Abstract
Purpose
FLASH radiotherapy (FLASH-RT) has been reported to spare skin from acute radiation toxicity. However, systematic quantification is needed to determine the dose and dose-rate thresholds necessary for optimal radioprotection in FLASH radiotherapy.
Materials and Methods
The hind limbs of male C57BL/6J mice were irradiated with a single-fraction dose (18-24 Gy) delivered using either FLASH-RT (ultrahigh dose rate: 250 Gy/s) or conventional radiotherapy (CONV-RT: 0.1 Gy/s) in the dose optimization experiment. Subsequent studies included a dose-rate optimization trial comparing FLASH-RT (40-400 Gy/s) with CONV-RT at 20 Gy; the antitumor efficacy of a single 20 Gy dose delivered via FLASH-RT (300 Gy/s) was compared with that of CONV-RT; and an assessment of the reirradiation response using FLASH-RT (400 Gy/s) or CONV-RT at 20 Gy following a prior hypofractionated CONV-RT regimen (9 Gy × 4). Skin injury scores, tumor volume, and body weight were monitored longitudinally for up to 30 days after irradiation or until the experimental endpoint was reached. Tumor weight and lung metastasis nodules were measured at the endpoint.
Results
Dry desquamation, alopecia, moist desquamation, hemorrhage, full-thickness necrosis, and ulceration were observed more frequently in the CONV-RT group than in the FLASH-RT group. Compared with CONV-RT, FLASH-RT resulted in significantly lower acute skin injury scores at doses ≤22 Gy (18 Gy, 20 Gy, 22 Gy, P < 0.001), but this protective effect was absent at doses ≥24 Gy. Dose-rate optimization (40-400 Gy/s) delivering a 20 Gy dose revealed that acute skin injury scores were significantly reduced only at the higher dose-rates (≥300 Gy/s, compared with CONV-RT, P < 0.001), whereas no significant reduction was observed at the lower dose-rates (≤200 Gy/s, P ≥ 0.05). No additional benefit was detected between 300 Gy/s and 400 Gy/s. Although 250 Gy/s provided significant radioprotection compared with CONV-RT (P < 0.01), it was not the minimal rate required for maximal protection, as evidenced by a significantly better outcome at 300 Gy/s at 24 days (P = 0.0296). Finally, in B16/F10 cell-transplanted mouse models, FLASH-RT (300 and 400 Gy/s) achieved tumor control equivalent to that of CONV-RT while also reducing lung metastasis nodules and significantly mitigating acute skin toxicity (P < 0.001) during reirradiation.
Conclusions
In this study, a dual-threshold framework was established to guide the clinical implementation of FLASH radiotherapy: a dose ceiling below 24 Gy to preserve the skin-sparing effect and a dose-rate threshold exceeding 200 Gy/s for efficacy, with further optimization achieved at rates above 250 Gy/s. These findings provide guidelines for minimizing radiotherapy-induced acute skin toxicity while maintaining comparable antitumor efficacy, thereby outlining a translatable pathway towards the clinical translation of FLASH-RT.
Introduction
Ultrahigh-dose-rate irradiation (UHDR), or FLASH radiotherapy (FLASH-RT), has drawn widespread research interest because of its tumor control comparable to that of conventional radiotherapy (CONV-RT), superior normal tissue sparing (the “FLASH effect”), ultrashort treatment time, and reduced behavioral interference during irradiation.1-4 The FLASH effect has been empirically validated in skin using electron beams across multiple species (including murine, feline, canine, mini pig, and human skin cancer models).5-9 Nevertheless, the potential mechanism of FLASH effect has not been clarified yet. At present, the mainstream physical and chemical mechanism hypotheses of FLASH effect include Oxygen Depletion Effect, ROS and Free Radical Effect, Fenton-type reaction and peroxidized compounds, etc., while the biological mechanism hypotheses include immune and inflammatory response, reduction of stem cell senescence, DNA and vascular injury. 10 Furthermore, the occurrence of the FLASH effect is not universal. Some studies have revealed an absence of protective effects or even increased late toxicity following electron FLASH irradiation. For example, Duval et al 11 reported a delayed onset of moist desquamation at 25 Gy not at 30 Gy, with no overall difference in the incidence or severity of acute skin damage between FLASH and CONV irradiation. In contrast, Rohrer Bley et al 12 reported severe late skin toxicity, such as dermal necrosis and osteoradionecrosis in mice after FLASH exposure. Clinical observations by Gaide et al 13 also revealed comparable tumor control and toxicity between FLASH-RT and CONV-RT in lymphoma patients. The literature highlights dose and dose rate as crucial parameters influencing the FLASH effect.14-16 However, systematic studies focusing on pulsed electron beam FLASH and its impact on radiation-induced skin injury remain limited.
