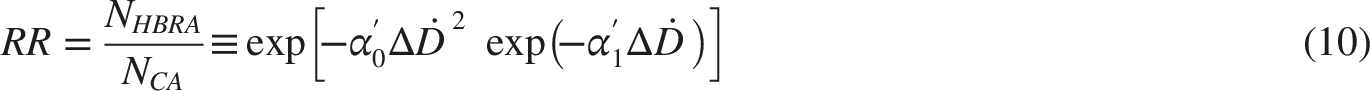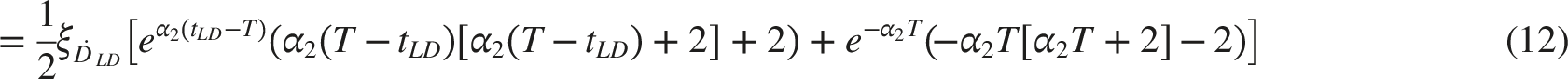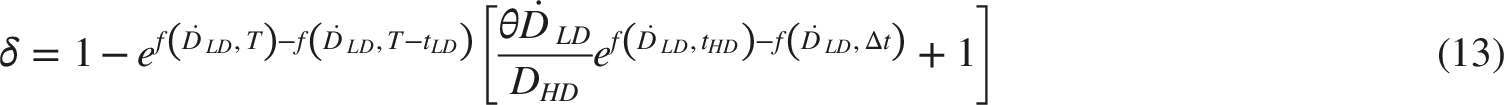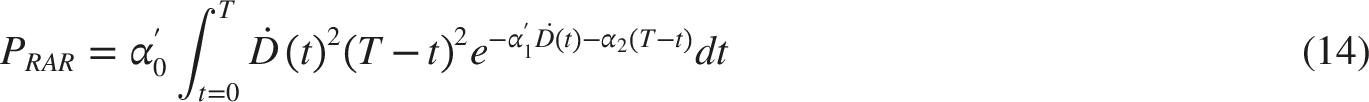Abstract
Objectives
The radiation-induced adaptive response (RAR), also referred to as radioadaptation, describes modifications of biological radiation sensitivity following prior exposure to low doses or low dose-rates of ionizing radiation. Despite extensive experimental evidence, RAR remains difficult to reproduce consistently and lacks a unified quantitative and methodological framework. The objective of this study was to develop a systematic biophysical approach enabling coherent analysis and comparison of RAR experiments performed under different irradiation protocols.
Methods
We formulated a dose- and time-dependent adaptive response function characterized by transient, memory-like dynamics. On this basis, we derived analytical expressions describing RAR under multiple irradiation schemes, including priming–challenge protocols, radiation training, constant low dose-rate exposure, and variable dose-rate scenarios. A unified relative endpoint parameter was introduced to quantify the magnitude of the adaptive response across experimental designs.
Results
The proposed framework yields explicit expressions for the adaptive response parameter under diverse exposure conditions and demonstrates how RAR magnitude depends on dose, dose-rate, and time interval between exposures. The methodology enables consistent normalization of experimental endpoints, facilitates parameter estimation from empirical data, and clarifies conditions under which adaptive effects are expected to emerge or vanish.
Conclusion
This work provides a coherent and transferable methodological foundation for quantitative RAR research. The framework improves comparability between experimental studies and supports mechanistic interpretation of low dose adaptive effects, while remaining primarily applicable to controlled experimental systems rather than population-level radiation risk assessment.

1. Introduction
Every living system is characterized by its ability to adapt to external factors. This adaptive capacity is one of the key determinants of an organism’s survival, energy-processing efficiency, and reproductive potential. Even if we generalize the concept of a living system to a thermodynamic system far from equilibrium, it becomes evident that any perturbation from a steady state triggers mechanisms aimed at restoring the original condition.
From a physical standpoint, adaptation is described in terms of two mechanisms: activity and memory.1,2 The first, activity, represents an immediate response - an abrupt change in the system’s state under the influence of an external stressor (e.g., detecting a virus entering the organism and initiating antibody production). The second, memory, depends on past events that shape how the organism reacts to a new external stimulus. The efficiency of this memory-type response determines how effectively the organism can mitigate the negative effects caused by external stressors. In the viral infection example, memory corresponds to antibodies produced and retained in the body - either from previous infections or vaccinations. In many cases, this memory response reaches its peak after some time and then decays exponentially.1,3,4 Another well-known case in which adaptation is formally divided into activity and memory components is sensory adaptation, a phenomenon well described within the framework of stochastic thermodynamics. 2
In biology, the term adaptive response (AR) is often restricted to the memory-type response, since this component is easier to observe under laboratory conditions given sufficiently long observation periods. For instance, behavioral or physiological adaptation in laboratory animals exposed to potentially toxic substances may only become evident after several hours or weeks of monitoring, typically through comparisons between exposed and control cohorts (as in epidemiological studies). 5
A particular case of adaptation is the Radiation Adaptive Response (RAR), also known as radioadaptation.6,7 It is characterized by the organism’s improved tolerance to ionizing radiation - meaning a better ability to cope with its harmful effects - if it has been pre-exposed to very low radiation doses. Although many radiobiologists remain skeptical about RAR due to its unpredictability and relatively weak magnitude, the phenomenon remains undeniably important, intriguing, and scientifically significant.5-8
The earliest experimental studies on radiation-induced chromosome damage and repair began in the 1930s with Karl Sax, who showed that radiation effects depend on both dose and time, and that cells can recover after low exposures. 9 During the Manhattan Project, John Raper discovered that animals pre-exposed to a sublethal dose became more resistant to a subsequent lethal dose - a phenomenon termed “recovery from radiation damage” 10 and later confirmed by Eugene Cronkite and Colleagues. 11 Although early research lacked mechanistic understanding, later studies by Pohl-Rüling and Fisher 12 demonstrated that low dose radiation could induce repair enzymes, suggesting endogenous protective mechanisms. In the 1980s, the concept of “adaptive response” was formally introduced by Gregorio Olivieri and colleagues, 13 while Morio Yonezawa’s experiments in Japan established the “priming dose effect” or “Raper–Yonezawa effect”. 14 The United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) summarized the phenomenon in its 1994 and 2020/2021 reports.8,15 Although research activity declined after 2000, recent years have seen renewed interest,5,16 focusing on the relationship between adaptive response and radiosensitivity under diverse irradiation schemes. 17
Today, many research teams are once again trying to solve the key problem of the RAR: the ability (or failure) to consistently induce this effect. Experimental studies have long faced challenges with reproducibility, as results often vary depending on the biological model, radiation quality, dose, and timing between priming and challenge exposures.5-7 Some experiments clearly show protective effects of low dose pre-irradiation, while others fail to reproduce them, raising doubts about the robustness of the phenomenon. These inconsistencies may stem from cell-type specificity, dose–time dependencies, and differences in experimental conditions. Translating laboratory findings to human health contexts remains uncertain, as epidemiological data are inconclusive. Moreover, RAR may challenge the assumptions of the linear no-threshold (LNT) model that underpins radiation protection, creating controversies in risk assessment and therapeutic applications, especially given the possibility that tumor cells might also adapt in ways that promote survival. 18
Mechanistically, the radiation adaptive response involves multiple interlinked biological pathways that enhance cellular resilience. Low dose or low dose-rate exposure can stimulate DNA repair systems, trigger transient cell cycle arrest, boost antioxidant defenses, and induce protective gene expression profiles. Additional processes include improved detoxification of free radicals, enhanced immune and inflammatory responses, and induction of apoptosis to eliminate damaged cells. Together, these mechanisms represent a coordinated, multi-level adaptation aimed at maintaining genomic stability and cellular homeostasis. 5 However, the precise molecular trigger initiating RAR remains elusive, underscoring that the phenomenon, though well-documented, is still not fully understood and requires further mechanistic clarification.
