Abstract
High-risk human papillomavirus (HPV) infection and viral persistence is a major risk factor in the development of squamous intraepithelial lesions and invasive carcinoma of the cervix. In the United States, deaths due to squamous cell carcinoma of the cervix have fallen by 75% since the 1960s because of Papanicolaou (Pap) smear screening. However, the traditional Pap had a sensitivity of about 70% for detecting clinically significant precancerous lesions and cancer because of sampling and interpretive errors. The introduction of 2 liquid-based Pap smear collection systems in the 1990s, the use of HPV testing as a triage and co-testing with Pap smear, and the introduction of 2 automated screening devices have had a significant impact on improving the detection of such precancerous lesions. This review provides an analysis of the changes in Pap smear collection, improvements in screening, the evolutionary changes of high-risk HPV testing, reporting terminology of Pap smears, and clinical management guidelines. The future impact of 2 prophylactic HPV vaccines on the incidence of cervical carcinoma is also discussed. This article also discusses alternatives such as primary screening for high-risk HPV testing with visual inspection for cervical cancer detection used in resource-poor settings with a high incidence of cervical cancer.
Cervical cancers are caused by HPV infection, viral persistence, progression of a clone of persistently infected cells to precancer (cervical intraepithelial neoplasia or CIN 3), and finally invasion.
Epidemiology of Human Papillomavirus
Infections with 15 types of oncogenic anogenital human papillomavirus (HPV) cause almost all the precancerous intraepithelial lesions and cancers in the cervix.1,2 Among all HPV types, HPV 16 is responsible for causing 56% of all cervical squamous cancers, whereas HPV 18 causes 10% to 15% and HPV 45 approximately 7%.2-4 HPV 18 also causes 35% of cervical adenocarcinomas. 4 The incidence of different types of HPV is more or less common in the various parts of the world. Understanding the epithelial biology of HPV has greatly enhanced our current understanding of cervical carcinogenesis.5,6 Cervical cancers are caused by HPV infection, viral persistence, progression of a clone of persistently infected cells to precancer (cervical intraepithelial neoplasia or CIN 3), and finally invasion 6 (Figure 1). HPV infection is a common sexually transmitted infection with an overall incidence of 26.8% in a representative sample of females in the United States, whereas lifetime incidence of HPV has been reported to be as high as 80%. The highest prevalence of 44.8% is seen in women aged 20 to 24 years, 7 and it declines after age 30 years, perhaps because of the high rate of viral clearance in young women and possibly because of HPV-type-specific acquired immunity. 1 The overall prevalence reported for high-risk HPV was 1.5% and 0.8% in types 16 and 18, respectively. 7 Concurrent infection with multiple types of HPV is a very common phenomenon, since all types of HPV share the same transmission route. Most of the cervical HPV infections with or without cytologic abnormality tend to be cleared through cell-mediated immunity within 1 to 2 years after exposure. Viral persistence lasting more than 1 to 2 years is a major risk factor for progression to neoplasia.1-8 It is still debated whether the infections disappear by complete clearance of viral particles or whether the virus is maintained in a latent state in basal cells of the nonkeratinizing squamous epithelium of the cervix at extremely low levels resulting in a failure of full viral expression. 6 The theory that the virus is maintained in a latent state even when women have no visible signs of HPV infection might explain the reemergence of HPV infection in renal transplant patients and HIV-positive women. 1 Among the oncogenic types, HPV 16 generally tends to persist longer than others.1,6 Smoking, multiparity, and long-term oral contraceptive use can double or triple the risk of viral persistence and progression to precancer and cancer in women with oncogenic types of infection.1,6 However, the role, if any, of other sexually transmitted diseases, nutritional factors, and chronic inflammation is less clear. 1
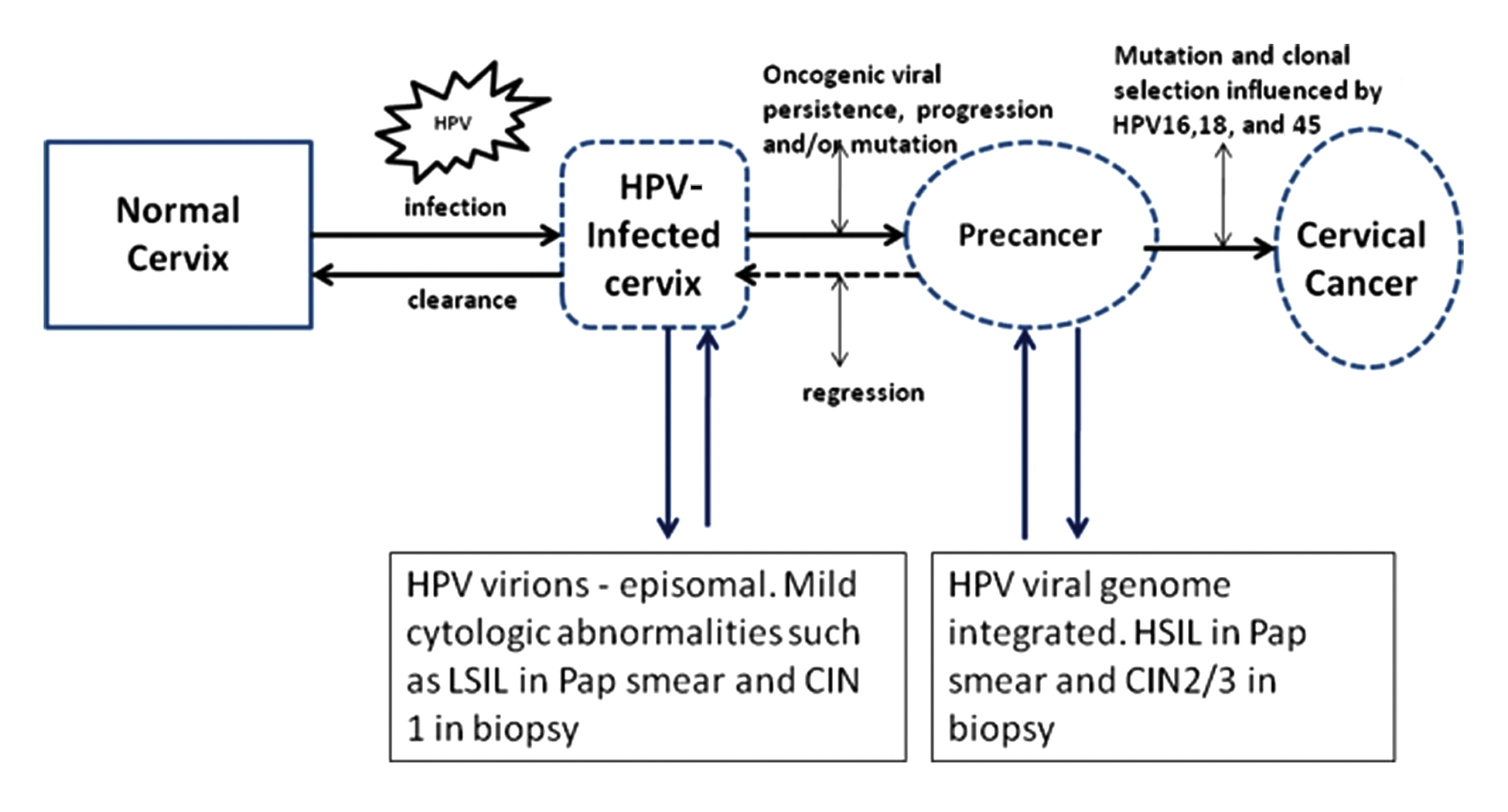
A schematic model of HPV-induced cervical carcinogenesis.
