Abstract
Objective
Ultra-high dose rate radiotherapy, commonly referred to as FLASH radiotherapy, is a transformative focus in radiotherapeutic research. This study aimed to identify intestinal contents based metabolic biomarkers of photon FLASH.
Methods
C57BL/6J mice were exposed to 0 Gy, 10 Gy, 12 Gy, or 14 Gy of photon FLASH irradiation (FLASH-IR) without a conventional irradiation. The intestinal contents were collected 3.5 d post-iiradiation, and subjected to liquid chromatography-mass spectrometry.
Results
Metabolomic profiling revealed 25 differential metabolites that were consistently differentially regulated across the comparisons of 10 Gy vs. 0 Gy, 12 Gy vs. 0 Gy, and 14 Gy vs. 0 Gy groups. Notably, a dose-dependent positive correlation was observed for 3-hydroxyanthranilate, while 13 metabolites—including guanosine, adenosine monophosphate, and hydroquinone—exhibited a significant negative correlation with increasing FLASH-IR dose. Further enrichment analysis identified several perturbed metabolic cascades, prominently involving the PI3K-AKT signaling pathway, tyrosine metabolism, and purine metabolism.
Conclusions
These findings illuminated the intestinal metabolic signatures post photon FLASH-IR, offering insights to optimize FLASH radiotherapy parameters and enhance its therapeutic ratio.

Introduction
Radiotherapy remains a cornerstone in the management and eradication of malignant tumors; however, its therapeutic efficacy is often constrained by collateral damage to adjacent normal tissues. In recent years, ultra-high dose rate radiotherapy (commonly referred to as FLASH radiotherapy) has emerged as a transformative innovation in radiation oncology.1,2 Defined by the delivery of an entire therapeutic dose to the target volume within an ultra-short timeframe (<1 second) at dose rates exceeding 40 Gy/s, FLASH radiotherapy demonstrates remarkable potential to preserve tumor control while mitigating normal tissue toxicity compared to conventional radiotherapy, thereby substantially widening the therapeutic index of radiation therapy. 3 Preclinical investigations have consistently validated the efficacy of FLASH radiotherapy. 4 The landmark FAST-01 clinical trial, which evaluated proton FLASH radiotherapy for symptomatic bone metastases, reported promising outcomes with no overt adverse events. 5 Together with the ongoing FAST-02 trial, it will provide critical evidence for its clinical translatability.6,7
Nonetheless, the successful translation of FLASH radiotherapy into routine clinical practice hinges on a comprehensive understanding of its underlying biological mechanisms. In our previous work, we compared the impacts of FLASH-IR and CONV-IR on metabolic profiles and gut microbiota perturbations in colon cancer models. 8 However, study exploring metabolic and cellular responses to variable FLASH-IR dosing remains scarce. To address this knowledge gap, the present study employs an untargeted metabolomics approach to investigate dose-dependent metabolic perturbations in the intestinal contents of mice following FLASH-IR at doses ranging from 10 to 14 Gy without a CONV-IR comparator. By elucidating the metabolic signatures induced by photon FLASH-IR, this work aimed to dose-dependent intestinal metabolic changes under high-dose single-fraction irradiation delivered at photon FLASH dose rates in mice and accelerate the clinical implementation of photon FLASH radiotherapy.
Materials and Methods
Experimental Animals and Groups
This study was performed from January 2025 to June 2025. Five-week and age-matched 20 male C57BL/6J mice were purchased from the Soochow University Laboratory Animal Centre. All mice were housed under SPF conditions with 12/12-h light/dark cycles and were freely accessed to water and food. After a one-week acclimation period, all mice were randomly divided into four groups (five animals in each group): control, not irradiated mice (F0), mice irradiated with a single-dose 10 Gy of FLASH-IR (F10), mice irradiated with a single-dose 12 Gy of FLASH-IR (F12), mice irradiated with a single-dose 14 Gy of FLASH-IR (F14).