Despite many decades of research on the RAR effect, there is still no consistent methodology for conducting such studies. Although the Raper–Yonezawa (priming dose) scheme remains the most commonly used experimental approach, it lacks coherence due to its inherent model limitations. The RAR effect can be investigated through various strategies, including priming/challenging dose experiments, correlations with non-radiation stressors (e.g., chemicals), consideration of individual radiosensitivity, and adaptation to low constant dose-rates. The purpose of this article is to propose a more unified methodology for RAR research, suggest standardized experimental frameworks, and discuss different approaches for studying this phenomenon. We hope that these efforts will help advance research on this intriguing effect and clarify the precise mechanisms responsible for its unequivocal induction.
2. Methods
2.1. Memory Effects of RAR
As described in the previous chapter, the key to understanding adaptive mechanisms in biological (or more broadly, biophysical) systems lies in the concept of memory mechanisms. 1 Prior conditioning of a system increases its adaptive capacity at later times, although such adaptation is not always permanent and often decreases with time. In the case of the RAR, the factor that triggers memory mechanisms is a low dose (or low dose-rate) of ionizing radiation. The memory mechanisms themselves include DNA repair enzymes (or, more generally, enhanced DNA repair efficiency), increased antioxidant and anti-inflammatory responses, and effective regulation of the cell cycle and apoptosis as a means of eliminating mutated cells. There is, therefore, no single homogeneous memory mechanism responsible for RAR - what we call the adaptive response is the emergent outcome of many interacting processes whose spatiotemporal superposition is extremely difficult to predict. 5 This is likely the central problem associated with RAR - the challenge of predicting when and under what conditions the effect will or will not occur. A proper understanding of these interrelations - given the laws governing complex systems - appears very difficult, if not impossible. Thus, such systems must be studied holistically, by analyzing their overall behavior and dynamics as functions of individual characteristics, time, and radiation dose distribution (emergence). 17
We can attempt to describe the memory mechanism mathematically, for example through an effective repair function, denoted as f repair . 19 This function fundamentally depends on three factors: individual susceptibility (commonly referred to as radiosensitivity), absorbed dose, and time. Individual radiosensitivity can, in principle, be accounted for by defining a unique repair function for each single cell / organism. However, when averaging across a population, an adaptive potential function must be introduced to describe the likelihood and magnitude that a given individual exhibits an adaptive response, expressed as <f repair > = p RAR (D,t) - a function of the radiation dose D received t time units earlier.
Thus, the dose- and time-dependent function p RAR (D,t) describes the adaptive potential per unit of time; in other words: this is the adaptive response signal that modulates biological repair dynamics, and quantifies the effectiveness of radiation-induced adaptation in a biological system. This function must be determined empirically, based on a sufficient number of experimental data points obtained for various doses and times. In general, p RAR (D,t) exhibits a hump-shaped (bell-shaped) curve: after the inducing factor appears, the effective memory of the system increases rapidly, reaches a maximum, and then exponentially decays as memory fades and the enhanced efficiency of DNA repair mechanisms diminishes over time. 4
The hump-shaped curve was first observed by John Raper during his radioadaptation experiments within the Manhattan Project. 10 The explicit dependence of this curve on both dose and time (initially as separate repair functions, one for D and one for t) was later introduced by Prof. Ludwig Feinendegen, the pioneer of theoretical descriptions of RAR. His works in the 1990s laid the foundation for the modern mechanistic understanding of the phenomenon.20,21 Since then, mathematics, modeling, and physics have become integral parts of RAR research - an evolution not always fully appreciated within traditional biology.
In general, p
RAR
(D,t) can be represented as a combination of activation functions A (increasing with dose/time) and relaxation functions R (decreasing with dose/time):
The activation function, for any variable ε, can be expressed as an increasing polynomial of degree n, A(ε)=W
n
(ε), in which the dominant term is the highest-order component, hence A(ε)∼ε
n
. Of course, this polynomial term is used as a phenomenological approximation capturing the initial activation of adaptive mechanisms prior to saturation. Next, the relaxation function typically follows an exponential decay, R(ε)=exp(−λε). Hence, Eq. (1) can be rewritten as3,4:
Equation (2) represents the classical hump-shaped curve, consistent with the empirical forms proposed by Feinendegen, and confirmed analytically by Kino.22,23 The free parameters (all positive) may take arbitrary values depending on the organism, tissue, or cell type under investigation (the model describes the adaptation of a “system” regardless of what that “system” is). Mathematically, one may also impose boundary conditions such as p
RAR
(ε)=0 for ε=0 or ε→∞. Based on deeper analysis and including the Kellerer–Rossi model, Fornalski reformulated the function into the specific form with
The function in Eq. (3) thus has three free parameters {α} reflecting the individual radiosensitivity of the system and depends on two variables: the radiation dose D received t time units earlier. In other words, Eq. (3) defines the chance that the analyzed system exhibits an adaptive response if it was exposed to a single dose D of radiation t time units before observation.
