Abstract
Chronic pain has a significant economic and social impact on the community. The most common medical treatments for it include paracetamol, anti-inflammatory agents, and opioid analgesics. However, many of these medications cause side effects, and their long-term effectiveness is questionable. The traditional alternative to the biomedical approach is cognitive behavioral therapy. However, this has also been shown in recent studies to have only modest benefit. It is becoming clear that the effective management of chronic pain requires a more holistic, systems-based approach, hence the emerging interest in the relationship between pain and lifestyle. The authors aim to review the literature regarding the relationship between comprehensive lifestyle changes, markers of systemic inflammation, and the perception of chronic pain. An extensive search of bibliographic databases, including MEDLINE, PubMed, Web of Science, and Cochrane Library databases was made. A total of 2197 articles were identified using the search strategy. Only 44 articles were retrieved for critical appraisal, of which only 2 studies met the prespecified primary inclusion criteria and were included in the final review. These data provided some evidence that a single lifestyle factor (sleep restriction or disturbance) can produce elevated levels of interleukin-6, which is associated with higher pain intensity ratings. However, this review has highlighted a paucity of research based around the relationship between lifestyle, metaflammation, and chronic pain. There is a clear need for well-designed trials examining comprehensive lifestyle interventions and their effect on both pain intensity and markers of metaflammation.
…It has been demonstrated that chronic pain is strongly associated with other chronic diseases (eg, heart disease and type 2 diabetes) that have been linked to lifestyle.
Introduction
Chronic pain is defined as pain that extends beyond the expected healing period or for more than three months 1 and can develop even without a clear pathological trigger. 2 Approximately 20% of adults in Western countries experience chronic pain.3-5 Although it can be a debilitating condition for the individual, chronic pain also has a significant economic and social impact on the community.1,3,5
Currently, the most common treatments for chronic pain include paracetamol, anti-inflammatory agents, and opioid analgesics. However, all these medications have been associated with side effects, and their long-term effectiveness in treating chronic pain is questionable.6,7 The traditional alternative to biomedical treatments is cognitive behavioral therapy. However, this has also been shown in recent studies to have only modest benefit.8,9
It is becoming clear that the effective management of chronic pain requires a more holistic, system-based approach than has been used to date.7,10 Hence there has been an emerging interest in the relationship between chronic pain and lifestyle. It has been demonstrated that chronic pain is strongly associated with other chronic diseases (eg, heart disease and type 2 diabetes) that have been linked to lifestyle.
11
In addition, it has been shown that systemic low-grade inflammation, or
Although metaflammation was initially implicated in atherosclerosis through its effects on the vascular endothelium, other sites and diseases have been recently identified.14,18,19 The inflammatory process may also spread across the neurovascular junction to activate glia, resulting in sensitization of central neural pathways and facilitation of chronic pain.7,20-27 Attention is thus turning to those lifestyle factors that may induce this neurovascular inflammation, with the suggestion that they may be among the same as those that produce metaflammation. Accordingly, it has been suggested that we need to consider chronic pain, at least in part, as a lifestyle-related disorder.10,28 This review aims to identify and examine studies that have measured the relationship between lifestyle factors (diet, physical activity, sleep, stress, obesity, and smoking), which are known inducers of metaflammation, and intensity of chronic pain.