HPV types 6 and 11 generally cause genital warts (condyloma acuminata) and are very rarely encountered in high-grade squamous intraepithelial lesions (CIN 3) or invasive squamous cancers. HPV 16 followed by HPV 18 are the most prevalent types to infect the uterine cervix as well as to persist and are responsible for the majority of intraepithelial and invasive squamous neoplasias. HPV 18 is more frequently associated with adenocarcinomas and small cell neuroendocrine cancers of the cervix. 5 Khan et al 4 reported that HPV screening for types 16 and 18 may identify those women at increased risk of developing significant cervical intraepithelial lesion (CIN 3 or cancer) and might facilitate less aggressive management of other women with other types of oncogenic HPV infections. Estimates of prevalence of the other oncogenic HPV types (eg, types 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, and 69) cannot be estimated accurately because HPV detection methods categorize high risk or low risk rather than individual types. 5 The total risk for developing CIN 3 of the cervix for women is increased when they carry multiple oncogenic HPV types compared with women with any 1 of the HPV types alone. Currently, it is not clearly understood whether a woman’s risk is greater than the sum of the risks of individual types of HPV. 6 Viral load measurements are not good outcome predictors for development of CIN 3 or cancer except in the case of HPV 16. 3
There appears to be a strong correlation not only with specific oncogenic HPV types but also between the physical state of HPV DNA within the host cells and the potential for development of cancer of the cervix. In women with HPV infections with low oncogenic risk, the viral DNA exist in the host cell as extrachromosomal plasmids, monomeric circular molecules, whereas in cancers, HPV DNA is seen as multimeric circular molecules with detectable deletions and most commonly integrated into the host chromosomes. All HPV genomes are collinear and have open reading frames encoded on 1 DNA strand. The open reading frames are divided into 2 regions, early (E) and late (L); within the E region there is a third upstream regulatory region (Figure 2). Proteins encoded by E6, E7, and E5 are responsible for the transformation where E1 codes for 2 or more proteins needed for regulation of episomal DNA replication and E2 codes for 2 or more proteins responsible for regulation of viral transcription. L1 and L2 encode for major and minor capsid proteins. 5 E4 protein is thought to modulate structural changes in the cytoplasm of the infected cells. The HPV genome codes for 8 genes (Figure 2) where E6 and E7 are the primary HPV oncoproteins each with multiple cellular targets with p53 and retinoblastoma tumor suppression protein (pRB) being the most significant. E6 inhibits p53 thus blocking apoptosis, whereas E7 inhibits pRB abrogating cell cycle arrest. 6 E7 acts as a primary transforming protein. Both E6 and E7 proteins are expressed at low levels during the process of HPV infection, and at some point after viral integration into the host, E6 and E7 expression is deregulated because of genetic or epigenetic changes resulting in overexpression and development of CIN 3 and cancer.5,6
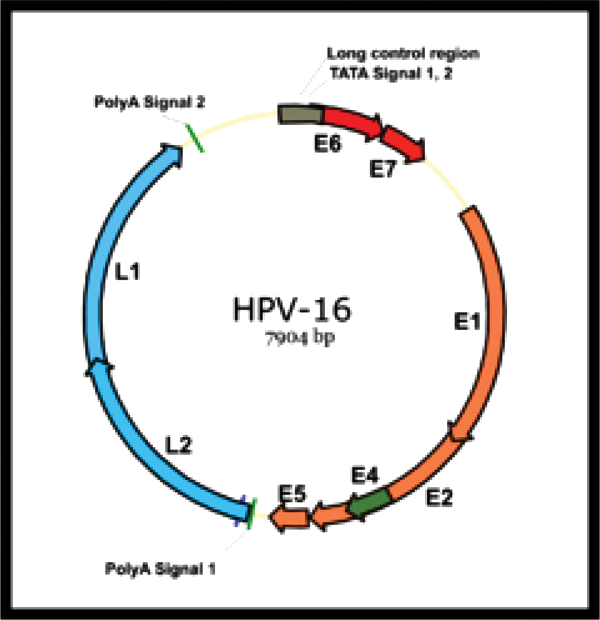
Human papillomavirus 16 genome.
Human Papillomavirus and a Model for Cervical Carcinogenesis
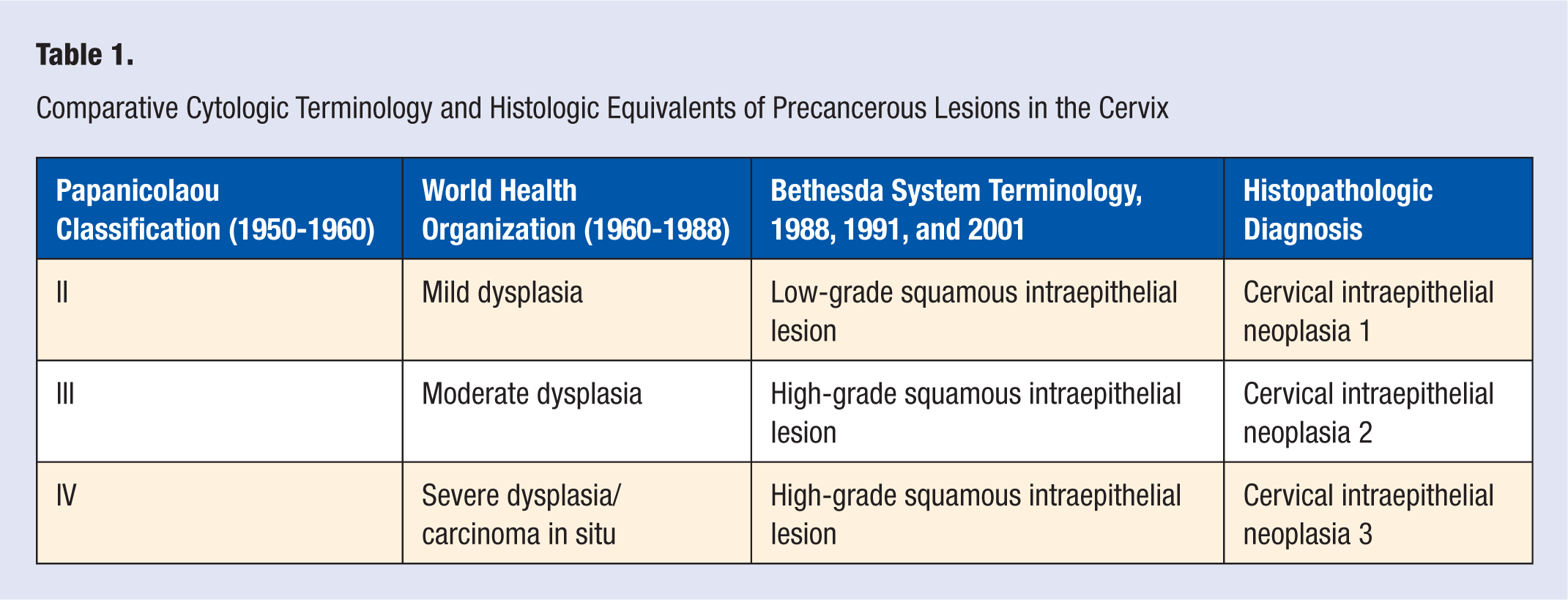
Cervical transformation zone is a ring of mucosa highly susceptible to HPV infections and persistent HPV infections, especially types 16 and 18 capable of causing squamous cell carcinoma and also adenocarcinoma (mostly due to HPV 18) of the uterine cervix. Histologically, HPV infections infect the basal, reserve, or stem cells of the transformation zone as a result of microscopic tears during intercourse. These basal-type cells have the potential to differentiate into squamous, glandular, or neuroendocrine cells that maintain epithelial integrity. 