Please note that such the polynomial–exponential form adopted for the adaptive-response function provides a phenomenological yet analytically convenient approximation to saturating biological kinetics, such as Michaelis–Menten or Hill-type responses.25,26 For moderate dose and time ranges, this functional form reproduces key qualitative features of sigmoidal activation, including threshold-like onset, a well-defined maximum, and effective saturation. Unlike explicit Hill formulations, the chosen representation allows closed-form analytical treatment of transient behavior and cumulative effects under variable irradiation schemes.
Figure 1 shows the temporal shape of the curve defined by Eq. (3) for the adaptive response of human peripheral blood lymphocytes irradiated in vitro with X-rays for various inducing doses D. Notably, the function reaches its maximum values for the optimal parameters D
max
= 2/α
1
and t
max
= 2/α
2
.
24
Potential functions of the radiation adaptive response as a function of time for three initial doses DLD of 25 mGy (upper curve), 10 mGy (middle curve) and 100 mGy (lower curve), for RAR parameters obtained for human lymphocytes as 
The hump-shaped form of Eq. (3) defines the dose- and time-dependent adaptive response signal, quantifying the effectiveness and temporal persistence (memory) of the RAR. It also represents an effective repair function for radiation-induced damage, a concept that will be discussed in further detail later.
2.2. Experimental Schemes
There are many possible experimental approaches to study the radiation adaptive response. They include the priming dose effect (also known as the Raper–Yonezawa effect), the combined effect (involving non-ionizing stressors in addition to ionizing radiation), radiation training (where the system is exposed to more than two fractions of low radiation doses), constant dose-rate exposure (irradiation under a stable low radiation field), and the variable scheme, in which the dose-rate changes over time.
As for the analyzed biological end-point, the range of options is extensive. The most frequently examined parameters include cell survival curves, mutation frequency, chromosomal aberration rates, and standard radiobiological assays, such as the comet assay, which assesses the time-dependent dynamics of DNA repair. In epidemiological studies, researchers typically analyze radiation risk, expressed in terms of cancer incidence or mortality rates. Please note that - from the perspective of model applicability - the specific biological endpoint is not arbitrary: the model parameters {α} must be calibrated within the chosen experimental framework and interpreted consistently with that endpoint. Each parameter set {α} is therefore intrinsically endpoint-specific and should not be transferred across different biological endpoints without re-calibration.
Let us begin, however, with a discussion of the irradiation schemes applied to biological systems, focusing on their methodological relevance for observing the RAR phenomenon.
2.2.1. Raper-Yonezawa Scheme (Priming Dose Effect)
In the past, this scheme - originally called the priming dose effect - was often treated by many researchers as synonymous with the RAR as a whole. This was due to the fact that the Raper–Yonezawa scheme is the most effective and most commonly used irradiation protocol among radiobiologists. 8 Today, however, it is clear that the concept of RAR is broader, 5 and the Raper–Yonezawa (priming dose effect) represents only a special case of radioadaptation. 24
The main idea of the Raper–Yonezawa scheme is to examine a selected biological end-point by irradiating two subgroups of biological samples. One group receives only a high dose (the so-called challenging dose, denoted D
HD
), while the other group is first irradiated with a low dose (the priming dose, denoted D
LD
) and then, after a defined time interval Δt, with the same high dose (D
HD
) as the first group. In this way, we obtain two comparable groups exposed to the same high dose, but only one of them has been pre-exposed to a small priming dose. By comparing the measured values of the biological end-point N between both groups, we can quantify the relative adaptive response as
24
:
The parameter delta δ defined in Eq. (4) thus provides a useful, objective, and universal measure of the occurrence and magnitude of RAR. It was originally proposed in 24 and later applied in several other RAR studies.27,28
In practice, experimental setups often also include a control group (not irradiated), and sometimes an additional group exposed only to the D LD dose. As mentioned earlier, the measured biological end-point N may represent virtually any parameter - most commonly, however, it is the frequency of radiation-induced mutations in cells. Naturally, both the method of measuring the end-point N and the entire experimental protocol must remain identical for all subgroups (D HD only, D LD +D HD , control, and possibly D LD only). Importantly, all subgroups should originate from the same colony or culture, to eliminate potential bias due to differences in intrinsic radiosensitivity. Likewise, environmental factors - such as feeding, hydration, or light exposure - must be kept identical across all groups, so that ionizing radiation remains the only variable factor.
Once the end-point values N have been measured for each group, the most convenient way to present RAR results is a bar chart (Figure 2). This graphical representation is widely used in RAR studies because it allows for a straightforward estimation of the δ parameter (Eq. (4)). Typically, the x-axis represents the dose, while the y-axis corresponds to the measured biological end-point (e.g., mutation frequency). The first bar usually corresponds to the D
LD
group; the second, significantly higher bar, to the D
HD
group; and the third, to the combined D
LD
+D
HD
exposure, in which the sample is irradiated first with D
LD
and, after a time interval Δt, with D
HD
. If RAR occurs, the third bar is lower than the second, indicating a positive value of δ (Eq. (4)). A schematic representation of this result presentation is shown in Figure 2. It is also assumed here that both doses are delivered as instantaneous pulses, meaning that the irradiation time is negligible compared with the time interval Δt between them. Example of the radiation-induced adaptive response (RAR) in the priming dose scheme (Raper–Yonezawa effect). A given living organism is divided into at least two colonies (lower plot). The first colony (scenario 1) is irradiated only with a high challenging dose (DHD), while the second colony (scenario 2) is irradiated with a combination of a low priming dose (DLD) followed, after a certain time interval, by the same high challenging dose (DHD). If RAR occurs, the frequency of the biological end-point N in the second scenario is lower than in the first scenario. This effect is described by the parameter delta (δ), which represents the relative difference between the N values obtained for both scenarios (upper plot)
In summary, at this stage, the essential parameter for quantifying the RAR level is the δ parameter, representing the relative difference between the number of end-points for a single D HD dose and the combined D LD +D HD doses (see Figure 2 and Eq. (4)). Naturally, the value of δ depends on both doses and on the time between them, δ→δ(D LD , D HD , Δt), and its maximum value (i.e., strongest RAR) occurs at D max = 2/α 1 and t max = 2/α 2 , as derived in the previous section.
