Abstract
High-intensity interval training (HIT) is characterized by intermittent periods of work and rest and may include work bouts lasting seconds to minutes. HIT has typically been applied to older, diseased, and at-risk populations using longer work intervals (2-4 minutes), whereas more recent definitions of HIT include work intervals of 30 to 60 s. Both traditional endurance training (TET) and HIT exert a peripheral affect increasing the capacity of muscle cells to oxidize substrate via signaling cascades that support the activation of transcription factors that orchestrate the coexpression of nuclear and mitochondrial genes, with HIT triggering these benefits following minutes of training. With 1 exception, reports of central adaptations (eg, increased stroke volume) have been based on longer work intervals (eg, 4 minutes). Recent investigations have tied HIT to increased lipolysis and enhanced insulin sensitivity. HIT favors the activation of oxidative as opposed to hypertrophic pathways. Although the length of the work interval may need to be adjusted to fit the needs and capacity of the participant, HIT should be considered as an alternative to TET for older adults with the expectation that it requires less time to execute, yet promotes peripheral and perhaps central adaptations.
High-intensity interval training (HIT) is a low-volume exercise regime characterized by intermittent periods of work and rest. It is not uncommon for work intervals to approach 80% to 95% of one’s maximum heart rate (MHR), whereas the rest interval is marked by a much lower effort (eg, 40%-50% of MHR).1-4 In contrast to the high-volume lower-intensity exercise typical of traditional endurance training (TET), HIT’s viability as a form of exercise for older adults, and even at-risk older adults, has until more recently received little attention. 5 Not surprisingly, HIT’s high metabolic stress6,7 is viewed as increasing the risk of an adverse event in deconditioned older adults and in particular deconditioned at-risk older adults. Rather, a more conservative approach has been used in which the exercise intensity is lower (50%-70% MHR) and the duration increased over time with the expectation that physical work capacity will increase according to phenotypic adaptations, both centrally and peripherally. 8 By comparison, TET generally requires more time than HIT per session as well as total time over the course of a training period or block. Time commitment is noteworthy given that time is a commonly reported reason why people are not habitual exercisers.5,9 Therefore, apart from an apparent reduction in the time needed for exercise, the benefits of HIT for an older population are not well known.
. . . these data are remarkable considering that HIT [high-intensity interval training] promotes a reorganization of skeletal muscle oxidative pathways following, literally, minutes of total work.
The primary focus of this article is to report the similarities between TET and HIT in terms of selected peripheral adaptations, including muscle cell signaling specializing in oxidative capacity and mitochondrial biogenesis. It is interesting to note that the application of HIT to older adults appears to be limited primarily to at-risk persons and those with disease. As such, research supports HIT as a viable solution for improving physical work capacity as well as desirable metabolic responses even in older adults with disease-imposed limitations.
Peripheral Adaptations and TET