Method
The search strategy sought to identify original articles published between January 2000 and February 2011 by searching the MEDLINE, PubMed, Web of Science, and Cochrane Library databases. The following keywords were used in the search: lifestyle, lifestyle interventions, physical activity, physical inactivity, sedentary, nutrition, diet, cafeteria diet, Mediterranean diet, fish oil, overweight, obesity, sleep, disturbed sleep, sleep disorders, insomnia, stress, anxiety, depression, smoking, cigarettes; inflammation, systemic inflammation, subclinical inflammation, metaflammation, neurovascular inflammation, neural plasticity, gliosis, pain, chronic pain, persistent pain, neuropathic pain. Hand searching was also conducted, and original articles referenced in the retrieved articles were screened for inclusion. To be included in this review, studies needed to involve humans and specifically examine the relationship between chronic pain and lifestyle factors (nutrition, physical activity, sleep, smoking, stress/anxiety/depression, and overweight/obesity) by including measurement of low-grade systemic inflammation through established inflammatory markers. In addition, studies that investigated single or multiple lifestyle interventions and chronic pain, but without measuring inflammation, were also retrieved in a secondary search but not included in the discussion (Table 1). Original articles prior to January 2000 were excluded, as were articles that related to acute pain, cancer pain, or pain related to rheumatoid arthritis, osteoarthritis, fibromyalgia, Complex Regional Pain Syndrome (CRPS), and migraine. Studies that involved participants <18 and >65 years of age were also excluded. The PersonalBrain 6 software (http://www.thebrain.com/) was used to mind-map all the results and to organize these into categories for analysis.