To accurately determine the potential function p RAR (Eq. (3)), we must experimentally measure many δ values for different doses and time intervals. This will allow us to obtain the exact functional form of p RAR , and consequently, the parameters {α} characterizing the system’s susceptibility to RAR induction.
There are various ways to determine the parameters {α} from δ values. For a small number of data points, the problem can be solved analytically, but in most cases, numerical methods are used (see Appendix 2 in 24). The detailed procedure for estimating {α} from δ can be derived from Eqs. (3) and (4), assuming that the end-points N undergo effective repair dynamics governed by p
RAR
, expressed by the differential equation dN = −N p
RAR
dt. The current value of p
RAR
at the moment of applying the high dose D
HD
is determined solely by the value of the low dose D
LD
(Eq. (3)). After some algebraic manipulations,
24
we obtain δ correlated with both doses and the time interval Δt:
2.2.2. Combined Effects (Different Stressors)
The Raper–Yonezawa scheme described above can also be implemented in a mixed-exposure design, in which one of the doses (either D LD or D HD ) corresponds to a non-ionizing stressor, such as a chemical agent. 5 The key issue, of course, is whether the adaptive response function for the additional stressor follows the same form as proposed in Eq. (3).
An analysis of numerous studies of this type5-7 indicates that, qualitatively, these responses are also well described by a bell-shaped curve. Therefore, the adaptive potential (control) function p RAR given by Eq. (3) can also be applied in such cases. Naturally, the parameter set {α} will differ between ionizing radiation and the secondary stressor. Nevertheless, the overall irradiation protocol and the analysis of the δ parameter remain identical to those in the standard Raper–Yonezawa setup. It is essential, however, to ensure that in Eq. (5), the parameters {α} correspond specifically to the D LD dose, meaning that if D LD represents a non-radiation stressor, the parameters {α} must be those characterizing that particular stressor.
2.2.3. Radiation Training
Radiation training is essentially the irradiation of a biological system (cells, tissues, or entire organisms) with a series of individual low dose pulses (fractions), typically delivered at regular time intervals. This dosing scheme is sometimes referred to as dose fractionation; however, that term is primarily reserved for radiotherapy procedures involving high or very high doses. Radiation training, by contrast, concerns only low dose exposures.29-32
A single dose is assumed to generate an independent adaptive-response signal described by Eq. (3), in case of multiple doses the total adaptive function is taken as the linear superposition of these contributions therefore at any given time, the overall potential of RAR represents the sum of all preceding signals (i.e., the system’s memory), which can be written simply as P RAR =∑p RAR . It is worth noting that in the linear regime any potential function in physics is additive; therefore, we apply this principle to the additivity of the function defined in Eq. (3) for different independent pulsed radiation doses.
In mathematical terms, this additive formulation represents the linear-response (first-order) approximation of a more general nonlinear adaptive dynamics, and is justified provided that the cumulative adaptive signal remains small compared with the intrinsic nonlinear saturation scale of the underlying cellular repair and signaling pathways, beyond which resource competition or feedback mechanisms would invalidate linear superposition.
Accordingly, the additive formulation should be understood as a minimal phenomenological description of cumulative adaptive memory, valid under conditions where inter-signal competition remains weak. The transient, bell-shaped form of the elementary adaptive signal ensures temporal decay and effective memory loss, but does not by itself introduce nonlinear coupling between distinct exposure events.
It should again be emphasized that the functional form of p RAR proposed in Eq. (3) is not unique for the radiation training; other formulations of the cumulative adaptive response have been proposed.29-32 Nevertheless, for the sake of consistency with previous sections, we continue to use the form given by Eq. (3).
To better illustrate the issue of additivity in the adaptive response function, let us extend the Raper–Yonezawa scheme by replacing the single priming dose D
LD
with two equal priming doses, D
LD1
and D
LD2
.
24
To calculate the value of the RAR function at the moment when the high dose D
HD
is delivered (assumed to occur at T=Δt+ΔT after the first irradiation), we can write: Potential function (left axis) of the radiation adaptive response, 
It becomes clear that the effectiveness of post-radiation damage repair - and thus the value of the δ parameter - is greater for a system exposed to two priming doses than for a single one. Naturally, this case can be generalized to multiple priming doses, which, after a certain point, will lead to saturation of the adaptive response function - this saturation effect is precisely what is referred to as radiation training (Figure 4).
Let us now assume that the system is irradiated with n successive low doses, D
LN,i
, each delivered at equal time intervals Δt. This means that, after a sufficiently long period, the system reaches a steady-state value of the adaptive response potential function, which is the sum of contributions from all individual doses
24
:
A practical implementation of the radiation training scheme in a potential clinical context was presented by Socol et al. 29 In this case, the parameter set {α} is identical for each individual dose, since the irradiation conditions are the same. It should also be emphasized that the duration of irradiation per single dose is negligibly short compared with the time interval Δt between doses.
If we decrease the time interval between doses (Δt→0), the system gradually transitions from pulsed-dose irradiation to continuous irradiation at a constant dose-rate. This represents a distinct RAR scheme, which will be discussed in the following section. Likewise, no explicit expression for the δ parameter is provided here, as its value coincides with that obtained for the constant dose-rate case.