5 In the basal cells committed to squamous differentiation, a highly regulated process of maturation and differentiation occurs throughout the thickness of the epithelium at a morphologic as well as molecular level where cell division is restricted to cells in the basal and parabasal layers. In patients with HPV infection detected with molecular assays such as polymerase chain reaction, Hybrid Capture II, or Invader Technology using Cervista but with no morphologic abnormality in the Pap smear, HPV replication and gene expression are inhibited to essentially maintenance levels. 5 Viral replication and gene expression with HPV continue in cells that have started squamous maturation, and in the parabasal zone, expression of the early regions of the virus occur, leading to viral DNA synthesis and the formation of increased number of virions occurring episomally. This results in altered maturation and differentiation where the mature cells at the surface of the epithelium show nuclear enlargement and hyperchromasia due to E6/E7-mediated host DNA synthesis. This type of abnormality is referred to as mild dysplasia or low-grade squamous intraepithelial lesion (LSIL) in the current Bethesda System 2001 or cervical intraepithelial neoplasia 1 (CIN 1 in histology; see Table 1 for terminology comparison). The life cycle of HPV in nonkeratinizing squamous epithelium of the cervix is illustrated in Figure 3. Currently, the CIN terminology is used in histopathologic diagnoses, whereas the Bethesda System 2001 terminology of LSIL and high-grade squamous intraepithelial lesion (HSIL) is used for reporting squamous intraepithelial lesions in cytology. Some of these HPV-infected cells might show koilocytosis with characteristic perinuclear halo and nuclear atypia (Figure 4). It is believed that the correct amount and type of HPV E4 expression results in cytoskeletal changes in the cytoplasm and causes the koilocytosis. 1 LSILs regress in many patients, and in the scheme of cervical carcinogenesis LSILs or CIN 1 do not predict a higher risk for development of severe dysplasia/carcinoma in situ, HSIL, 6 or CIN 3 in histology. If the process of LSIL is not fully developed, the cells at the surface of the epithelium often show lesser nuclear abnormality and result in atypical squamous cells of uncertain significance (ASC-US). 5
Comparative Cytologic Terminology and Histologic Equivalents of Precancerous Lesions in the Cervix
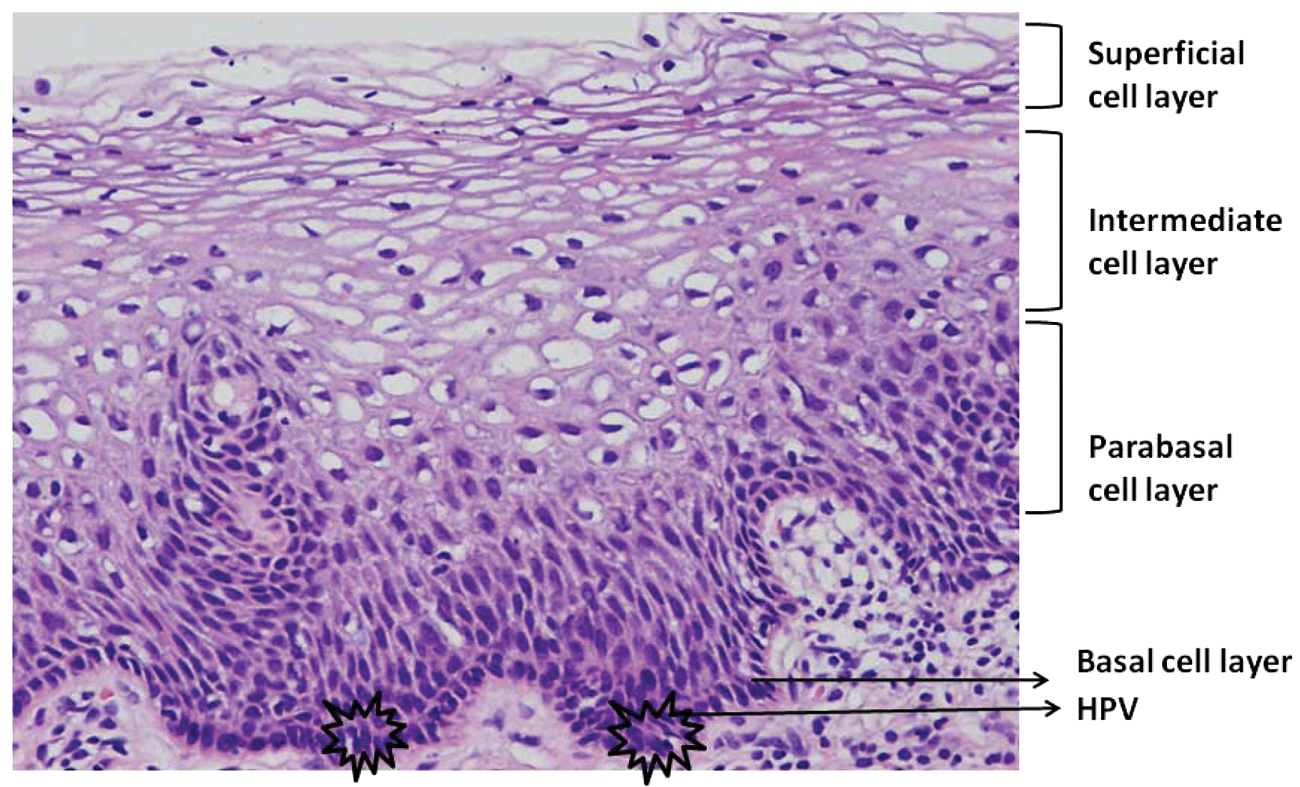
Human papillomavirus life cycle in the nonkeratinizing squamous epithelium of the cervix.
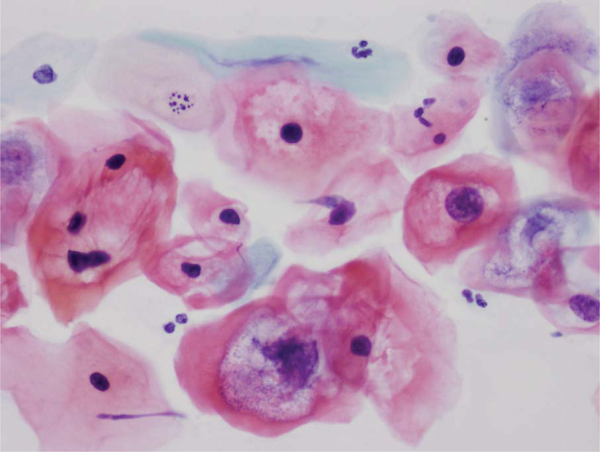
Cervical smear showing low-grade squamous intraepithelial lesion with perinuclear cavitation or koilocytosis and enlarged, hyperchromatic nuclei (papanicolaou stain 400×). The corresponding biopsy will show cervical intraepithelial neoplasia 1.
In the development of HSIL, a critical event of viral integration or mutations resulting in loss of E6/E7 regulation causes uncontrolled cell proliferation of basal-type cells with mitotic activity up to two-third thickness of the nonkeratinizing squamous epithelium with minimal differentiation at the surface (HSIL or moderate dysplasia in cytology and CIN 2 in histology) or the entire thickness of the epithelium showing no differentiation (HSIL, severe dysplasia/carcinoma in situ, and CIN 3 in histology). Promoters that play a role in this step include smoking, infections with other viruses, inflammation, or other random mutation. 5 In this process of cervical carcinogenesis, the term precancer refers to a morphologic diagnosis of HSIL or CIN 3 or severe dysplasia/ carcinoma in situ (Figure 5) where the undifferentiated basal-type cells with fixed genetic abnormalities replace almost the entire thickness of squamous epithelium of the cervix. 6 But from the biological standpoint, a vital distinction happens between LSIL or CIN 1 and HSIL or CIN 2, with a switch to a proliferative as opposed to a differentiated and virally productive phenotype, and this is consistent with the Bethesda System lumping CIN 2 and CIN 3 together as HSIL for clinical management purposes. 5
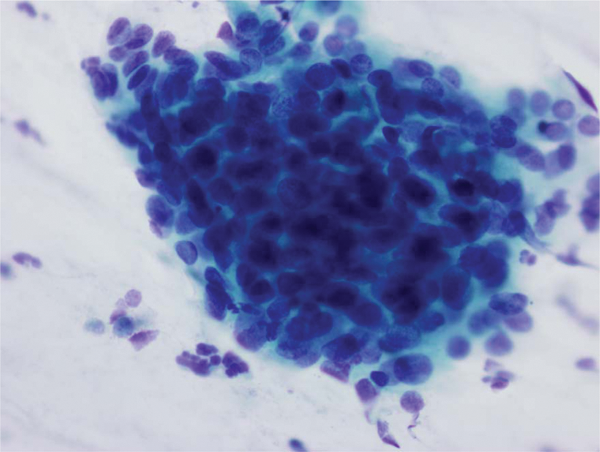
Cervical smear showing precancerous high-grade squamous intraepithelial lesion in which the corresponding biopsy will show cervical intraepithelial neoplasia 3. Note the syncitial aggregate of cells with hyperchromatic nuclei and scanty cytoplasm (Papanicolaou stain 400×).