Studies That Examined the Association Between Lifestyle Interventions and Chronic Pain (but not Metaflammation).
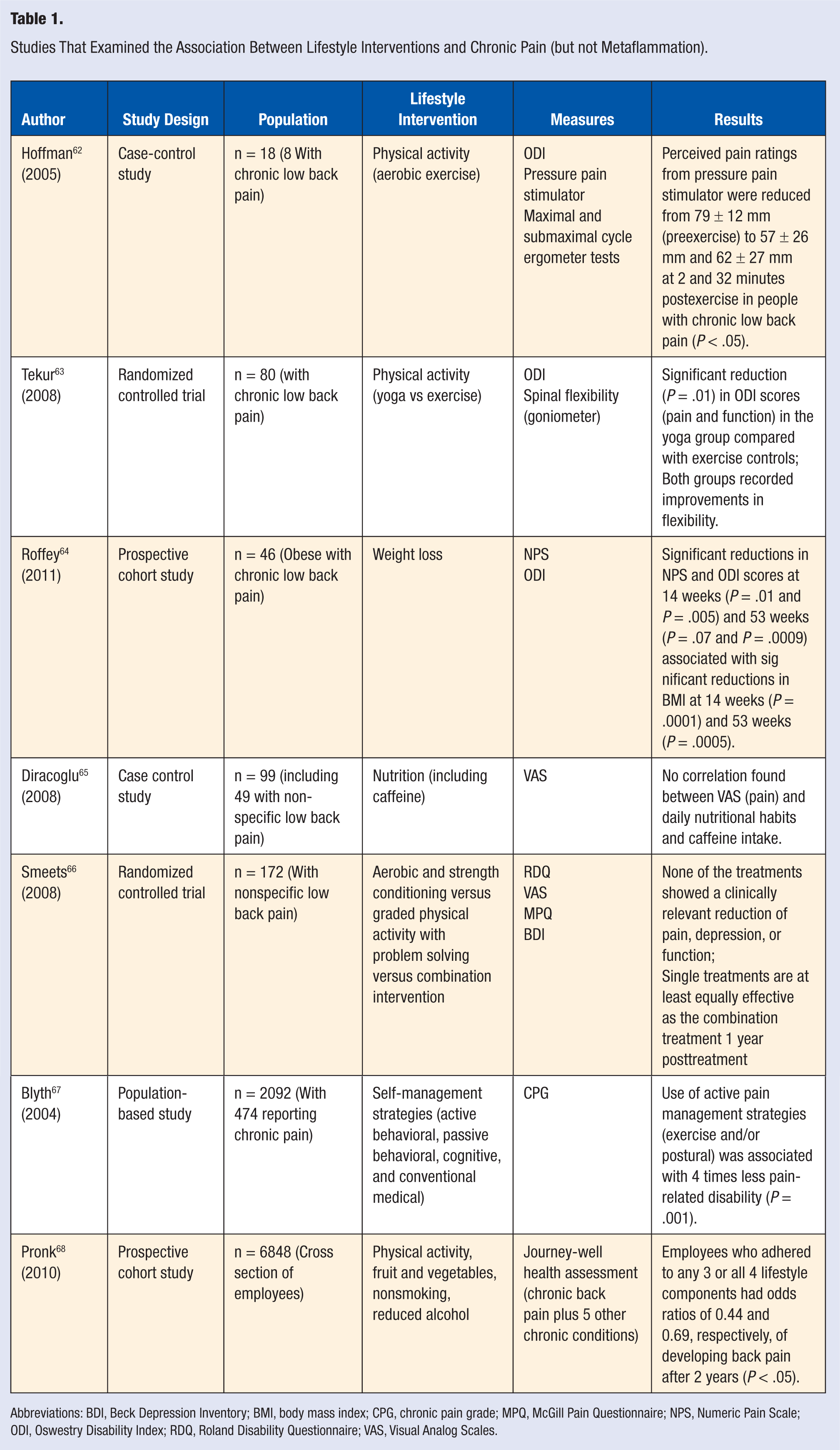
Abbreviations: BDI, Beck Depression Inventory; BMI, body mass index; CPG, chronic pain grade; MPQ, McGill Pain Questionnaire; NPS, Numeric Pain Scale; ODI, Oswestry Disability Index; RDQ, Roland Disability Questionnaire; VAS, Visual Analog Scales.
Results
A total of 2197 articles were identified using the search strategy. Following the removal and/or exclusion of articles based on title, duplication, or abstract, only 44 articles were retrieved for critical appraisal. A further 35 articles were excluded that did not meet the primary inclusion criteria. Of these articles, 7 satisfied the secondary search criteria, and the summary data from these presented in Table 1. However, only 2 studies met the prespecified primary inclusion criteria and were included in the final review. Numerous studies examined the associations between lifestyle and metaflammation, but only these 2 studies addressed all 3 variables and fully satisfied the primary selection criteria. In Table 2, we summarize the study design, study populations, lifestyle factors, subjective and objective measures, and results for each of the 2 studies.
Studies That Examined the Association Between Lifestyle and Metaflammation and Chronic Pain.
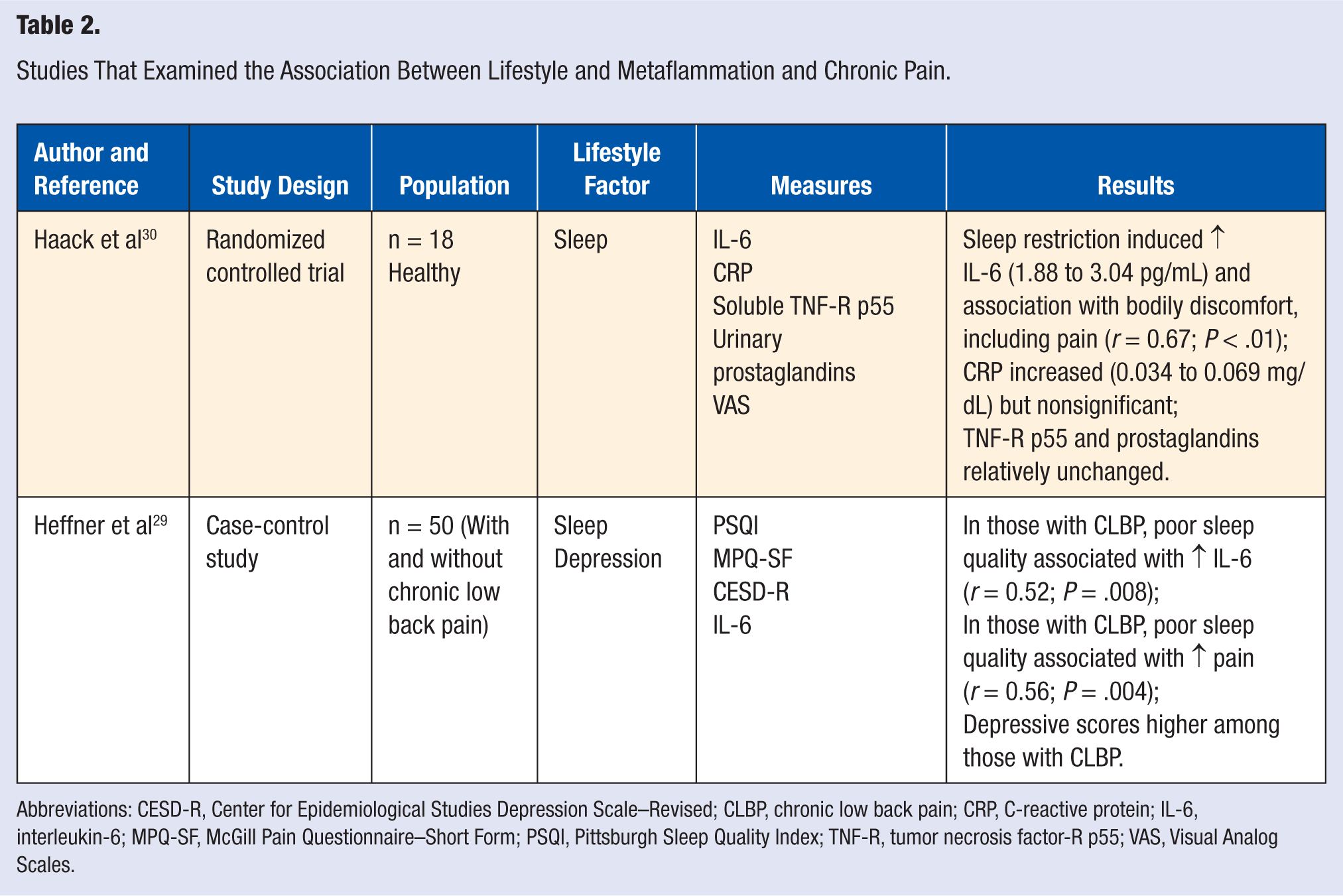
Abbreviations: CESD-R, Center for Epidemiological Studies Depression Scale–Revised; CLBP, chronic low back pain; CRP, C-reactive protein; IL-6, interleukin-6; MPQ-SF, McGill Pain Questionnaire–Short Form; PSQI, Pittsburgh Sleep Quality Index; TNF-R, tumor necrosis factor-R p55; VAS, Visual Analog Scales.