Peripheral adaptations to high-volume, lower-intensity exercise or TET are well known and are consistent across gender, age groups, and level of physical conditioning.10-14 That is, given that TET is a sustained activity, adaptation reflects an increase in the capacity of muscle cells to generate energy via oxidative phosphorylation. 15 This oxidative capacity is directly mediated by an increase in mitochondria with key signaling cascades set in motion during and following TET, which support mitochondrial biogenesis. Specifically, muscle contraction requires the continual and relatively rapid use of ATP with an associated increase in ADP, Pi, AMP, and cytosolic calcium concentration. 16 This disruption in cell homeostasis prompts gene-specific transcriptional activation, a coexpression of nuclear and mitochondrial genomes regulated by the transcription factors nuclear respiratory factors 1 and 2 (NRF1, NRF2), which in turn bind promoter regions triggering transcription of mitochondrial proteins.17,18 Additionally, NRF1 activates its downstream target, mitochondrial transcription factor A, which regulates the transcription of mitochondrial DNA. An additional transcription factor known as peroxisome proliferator–activated receptor gamma coactivator (PGC-1α) regulates the expression of a number of factors (eg, NRF1), including itself, ultimately acting as a master regulator of both nuclear and mitochondrial genomes. 19 PGC-1α promotes mitochondrial biogenesis in response to increased AMP and p38 mitogen-activated protein kinase (p38 MAPK). That is, as AMP rises and energy (ATP) declines during exercise, the enzymes AMP-activated protein kinase (AMPK) and p38 MAPK are triggered with an associated increase in PGC-1α.15,20
Peripheral Adaptations and HIT
Evidence continues to mount that suggests that HIT promotes many of the same peripheral adaptations typically attributed to TET. 5 However, although the idea of using interval training to leverage greater work capacity is not new, 21 not all HIT protocols are the same. For example, Table 1 illustrates the variability inherent in HIT paradigms with protocols using longer work intervals (eg, 4-60 minutes), particularly with persons who have diseases, whereas a more recent definition of HIT uses work intervals of much shorter duration (30 s to 1 minute). 5 It is interesting to note that these 2 approaches to HIT are relevant in perhaps helping explain the reported benefits, including both central and peripheral adaptations. For example, Burgomaster et al 22 used 30-s Wingate tests (4-6 in number, with 4.5 minutes of rest in between) versus 40 to 60 minutes of continuous training 23 at 65% maximum oxygen consumption VO2max to compare the oxidative and metabolic responses to HIT and TET in young untrained individuals. Although there was substantial difference in terms of training duration and intensity, both groups enjoyed significant increases in mitochondrial markers of oxidation as well as PGC-1α nuclear protein content. Similarly, Hood et al 23 had middle-aged women perform 10 bouts of 1-minute cycling at 80% to 95% or heart rate reserve, with a 1-minute rest between bouts, 3 times per week for 6 weeks. After training, oxidative enzymes (ie, citrate synthase and complex IV) and PGC-1α increased significantly as did GLUT 4 and insulin sensitivity. Moreover, Bartlett et al 24 reported that HIT (6 × 3 minutes at 90% VO2max) as compared with continuous running at 70% of VO2max did not differ in terms of the extent to which they induced cell signaling (ie, p38 MAPK, PGC-1α) and gene expression (ie, HSP72 mRNA) posttraining. Thus, consistent with TET,15,25 the disruption of muscle cell homeostasis (ATP: ADP/AMP) during short duration, very-high-intensity HIT activates key upstream signaling (ie, AMPK and p38 MAPK), thereby promoting PGC-1α and mitochondrial biogenesis.26-28 Finally, from a practical standpoint, these data are remarkable considering that HIT promotes a reorganization of skeletal muscle oxidative pathways following, literally, minutes of total work.