2.2.4. Constant Dose-Rate Exposure
This case is a direct continuation of the previous scenario, namely radiation training, in which the biological system is irradiated with a continuous, constant low dose-rate
The approach to saturation follows a sigmoidal pattern and can be calculated analogously to the integral in Eq. (8). It should also be noted that the parameters {α} in Eq. (8) differ from those in Eq. (3), although they can be interconverted based on the saturation time (i.e., the time at which Eq. (8) reaches a constant value) and the change of dose into dose-rate. In practice, these parameters are typically determined experimentally by fitting the model to empirical data.3,24,33,34
Let us now consider an analogous situation to that shown in Figure 2, but this time a system is exposed for a sufficiently long time to a constant low dose-rate (i.e., one for which P RAR has reached the saturation level given by Eq. (8)) is subsequently subjected to an additional large dose D HD . The reference group in this case can be a cohort not exposed to elevated background radiation, i.e., the control area (CA).
From an experimental standpoint, it is practically impossible to find conditions where the background radiation is strictly zero, since a natural radiation background exists everywhere on Earth. Therefore, the only feasible approach is to determine the relative difference in biological endpoints between populations residing in High Background Radiation Areas (HBRA) and those living in control (CA) regions. This analysis assumes that the system is subjected to the same large dose D HD : once under HBRA conditions and once under CA conditions.
Although experimentally achievable, such a setup requires significantly greater effort and time than the classical Raper–Yonezawa scheme. Nevertheless, the calculation of the δ parameter proceeds analogously to that in Eq. (4) but with constant irradiation.
However, the dynamics of DNA repair differ in this case: while in the Raper–Yonezawa scheme the analysis relied solely on Eq. (3) to derive Eq. (5), here we deal with continuous irradiation, which modifies the formalism somewhat. This issue was discussed in 24 (see there Eq. (34) in 24). Based on Eq. (4), the new δ parameter can be expressed as:
Studies of the adaptive response under elevated constant dose-rate conditions also have epidemiological applications, particularly for populations residing in HBRA regions. In such analyses, the biological endpoints include cancer incidence, cancer mortality, or the frequency of chromosomal aberrations. 33
In these cases, however, the RAR is not tested experimentally via a high-dose challenge (D HD ), since we are dealing with human populations, not laboratory systems. Consequently, such studies are inherently more uncertain, yet several RAR models have been developed to describe these effects, showing a remarkable level of consistency.30-34
It is important to emphasize that the transition from cellular or tissue-level adaptive responses to epidemiological observations in human populations is not straightforward and should not be interpreted as a direct mechanistic mapping. The RAR function introduced in this work is primarily formulated to describe dose- and time-dependent modulation of biological endpoints at the cellular or sub-organismal level, where radiation exposure, temporal structure, and biological response can be relatively well controlled.
In the context of HBRAs, the proposed framework should therefore be understood as providing an effective, population-averaged description rather than a deterministic prediction of individual cancer risk. Epidemiological endpoints such as cancer incidence or mortality integrate a wide range of biological processes acting over long timescales, including cellular repair, tissue remodeling, immune surveillance, latency effects, genetic heterogeneity, and non-radiation-related environmental factors. The adaptive response signal considered here represents only one of several contributing mechanisms and does not account for all sources of variability present at the population level.
Consequently, parameters characterizing RAR at the cellular level cannot be transferred directly to epidemiological settings without reinterpretation. In HBRA studies, the adaptive response formalism should be viewed as a phenomenological tool that captures the net effect of long-term, low dose-rate exposure on population-level biological outcomes, rather than as a direct extrapolation of in vitro radiosensitivity parameters. Any comparison between model predictions and epidemiological data must therefore be interpreted cautiously and within the limits of this effective description.
Therefore, within the present model, the averaged differences in biological endpoints between HBRA and CA populations can be correlated with the steady-state probability of RAR occurrence, expressed through the relative risk (RR) commonly used in epidemiology
33
:
This equation, however, has significant limitations, such as only allowing modelling cases where adaptive response was observed, which are discussed in greater detail in Ref. 33.
2.2.5. Variable Scheme of Irradiation
All the cases described above are special cases: they describe specific, fixed irradiation schemes. In general, however, a system may be irradiated according to any protocol - for example, the Raper–Yonezawa scheme with the dose delivered not as a short pulse but spread out over time. One may also encounter variants with the low dose given continuously as D LD and the high dose as a pulse D HD , or vice versa. In any event, the procedure for calculating the δ parameter is analogous to those presented in the preceding sections. The key task is to write down correctly the dependence p RAR (or P RAR , if it is composed of more than one p RAR ) and the time-evolution function of the biological end-points, N(t).
Let us now consider the case in which the priming dose, D
LD
, is delivered over appreciable duration; denote the delivery time of the low dose by t
LD
and the moment of delivery of the short-pulse high dose by t
HD
. The first (low) dose causes a build-up of P
AR
according to a relation analogous to Eq. (8)
24
:
Whether or not the adaptive-response function has saturated, in our scheme the irradiation then stops for a period Δt. This means we must subsequently describe the decaying P
RAR
over the interval Δt measured from the end of t
LD
. In other words: the potential function of the adaptive response for irradiation at dose-rate
We now assume that at time T (measured from the start of the entire irradiation sequence, i.e., Δt after its end) a second irradiation with the large dose pulse D
HD
occurs. When the second irradiation takes place, the adaptive-response function is given by the sum of Eqs. (12) and (11). At the moment when irradiation finishes and biological end-point is measured (the moment of T > t
LD
+Δt = t
HD
), the δ parameter therefore takes the form:
It is, of course, impossible to list all possible irradiation schemes. In particular, substantial difficulties arise when a system is irradiated continuously with a time-varying dose-rate (
In this case, all calculations are most conveniently performed using numerical methods.
2.3. Practical Application
There are many possible practical applications of the model described above. Everything depends, of course, on what exactly we want to demonstrate and what input (experimental) data we have at our disposal. If our goal is to determine the RAR parameters (i.e., the set of α parameters) with high precision, then we naturally apply the given equations directly - most importantly, by evaluating different values of delta (δ) for various dose-and-time scenarios, we can accurately estimate the parameters {α}. However, this necessarily restricts us only to datasets that exhibit RAR (even if weak or statistically insignificant).
