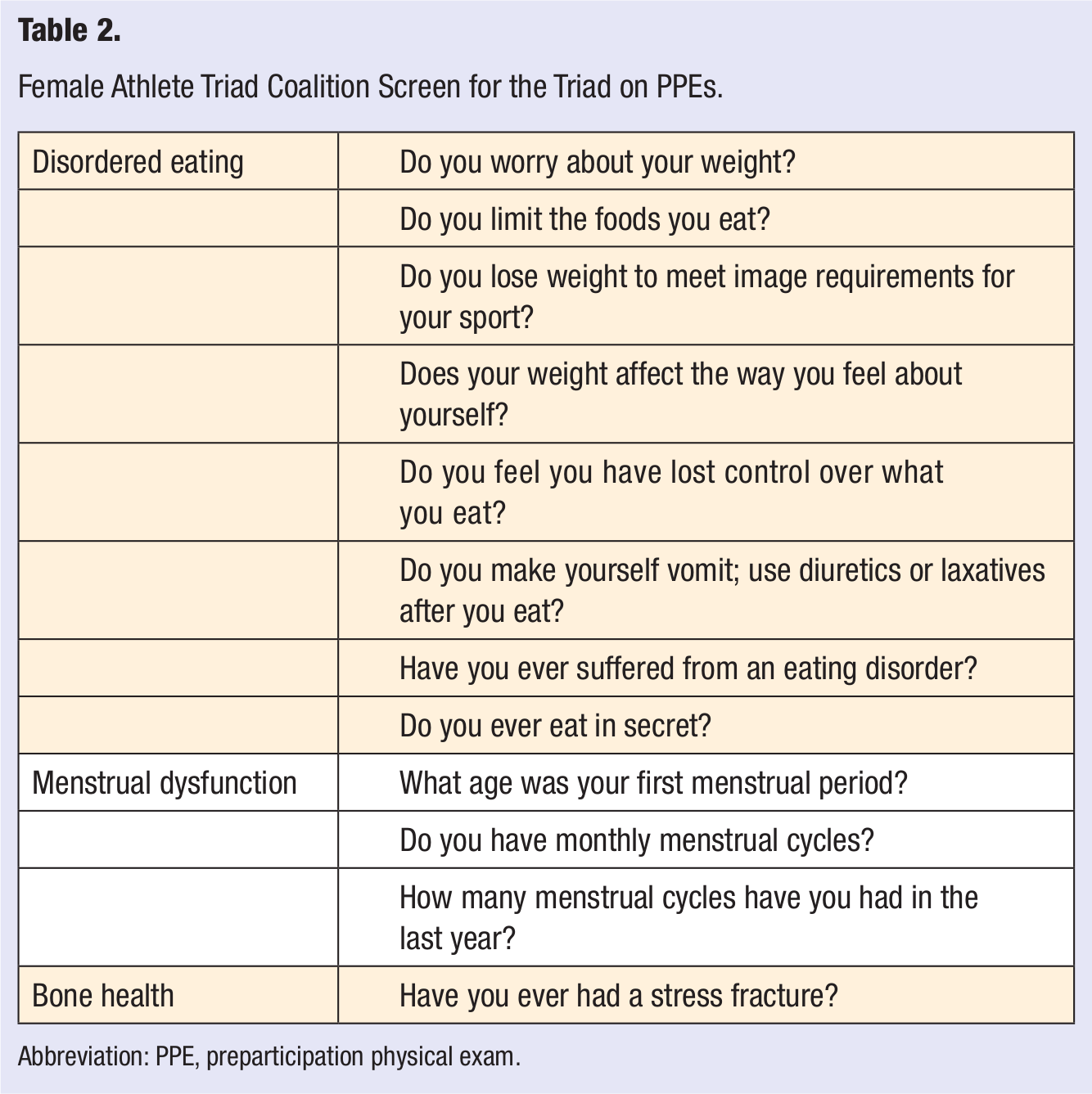
Abbreviation: PPE, preparticipation physical exam.