HSIL results in mitotically proliferating basal-type cells that have a greater potential for acquiring additional mutations. Clonal selection influenced by high-risk HPV infections, predominantly of types 16, 18, and 45, results in invasive squamous carcinoma (Figure 6) in a genetically susceptible host. The different subtypes of invasive squamous carcinoma of the cervix, that is, keratinizing type on the ectocervix, nonkeratinizing around the transformation zone, and small cell neuroendocrine carcinoma high in the endocervical canal, mostly reflect different genetic pathways modulated by the specific environment in which the cancer develops. 5 HPV 18 exhibits a greater predilection of viral integration into the host genome and may show affinity to cells not differentiating toward squamous epithelium4-7 and has been associated with the development of endocervical adenocarcinoma and small cell neuroendocrine carcinoma of the cervix. For productive viral replication, an environment of squamous differentiation is critical. Hence, it is theorized that an abortive or latent infection of morphologically normal endocervical cells leading to dysregulation of viral gene expression resulting in a hyperproliferation of glandular cells is recognized as severe endocervical dysplasia/adenocarcinoma in situ and has a potential to lead to invasive adenocarcinoma. 5 A similar argument can be put forth for the development of small cell neuroendocrine carcinomas of the cervix. In addition to cervical cancer, malignancies such as esophageal, oropharyngeal, anal, penile, vulvar, and vaginal have been causally associated with various subtypes of HPV infection. 9
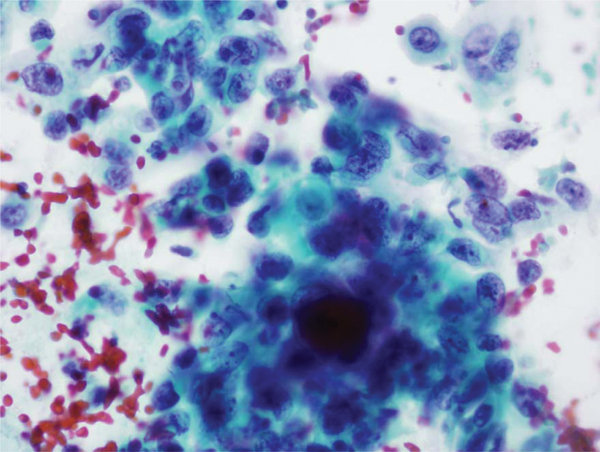
Cervical smear showing nonkeratinizing squamous carcinoma with malignant cells singly and in syncitial aggregates with hyperchromatic nuclei and prominent nucleoli. Note the evidence of bleeding and tumor diathesis in the background (Papanicolaou stain 400×).
Role of Pap Smear in Cervical Cancer Screening
Cervical cancer with an annual global prevalence of 2.3 million cases is second only to breast cancer as the leading cause of cancer in women and the third most common cause of cancer-related mortality worldwide in women. 10 In the United States, cervical cancer rates have fallen by 75% or more since the 1960s because of the success of the cytology-based cervical cancer screening program using the Pap smear, 11 whereas 82% of the new cervical cancer cases occur in developing countries that do not have screening programs. 10 George Papanicolaou, MD (Dr Pap), originally presented his screening smear technique at a 1928 conference in Battl Creek, Michigan, in his paper titled “New Cancer Diagnosis,” 12 and a New York newspaper article covering the conference reported Dr Pap’s intention of his technique as follows:
Although Dr. Papanicolaou is not willing to predict how useful the new diagnostic method will be in the actual treatment of malignancy itself, it seems probable that it will prove valuable in determining cancer in the early stages of its growth when it can be fought and treated. There is even hope that pre- cancerous conditions can be detected and checked.
13
Dr Pap’s article unfortunately did not elicit any interest in the medical community until he, along with a gynecologist coinvestigator, Dr Herbert Traut, MD, published findings regarding the Pap smear in 1941. 14 The American Cancer Society widely promoted this screening technique, which led to the establishment of cervical cancer screening programs in many places in the United States and in Europe. The initial reports establishing the validity of the Pap smear in reducing morbidity and mortality due to cervical cancer were published in 1955 by Eriksson and colleagues in Tennessee 15 and by Christopherson et al 16 in Kentucky. Christopherson et al16,17 reported their data on the significant reduction in morbidity and mortality collected over a period of 21 years since the mid-1950s when the cervical cancer screening programs were established in Kentucky. Soon Pap smear became a lexicon of woman’s health and enjoyed remarkable success in the United States, and Europe, as well as in a few developing countries with a successful screening program. The screening program’s success is based on repeated screening of women within short intervals during the long process of carcinogenesis, from incident HPV infection to development of HSIL or CIN 3 (typically 2-15 years) and from HSIL or CIN 3 to cancer (typically 10 or more years). 11 The method used by Dr Pap of scraping cells from the cervix and smearing onto a glass slide with patient identifiers and fixing it in alcohol is referred to as the conventional Pap smear. The conventional Pap smear was used as is for more than 45 years. A Pap smear is intended as a screening program, and from a clinical performance perspective, a conventional Pap smear has a sensitivity of about 70% for detection of clinically significant HSIL or carcinoma 18 and warrants repeats over short intervals to improve efficacy. Second, there was poor reproducibility of results even among experts as illustrated in the ASCUS LSIL Triage Study (ALTS) study conducted by the National Cancer Institute. 19 Third, cytology is a labor-intensive test, and human errors in interpretation do occur. But in the eyes of the public, even a single Pap smear should be effective and almost equivalent of a cervical cancer insurance policy capable of paying a substantial monetary benefit if cervical cancer were to develop. 12
In the United States, the cervical screening landscape changed dramatically after the November 2, 1987, publication by Walt Bogdanich in the Wall Street Journal titled “Lax Laboratories: The Pap Test Misses Much Cervical Cancer Through Labs’ Errors.” 20 This article magnified the public scrutiny on Pap tests and cervical cancer screening litigation increased, and the Centers for Medicare and Medicaid Services instituted major changes under the Clinical Laboratory Improvement Amendments of 1988 (CLIA’88) for all types of clinical laboratories including cytology. 21 Public’s expectation of an impossible 100% screening efficacy for Pap smear and poorly understood rules of CLIA’88 led to a marked increase in Pap smear–related litigation. But a positive outcome of all these led to improvement of the sensitivity of Pap smear in the areas of preparation, and screening, as well as reporting of results.
Dawn of Liquid-Based Pap Tests
To minimize inaccurate and equivocal Pap smear diagnoses related to sampling errors and suboptimal specimen preparations, a new method of collecting all the cells from the Pap smear collection devices in a liquid preservative came into practice with the FDA approval of ThinPrep (Hologic Corporation, Boxborough, Massachusetts) in 1996. In the ThinPrep system, the collection device with the cervical specimen is placed into a vial containing 20 mL of liquid preservative (PreservCyt), instead of being smeared on a glass slide and transported to the cytology laboratory, and is prepared using the company’s proprietary equipment and process to produce a 20-mm monolayer of cells for optimal visualization. This specialized process removes excess blood, inflammatory cells, and any other debris that might obscure cellular details, and the smears are stained with Pap stain for microscopic review. The collection method in liquid preservative also facilitated testing for sexually transmitted diseases caused by Chlamydia, gonorrhea, and also HPV from the same vial without requiring another clinical specimen. Since FDA approval, many studies have been conducted comparing conventional and ThinPrep Pap smears and have reported that ThinPrep reduces the number of unsatisfactory smears and enhances the sensitivity for diagnosing all abnormalities, especially the clinically significant HSIL.22-24 Two major population-based studies in Costa Rica and China compared the sensitivity of conventional Pap smear with ThinPrep with biopsy correlation (gold standard) for diagnostic accuracy.25,26 Belinson’s Cleveland Clinic group 26 study from Shanxi Province documented a sensitivity for detecting biopsy-proven HSIL for ThinPrep of 94% for CIN 2 and 98% for CIN 3. In 2000, a second BDSurePath liquid-based Pap test was approved by the FDA. BD SurePath liquid-based Pap test uses a similar liquid-based collection method, a proprietary process to remove excess inflammatory cells, blood, and so on using BD PrepStain slide processor. The prepared slides are then stained in an automatic stainer to produce a cell-enriched 13-mm monolayer cell preparation. Bishop et al 27 reported their comparative analysis between BD SurePath liquid-based Pap test (formerly referred to as AutoCyte Prep) and conventional Pap smear and described the significant reduction of unsatisfactory and satisfactory but limited due to lack of transformation zone smears and also a 64.4% increase in HSIL detection when compared with conventional smear. A comparative account of conventional, ThinPrep, and BD Surepath is provided in Table 2.