To address this, in the present study, murine models of acute radiation-induced skin injury and tumor radiotherapy were established by delivering a single-fraction dose at varying dose and average dose rate using a pulsed electron beam FLASH or CONV irradiation technique to the hind limbs of C57BL/6J mice. The aim of this study is to evaluate the safety and efficacy of FLASH-RT and to identify optimal single-fraction dose and dose-rate thresholds for eliciting the skin FLASH effect by comparing acute injury scores, local tumor control, and distant metastasis inhibition.
Materials and Methods
All experiments were implemented from March 2024 to August 2025.
Animal Resources and the Administration of Irradiation
Experimental Models, Irradiation Modes and Parameters, and Numbers of Mice in Each Group

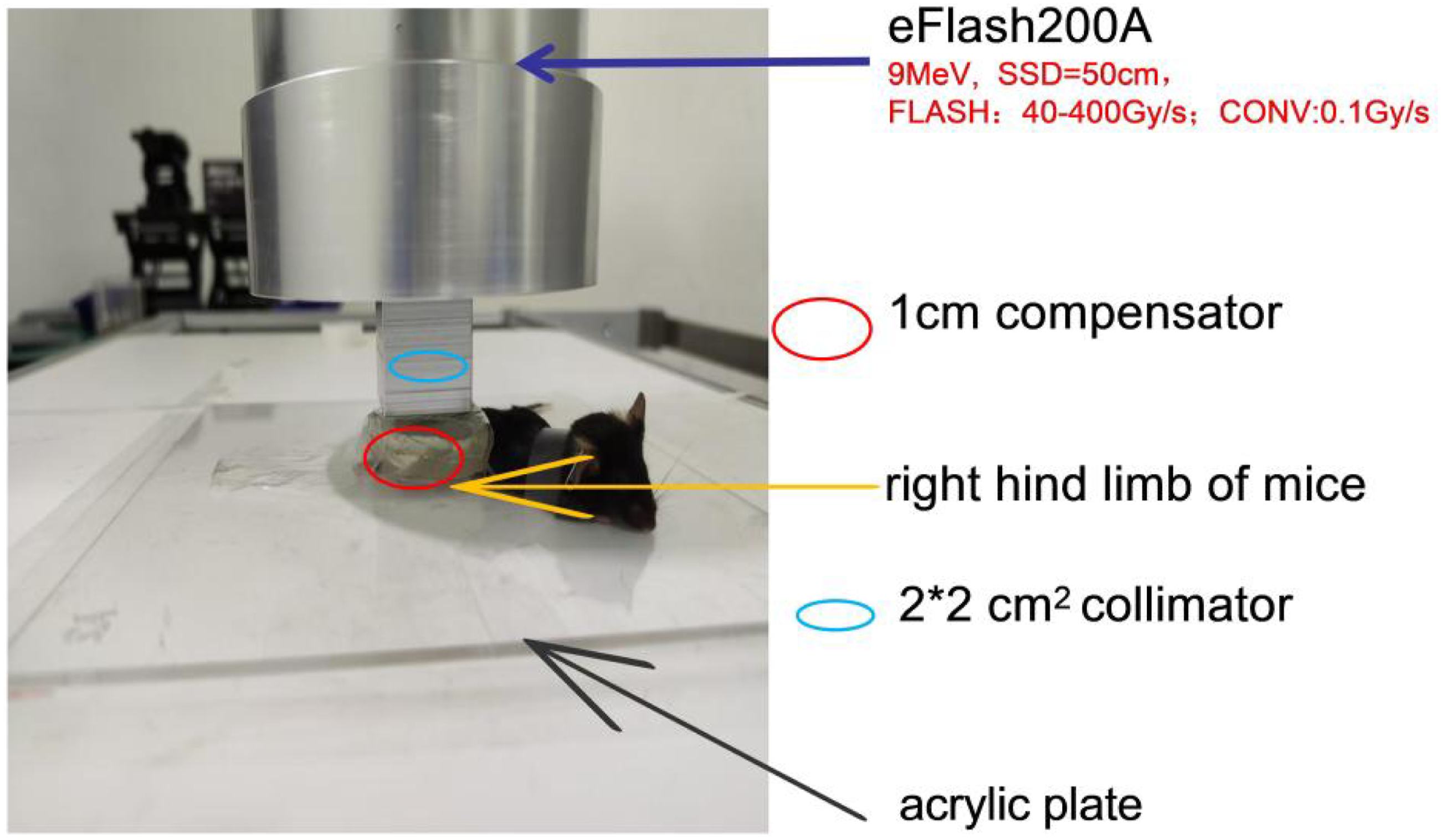
Irradiation Setup and Procedure. The Right Hind Limb (Covered by a 1 cm Thick Tissue Compensator) of an Anesthetized Mouse was Fixed on an Acrylic Plate, and a 2 × 2 cm2 Irradiation Area was Exposed at a Fixed Source-To-Skin Distance (SSD, 50 cm). The Mice Were Irradiated at Different Doses or Dose Rates according to the Experimental Design
B16/F10 Cell Subcutaneous Transplantation Model
B16/F10 cells were maintained in high-glucose DMEM (Gibco) supplemented with 10% FBS (Corning). At 80% confluence, the cells were trypsinized, resuspended in ice-cold sterile phosphate buffered saline (PBS) at 5 × 106 cells/mL, and inoculated into the right hind limbs of the C57BL/6J mice (5 × 105 cells per mouse). The tumors were allowed to grow until they reached a basal diameter or a volume (about 5 mm, 80-100 mm3). The mice that reached the predefined humane endpoints or experimental endpoints were euthanized, and tumor tissues were collected for hematoxylin and eosin (H&E) staining or Masson’s trichrome staining analysis.