A recent study in 2012 by Mencias et al looked at 257 of the NCAA Division I Universities’ PPE forms, and they found that only 25 universities (9%) had 9 or more of the 12 recommended items included in their forms, whereas 127 universities (44%) included 4 or less items. 49 The 12-item questionnaire has not been validated but is quick and easy to complete to help raise red flags for more thorough evaluations. Other screening questionnaires may be used during annual exams to detect disordered eating; the Bright Futures guidelines and the SCOFF Questionnaire are 2 validated options.50,51
Physical Exam
Exam findings suggestive of the triad include bradycardia, orthostatic hypotension, low body mass index, acrocyanosis of the hands and feet, hypothermia, hypercarotenemia, lanugo hair or thinning scalp hair, oral ulcerations, dental enamel erosions, and parotid gland enlargement. Pelvic exam may reveal signs of hypoestrogenism with vaginal atrophy. If an electrocardiogram (EKG) is obtained, it may reveal signs of electrolyte disturbances, such as prolonged QT or U waves. However, in most cases the physical exam will be normal, which should not rule out the presence of any of the components of the triad. 52
Laboratory Testing and Imaging
A standard set of laboratory tests should be obtained if one component of the triad is suspected. These labs should include a complete blood cell count with differential, complete metabolic panel; erythrocyte sedimentation rate; thyroid function tests; and urinalysis. 53 In cases of disordered eating, a stool guaiac in those with a history of laxative abuse and urine electrolytes and EKG and salivary amylase in those with a history of self-induced vomiting would be appropriate. In cases of amenorrhea, a pregnancy test, serum FSH, LH, prolactin, and thyroid stimulating hormone should all be obtained to rule out other causes of secondary amenorrhea, as functional hypothalamic amenorrhea is a diagnosis of exclusion. 54 If the pregnancy test is negative and the laboratory tests are normal, a direct measurement of estrogen can be obtained by ordering a serum estradiol, or a progesterone challenge test can be performed to assess estrogen levels indirectly. This involves the administration of medroxyprogesterone acetate 10 mg once daily for 7 to 10 days with subsequent withdrawal bleeding thereafter.
The ACSM recommends a baseline DXA test in any athlete with evidence of greater than 6 months of amenorrhea, oligomenorrhea, and/or disordered eating. Also, any history of stress fractures should warrant a DXA. 55 In addition, reevaluation with DXA is recommended after 12 months for any persistent components of the triad. 6
Prevention and Treatment
Prevention
A multidisciplinary approach to prevention is key when approaching the female athlete triad. A team may consist of health care providers, mental health practitioners, dietitians, coaches, parents, athletic trainers, exercise physiologists, physical therapists, and so on. Educating athletes about energy availability, nutritional intake, and balancing intake with energy output is a key component to deter the sequelae. 6 As stated previously in the discussion of prevalence, components of the female athlete triad are present in both athletes and nonathlete adolescent females. Athletic demands compounded with the societal and media pressures to be thin are all likely to play a role in the prevalence of low energy availability and disordered eating 13 ; therefore, a targeted audience of early adolescent females would be the most logical place to begin education on prevention. Specific teaching should include daily caloric needs and caloric expenditure with activities, along with intake of nutrients such as vitamin D and calcium. Time should be spent on a discussion of normal menarche, menstrual function, and cycle frequency. It should also be emphasized that a decision to try to lose weight should not be based on a coach’s recommendation, nor should the coach determine the athlete’s ideal body weight. Instead, these decisions should be made in concert with a nutritionist’s guidance to ensure adequate energy availability. If a decision is made that the athlete may safely make changes in her diet and/or training to improve performance, then the athlete should be followed closely. 56
Treatment
Nonpharmacological
The recommended first-line treatment strategy for athletes with oligomenorrhea and amenorrhea consists of increasing energy availability by either increasing dietary intake or decreasing energy expenditure. 6 With stabilization of energy availability, Miller et al were able to demonstrate increases in BMD without the resumption of menses, suggesting an independent or additive effect on bone health. A 38% increase in body weight over 3 months resulted in a BMD increase of 2.6% (hip) and 1.1% (spine) in women who remained amenorrheic. 57 Comparatively, Dominguez et al, in 2007, had participants regain 90% of their ideal body weight, which resulted in increases in spine and hip BMD by 4.38% and 3.77%, respectively, after 2.2 months. 58 In addition, Warren et al, in 2003, demonstrated an 11% increase in BMD after reversal of amenorrhea. 59 Further studies with larger cohorts are necessary to determine the relationship between the increase in energy availability and timing of return of menses and how it correlates with bone restoration. 25
Nutritional counseling is vital to the achievement of restoring adequate energy availability. Counseling should include recommendations on daily intake of calcium and vitamin D. For athletes who do not routinely meet these goals or display resistance to counseling or monitoring, restrictions on practice or competition may be necessary and enforced with a written contract. 6 Written contracts may be based on weight goals, return of menses, or DEXA Z-score improvements by a certain time period determined by the supervising physician. These contracts are athlete specific.