Comparative Analysis of Pap Smear Methods
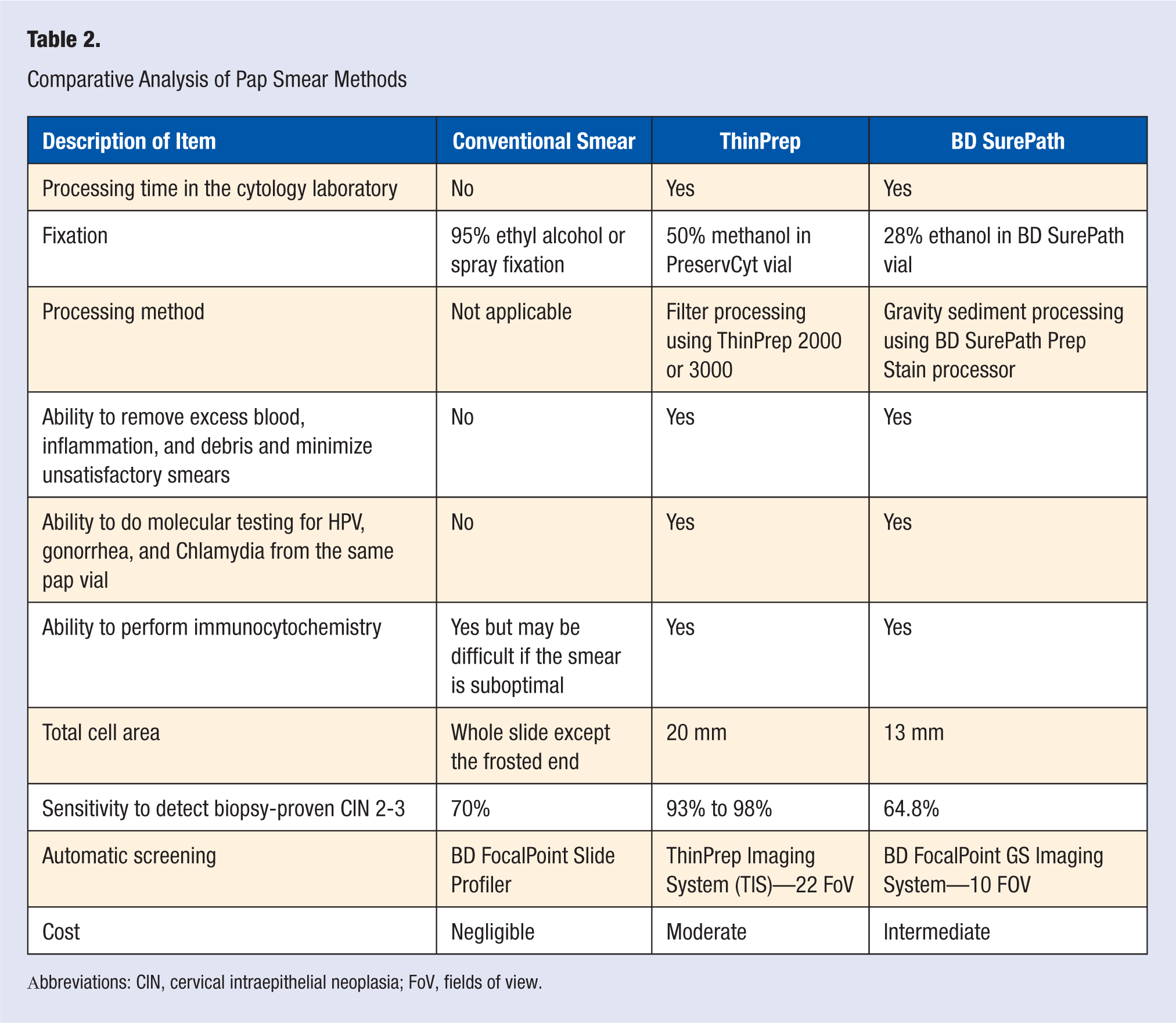
Abbreviations: CIN, cervical intraepithelial neoplasia; FoV, fields of view.
Automated Screening in Cytology
In 2003, Hologic Corp introduced the ThinPrep Imaging System (TIS) with Dual Review into diagnostic cytopathology in an attempt to further enhance the sensitivity of ThinPrep Pap smears as well as the productivity of laboratories. The TIS Dual Review scans the ThinPrep Pap smears using special computer algorithms, scans the entire slide, and preselects 22 fields containing the most significant cells in each slide to be reviewed by a cytotechnologist using a review scope. If the cytotechnologist, on careful review of the 22 fields of view (FOV), does not find any cytologic abnormality, the smear can be reported as negative for intraepithelial lesion or malignancy. If abnormal cells are detected within the 22 FOV, the cytotechnologist can revert to a manual screening for careful evaluation of the smear, mark the abnormal cells, and report the findings appropriately. Dziura et al 28 screened 27 525 smears, both manually and with TIS, and found that TIS performed significantly better than manual screening in the detection of biopsy-confirmed HSIL. Dawson 29 described the improvement of cytotechnologist productivity with TIS and its potential to address the screening/ interpretive portion of the false-negative fraction, thus improving patient care outcomes.
The BD FocalPoint Slide Profiler (AutoPap Primary Screening System) has been in use for primary screening of conventional Pap smears and recently for BD SurePath liquid-based Pap smears. Using proprietary computer algorithms, this screening system is capable of scanning the entire slide, assessing the cellularity, and providing a quintile rank for each slide corresponding to the slide’s likelihood of containing abnormality as a number from 1 to 5 where quintile 1 is most significant for containing abnormal cells to assist the cytotechnologist for careful further review. Recently, the FDA has approved the BD FocalPoint GS Imaging System, which includes the BD FocalPoint Slide Profiler and BD FocalPoint GS Review Station. The BD FocalPoint GS Imaging System assists in the cervical screening of BD SurePath liquid-based Pap smears to detect squamous carcinoma, adenocarcinoma, and its precursor conditions. The slides are initially scanned and rank ordered in quintile according to the likelihood of abnormality and provide relocation and visual review of 10 fields of view most likely to contain the abnormal cells to the cytotechnologist for diagnosis using the FocalPoint GS Review Station. This also identifies 15% of all successfully processed slides using the BD FocalPoint Slide Profiler Directed QC Technology for a directedquality control rescreen. Kardos 30 presented his findings on the effectiveness of BD FocalPoint Slide Profiler and BD FocalPoint GS Review Station during the Annual Meeting of the American Society of Cytopathology in 2003.
Bethesda System (TBS) for Reporting Cervical/Vaginal Cytological Diagnoses
In December 1988, 31 the National Cancer Institute sponsored a workshop in an attempt to develop a standardized lexicon for cervical/vaginal cytopathology reports specifically emphasizing provision of clear guidance for clinical management. The main intent of the first Bethesda workshop in 1988 was to reduce the clinical confusion caused by the use of multiple classification systems from the original Papanicolaou’s terminology and the World Health Organization’s terminology using mild, moderate, and severe dysplasia and carcinoma in situ for precancerous lesions (Table 1). A second Bethesda conference was held in 1991 32 to modify the terminology of Pap smear reporting based on the laboratory and clinical experiences using TBS 1988 reporting for Pap test results. Extensive research regarding the TBS 1991 terminology as well as the new and expanded role of adjunct technologies, and a much greater understanding of HPV biology in cervical carcinogenesis, paved the way for a third Bethesda conference in 2001. One major milestone was understanding the role of high-risk HPV testing as a triage tool for about 2 million women in the United States receiving the diagnosis of atypical squamous cells of undetermined significance (ASCUS) each year. A diagnosis of ASCUS renders increased risk for those patients for prevalent CIN 2, CIN 3, and cancer while colposcopic evaluations reveal that >70% are CIN 1, a sign of benign HPV infection that will most likely be cleared. 33 Therefore, satisfactory colposcopy is necessary for all these women to identify a small minority who might have clinically significant disease and warrant close follow-up. This causes excessive burden on colposcopy clinics and also a major financial drain in these times of escalating medical cost.