If, instead, we wish to examine the problem more broadly - for example, by investigating how RAR behaves in terms of effect reproducibility, its relative influence on an entire organism or on a population - then the theoretical framework described above must be embedded within a larger model, such as one based on Monte Carlo techniques or Markov chains. Such an approach may be applied, for instance, to analyze the impact of RAR on an organism (radiation risk), on radiotherapy procedures (potential priming of cancer cells), or on entire human populations (epidemiology). Selected areas of such practical applications are discussed below.
2.3.1. Monte Carlo Model of Cells Colony Irradiation
Let us assume that we have a colony of cells that we plan to study in terms of their behavior under exposure to ionizing radiation, in particular the radiation-induced adaptive response. Monte Carlo modeling enables the creation of a virtual in-silico laboratory, within which we can perform any analysis of such cellular behavior. The colony may be one-, two-, or three-dimensional, depending on our needs. Likewise, we can simulate cells of any type and any state (e.g., healthy or cancerous). Monte Carlo techniques make it possible to simulate essentially any process, provided that it is mathematically well-defined and calibrated against real experimental data.
The technical details of the Monte Carlo model under discussion were presented in Ref. 35 and will not be described here in depth. However, it is useful to mention a few fundamental elements - most importantly, that the cell colony is subject to two principal numerical loops: one over individual cells, and another over discrete time steps. As a result, at each time step every cell can traverse the full probability tree corresponding to its current state (possible states include: healthy, damaged, mutated, and cancerous). This probability tree describes each radiobiological process that a cell in a given state may undergo (e.g., a cell may divide, be killed, die naturally, transition to another state, generate an adaptive response signal, or - most commonly - remain unchanged). By repeating this procedure numerically over a very large number of iterations, we can simulate arbitrary radiobiological processes in any scheme and over any time interval determined by successive time steps. 35
Naturally, our main objective is to simulate RAR. With the Monte Carlo framework described above, this phenomenon can be embedded within a broader context of various intercellular interactions and radiobiological effects. Such an approach allows us to recover the actual scale of the phenomenon, which is generally quite small. The adaptive response is significant only within a narrow window of dose and time, because otherwise it is too weak to rise above the noise associated with natural fluctuations of other processes. 36 Thus, while this approach is fully justified and necessary for radiobiological studies or for estimating an individual organism’s response to radiation (with which RAR is undoubtedly associated), considering RAR from a population-level perspective - such as for the purposes of radiation risk assessment - appears to be an overly far-reaching application.
Regardless, the use of the RAR model in Monte Carlo simulation allows for its capture in a broader context, e.g. at the level of a tissue or the whole organism, of course depending on how precisely the intercellular relationships are described in the stochastic probability tree.
2.3.2. Thermodynamical Model
The adaptive response phenomenon - including the formulation proposed in this work - can also be expressed within the formalism of statistical physics and nonequilibrium stochastic thermodynamics. This is neither an easy nor an immediately obvious approach; nevertheless, it allows one to draw remarkably interesting conclusions.
First, it must be emphasized that a living organism is a physical thermodynamic system far from equilibrium with its environment. This means that such a system continuously exchanges energy with its surroundings and is subject to external driving forces that maintain it in a steady state far from equilibrium. In such a case, the entropy of the system is lower than that of its environment and is continuously maintained at that reduced level. This implies that adaptation of the system can be considered from the perspective of entropy - that is, a thermodynamic state function describing the system’s degree of disorder.
How can entropy be linked to the adaptive response? At first glance this seems nontrivial, but entropy may be understood as a marker of an organism’s behavior. 37 Every organism has its own low-entropy minimum, within which it operates optimally. Any disturbance of the system (the organism) moves it away from this minimum, causing at least a local and temporary increase in entropy. For example, the onset of a disease or infection may generate a slight rise in entropy, which is quickly compensated by the organism so that entropy returns to its original low value. In this way, adaptation can be interpreted as the action of the system that suppresses an entropy increase - or, if the increase has already occurred, quickly reduces it and restores the initial state.2,38
For the thermodynamic system under consideration, it is convenient to use the mathematical formalism of Markov chains. This approach is frequently employed to model the physics of living systems, because Markov chains exhibit many features characteristic of evolving and self-adapting systems - features that any living system possesses.
A Markovian thermodynamic system that can serve as an analogue of a living system is shown schematically in Figure 5. It consists of a series of states (for now described only qualitatively) separated by potential barriers. These states may be interpreted in a broad sense (as states of the entire system, where transitions represent system evolution), or in a detailed sense (e.g., as specific molecular configurations such as different states of a DNA segment). In either case, changes and adaptations of the system correspond to transitions between states across potential barriers. Visual representation of one dimensional Markov chain, with potential wells 
The height of the potential barrier is crucial. Depending on the barrier height, the transition probabilities between states differ. If the barrier between two neighboring states is high, the transition probability is small - thus the system is resistant to external perturbations and therefore exhibits strong adaptation. Here we consider only those external perturbations that tend to drive the system toward a specific state transition, effectively imposing a preferred direction (the so-called driving force), typically opposite to the direction that maintains order in the system. On the other hand, if an unfavorable external interaction has already occurred (disturbing the state probability distribution and thereby increasing entropy), then low potential barriers allow for a rapid and easy return to the original state. Thus, the idea of adaptation in such a Markov system reduces to appropriate manipulation of potential barriers: lowering them once the system has been perturbed, and raising them to counteract further perturbations.
In stochastic thermodynamics, these two adaptive processes are referred to as activity and memory mechanisms (see the discussion in Chapter 2). Only when activated in the correct manner and sequence do they provide the key to effective system adaptation. In this way, one can elegantly and in a purely physical manner illustrate the mechanism of the radiation-induced adaptive response described in the preceding chapters.