Dosimetry and Irradiation Parameters
For dose-effect study, eight-to-nine-week-old mice were irradiated with single-fraction doses of 18-24 Gy. For dose-rate investigations, a single-fraction dose of 20 Gy was used. Doses were delivered via FLASH-RT (40-400 Gy/s) or CONV-RT (0.1 Gy/s), with dose rates adjusted via pulse repetition frequency or dose per pulse modulation. Prior to irradiation, dosimetric verification was conducted using Gafchromic EBT-3 films (Ashland Inc.), which were calibrated and placed between solid water templates in a mouse-mimicking phantom. The films were scanned and analyzed 5 minutes after irradiation, with the dose averaged over an 18 × 18 mm2 central region. No significant difference in dose distribution was observed between FLASH-RT and CONV-RT (Figure 2). Dose Distribution Profiles Across FLASH-RT and CONV-RT Irradiation Fields. (A) EBT3 of CONV-RT. (B) EBT3 of FLASH-RT. (C) Horizontal (X-Axis) Dose Distribution Curve. (D) Vertical (Y-Axis) Dose Distribution Curve
Scoring for Injury to Irradiated Skin Tissues
The skin of the right hind limb was evaluated for acute radiation-induced toxicity according to the Radiation Therapy Oncology Group (RTOG) scoring criteria. 18 Assessments were performed on the day of irradiation and every 3 days thereafter.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism V.10. Normally distributed data with homogeneous variance was analyzed using unpaired two-sided Student’s t-test, one-way ANOVA, or two-way ANOVA, whereas nonnormally distributed or unequal-variance data were analyzed with the Mann‒Whitney U test. For comparisons among 3 or more groups, ANOVA followed by Tukey’s post hoc test was applied. The data were expressed as the mean ± SD, and the criterion for significance was P < 0.05.