Dawn of HPV Testing as a Triage Tool to Cytology
Sponsored by the National Cancer Institute, a multicenter randomized clinical trial of the management of women with low-grade and equivocal cytology, called the ASCUS/LSIL Triage Study (ALTS), was undertaken from November 1996 through December 1998. 33 The goal of this study was to compare the sensitivity and specificity of immediate colposcopy, repeat cytology, and HPV testing for a timely detection of histologically confirmed CIN 3 with a high risk of progression to invasive cancer and therefore warranting immediate treatment. A total of 3488 women with a diagnosis of ASCUS and 1572 women with a diagnosis of LSIL from 4 major medical centers in the United States were enrolled in the study and were randomly assigned to 1 of 3 management arms: (a) immediate colposcopy following cytologic diagnosis of ASCUS or LSIL, (b) conservative management with follow-up cytology and colposcopy only if follow-up cytology is HSIL or above, or (c) triage to colposcopy based on high-risk HPV-positive results from Hybrid Capture 2 (HC2) and ThinPrep cytology results. The HPV triage arm of women with a cytologic diagnosis of LSIL was suspended early since the interim analysis revealed a substantial majority of women (83%) would be triaged to colposcopy-based HC2 results while it is understood that majority of the LSILs might regress spontaneously. The immediate colposcopy arm was a priori assumed to provide a standard for detecting HSIL (CIN 3 in biopsy) to which the HPV testing and follow-up with cytology arms would be compared. The major conclusion of ALTS was that HPV testing using HC2 for high-risk types had a greater sensitivity to detect CIN 3 or above and specificity comparable with a single additional cytologic testing with ASCUS or above and therefore provides a viable option for management of women with ASCUS. 33 Patients with atypical squamous cells, where HSIL (ASC-H) cannot be ruled out, were directly referred to colposcopy since these lesions have a significant predictive value for HSIL and oncogenic HPV DNA is detected in up to 70% to 85% of the cases. 19
With an improved understanding of the process of cervical neoplasia, the role of high-risk HPV testing, and changing needs of clinicians and cytopathologists, there was a need for a third Bethesda conference 34 in 2001 to further modify the TBS 1991 system. The 2001 TBS developed many forums on each with members from all communities of interest, including cytotechnologists, cytopathologists, clinicians, and patient advocates. One of the forum groups addressed the use of ancillary tests, most common among them being HPV testing. 35 This forum used HPV testing as a model for HPV tests, which can also serve as a model for other ancillary tests that might arise in the future. This forum recommended that the specific HPV test used be clearly mentioned and the results reported for HPV as positive or negative for a certain type or class (eg, high-risk HPV). The Pap test and the HPV test results should refer to each other and preferably be reported together. Some of the reporting schemes include probabilistic reporting, integrated reporting, reporting as a result, and reporting with clinical management recommendations. 35 Although there are many molecular methods, including in situ hybridization, Southern blot hybridization, ligase chain reaction, branch signal DNA, and polymerase chain reaction, the 2 FDA-approved and commonly used HPV tests are Hybrid Capture II or HC2 and Cervista HPV HR to be used with ThinPrep. Validation can be done in the laboratory for BDSurePath-type Pap smear. HC2 technology is based on the principle of signal amplification and detected by chemiluminescence and the results expressed as RLU (relative light units), but it cannot determine whether sufficient human DNA was present for authentication of HPV testing results. Cervista HPV HR uses patented Invader chemistry using signal amplification and fluorescence detection. It is unique in that it contains an internal control to validate that sufficient DNA is present for testing and can detect 14 oncogenic strains of HPV with low risk of cross-reactivity. It is also the first test that can perform specific genotyping for HPV 16 and HPV 18 associated with more than 70% of all cervical cancers and recommended as a follow-up tool in risk stratification for development of invasive carcinoma.3,4,36,37 A comparative study of these 2 techniques by Johnson et al 38 found that generally the results from both assays were comparable when comparing the cytologic evaluation but the Invader-based test demonstrated a clinical sensitivity of 98% in ASC-US cases for CIN 2 or worse and 100% for CIN 3 or worse and a negative predictive value of 96.9%. The authors felt that since Digene HC2 has a false-positive rate from 10% to 19% due to cross-reactivity with low-risk HPV DNA, the Invader method is more advantageous for the laboratory.
Bolick 39 described ways for successful implementation of HPV testing in the laboratory, and Seabrook and Hubbard 40 described how to successfully add HPV testing that achieves high-quality reproducible results and at the same time is compliant with CLIA’88 requirements in molecular testing of HPV. Zuna et al 41 reported how HPV testing done from the residual volume of liquid-based Pap tests can be used clinically for patient follow-up and that it can also function as a quality assurance monitor for the cytology laboratory.
Changes in Clinical Management Guidelines and the Use of HPV Co-testing With Pap Smear
American Society for Colposcopy and Cervical Pathology (ASCCP) hosted a consensus conference following the TBS 2001 conference to develop evidence-based guidelines for the management of women with cervical abnormalities spanning from equivocal diagnoses such as ASC-US to HSIL (CIN 3) based on the findings of the ALTS study and the subsequent longitudinal studies authenticating the original findings of ALTS42,43,44 that a single HPV testing is equal in sensitivity to colposcopy and 2 follow-up cytology 6 months apart. This led ASCCP to recommend that if liquid-based Pap smear is used in patients with a cytological diagnosis of ASC-US, high-risk HPV testing is the preferred method for following those patients to detect the 8% to 20% of the subset at risk of having HSIL or cancer to be referred to colposcopy and further treatment. 45 HPV testing was not recommended for those patients with a diagnosis of LSIL (CIN 1) or ASC-H due to the high prevalence of HPV positivity and therefore referral to colposcopy is preferred to HPV reflex test. It also recommended that HPV testing is a valid marker as a follow-up of women not treated for ASC-US or LSIL and as a posttreatment management of women with LSIL, HPV-positive ASC-US and ASC-H, and for those treated by conization for HSIL to detect evidence of HSIL recurrence high in the endocervical canal. 46 The substantial clinical benefits of HPV reflex testing include immediate referral to colposocopy if HPV is positive for further management without any delay for waiting for repeat cytology results after 6 months, lower attrition to follow-up compliance, cost effectiveness, and less anxiety for those patients with HPV-negative results due to the nearly 100% negative predictive value of HPV testing.
The sensitivity of a conventional Pap smear was about 70% and 93% with liquid-based ThinPrep, but not the 100% the public expects from a cervical cancer screening program. With the advent of HPV testing combined with liquid-based Pap smear testing, 100% sensitivity and a 100% negative predictive value if both tests are negative was feasible. This will be a great advance for minimizing patient and laboratory risk and better outcome for patients. 46 Lorincz and Richart, 47 in their-meta analysis of 10 large studies using HC2 and 3 studies using polymerase chain reaction test to predict rare underlying HSIL (CIN 2/3) using HPV triage in patients with ASCUS diagnosis, concluded that HPV DNA testing was more sensitive than conventional or liquid- based Pap smear cytology. They also reported that the combined HPV DNA and Pap test had a sensitivity and negative predictive value of about 100%. The specificity of the combined tests was reported to be slightly lower than that of the Pap test but this decrease could be offset by the greater protection and cost savings from extending screening intervals. A single “double-negative” HPV DNA and Pap test can provide a better prognostic assurance against risk of future development of HSIL (CIN 3) than 3 subsequent conventional Pap tests and therefore may allow 3-year screening intervals for these low-risk women. Similar results were reported in population- based studies from Sweden 48 and Canada. 49 All these findings led to FDA approval of HPV testing in conjunction with cytology for women >30 years (the DNA Papanicolaou test) on March 31, 2003. 50 A comparative summary of the different screening and management guidelines is provided in Table 3.