Comparison of HIT Protocols/Outcomes: Diseased and Healthy Populations
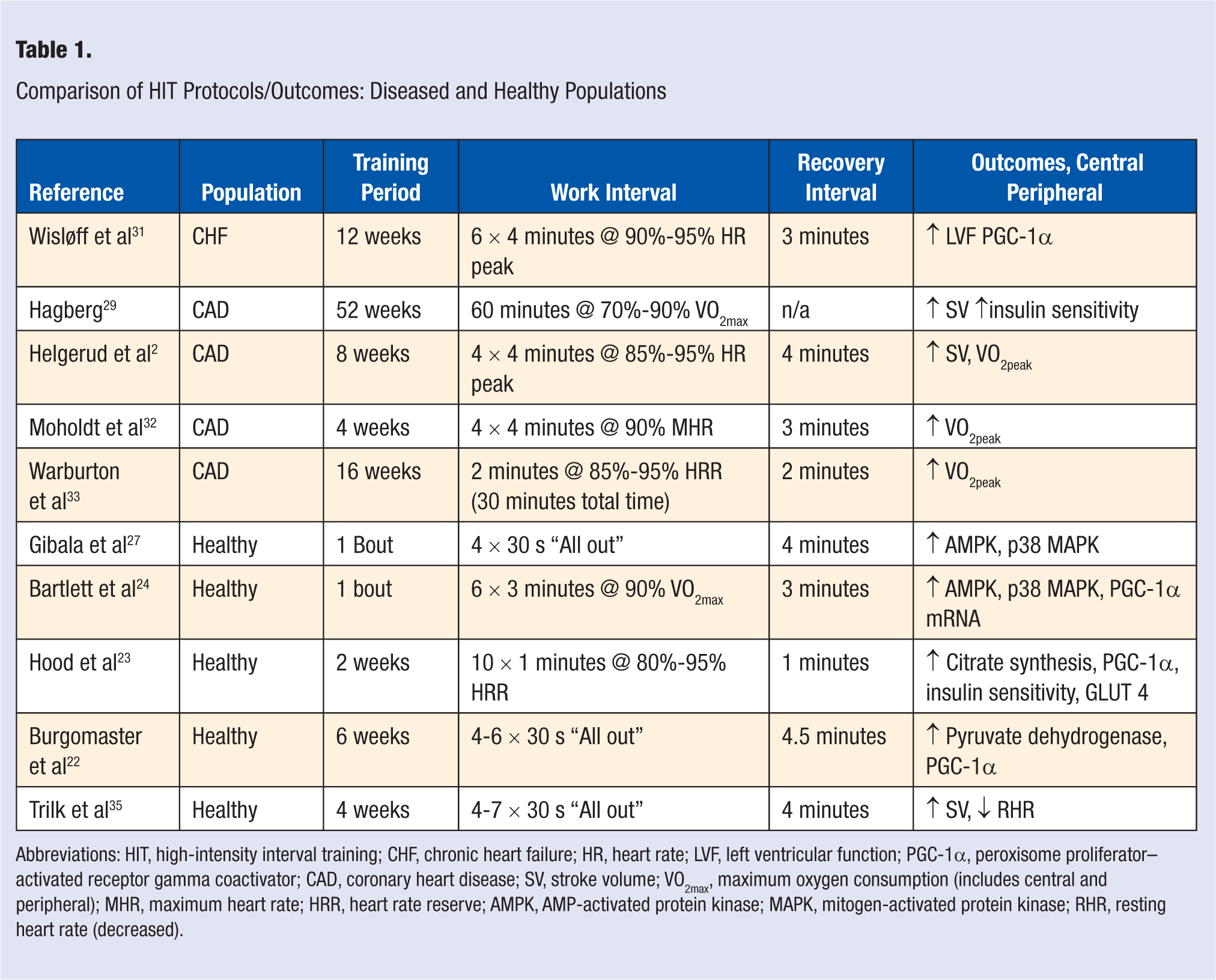
Abbreviations: HIT, high-intensity interval training; CHF, chronic heart failure; HR, heart rate; LVF, left ventricular function; PGC-1α, peroxisome proliferator–activated receptor gamma coactivator; CAD, coronary heart disease; SV, stroke volume; VO2max, maximum oxygen consumption (includes central and peripheral); MHR, maximum heart rate; HRR, heart rate reserve; AMPK, AMP-activated protein kinase; MAPK, mitogen-activated protein kinase; RHR, resting heart rate (decreased).
HIT and At-Risk Older Adults: Determining Training Intensity
Significant improvement in stroke volume29,30 and desirable changes in left-ventricular morphology 31 have been observed in patients with heart disease following HIT. Moreover, when HIT and TET groups were compared, HIT produced greater cardiorespiratory benefits and better endothelial function as assessed via flow-mediated dilation of the brachial artery.31,32 Finding central circulatory adaptations following HIT may be a function of the duration of the work period. That is, the HIT studies reporting central adaptations used longer-duration (at least 4 minutes) work periods,29,31,33,34 whereas only 1 study has reported central changes, including increased stroke following 4 weeks of HIT (4-7 in number, 30-s sprints). 35 Although more studies are needed, the short-term ventricular stress imposed by the 30-s HIT may be too brief to induce ventricular wall remodeling and other circulatory adaptations associated with TET. Additionally, the 1 study that reported central changes included women (ie, 45 years) with no history of heart disease. As such, it may be unrealistic to assume that individuals with disease and deconditioned individuals can sustain the high power output (eg, 90%) required for an “all out” or near all out 30-s work interval.
Although interval training is a commonly used exercise paradigm with low-functioning older adults (see Table 1.), there is no standard by which the exercise training intensity is actually determined. 36 Based on data from middle-aged patients with heart failure and chronic heart disease, Meyer 37 recommends identifying the maximum short-term exercise capacity (MSEC) using a ramp protocol that begins with unloaded cycling for 3 minutes followed by 25-W increases every 10 s until failure. This protocol, although obviously aggressive, did not produce an excessive hemodynamic response (as assessed by rate-pressure product) because of the short duration of the test. Based on MSEC or maximum power output (eg, 150-200 W) achieved during testing, an exercise prescription is established. Typically, the interval training program includes work intervals ranging from 10 to 30 s, with 60 s of active recovery (pedaling at 15 W) across MSEC from 50% to 80%. Specifically, the recommended work phases are 30 s using an exercise intensity of 50% of MSEC, 15 s using an exercise intensity of 70% of MSEC, and work phases of 10 s using an exercise intensity of 80% of MSEC. Surprisingly, training at these high intensities elicited a lower hemodynamic and metabolic stress than TET at 75% maximal aerobic capacity.