Instrumentation and FLASH-IR
Mice were anesthetized via intraperitoneal injection of amobarbital. The mice were immobilized on plastic boards using adhesive tape to prevent movement during radiation exposure. Whole-abdomen FLASH-IR was performed using a 10 MeV photon FLASH radiotherapy X-ray source in Mianyang, China without a CONV-IR comparator. 9 This compact FLASH X-ray source system that utilizes an S-band accelerator to generate electron beams with energies of up to 10 MeV. 9 These beams then bombard a high-speed rotating tungsten target, converting into ultra-high dose-rate X-rays via bremsstrahlung radiation. Real-time monitoring of the electron beam was achieved using a high-speed current transformer (Bergoz, ICT-082-012-05) mounted upstream of the target. X-ray output was tracked with a diamond detector (PTW, Ref. TW60025) positioned at the collimator inlet. The detector signal was transmitted via a coaxial cable to a digital oscilloscope (Rigol, DS7034) for data acquisition. This oscilloscope featured automated capture, storage, and integration of transient pulse signals. The integrated pulse value served as a monitoring metric for accelerator output, while beam current was finely tuned by adjusting the drive laser parameters. A single dose of 10 Gy, 12 Gy or 14 Gy was delivered to the whole abdomen at a dose rate of 200 Gy/s. The dose was selected based on the FLASH-IR lethal dose (LD50) values of Evans et al. 10 The source-to-skin distance (SSD) was 19 cm, and the radiation field size was 4 cm × 3 cm. Healthy control mice were treated with 0Gy sham irradiation under the same conditions. EBT3 film (Ashland Inc, USA) was used for dose verification to monitor the stability and reliability of the radiation beam output.
Intestinal Tissue Processing and HE Staining
Considering that many preclinical studies consistently identify 3–4 days post-abdominal irradiation as the peak of acute intestinal mucosal injury in murine models, characterized by maximal villous atrophy, crypt apoptosis, and inflammatory cell infiltration,11,12 all surviving mice were killed after 3.5 d of irradiation. The ileal ends of interest for the study were removed and fixed in 4% paraformaldehyde fixative. Intestinal specimens were paraffin-embedded, sectioned at 3 µm, oven-dried, dewaxed and rehydrated, stained with hematoxylin-eosin, sealed with neutral resin and coverslips. We adopted a validated semi-quantitative scoring system originally described by Chiu et al, 13 which is widely recognized and utilized in assessing intestinal mucosal injury. This system grades intestinal damage on a scale of 0 to 5, with each grade corresponding to distinct histological features observed under light microscopy. All histological slides were independently evaluated by two pathologists who were blinded to the treatment groups. In cases of discrepancy between the two evaluators, a third senior pathologist was consulted to reach a consensus score. This approach ensured the objectivity and reliability of our intestinal injury assessment.