Results
FLASH Irradiation Mitigates Acute Skin Toxicity within a Defined Dose Range
To determine whether FLASH irradiation reduces radiation-induced skin toxicity and to determine its dose-dependent efficacy, the right hind limbs of C57BL/6 mice were irradiated with single-fraction doses ranging from 18 Gy to 24 Gy using either FLASH or conventional (CONV) dose rates. Compared with those observed in the FLASH-RT mice, significantly more severe and earlier-onset toxicities, such as erythema, alopecia, dry and moist desquamation, hemorrhage, full-thickness necrosis, edema, and ulceration, were observed in the CONV-RT mice. In general, the time for the acute skin injury to become visible to the naked eye after irradiation ranged from approximately 9-15 days; the acute skin injury peaked approximately 21 to 27 days after irradiation, and more skin lesions were unhealed on day 30 after irradiation in the CONV-RT group. Longitudinal assessment of acute skin injury in murine hindlimbs following a single 22 Gy irradiation revealed marked differences between FLASH-RT group and CONV-RT group. While both groups developed dry desquamation (green arrow) by day 15, the FLASH group exhibited a notably reduced affected area and lower incidence. By day 21, the injury in the CONV group had progressed to moist desquamation or ulceration, a transition that was not observed in the FLASH-RT group. At day 30, almost skin lesions had resolved in FLASH-irradiated limbs, whereas pronounced moist desquamation or ulceration (red circle) persisted in the CONV-RT group (Figure 3(A) and (B)). The RTOG acute skin injury scores indicated a significant protective effect of FLASH-RT at doses ≤22 Gy (maximum difference: 18 Gy, 0.00 ± 0.00 vs 2.00 ± 1.67; 20 Gy, 0.67 ± 1.03 vs 2.83 ± 0.75; 22 Gy, 0.17 ± 0.41 vs 3.00 ± 1.26; all P < 0.001; Figure 3(C)–(E)), with sustained reductions in injury scores relative to those of CONV-RT. In contrast, no significant radioprotective effect was observed at 24 Gy (maximum difference: 1.50 ± 1.22 vs 2.83 ± 1.33, P = 0.1066; Figure 3(F)). Combined with our preliminary findings, these findings revealed no significant difference in acute skin damage between FLASH-RT and CONV-RT at single doses of 25 Gy, 30 Gy, and 35 Gy (Figure S1). These results indicate that the FLASH effect provides robust protection against acute skin injury within a defined dose range (≤22 Gy), but this effect is nonexistent at higher doses (≥24 Gy), suggesting that an upper threshold dose below 24 Gy is needed to elicit the FLASH-sparing response. Acute Skin Injury after Single-Fraction Dose Irradiation With FLASH-RT Versus CONV-RT. (A-B) Representative Photographs of Irradiated Skin on Day 15, 21, and 30 after Irradiation (22 Gy; n = 6 per Group; Red Circle: Grade ≥2; Green Arrow: Grade = 1). Compared With CONV-RT, FLASH-RT Resulted in Milder Erythema, Desquamation, Ulceration and Improved Tissue Regeneration. (C–F) Acute Skin Injury Scores Over Time in 8-9-Week-old Mice (n = 6 per Group) Irradiated at the Indicated Doses (Two-Way ANOVA; Error bars: SD; ns: P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001)
Higher Dose Rates Enhance FLASH-Mediated Skin Protection
To determine the dose-rate dependency of the FLASH effect, the right hind limbs of male C57BL/6 mice were irradiated with a single-fraction dose of 20 Gy at average dose rates ranging from 40 to 400 Gy/s, modulated via pulse repetition frequency. Comparative analysis of skin reactions at different dose rates revealed that by day 15 post-irradiation, dry desquamation (green arrows) was observed in the CONV and low-dose-rate (≤200 Gy/s) FLASH groups, but not in the higher dose-rate FLASH-irradiated mice. By day 21, more severe skin lesions, including moist desquamation and ulceration (red circle), developed in the CONV-RT