Both studies were conducted in the United States since 2006. The total number of participants across the 2 studies was 68, with the age range being 21 to 40 years (6 women and 12 men) in 1 randomized controlled study and 19 to 55 years (30 women and 20 men) in the other case-controlled trial (CCT). It is interesting to note that of all the potential lifestyle factors under consideration in this review, both these studies were limited to investigations of sleep disturbance or restriction, although the study conducted by Heffner et al 29 included an analysis of depression as a covariate.
Haack et al 30 used 18 healthy volunteers, whereas Heffner et al 29 monitored 50 participants, 25 of whom suffered from chronic low back pain (CLBP) for greater than 6 months. Both studies established significant exclusion criteria to minimize confounding variables. Whereas they both measured perceptions of pain, they each used different instruments to do this. Haack et al 30 used a computerized Visual Analog Scale to measure bodily discomfort and tiredness/fatigue every 2 hours of wakefulness during the study. Heffner et al 29 used the McGill Pain Questionnaire-Short Form, which was administered once on the second and final day of the study. To measure sleep quality, the first study restricted sleep for 12 consecutive days and applied the Visual Analog Scale consistently over the study period, whereas the second study used the Pittsburgh Sleep Quality Index to measure the participants’ perceptions of their sleep on 2 occasions, measuring both the previous month’s sleep and sleep on the night before measurement. Each study measured the cytokine IL-6, although Haack et al 30 also measured CRP and TNF-R p55 as well as urinary prostaglandins.