To formulate it in more physical way: the thermodynamic considerations introduced in this section serve to establish a conceptual and functional link to a previously developed nonequilibrium thermodynamic framework, 37 in which radiation-induced adaptive responses are described in terms of transitions between metastable states in a driven stochastic system operating in a non-equilibrium steady state. Within that framework, the RAR function introduced here plays a specific and well-defined role: it modulates the effective potential barriers separating system states as a function of dose and time. Following exposure to an external stressor, which perturbs the driving forces maintaining the non-equilibrium steady state, the adaptive response acts to dynamically adjust these barriers, either enhancing or suppressing transition rates between states (Figure 5). This modulation promotes trajectories that minimize detrimental outcomes, corresponding to reduced entropy production and accelerated relaxation back to the steady state.
Accordingly, the RAR function should be interpreted as an effective control parameter that regulates the system’s resilience to radiation-induced perturbations by shaping the underlying potential landscape. The detailed thermodynamic formulation, including explicit definitions of state variables, transition rates, and entropy production, is presented elsewhere; the present work focuses on providing a phenomenological, dose- and time-dependent parametrization of this adaptive modulation suitable for radiobiological modeling and experimental analysis. Therefore, within a stochastic nonequilibrium framework, the adaptive-response function enters naturally as a time-dependent modifier of transition barriers, appearing in the exponential factors governing state-to-state transition rates in master-equation or Kramers-type descriptions.
2.3.3. Radiobiological and Epidemiological Investigations
The RAR model described above can, of course, be directly applied to dedicated radiobiological experiments or to collected epidemiological data. The methodology for conducting specific experiments was discussed in earlier chapters, particularly those involving the Raper–Yonezawa scheme (i.e., priming dose + challenging dose and calculation of the delta parameter), as well as other schemes (e.g., constant dose-rate irradiation).
In epidemiological data analysis, one typically considers the so-called dose–effect curve, which describes radiation risk - that is, the epidemiological probability of cancer occurrence (most often defined as cancer mortality) as a function of dose. This approach underpins modern radiation protection, which relies on a linear dose–effect relationship. This is the well-known Linear No-Threshold (LNT) hypothesis, originating from epidemiological data on survivors of the atomic bombings in Hiroshima and Nagasaki. The LNT model is today the foundation of worldwide radiation risk assessment, the basis of radiological protection principles, and the conceptual core of the ALARA (As Low As Reasonably Achievable) principle. However, the LNT model (or, more precisely, the LNT hypothesis) is more of a mathematical construct used for regulatory purposes than a true scientific model. It is difficult, for example, to relate occupational radiation exposure to the situation in Hiroshima and Nagasaki, where victims received their doses within an extremely short moment in time, at enormous dose-rates. Moreover, many scientific reports indicate a breakdown of linearity in the dose–effect curve at low radiation doses (below 100 mSv).39,40
Where, then, does RAR fit into all of this? As we have already shown, the adaptive response can appear under conditions of continuous chronic irradiation, for example in HBRA regions. This may manifest as a reduced risk of DNA mutations and, consequently, a reduced risk of developing cancer. Thus, RAR may (under certain specific conditions) influence carcinogenesis probability and thereby the dose–effect curve. Since many datasets show a deviation from linearity predicted by the LNT model, RAR may - though of course it need not - be one of its main causes.
Let us therefore consider how the adaptive response mechanism can be introduced into epidemiological dose–risk modeling.20-22,40 Equation (10) already offers a hint, but it is specific rather than general. If the radiation risk in the simple LNT model is linearly proportional to dose, R ∼ aD, then by introducing appropriate modifications we can obtain a dose–effect curve that incorporates the RAR mechanism
41
:
Of course, this does not mean that these equations can be applied automatically in every radiation-risk assessment. Their use is restricted to situations in which RAR is undoubtedly present. As mentioned earlier, RAR does not occur in every individual; therefore, eqs. (15) and (16) are not universal for the general population and cannot be used for population-level radiation risk assessment, which is the primary domain of epidemiology. However, they can effectively capture the nonlinear dose–effect relationship in cases where RAR does occur, provided that the curves given by eq. (15) or eq. (16) are fitted directly to epidemiological data and the α-parameters - characteristic of the adaptive response and its associated radiosensitivity - are estimated accordingly.
Thus, the epidemiological formulations presented here are intended to explore consistency with observed trends rather than to replace established risk models or to provide individual-level risk estimates.
2.4. Inclusion/Exclusion Criteria for Model Application
This paper is theoretical and methodological in nature and does not involve recruitment of human participants or experimental animals. Therefore, classical clinical inclusion and exclusion criteria are not applicable. Instead, we define methodological inclusion and exclusion criteria related to the experimental designs and datasets to which the proposed framework can be applied.
Inclusion criteria (methodological) - the proposed RAR formalism is applicable to: • Experimental systems (in vitro, ex vivo, or in vivo laboratory models) in which: ◦ radiation dose (D) or dose-rate (Ḋ) is quantitatively defined, ◦ exposure timing is controlled, ◦ biological endpoints are measurable and comparable between subgroups. • Irradiation schemes including: ◦ priming–dose (Raper–Yonezawa) protocols, ◦ radiation training (multiple low dose pulses), ◦ constant low dose-rate exposure, ◦ variable dose-rate exposure scenarios. • Studies in which the biological endpoint is quantifiable in relative terms (e.g., mutation frequency, chromosomal aberrations, survival fraction, relative risk). • Epidemiological datasets where dose or dose-rate differences between populations are reasonably estimated and biological endpoints are expressed in relative risk (RR) or comparable normalized measures.
Exclusion criteria (methodological) - the framework is not intended for: • Situations where radiation exposure is poorly characterized or dose reconstruction is unreliable. • Endpoints that are not radiation-related or cannot be normalized between groups. • Systems in which adaptive response is biologically implausible or experimentally excluded. • Direct individual-level risk prediction in heterogeneous human populations without proper parameter recalibration.
Thus, the model is primarily designed for controlled experimental systems and for phenomenological description of population-averaged trends.
3. Results
The proposed RAR framework yields explicit analytical expressions describing the magnitude of radiation-induced adaptive response under multiple irradiation schemes, including priming–dose protocols, radiation training, constant low dose-rate exposure, and variable dose-rate scenarios.