Extraction of Intestinal Contents
Intestinal contents were obtained by rinsing ileocolonic segments with 0.5 mL phosphate buffered saline (PBS) pre-cooled at 4 °C. The sample was accurately weighed into a 2 mL centrifuge tube, and then 600 µL MeOH was added and vortexed for 30 s. Steel balls were added and stirred for 120 s at 50 Hz in a tissue grinder. The reaction mixture was sonicated at room temperature for 10 min, followed by centrifugation at 12000 rpm for 10 min at 4 °C. The supernatant was filtered by 0.22 μm membrane and transferred to the detection bottle for liquid chromatography-mass spectrometry (Figure 1). The workflow of liquid chromatography-mass spectrometry for intestinal metabolomics analysis in mice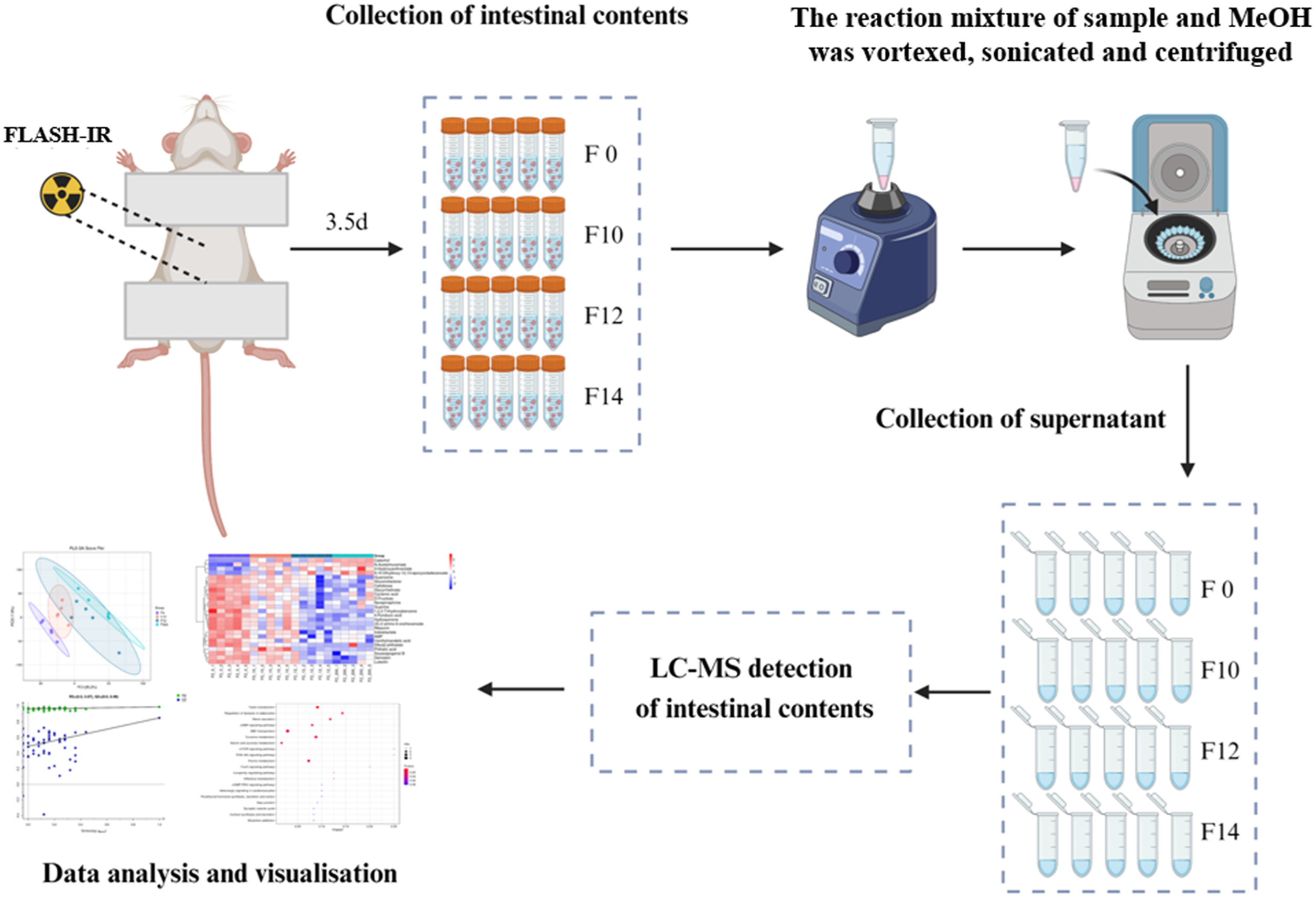
Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis of Intestinal Contents
Liquid chromatography analysis was performed on a Vanquish UHPLC System (Thermo Fisher Scientific, USA). Chromatography was carried out with an ACQUITY UPLC ® HSS T3 (2.1×100 mm, 1.8 µm) (Waters, Milford, MA, USA). Mass spectrometric detection of metabolites was performed on Orbitrap Exploris 120 (Thermo Fisher Scientific, USA) with ESI ion source. The differential analysis in this study involved identifying differential metabolites from a list of primary metabolites. Consequently, all differential metabolites included in the final screening and discussion were those achieving MSI Level 1 status. This was accomplished by matching exact mass, retention time, and MS/MS fragment spectra against chemical reference standards on the same platform. The detailed liquid chromatography conditions and masspectrum conditions were depicted in supplementary methods part.
Differential Metabolites Identification and Pathway Analysis
The raw data were firstly converted to mzXML format by MSConvert in Proteo Wizard software package (v3.0.8789) 14 and processed using R XCMS (v3.12.0) for feature detection, 15 retention time correction and alignment. The batch effect was then eliminated by correcting the data based on QC samples. Metabolites with RSD>30% in QC samples were filtered and then used for subsequent data analysis. The statistical significance of P.value was obtained by statistical test between groups. Combined with P.value, VIP (variable importance in projection) and FC (multiple of difference between groups) to screen biomarker metabolites. By default, when uncorrected P value < 0.05 and VIP value > 1, we think that metabolites were considered to have significant differential expression. Results from univariate comparisons were adjusted to account for the false discovery rate (FDR) using Benjamini and Hochberg. Differential metabolites were subjected to pathway analysis by MetaboAnalyst. 16 The metabolites and corresponding pathways were visualized using KEGG Mapper tool.
Statistical Analysis
R version 4.0.3 was used for all statistical analyses. Student’s t-test was used to conduct two-group comparisons. P<0.05 was considered statistically significant.