and the lower dose-rate (≤200 Gy/s) FLASH-RT groups. Importantly, within the FLASH regimen, the incidence and severity of these skin injuries exhibited a clear inverse correlation with the dose rate (Figure 4(A)). Then, we also observed that both CONV-RT (0.1 Gy/s) and FLASH-RT (≤200 Gy/s) induced persistent skin lesions and incomplete recovery on day 30. In contrast, the mice that were irradiated at FLASH-RT rates ≥300 Gy/s presented only mild, transient damage (grade ≤1), with most lesions fully resolved. Quantitative analysis confirmed that compared with CONV-RT, dose rates ≤200 Gy/s did not significantly reduce skin injury (maximum difference: 40 Gy/s, 0.79 ± 0.91 vs 1.57 ± 0.79; 100 Gy/s, 0.93 ± 1.35 vs 1.79 ± 1.52; 200 Gy/s, 0.50 ± 0.76 vs 1.57 ± 0.79; all P > 0.05; Figure 4(B)–(D)). In contrast, significant radioprotection was achieved at ≥300 Gy/s (maximum difference: 300 Gy/s, 0.00 ± 0.00 vs 1.79 ± 1.52; 400 Gy/s, 0.00 ± 0.00 vs 1.79 ± 1.52; all P < 0.001; Figure 4(E) and (F)), with no additional benefit between 300 and 400 Gy/s (maximum difference: 0.64 ± 0.38 vs 0.36 ± 0.38, P > 0.05; Figure 4(G)). Although 250 Gy/s also conferred significant protection compared with CONV-RT, it was inferior to 300 Gy/s at later time points (1.33 ± 1.75 vs 0.14 ± 0.24, P = 0.0296 on day 24). Collectively, these results indicate that a dose rate exceeding 200 Gy/s is necessary to initiate the FLASH skin-sparing effect, while rate exceeding 250 Gy/s is required to achieve maximal protection. Dose-Rate Dependency of FLASH-RT-Mediated Skin Protection at 20 Gy. (A) Representative Skin Images on Days 15 and 21 after Irradiation From Each Group, including CONV-RT (0.1 Gy/s) and FLASH-RT (40-400 Gy/s), Red Circle: Grade ≥2; Green Arrow: Grade = 1. (B–G) Acute Skin Injury Scores Across Time for Mice (n = 6, 8-9 weeks Old) Irradiated at Varying Average Dose Rates (Two-Way ANOVA; Error bars: SD; ns: P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001)
FLASH-RT Preserves Tumor Control while Reducing Skin Toxicity and Pulmonary Metastasis
On the basis of the results of the dose and dose-rate optimization study, we further investigated the antitumor efficacy and normal tissue toxicity of FLASH-RT at a single-fraction dose of 20 Gy. Additionally, the efficacy and safety of FLASH-RT vs CONV-RT were evaluated in a reirradiation setting, where a single-fraction 20 Gy dose was delivered following a priming hypofractionated course (CONV-RT, 9 Gy × 4). The results revealed that compared with no irradiation, both FLASH-RT and CONV-RT effectively suppressed tumor growth (P < 0.05), with no significant differences in tumor volume (Figure 5(A) and (B)) or weight (Figure 5(C)) observed between the 2 irradiated groups (tumor weight: FLASH-RT vs CONV-RT vs Control = 1.96 g ± 1.09 g vs 1.85 g ± 1.09 g vs 3.67 g ± 1.00 g). Notably, the number of lung nodules was lower in the FLASH-RT group than in the CONV-RT group (average: 0.33 ± 0.52 vs 2.00 ± 3.63; Figure 5(D) and (E)). Moreover, compared with nonirradiated mice, mice treated with CONV-RT exhibited less body weight loss (maximum difference: 25.3 g ± 1.64 g vs 26.93 g ± 3.13 g; Figure 5(F)). Thus, FLASH-RT achieved tumor control comparable to that of CONV-RT while potentially reducing systemic toxicity. Antitumor Response and Lung Metastasis Following Single 20-Gy Irradiation. (A) Postirradiation Growth Curve. (B) Representative Images of Subcutaneous B16F10 Tumors From the Nonirradiated (Control), CONV-RT, and FLASH-RT Groups. (C) Tumor Weights on Day 10 after Irradiation. (D). Representative Images of Lung Metastasis Nodules From the CONV-RT and FLASH-RT Groups. (E) Number of Lung Metastasis Nodules in the CONV-RT and FLASH-RT Groups. (F) Postirradiation Body Weight Curves. (A, F) Analysis by Two-Way ANOVA; (C) Analysis by One-Way ANOVA. (E) Analysis by an Unpaired t Test. All Values are Presented as Mean ± Standard Deviation (SD). n = 6 per group, ns: P > 0.05,*P < 0.05