Discussion
We concluded from the available studies that sleep disturbance or restriction was associated with higher levels of IL-6, which was in turn associated with higher levels of perceived pain. There is the possibility that the association could be bidirectional. In the sleep-restricted group, Haack et al 30 showed a significant increase in IL-6 along with higher ratings of bodily discomfort (pain) and tiredness/fatigue but a nonsignificant increase in CRP and no change in TNF-R p55. Heffner et al 29 demonstrated that poor sleepers had higher levels of IL-6 compared with good sleepers and that this was strongly associated with higher perceived pain ratings. Both authors demonstrated increases in IL-6 consistent with metaflammation; however, in the study by Heffner et al, 29 IL-6 was the only marker that was measured, whereas Haack et al 30 used IL-6, CRP, and TNF-R p55 as their markers.
It is acknowledged that metaflammation differs from classical inflammation, in that it is systemic and chronic and typically only produces a modest increase in inflammatory mediators such as TNF-α, interleukin-1β (IL-1β), IL-6, CRP and some transcription factors.16,17,21,31,32 In analyzing these modest increases in inflammatory mediators and determining what is outside the normal range, there will always be some degree of uncertainty because there are a range of potential confounding factors, including age, gender, ethnicity, and medications.33,34 However, with respect to CRP, Pearson et al 35 have identified that levels >3.0 mg/L could be reflective of systemic inflammation, placing individuals at a high risk of cardiovascular disease. There do not appear to be similar reference intervals for the ILs and TNF- because these are mostly tests used within the research and not diagnostic fields.
In relation to chronic pain, Milligan and Watkins 26 point to IL-1β, TNF-α, and IL-6 playing key roles in central sensitization and hyperalgesia. McCooey and Wilmarth 36 have indicated that CRP, although correlated with acute pain, has not been shown to be associated with CLBP, which was supported by Haack et al 30 in their study. Although there were significant increases in IL-6 in both studies, neither examined the role of IL-1β or TNF-α. Haack et al 30 acknowledged that measuring TNF-α rather than TNF-R p55 may have been more enlightening for their study. Although both studies identified elevated IL-6 as a consequence of sleep deprivation and a key link to chronic pain, neither study explored the possible neurovascular mechanisms to help explain this connection between metaflammation and chronic pain. Heffner et al 29 did acknowledge that this deserves further scrutiny, and both suggested that IL-6–induced hyperalgesia may result from the inflammatory effects on pain sensory pathways. The comprehensive exclusion criteria in the CCT 29 did reduce the likelihood that the elevation of IL-6 levels could be attributed to inflammatory-related conditions, although they did not exclude episodic pain as a possible confounding factor.
There is substantial evidence that a range of lifestyle factors can induce metaflammation,37-45 but it is still unclear how this inflammatory response can facilitate, aggravate, or mediate chronic pain. It was once thought that chronic pain was confined to neural pathways; however, glial cells (particularly microglia and astrocytes) have recently been identified as potential mediators of inflammatory responses, with the connections between the neural, vascular, and glial systems suggesting a tripartite link between lifestyle and chronic pain.22,32,46,47 It has been proposed that chronic inflammation of glial cells in the neurovascular junction and the subsequent immune system reaction may stimulate nearby neural synaptic tissue, resulting in a central sensitization and pain.7,10,23,47-49 The activation of glial cells occurs to some extent in acute pain, but it is the chronic activation of these cells that may be implicated in persistent or “locked in” 50 pain states.7,22,51 It appears that following peripheral nociceptive stimulation, microglia become activated and release proinflammatory substances, including IL-1β, TNF-α, and IL-6, which subsequently activate more microglia and also astrocytes, facilitating the development of chronic pain by modulating neuronal activity.21,27,32,46,52
Increasing evidence indicates that these proinflammatory substances also target the endothelial cells within the neurovascular unit to induce changes in the permeability and integrity of the blood-brain barrier, which may have an etiological role or exacerbate neuropathological conditions.7,49,53 If metaflammation in vascular endothelial tissue is communicated to the neurons via glial cells and is reflected in the early stages as acute pain, long-term glial activation could result in chronic pain associated with ongoing lifestyle-related stimuli.10,22 Consequently, it has been speculated that metaflammation may extend to both central and peripheral glial connections associated with pain perception, thus linking lifestyle-related inducers (at least in some cases) to the neural sensitization underlying chronic pain.10,28
Haack et al 30 agreed that proinflammatory cytokines may be one of the key factors in the development of chronic pain, although they acknowledged that most of this evidence to date has come from animal studies. For example, it has been shown that exogenous administration of proinflammatory cytokines in animal studies can produce hyperalgesia and pain.54,55 In so doing, these and other studies have examined the therapeutic benefits of targeting glial cells to ameliorate chronic pain conditions; however, they have tended to focus specifically on potential pharmacological and not lifestyle interventions.7,21,26,47,49,54,56,57 However, Gosselin et al 23 have cautioned against embracing the universal association between glial cells and chronic pain because the data available for humans are very limited.