Results
The Effect of FLASH-IR on the Morphology Changes of Intestinal Tissues in Mice
To explore the effects of different FLASH doses on the damage of intestinal tissues, FLASH-IR was used to create intestinal injury models in healthy C57BL/6J mice. Morphological changes in the intestinal tissue were observed using HE staining. Compared to non-irradiated controls (Figure 2A), mice exposed to FLASH-IR exhibited varying degrees of intestinal tissue damages, which worsened with increasing radiation dose (Figure 2B–E). The pathological changes included: widening of the subepithelial space, detachment of the mucosal layer from the lamina propria, shedding of villus tips, degeneration of the lamina propria, and hemorrhage. These data suggested that the extent of small intestinal damage varies with changes in the dose of FLASH-IR. Changes in intestinal injury in mice after whole-abdominal FLASH-IR with different doses. (A): Control group (Magnification 200×). (B-D): FLASH-IR groups received 10, 12 or 14 Gy X ray irradiation (Magnification 200×). (E): Statistical chart of intestinal injury scores in the control group and FLASH irradiation groups with different doses (**P<0.01,***P<0.001)
Quality Analysis of the Metabolome for Intestinal Contents
We previously have compared the effects of FLASH-IR versus CONV-IR on metabolites and gut microbiota alterations in colon cancer models. 8 However, studies about the metabolic and cellular responses to varying doses of FLASH-IR remain limited. In this study, we focused on evaluating the impact of different doses of FLASH-IR on metabolite profiles in murine intestinal contents.
To understand the changes of intestinal differential metabolites in mice during different doses of FLASH-IR, multivariate statistical analyses such as PCA, PLS-DA, and OPLS-DA were used to explore the relationship between biological characteristics and metabolomics. Figure 3A presents the results of unsupervised PCA analysis, serving as the primary reference for evaluating the inherent structure of the data and the model’s validity. As shown in Figure 3B–C, differences in metabolite profiles were observed between the control group and the other three FLASH-IR groups. The differences became more significant as the irradiation dose increased, with the most pronounced difference between 14Gy and the control group. The classification parameters R2Y = 0.992 and Q2 = 0.852 indicated a good fit and predictive power. A permutation test of 100 iterations showed that the intercept values of R2 and Q2 were (0, 0.97) and (0, 0.49), respectively, thus indicating that the PLS-DA mode was reliable. Multivariate statistical analysis of identified metabolites in different irradiation doses. (A): Principal component analysis (PCA) score plot for all grouped samples. (B): The degree of separation of the intestinal contents samples in the partial least squares discriminant analysis (PLS-DA) score plot. (C): The validation of the PLS-DA model by permutation testing (100 iterations)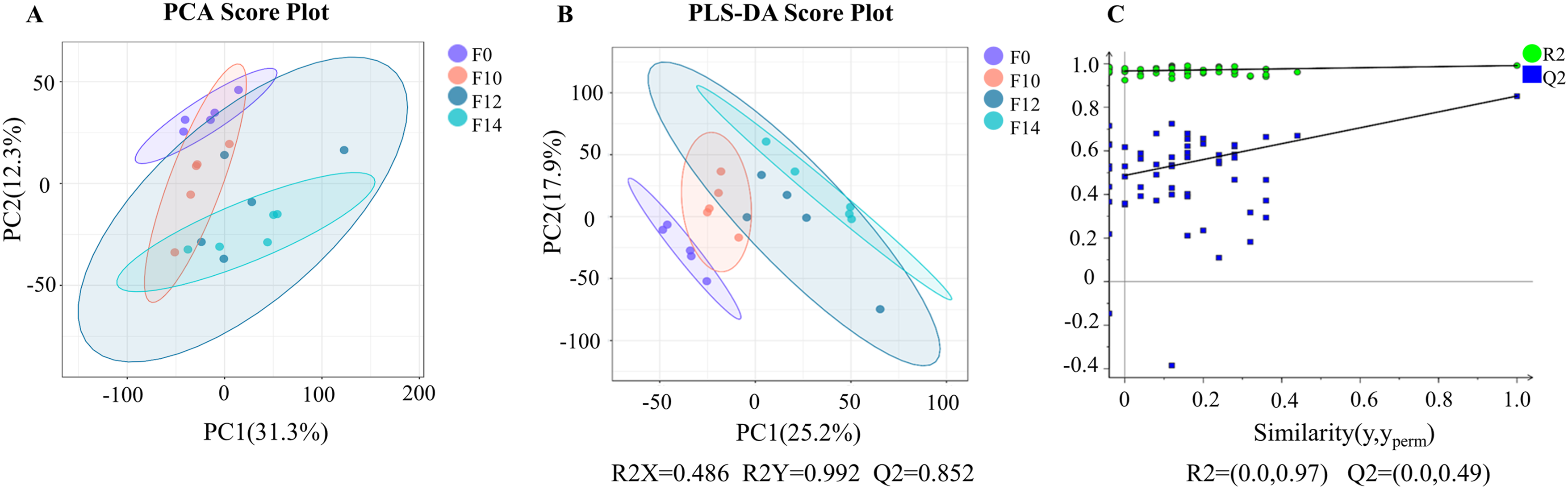
It should be noted that, although the validity of the model has been assessed through substitution tests, readers should exercise caution when interpreting the results due to the risk of overfitting. If a larger independent validation cohort becomes available in the future, it will provide more reliable confirmation of the preliminary findings regarding the differences in metabolites identified in this study.