More recently, Beale et al 36 evaluated the maximal exercise capacity of chronic heart failure patients by having them complete 2 incremental cycle tests, one in which the load was increased by 10 W/min and a second test in which load was increased by 25 W/min. Interestingly, there was no significant difference between conditions for maximum watts achieved or VO2max. Collectively, these investigations lend some support for the use of either a moderate or aggressive cycle ramp protocol to identify maximum work capacity in hopes of establishing a HIT exercise intensity. Still, given the scarcity of data on the subject, the question of exactly how much intensity to safely elicit via interval training in an at-risk population remains largely unanswered.
Other Benefits of HIT Training
Other reported HIT-related benefits include subcutaneous fat loss30,33,34,38 as well as improved insulin sensitivity.34,35 For example, there is an expectation that an acute bout of TET will result in elevated catecholamine and subsequent adrenoceptor-mediated lipolysis. 39 This same expectation is less apparent for HIT given that the extremely brief work bout favors glycolysis. 15 However, very brief work and rest intervals (8 by 12 s and 12 by 24 s) performed on the cycle ergometer produced significantly elevated catecholamine levels 20 minutes postexercise. 40 Bracken and Brooks 41 found similar results in young men who performed 6-s sprints on the cycle ergometer. That is, compared with baseline, there was more than a 6-fold increase in epinephrine and a 14.5-fold increase in norepinephrine. Because catecholamines, particularly epinephrine, drive lipolysis, these findings are noteworthy, in that HIT may contribute to lipolysis.
Regarding insulin sensitivity, it may be that HIT promotes an increased expression of GLUT 4 as is seen in TET.15,42,43 Several studies now confirm increased skeletal muscle GLUT 4 content following HIT of the short work bout type.5,26,34 Thus, insulin sensitivity as a product of GLUT 4–activated glucose transport via HIT may offer an additional exercise strategy to combat a growing epidemic of type II diabetes (see Gibala et al 5 for more details).
Given the brief, intense nature of HIT, it is tempting to speculate that anabolic pathways typically associated with muscle hypertrophy are activated. But contradicting this notion is the work by Gibala et al, 5 in which he reports a decrease in akt as well as downstream targets (P70-S6 kinase) of muscle hypertrophy. Gibala’s findings may be explained on the basis of calcium signaling, where calcium flux corresponds to the activation of specific pathways. Recent investigations suggest that calcium release and reuptake can be characterized by the type and intensity of the muscular contraction.44,45 That is, sustained, high-metabolic-demand activities such as running and cycling are underwritten by more moderate cytosolic calcium fluctuations, which support cell signaling (eg, AMPK and PGC-1α) associated with an increased oxidative capacity. 5 Conversely, the high load and high mechanostress inherent in resistance exercise triggers greater and more substantial calcium fluctuations, which tend to activate hypertrophic (akt/mTOR) pathways. 5
Conclusions
Given the available data, HIT offers considerable benefits, particularly in light of the time savings. For example, HIT induces peripheral adaptations consistent with changes seen in TET, including the disruption of cell homeostasis, prompting gene-specific transcriptional activation, a coexpression of nuclear and mitochondrial genomes via a cell signaling cascade, resulting in mitochondrial biogenesis and increased oxidative capacity. These changes in peripheral architecture have been reported with HIT work intervals ranging from 30 s to more than 4 minutes. Additionally, HIT has been successfully used with older adults with cardiovascular disease and, when compared with TET, produces as good or better cardiorespiratory benefits. Moreover, HIT offers additional benefits, including increased lipolysis and enhanced insulin sensitivity. Finally, one study of young healthy individuals reports central changes (improved stroke volume and reduced RHR) following 4 weeks of 30-s “all out” work bouts. Whereas more study is required to validate the central influence of the very brief HIT protocol, it is hard to discount the efficacy of HIT, both in terms of inducing a host of desirable physiological phenotypes as well as from an efficiency standpoint. It is important to note that although work bouts vary, even older adults with limited capacity appear to tolerate HIT. The actual intensity prescribed during HIT may be identified from the results of a moderate to aggressive cycle ramp protocol. It is important to point out that “all out” HIT of less than 1 minute may be unrealistic for older deconditioned persons and those suffering from diseases. The effort required for the very brief work bouts may be beyond both the muscular and cardiovascular capacity of this population. Finally, considering the peripheral and perhaps central adaptation to HIT, older individuals should consider HIT as a viable alternative to TET.