ASCCP Clinical Management Guidelines for HPV Testing and Abnormal Pap Smear Findings
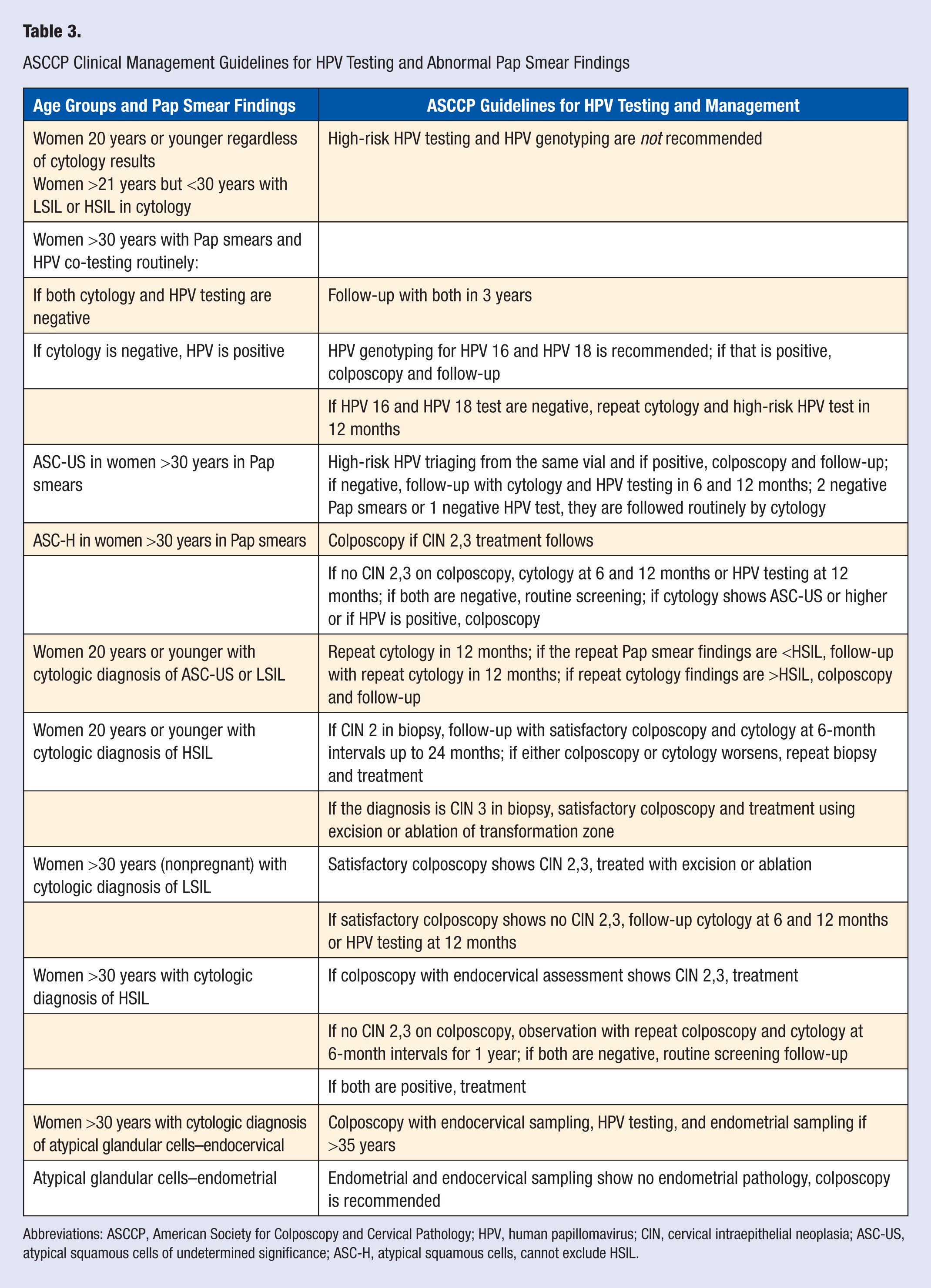
Abbreviations: ASCCP, American Society for Colposcopy and Cervical Pathology; HPV, human papillomavirus; CIN, cervical intraepithelial neoplasia; ASC-US, atypical squamous cells of undetermined significance; ASC-H, atypical squamous cells, cannot exclude HSIL.
Castle et al 51 and others52,53 substantiated the benefits of combining HPV DNA and Pap test for women >30 years for detection of HSIL (CIN 2/3) when compared with repeated Pap testing alone. But a major issue with the HPV test is the specificity of 83%, slightly less than the Pap test. Therefore, many authors emphasized the use of a combined HPV test and Pap test to get the 23% to 43% increased sensitivity of HPV test when compared with cytology and to get a 5% to 8% increase in specificity of Pap smear when compared with the HPV test, 42 and it is not yet time to use the HPV test as the only primary screening test.50,54-56
Patient education and compliance and development of a strong partnership between physicians and patients are important aspects for a successful screening program. Roach 57 stressed the importance of educating the public about HPV testing, deemphasizing the sexually transmitted nature of the disease, and developing strong partnerships between professional and patient communities to get optimal results in minimizing the incidence of cervical cancer.
Primary HPV Screening in Developing Countries
The benefits of clinical application of HPV testing as a primary screening modality in resource-poor settings with high incidence of cervical cancer and lacking screening programs due to lack of resources and trained personnel was first studied in a landmark study by Belinson et al 26 in Shanxi Province in China. In this study, each of the 1997 women aged 35 to 45 years underwent a self-test for HC2 assay for intermediate-risk and high-risk HPV, fluorescence spectroscopy, a liquid-based Pap test (read manually and by automated screening), a visual inspection (VIA) diagnosis, and colposcopy with multiple cervical biopsies. Based on the findings, the authors concluded that in rural areas in China, HPV testing can be successful as a primary screening tool followed by VIA in which acetic acid is applied to the cervix and ablation of the transformation in those women with suspected precancer to reduce the morbidity and mortality from cervical cancer. This option prevents delay in screening, diagnosis, and treatment of women from rural areas who might be lost to follow-up if HPV co-testing with Pap smear is touted as the only option for screening these women. Results from the POBASCAM trial 58 in the Netherlands also justified the use of HPV DNA testing as a routine primary screening with continual monitoring and longer screening intervals. Goldie et al 59 confirmed that the use of VIA and HPV DNA testing in 1 or 2 clinical visits are more cost-effective alternatives to the conventional 3 consecutive cytology-based screenings in resource-poor regions in India, Kenya, Peru, South Africa, and Thailand. The New Technologies for Cervical Cancer screening (NTCC) study 60 conducted a large, randomized controlled trial comparing liquid-based cytology combined with HPV DNA in the first phase and HPV DNA testing alone in the second phase. The conclusions of that study were to do HPV DNA testing and to use cytology to triage only those patients with positive HPV test and with HSIL (CIN 2/3) in cytology for colposcopy and further work up. Denny et al 61 reported from their study in Cape Town, South Africa, that HPV DNA test and direct visual inspection of the cervix with 5% acetic acid (ie, VIA) and cervicography identified similar numbers of HSIL and invasive carcinoma as Pap smear, but the false positives with HPV DNA test and VIA were considerably higher than Pap smear.
Current Status of HPV Testing in the United States
In 2006, ASCCP revised management guidelines for managing women with abnormal cervical cancer screening results based on the current understanding of HPV biology and on the large studies comparing the efficacy of HPV testing for reflex, as a primary co-test with Pap smear, and for follow-up of patients after treatment of significant precancerous lesions. 62 ASCCP consensus guidelines recommend HPV testing to triage patients with ASC-US for colposcopy, as a co-test for primary screening of women >30 years, and to monitor and follow-up patients who have had significant precancerous abnormalities in the squamous epithelium (HSIL) or endocervical glandular epithelium (atypical glandular cells favor neoplasia or adenocarcinoma in situ).