Screening for Intestinal Differential Metabolites
Parameters of Common Intestinal Differential Metabolites in Mice Irradiated at Different Doses of FLASH-IR
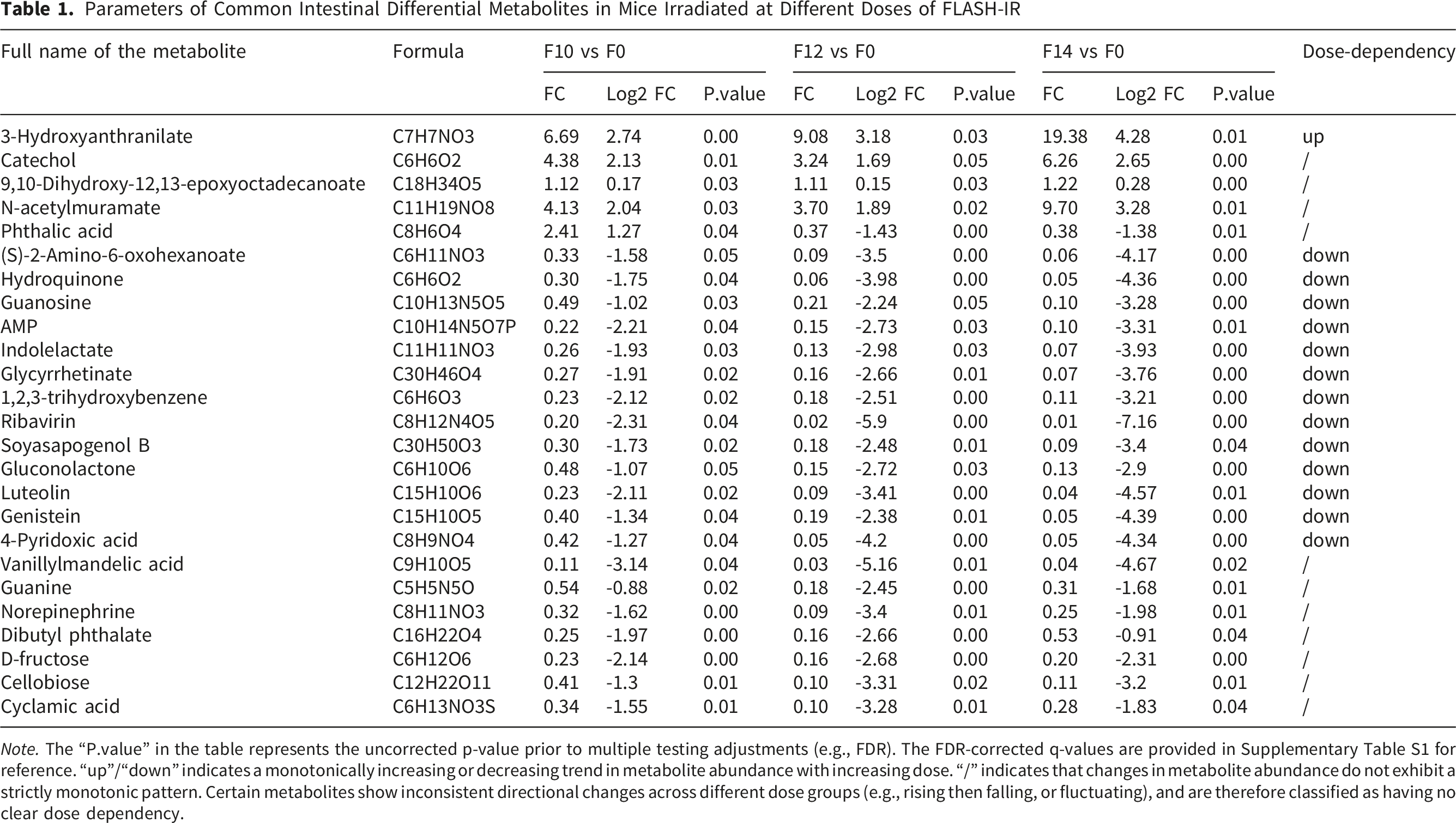
Note. The “P.value” in the table represents the uncorrected p-value prior to multiple testing adjustments (e.g., FDR). The FDR-corrected q-values are provided in Supplementary Table S1 for reference. “up”/“down” indicates a monotonically increasing or decreasing trend in metabolite abundance with increasing dose. “/” indicates that changes in metabolite abundance do not exhibit a strictly monotonic pattern. Certain metabolites show inconsistent directional changes across different dose groups (e.g., rising then falling, or fluctuating), and are therefore classified as having no clear dose dependency.
Within these 25 shared metabolites, 3-hydroxyanthranilate, catechol, 9,10-dihydroxy-12,13-epoxyoctadecanoate, and N-acetylmuramate were significantly elevated in FLASH-IR-exposed groups compared to the 0 Gy control. Notably, their levels were most pronounced at the highest dose (14 Gy). Phthalic acid displayed a distinct pattern, with up-regulation at 10 Gy followed by down-regulation at 12 Gy and 14 Gy relative to the 0 Gy group. The remaining 20 metabolites, including D-fructose, adenosine monophosphate (AMP), and guanine, were consistently decreased across all FLASH-IR doses compared to the control (Figure 4). These findings offered further evidence clarifying the mechanism of the effects on intestinal tissues by FLASH-IR. Heat map visualization of intestinal differential metabolites after different doses of FLASH-IR in mice
Dose-dependent Metabolite Analysis
To further explore the relationship between these 25 common differential metabolites and FLASH-IR dose escalation (10 Gy → 12 Gy → 14 Gy), we defined “dose dependency” as a monotonic trend in the relationship between metabolite abundance (Log2 FC) and radiation dose (0 Gy, 10 Gy, 12 Gy, 14Gy). It was observed that 3-hydroxyanthranilate was the sole metabolite exhibiting a regular, dose-dependent increase, with its levels positively correlated with escalating FLASH-IR doses (Table 1). Thirteen metabolites displayed a regular, dose-dependent decrease, where their levels were negatively correlated with increasing FLASH-IR doses (Table 1), including guanosine, AMP, and hydroquinone, and so on. These results highlighted 3-hydroxyanthranilate and the 13 down-regulated metabolites as potential dose-responsive biomarkers, offering insights into the metabolic pathways perturbed by FLASH-IR in a dose-dependent manner.