The American Physical Therapy Association recommends changing a workout routine from mainly cardiovascular endurance training to that with more resistance and weight training. 60 High-intensity resistance training in premenopausal women has shown an increase in the BMD of the lumbar spine and the forearm.62-63
Pharmacological
The American Academy of Pediatrics supports the initiation of estrogen therapy through oral contraceptive pills (OCPs) in adolescent females in primary amenorrhea at the age of 15 or in secondary amenorrhea occurring 3 years postmenarche. 64 Others have endorsed the use in female athletes with stress fractures. However, the use of OCPs and other forms of estrogen therapy remains controversial due to inconclusive studies. Several longitudinal cohort studies have demonstrated an increase in BMD while on a combined OCP.65-68 Three randomized control trials studying effects of combined OCP treatment on the BMD of oligomenorrhea/amenorrheic and eumenorrheic runners were inconclusive.59,69,70 Other studies have also demonstrated negative effects of estrogen therapy. Cutler in 1997 suggested that exogenous estrogen replacement may lead to premature closure of the physes (growth plates) in young athletes. 71 Also, Hartard et al demonstrated lower BMD in the spine and femoral necks of young female runners using OCPs. 72 No systemic reviews have been done specifically addressing initiation of OCPs in amenorrheic females and its effect on BMD. Further study is necessary to determine whether exogenous estrogen therapy is safe, effective, and for whom. Until then, health care providers will need to use their best judgment on a case-by-case basis for who may benefit from the use of exogenous estrogen therapy.
Calcium and Vitamin D supplementation are often prescribed for amenorrheic athletes; however, there is no consensus on necessary dosage. The ACSM recommends a calcium intake of 1000 to 1300 mg/day, 6 whereas the International Olympic Committee Medical Commission recommends at least 1500 mg/day. 57 Measuring the serum 25-Hydroxyvitamin D can assess vitamin D levels. Those falling below 50 mmol/L or 20 ng/mL should be supplemented. A dose of 400 to 800 IU per day in combination with calcium intake should suffice daily requirements. 73
With the emergence of a possible fourth component to the triad, endothelial dysfunction, folic acid has also emerged as a potential treatment. Folic acid has demonstrated potential cardiovascular benefits, with improved endothelial function, decreased arterial stiffness, improved blood pressure, and decreased thrombotic activity, as well as playing a role in upregulating the production of nitric oxide. 30 Several studies have demonstrated improvements in vasodilatory response with implementation of folic acid. Hoch et al, in 2009, improved amenorrheic runners vasodilator response from 3.0 ± 2.3% to 7.7 ± 4.5% after initiating 10 mg/day of folic acid supplementation. 12 Similar positive results were found in eumenorrheic runners initiating folic acid in comparison to placebo as well as professional ballet dancers.74,75 These studies suggest that low-dose folic acid supplementation, which has a very low adverse effect profile, may be an effective and safe treatment modality to combat the endothelial dysfunction associated with female athlete triad.
Bisphosphonates reduce bone turnover and are approved only for the treatment of postmenopausal osteoporosis. The International Olympic Committee Medical Commission does not approve the use of bisphosphonates in premenopausal women due to the long half-life (up to 10 years) and their potential teratogenic effects. 57
Conclusion
The tremendous increase in the number of female athletes since the introduction of Title IX has drawn researchers to study its effects on women’s health. Evidence reveals low energy availability leads to a cascade of menstrual irregularities and bone health disturbances. In the future, further emphasis may be needed on a potential fourth component to the female athlete triad, endothelial dysfunction. Further studies are necessary to determine whether endothelial dysfunction is necessary to screen for and treat.
With a better understanding of the pathophysiology, prevalence, and treatment of this triad, we can target specific audiences to engage in a discussion of preventative strategies. Not only can we improve prevention but also the treatment of these patients. At present, multiple organizations offer differing management and screening recommendations. A consensus among the multidisciplinary team of practitioners, using current and future studies, is needed to standardize treatment guidelines of female athlete triad.