In addition, severe acute skin injury (grade ≥(2) was observed exclusively in the mice that received CONV-CONV irradiation in reirradiation investigation. In contrast, the animals treated with CONV-FLASH exhibited only mild toxicity, such as dry desquamation or alopecia. Skin injury scores in the FLASH reirradiation group were significantly lower than those in the CONV reirradiation cohort (maximum difference: CONV-FLASH vs CONV-CONV vs CONV = 1.00 ± 0.00 vs 2.17 ± 0.75 vs 1.00 ± 0.00, P < 0.001; Figure 6(A) and (B)). No significant differences in tumor volume or tumor mass were observed between the treatment groups (Figure 6(C) and (D)). Moreover, the number of lung nodules was significantly lower in both reirradiation groups than in the nonreirradiated CONV-RT group. Furthermore, the CONV-FLASH group had the fewest cases of metastases among all the irradiated cohorts (CONV vs CONV-CONV vs CONV-FLASH = 7.67 ± 9.61 vs 5.00 ± 7.97 vs 2.00 ± 3.63, P < 0.001; Figure 6(E) and (F)). Notably, compared with nonreirradiated mice, mice treated with CONV-RT exhibited significant body weight loss (26.54 g ± 2.51 g vs 30.44 g ± 2.82 g, P < 0.05; Figure 6(G)), whereas FLASH-RT resulted in less weight loss, indicating improved systemic tolerance. Collectively, these results indicated that FLASH-RT not only maintained tumor control efficacy and normal tissue integrity upon reirradiation but also significantly suppressed distant metastasis and significantly reduced systemic toxicity. Normal Tissue Toxicity and Tumor Response after Reirradiation With FLASH-RT Versus CONV-RT. (A) Representative Skin and Tumor Images after Hypofractionated Radiotherapy (9 Gy x 4) and Reirradiation (With CONV-RT or FLASH-RT Following Reirradiation after Prior Hypofractionated Radiotherapy); Red Circle: Tumor; Green Arrow: Grade = 1; Orange Arrow: Grade ≥2. (B–D) Curves of Acute Skin Injury Score, Tumor Volume, and Tumor Weight after Reirradiation. (E) Representative Images of Lung Metastases. (F) Number of Lung Metastasis Nodules per Group. (G) Curves of Body Weight after Reirradiation With CONV-RT or FLASH-RT. (B, C, G). Data Represent Mean ± SD (n = 6 per Group). Analysis by Two-Way ANOVA; (D) Analysis by an Unpaired t Test; (F) Results Were Analyzed by One-Way ANOVA; Values are Presented as Mean ± Standard Deviation (SD), n = 6 per Group. ns > 0.05.*P < 0.05, *P < 0.001
Discussion
The dose and dose-rate thresholds required to elicit the FLASH effect appear to vary across studies. A growing body of evidence suggests that these parameters are influenced by factors such as radiation quality, beam energy, tissue type, and experimental model.19-23 For instance, FLASH effects observed at lower average dose rates often involve high-energy particle beams (eg, protons), which exhibit distinct physical dose deposition profiles. 19 Conversely, studies revealing higher dose thresholds often included elevated average dose rates. 11 Moreover, tissue-specific sensitivity plays an important role: Lung tissue has been shown to respond at dose rates as low as 40 Gy/s. 24 Whereas intestinal and brain tissues require 100 Gy/s or higher to manifest significant sparing.25,26 Notably, Ruan et al 25 reported a minimum average dose rate of 280 Gy/s for FLASH-mediated protection against acute intestinal injury, which aligns with our finding that a dose rate exceeding 250 Gy/s is necessary to maximize skin sparing. These studies underscore the need for organ-specific parameter optimization in future FLASH studies. Here, the results from our study primarily delineate 3 interlinked thresholds for the FLASH effect in murine skin: (1) A single-fraction dose constraint of less than 24 Gy is necessary to initiate the protective phenomenon. (2) The FLASH normal skin sparing effect begins at dose rates exceeding 200 Gy/s. (3) For maximal radioprotection to be achieved, a dose rate exceeding 250 Gy/s is mandatory. This parameter range defines the optimal window for eliciting the most pronounced skin-sparing effect in FLASH radiotherapy (mouse animal experiment). Moreover, FLASH-RT resulted in antitumor efficacy comparable to that of CONV-RT while reducing skin toxicity and suppressing pulmonary metastasis, particularly in a reirradiation setting.