Analysis of Signaling Pathways Associated With Differential Metabolites
To investigate the dose-dependent effects of FLASH-IR on intestinal metabolic pathways in mice, we conducted KEGG pathway enrichment analysis using Metabo Analyst on the 25 previously identified differential metabolites. This analysis predicted alterations in a total of 42 distinct metabolic pathways. Figure 5 presents the top 11 pathways exhibiting the most significant enrichment, defined by a p-value threshold of less than 0.05. Notably, three pathways demonstrated particularly high biological impact scores exceeding 0.15: the mTOR signaling pathway, the PI3K-AKT signaling pathway, and the FOXO signaling pathway. These pathways are critically involved in fundamental cellular processes including nutrient sensing, growth regulation, survival, and stress response, suggesting profound perturbations in these core mechanisms within the gut environment following varying FLASH-IR doses. Furthermore, the analysis highlighted several other crucial pathways providing deeper insight into the metabolic shifts induced by irradiation. These included the regulation of lipolysis in adipocytes, tyrosine metabolism, purine metabolism, and so on. Collectively, these enriched pathways paint a complex picture of the intestinal metabolic response to FLASH-IR, implicating disruptions in central signaling hubs governing cell fate and metabolism (mTOR, PI3K-AKT, FOXO), alongside significant changes in specific metabolic processes like lipid breakdown, amino acid utilization, and nucleotide turnover. Significantly changed pathways based on the enrichment and topology analysis. The x-axis represents the pathway impact score derived from topological analysis, which reflects the relative importance of the pathway in the network. The y-axis lists the enriched pathways
Discussion
Prevention or mitigation of radiation-induced damage to normal tissues has always been a theme of interest in radiotherapy research. FLASH radiotherapy is one of the most promising approaches based on the normal tissue-sparing effects of ultra-high dose rate irradiation (>40 Gy/s),17,18 and FLASH effects have been observed in various tissues and organs. A single dose of 17 Gy FLASH radiotherapy significantly inhibited the development and progression of pneumonia and pulmonary fibrosis compared to CONV radiotherapy, with an equivalent anti-tumor effect. 19 A comparative study by our group previously revealed that FLASH radiotherapy significantly reduced the inflammatory response in mouse liver tissue compared to CONV radiotherapy. 20
Metabolites are the final downstream products of gene expression, and the progression of radiation-induced injury is accompanied by changes in metabolism-related genes and metabolites. 21 In previous studies, we have used metabolomics techniques to screen FLASH-IR vs CONV-IR for metabolites differentially expressed in fecal samples from colorectal cancer mice 8 and plasma samples from glioma mice, 22 with the aim of identifying common or specific radiosensitisation markers in different modalities. However, how dynamic changes in key physical parameters of FLASH-IR (e.g., radiation source, dose, dose rate) would affect changes in metabolites in mice has not been explored. In this study, we irradiated the abdomen of healthy C57BL/6J mice with three different photon FLASH doses (10Gy, 12Gy or 14Gy) without a CONV-IR comparator. It showed that the different doses induced changes in the intestinal metabolites of the mice, and the difference became more significant with increasing dose, with the most obvious differences in the intestinal contents between 14Gy and the control group. A total of 166 differential metabolites were screened according to P value and VIP threshold. A total of 25 intestinal differential metabolites were present at all three irradiation doses of 10Gy, 12Gy or 14Gy compared to the control group, which have the potential to be radiosensitive markers common to different doses of photon FLASH-IR.
D-fructose plays an important role in promoting intestinal cell survival and lipid uptake. External studies have reported that it inhibits apoptosis of intestinal cancer cells and promotes intestinal tumor growth by stimulating the PKM2-HIF-1α-glycolysis pathway, 23 while high concentrations can disrupt the intestinal barrier, leading to inflammatory responses and tissue damage in the intestine. 24 Compared to the control group, d-fructose levels were significantly down-regulated in different dose groups of FLASH. Based on these external findings, we speculate that FLASH-IR may potentially inhibit intestinal tumor growth and reduce inflammatory responses in normal intestinal tissue by modulating glucose metabolism pathways, though this remains an extrapolation without direct evidence from our study.
Vanillylmandelic acid (VMA) is a metabolite of norepinephrine, which is converted from tyrosine as a precursor. 25 The variation trends of VMA and norepinephrine after irradiation with different doses of FLASH were relatively consistent and the contents were down-regulated compared to the control group, with the highest content at 10Gy and the lowest at 12Gy. It implies that the different effects of different doses of FLASH-IR may be related to tyrosine metabolism and its related pathways. Furthermore, we identified 14 dose-dependent differential metabolites in intestinal tissue. Among these, only 3-hydroxyanthranilate showed a progressive increase in content with escalating radiation doses (10 Gy, 12 Gy, 14 Gy). 3-hydroxyanthranilate is an intermediate of tryptophan metabolism, and recent studies have revealed that 3-hydroxyanthranilate significantly stimulates tumor cells to get rid of ferroptosis. 26 In our study, 3-hydroxyanthranilate levels were notably up-regulated in FLASH-irradiated mice across all doses, displaying a clear dose-dependent relationship. Based on this external evidence, we tentatively extrapolate that FLASH-IR may potentially modulate the efficacy of different radiation doses on tumor cells through regulation of the tryptophan metabolic pathway, though this hypothesis requires direct validation in future studies. In addition, the level of AMP, a key metabolite involved in energy metabolism, was down-regulated after FLASH-IR, showing a negative dose-dependent correlation.