For each scheme, closed-form or semi-analytical expressions for the relative adaptive response parameter δ were derived as a function of dose, dose-rate, and time interval between exposures. The model demonstrates that: • the adaptive response exhibits a well-defined maximum at optimal dose and time parameters (D
max
and t
max
); • multiple priming exposures produce cumulative adaptive signals within the linear-response regime; • constant low dose-rate exposure leads to saturation of the adaptive potential; • variable irradiation schemes can be handled through convolution-type formulations of the adaptive signal.
Applications to previously published datasets confirm that the framework allows consistent parameter estimation ({α}) and provides stable fits across distinct experimental scenarios.
4. Discussion
This article describes the phenomenon of the radiation-induced adaptive response (RAR), one of the non-targeted effects of low dose radiation exposure. This effect - studied for several decades8,15 - concerns the observed increase in resistance to ionizing radiation following stimulation by small doses or dose-rates. The focus of this article is the biophysical modeling of RAR, that is, the precise mathematical and physical description of this intriguing phenomenon. Naturally, this model, like any model, is still far from a complete and universally accepted scientific theory; nonetheless, it can already be stated that this approach captures RAR with high accuracy across virtually every relevant scenario and variant.
The model is explained in detail also from a methodological perspective - that is, how to apply it to selected experimental problems. This means that the article provides explicit equations (primarily for the delta parameter, δ) that can be directly applied to a given research scheme. The user does not need deep mathematical expertise to apply the proposed framework; however, many of the equations are much faster and easier to evaluate numerically - thus developing dedicated software is highly advisable, though not strictly necessary.
Because this article is purely theoretical and describes only the methodology for applying the proposed RAR model to specific experimental schemes (radiobiological or epidemiological), it does not present direct practical applications or specific data analysis. Indeed, the previous chapter discussed several practical approaches, but these were mainly descriptive and not linked to specific datasets. Nevertheless, it is important to emphasize that - even though the article intentionally does not include explicit experimental examples - applications of the model to real data have been carried out in the past. For example, the RAR model was applied to data collected using the Raper–Yonezawa scheme, yielding the α-parameters
The RAR functions discussed in this work describe the adaptation of the physical system (generally defined), and can be used on a level of a single cell, a cell population in vitro, a tissue fragment, or – in epidemiological applications – an averaged population of organisms. Consequently, parameter values obtained e.g. from in vitro lymphocyte experiments cannot be directly transferred to tissue-level or population-level analyses without reinterpretation and rescaling. However, the Monte Carlo modeling can be useful for wider application of the model: may allow its application for one level (e.g. cellular) in relation to a higher level (tissue or the whole organism), assuming a correct description of intercellular and tissue relationships in the Monte Carlo probability tree. 35
Although the scientific literature contains a large amount of data that can be used for RAR modeling, not all of it is conclusive. In particular - and this is important to highlight - the RAR phenomenon does not always occur. This is one of the major challenges for radiobiologists, because such variability complicates interpretation. Indeed, many radiobiologists criticize current research methodologies in the RAR field as insufficient for making real progress, as they do not answer the question of what specifically determines whether RAR appears or not. Is it simply randomness, or is there some yet-unidentified radiobiological mechanism behind it? The occurrence of RAR is certainly not due merely to statistical fluctuation, because whenever RAR does appear, the effect is consistent. Perhaps this is precisely why broad mathematical, physical, and numerical modeling is needed - to approach the subject from a completely different angle than traditional biological methods allow. For this reason, the proposed model - together with its full methodology of application - appears to be an important step forward, potentially enabling deeper insight into this fascinating phenomenon known as RAR.
However although the proposed model captures a wide range of experimentally observed RAR phenomena, several limitations must be acknowledged. The model assumes a well-defined hump-shaped adaptive function, whereas some experimental systems do not exhibit adaptive responses. In such cases, the parameters may become ill-defined or statistically unstable.
Additionally, due to the physical interpretation of potential function of adaptation, model assumes additive linear effects of RAR of the described living system, but neglects explicit nonlinear interactions between other systems (e.g. cells). However, many interactions between cells are possible to be described (e.g. bystander effect) by putting the model into the context of wider Monte Carlo simulation of the whole tissue/organism. 36 Another solution is to calibrate the model (selecting alpha parameters) in such a way that it mechanistically reflects the behavior of the entire tissue or organism(s).
Finally, the application to epidemiological datasets relies on effective population-averaged descriptions.
5. Conclusions
Radiation-induced adaptive response (RAR) is a biophysical effect associated with low doses of ionizing radiation. Despite decades of research, this effect remains not fully understood, primarily due to the still unclear factors that trigger (or suppress) it. While radiobiological studies have provided substantial information - especially regarding the correlation between RAR and individual radiosensitivity - they have not yet yielded a significant breakthrough. For this reason, considerable hope is placed in theoretical approaches, such as biophysical modeling.
One such biophysical model has been presented in this article, together with a detailed methodology for applying it to specific research schemes, including experimental designs and epidemiological modeling. This methodological framework of the RAR model constitutes the main focus of the present work, which is purely theoretical, offering only a general outline of potential practical applications. We hope that the methodology presented here will prove useful in future experimental studies, enabling them to rely on the common standards proposed in this publication and thereby facilitating meaningful comparisons among different experiments.
Footnotes
Acknowledgements
The authors have no acknowledgements to declare.
Ethical Considerations
This study is theoretical and methodological in nature and does not involve human participants, animals, or identifiable data. Ethical approval was therefore not required.
Author Contributions
Krzysztof W. Fornalski (KF) conceptualized the study, developed the methodological framework, prepared the appendices, and drafted the original manuscript. Joanna Krasowska (JKr) prepared the figures, developed the main text, verified the manuscript, and independently checked the mathematical derivations. Jan Kłos (JKl) verified the appendix calculations, contributed to the formal description of the equations, created figures in appendices, and critically evaluated the conceptual structure of the model. Jan Ostrowski (JO) contributed to the thermodynamic model description (including figure) and verified the appendix calculations. All authors contributed to manuscript revision and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No new data were created or analyzed in this study. The work is conceptual and methodological in nature. All relevant derivations and analytical results are provided within the manuscript and its appendices.