Further, our research team further investigated the differences in inflammatory factor induction and cell death patterns between FLASH-RT and CONV-RT, and found that, consistent with previous studies, FLASH-RT induces fewer inflammatory factors, apoptosis, and ferroptosis. Additionally, FLASH-RT exhibits differential autophagy induction intensity. Elucidating the mechanism of FLASH effect is crucial for the clinical translation of FLASH-RT. None of the existing mechanistic hypotheses in the literature can independently explain the FLASH effect. 27 Based on the study and summary of the mechanism of FLASH-RT and autophagy, we hypothesize that autophagy serves as a central regulatory node mediating this differential effect. The unique radiobiological environment created by FLASH-RT—characterized by transient ultra-high dose rate delivery leading to rapid oxygen depletion and a sharp but brief burst of reactive oxygen species (ROS) — may act as a “precisely dosed stress signal” that preferentially activates a protective autophagic response in normal tissues, avoiding autophagic flux collapse or destructive autophagy seen under certain stress conditions. 28 This optimally tuned autophagic activity is postulated to underpin the tissue-sparing effect through interconnected mechanisms: (1) It maintains redox homeostasis by selectively removing damaged organelles (eg, via mitophagy), limiting persistent ROS generation, and potentially stabilizing key anti-ferroptotic proteins like GPX4, thereby protecting against radiation-induced ferroptosis. (2) It promotes genomic stability by facilitating efficient DNA damage repair, possibly through clearing nuclear debris and providing metabolic precursors essential for repair complex function. (3) It preserves stem cell function and tissue regenerative capacity by mitigating oxidative stress and regulating inflammatory signaling (eg, reducing expression of pro-inflammatory factors like TGF-β), thereby maintaining the stemness and differentiation potential of tissue stem cells, which is crucial for long-term tissue recovery. 29 Notably, studies suggest cancer stem cells (CSCs), known for their radioresistance, exhibit higher basal levels of lysosome-mediated autophagy, which may contribute to their survival even under FLASH-RT. 30
In contrast, pre-existing conditions in tumor tissues, such as baseline metabolic stress, hypoxia, or genetic alterations in autophagy pathways, may render them unable to mount a similarly coordinated protective autophagic response upon FLASH-RT stimulation. Alternatively, the intense ROS burst might trigger excessive or dysregulated autophagy, leading to autophagic cell death and enhanced antitumor efficacy. 28 This differential modulation of autophagy—cytoprotective in normal tissues vs cytotoxic or non-protective in tumors—likely constitutes a fundamental mechanism underlying the widened therapeutic window of FLASH-RT. Future research should focus on identifying key molecular switches within this autophagic network and exploring combination strategies employing autophagy modulators (eg, inhibitors for tumor sensitization or careful activation for enhanced normal tissue protection) to further optimize the therapeutic index of FLASH-RT.