Recommendations were made for special populations such as adolescents with a high prevalence of transient HPV infection and clearance. In this population, for adolescent women with ASC-US and LSIL, HPV testing is not recommended, but a follow-up Pap smear in 12 months is recommended, and if that shows HSIL (CIN 2/3) then follow-up by colposcopy. This conservative approach is recommended to minimize aggressive ablation procedures such loop electrosurgical excision procedure or conization for lesions that might regress and to prevent complications of incompetent cervix in this age-group of women in their reproductive age. Recommendations were clearly outlined for special populations, such as women who are pregnant, postmenopausal, or immunosuppressed with a cytological diagnosis of LSIL or HSIL. More importance is placed on immediate “screen and treat” approaches for women with HSIL. It also recommended HPV testing for managing women with atypical glandular cells after colposcopic evaluation and endometrial sampling. HPV co-testing with Pap smear as a primary screening method for women >30 years was approved by FDA in 2003 and the interim guidelines of ASCCP in 2004 was formally adopted with clear guidelines of management if either of the test is positive was provided. It emphasized that if both HPV DNA and Pap smear are normal, repeat screening need be done only after 3 years.
Human Papillomavirus Vaccine and Its Effect on Cervical Cancer
Research into HPV biology and the role of high-risk types 16 and 18, causing about 70% of cervical carcinoma worldwide, and the role of low oncogenic risk types 6 and 11, causing predominantly anogenital warts, eventually led to the development of 2 FDA-approved prophylactic vaccines based on virus-like particles against capsid proteins L1: (a) a quadrivalent HPV vaccine (HPV 4; Gardasil, Merck, Whitehouse Station, New Jersey) that targets oncogenic types 16 and 18 and the HPV types 6 and 11 primarily responsible for anogenital warts and (b) a HPV 16/18 AS04-adjuvanted vaccine (Cervarix, GlaxoSmithKline Biologicals, Rixensart, Belgium), a bivalent vaccine targeting HPV types 16 and 18 only 63 with partial cross-protection against HPV types 31 and 45. Both vaccines have been shown to be relatively safe, immunogenic, and highly effective against type-specific infection, especially when administered to young girls before onset of sexual activity.64,65 Both Gardasil and Cervarix are recommended for girls around 11 to 12 years but can be given to those as young as 9 years. Gardasil is also approved as a catch-up vaccination provision for girls and women 13 to 26 years who did not receive all the 3 doses of the vaccine administered at day 1, month 2, and month 6. In 2009, the FDA licensed Gardasil for use in males aged 9 through 26 years for prevention of anogenital warts. Koutsky 66 of the Females United to Unilaterally Reduce Endo/Ectocervical Disease (FUTURE) II Study Group conducted a 1:1 randomized, double-blind trial of 12 167 women between the ages of 15 and 26 years in which the women in the vaccine group received 3 doses of the Gardasil quadrivalent vaccine. The primary composite endpoint was CIN 2/3, adenocarcinoma in situ, or cervical cancer related to HPV 16 or HPV 18. All subjects were followed for 3 years to determine vaccine efficacy. When the vaccine was administered to subjects who had not been exposed to either HPV 16 or HPV 18, the vaccine was highly effective (98%) in preventing HPV 16 or HPV 18 related CIN 2/3, adenocarcinoma in situ, or cervical cancer. The vaccine efficacy dropped to 44% for the population of women who had undergone randomization, which included women with HPV 16 or HPV 18 CIN 2/3 or infection with HPV 16 and HPV 18 before the first injection. 66 Many critics feel that the FUTURE II study had not gone long enough to prove the vaccine’s efficacy and claim that in a 3-year follow-up it is difficult to reach statistical significance in a disease process that takes about 10 years to develop. In a recent randomized, double- blind trial involving the per-protocol population, Manalastas et al 67 reported efficacy against disease or infection related to HPV types 6, 11, 16, and 18 was 90.5% and 83.1% for disease or infection related to HPV 16 and HPV 18 alone. GlaxoSmithKline Vaccine HPV-007 Study Group and collaborators reported that the sustained efficacy and immunogenicity of HPV-16,18 AS04-adjuvenated vaccine or Cervarix with a randomized placebo- controlled trial involving 27 sites in 3 countries was up to 6.4 years. 68 Schwartz 69 reported that Cervarix continued to demonstrate high levels of total and neutralizing antibodies against HPV 16 and HPV 18, 7.3 years after vaccination. The safety and immunogenicity of Gardasil and Cervarix have been recently compared with other study groups. 70 These studies were conducted to generate data that can support applications for regulatory approval for vaccination of individuals who fall outside the range for which efficacy data were obtained. In 2 such studies, Gardasil was shown to be safe and immunogenic in adolescent boys and girls with a 2-fold increase of VLP (virus like particles) specific antibodies than the responses in young women. Both vaccines have been evaluated for safety and immunogenicity in trials of “older” women (ages 24-45 years for Gardasil and ages 26-55 years for Cervarix) and results reported in scientific conferences. They affirm that these vaccines are safe in older women and seroconversion rates were high regardless of age. Cervarix was recently approved in Australia for girls from 10 years to women through age 45 years. Slade et al 71 summarized the reports to the Vaccine Adverse Event Reporting System and reported that most of the adverse effects following immunization with Gardasil were not greater than the background rates reported for other vaccines, with syncope (fainting) and venous thromboembolic events being the most common. Other minor side effects such as pain or soreness in the area of vaccination, fever, headache, and rarely Guillian–Barre syndromes have been reported.
Goldfarber-Fiebert et al 72 reported that vaccinating girls before age 12 years will likely be highly cost-effective in cervical cancer screening using an empirical model to assess the quality-adjusted life years, lifetime costs, and incremental cost-effectiveness of cervical screening, vaccination of teenage girls, and vaccination combined with screening. Initiation of cervical screening in this model was 21 years for unvaccinated women and 25 years for vaccinated women, and the screening interval was 3 years for the unvaccinated group and 5 years for the vaccinated group. For unvaccinated women, screening by cervical cytology with HPV triage between ages 21 and 25 years and primary HPV DNA testing with cytology triage or co-testing HPV and cytology for women older than 30 years was found to be more cost-effective. For the group of girls vaccinated by age 12 years, screening by cervical cytology with HPV triage every 5 years starting at age 25 years and switching to primary HPV DNA testing with cytology triage at age 35 years was found to be very cost-effective to maintain the same threshold as the unvaccinated group. Although it is accepted that HPV vaccines might offer a great advance in cervical cancer prevention, there are too many unresolved issues such as recommended age for vaccination, who should be vaccinated, public acceptability, cost, possible side effects, the length of period of immunity provided by the vaccine, the possible need for booster, and so on. However, in developing countries without successful screening programs vaccination might play a major role in reducing the morbidity and mortality associated with cervical precancerous conditions and cancer. It might also be more cost-effective when vaccination is used as a preventive strategy as reported in a predictive model from Mexico 73 and other countries. 74 Many therapeutic vaccines are in various phases of clinical trials and none are approved by the FDA. They hold promise since they are intended to promote antibodies against oncoproteins E6 and E7, which are constitutively expressed in HPV-associated precancers and cancers and play a critical role in induction, maintenance, cellular transformation, and progression of HPV-associated diseases.
Future of Cervical Cancer Screening in the United States
National Health Interview Survey estimated that there were approximately 66 million Papanicolaou cervical screening tests performed in the United States in 2000 and 65 million in 2005. Eltoum and Roberson 75 reported that with the advent of primary HPV co-testing with Pap smear for women aged 30 years and older, the reduced screening frequency to 3 years if HPV DNA test and Pap smear are both negative, other recommended uses of HPV testing by ASCCP, impact of HPV vaccine, and changing demographics, the Pap test volume will diminish annually by 43%. But an accurate estimate of the extent of reduction in Pap tests, changes in their role from screening to triage for HPV DNA testing as suggested in some studies, and the level of adoption of HPV vaccination and its impact provide a dynamic scenario for cervical cancer screening for the future.
Footnotes
Acknowledgements
I would like to acknowledge Dr Hassan El-Fawal, PhD, Chair of the Department of Health Sciences, Albany College of Pharmacy and Health Sciences, for reviewing and providing comments on this article.