The KEGG metabolic pathway enrichment analysis of the screened intestinal differential metabolites revealed that the top three pathways with the highest impact values were the mTOR signaling pathway, the PI3K-AKTsignaling pathway, and the FOXO signaling pathway, respectively. The downstream target of AKT is mTOR, and the downstream transcription factors of mTOR include FOXO, which is involved in the regulation of energy metabolism. External research has established that the PI3K/AKT/mTOR pathway promotes cell survival, cell growth, and cell cycle progression. 27 While dynamic changes in this pathway may theoretically influence cellular sensitivity to FLASH-IR, this is a speculative inference based on external literature rather than direct evidence from our study.
This study has several limitations that should be acknowledged when interpreting the findings. First, it focused solely on metabolite differences across FLASH-IR doses without including a comparative analysis against CONV-IR groups. This gap should be addressed in future research to contextualize the observed metabolic changes relative to CONV-IR protocols. Second, our analysis was conducted at a single acute time point post-irradiation, representing only an “acute phase” snapshot of metabolic changes. Long-term or chronic metabolic alterations in the intestinal microenvironment remain entirely unknown, and we cannot assume that the identified metabolites represent stable biomarkers of FLASH exposure. Validation of these findings across multiple time points is critical to understand the temporal dynamics of metabolic responses and determine the potential utility of these metabolites as robust biomarkers. Third, all experiments were performed using healthy, non-tumor-bearing mice, so the current data reflect metabolic changes in normal intestinal tissue of physiologically intact animals. Additionally, the small cohort size prevents subgroup analyses to explore potential age-related metabolic responses, future validation in larger cohorts (≥8 mice per group) is necessary to confirm the generalizability of our findings and reduce statistical bias.
Nevertheless, our results demonstrating a clear dependence of intestinal metabolite profiles on FLASH-IR dose represent a valuable step forward. Our findings characterize intestinal metabolic changes in response to high-dose single-fraction irradiation delivered at photon FLASH dose rates, which enhances our understanding of intestinal metabolic responses to high-dose single-fraction FLASH-IR and provides critical insights for optimizing physical parameters (e.g., dose selection) in FLASH-IR applications.
Conclusion
In summary, this study employed untargeted metabolomics to investigate the impact of varying doses of photon FLASH-IR on intestinal metabolites in mice. Our analysis identified a total of 25 distinct metabolites that may serve as common radiosensitive markers of photon FLASH-IR across different dose levels. Additionally, we observed dose-dependent changes in metabolite profiles: 3-hydroxyanthranilate exhibited a positive correlation with radiation dose, while the remaining 13 metabolites showed a negative dose-response relationship. These findings suggest that exposure to high-dose single-fraction photon FLASH-IR is linked to alterations in key metabolic pathways, including glucose metabolism, tryptophan metabolism, and tyrosine metabolism.
Footnotes
Acknowledgements
We thank Dr. Jianxin Wang and Dr. Yiwei Yang (Institute of Applied Electronics, CAEP, Mianyang, China) for their invaluable support with photon FLASH-IR.
Ethical Considerations
All animal experiments were conducted in strict accordance with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) 2.0 guidelines. The study protocol was approved by the Animal Welfare Ethics Review Board (Approval ID: YSZ-25-01).
Author Contributions
Conceptualization, Z.Y.S., and C.Z.F; Methodology, Z.S.Q., P.Q.L., Z.J., W.Z.M., and C.X.Y.; Formal Analysis, Z.S.Q., P.Q.L., and Z.J.; Investigation, Z.S.Q., P.Q.L., Z.J., W.Z.M., and C.X.Y.; Resources, Z.Y.S., and C.Z.F.; Writing—Original Draft Preparation, Z.S.Q., and C.Z.F.; Writing—Review and Editing, Z.S.Q., P.Q.L., Z.J., W.Z.M., C.X.Y., Z.Y.S., and C.Z.F.; Supervision, Z.Y.S., and C.Z.F.; Project Administration, Z.Y.S., and C.Z.F.; Funding Acquisition, Z.Y.S., and C.Z.F. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (12575369, 12275192), the project from State Key Laboratory of Radiation Medicine and Protection (GZK1202308, GZK12024046), the Nuclear Technology Application Excellent Experts Program of the Second Affiliated Hospital of Soochow University (XKTJ-HRC2021002), Nuclear Medicine Science and Technology Innovation Plan Project (ZHYLZD2025004), Discipline Priority of Suzhou City, Discipline Construction Support Project (XKTJ-XK202410), Central Guidance for Local Science and Technology Development Projects (2023ZYDF071).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