Several limitations of this study should be acknowledged. This study provides preclinical evidence supporting the concept of FLASH skin-sparing effect; however, the direct clinical extrapolation of the reported dose and dose-rate parameters is not warranted. Their applicability must be established through future studies in advanced models that bridge the translational gap, specifically addressing the interspecies differences in radiation response. Then, a formal a priori power analysis to determine the required sample size was not performed. All sample sizes were chosen based on common laboratory practice and precedent in the literature. 17 While our results showed statistical significance for key outcomes, the study may be underpowered to detect smaller, yet biologically relevant, effects or may be overpowered for large effects. For instance, the sample size may have constrained the statistical power of the subgroup analyses (particularly regarding the re-irradiation cohorts). Additionally, the focus on acute toxicity and the lack of long-term survival data limit insight into late radiation effects in this study. Future work should benefit from a preliminary power analysis to optimize animal use, ensure robust statistical conclusions, incorporate larger cohorts, investigate the late effects, add beam qualities or fractionation schemes. Transcriptomic approaches (eg, mRNA sequencing) would be employed to elucidate the molecular mechanisms (eg, oxidative stress, immune microenvironment and stem cell preservation as the key direction) underlying the FLASH effect. Extended follow-up studies will also be essential for evaluating late toxicity and normal tissue complication probabilities.
Conclusion
Our results demonstrate that the FLASH-RT-mediated tissue-sparing effect requires optimized single-fraction dose and average dose rate parameters, where FLASH-RT maintained tumor control equivalent to that of CONV-RT in both the primary and reirradiation scenarios. Hence, these findings refined the radiobiological characterization of electron FLASH-RT and supported its accelerated clinical translation, especially in cases requiring repeated irradiation.
Supplemental Material
Supplemental Material - Towards Clinical Translation: Establishing Optimal Dose and Dose-Rate Parameters for the FLASH Skin-Sparing Effect
Supplemental Material for Towards Clinical Translation: Establishing Optimal Dose and Dose-Rate Parameters for the FLASH Skin-Sparing Effect by Yufeng Shen, Jie Zhou, Jinyi Lang, Lintao Li, Yongjie Li, Chenxi Yang, Shuo Wang, Xiaohua Chen, Siyu Xie, Wei Tang, Shun Lu in Dose-Response
Footnotes
Acknowledgments
The authors would like to extend their sincere appreciation to Zhongjiu Flash Medical Technology Co., Ltd., for providing the irradiation equipment and to Applied Electronics of the China Academy of Engineering Physics (CAEP) for irradiation guidance.
Author Contributions
Conceptualization, methodology, and formal analysis: Shun Lu, Jinyi Lang, Jie Zhou, Lintao Li, and Yongjie Li. Investigation: Yufeng Shen. Statistical analysis: Yufeng Shen, Chenxi Yang, Shuo Wang, Wei Tang, and Siyu Xie. Resources: Shun Lu, Jinyi Lang. Writing—original draft preparation: Yufeng Shen. Writing—review and editing: Shun Lu, Jie Zhou, and Xiaohua Chen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation Project of Science and Technology Department of Sichuan Province; No. 2025ZNSFSC0787, the Chengdu Science and Technology Projects; No. 2024-YF05-02201-SN, Sichuan Science and Technology Program; NO:2024ZDZX0027.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