Both studies included in this review, although instructive, had some limitations. First, the use of relatively small sample sizes was a limitation in both studies. Second, Haack et al 30 conducted their trial in a clinical environment (research center) for 16 consecutive days, so it may not be possible to generalize their results to naturally occurring sleep disturbances. Finally, the CCT 29 recruited a relatively healthy and young population with CLBP; used a modified version of the Pittsburgh Sleep Quality Index on day 2, which had not yet been validated; measured only 1 inflammatory cytokine (IL-6) on a single occasion; did not identify BMI or preblood diet as possible confounders; and did not exclude episodic pain. Despite these limitations, both studies have demonstrated a relationship between a lifestyle factor (sleep deprivation), metaflammation (as evidenced by elevated levels of IL-6), and subjective ratings of pain in those with CLBP.
As previously mentioned, there are numerous studies that support the view that metaflammation may be associated with the sensitization of central neural pathways and facilitation of chronic pain.7,20-27,58 Furthermore, it is worth noting that the following studies have also provided some evidence to suggest that there may be an association between metaflammation and various aspects of lifestyle. The traditional Mediterranean style diet has been associated with a reduced risk of cardiovascular disease and most likely mediated by improvements in the inflammatory profile.38,40,45 Maroon and Bost 59 demonstrated the anti-inflammatory effects of omega-3 essential fatty acids typically found in fish and/or fish-oil supplements. The consumption of a new form of hybridized beef (wagyu) was shown to produce a significantly higher postprandial inflammatory response than eating a game meat (kangaroo). 60 In their review of 66 studies, Forsythe et al 61 concluded that weight loss interventions can lead to significant reductions in CRP, TNF-α, and IL-6, particularly when the weight loss exceeds 10%.
Conclusion
This literature review has highlighted a paucity of studies demonstrating a possible relationship between lifestyle, metaflammation, and chronic pain. A review of relevant data, however, suggests that such a link is physiologically feasible. Many studies that were excluded from this review had identified a strong link between lifestyle and chronic pain or lifestyle and metaflammation, but only the 2 studies identified29,30 had extended the research to examine lifestyle, metaflammation, and chronic pain. These studies provided some evidence that sleep restriction or disturbance is associated with elevated levels of IL-6 and higher ratings of perceived pain. It is likely, however, that other lifestyle factors (obesity, poor diet, inactivity, inadequate sleep, smoking, and stress) that are known to cause metaflammation could also have neurovascular effects and facilitate the development of chronic pain. 10 Although there is a strong theoretical basis, there is currently limited research evidence to support this hypothesis. The further investigation of lifestyle factors as a treatment option in chronic pain is an area of research that will provide interesting and valuable insights. If new approaches in the management of chronic pain can be identified, there is the potential to provide significant reductions in the economic and social costs of such pain.
Footnotes
Authors’ Note
None of the authors has any professional affiliation, financial agreement, or other involvement with any company that might pose a conflict of interest. No financial disclosures were reported by the authors of this article.
