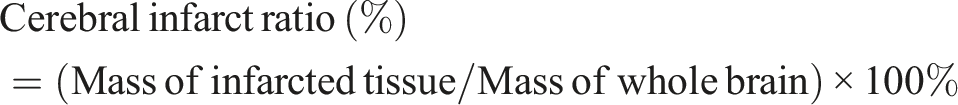Abstract
Objective
This study aimed to establish a high-performance liquid chromatography (HPLC) method for the simultaneous quantification of six components in Zhuqi Piantan Granules, and to investigate the protective effect and underlying mechanism of Zhuqi Piantan Granules alone or combined with electroacupuncture on cerebral ischemia-reperfusion injury in rats.
Methods
Zhuqi Piantan Granules were extracted with 80% methanol. HPLC analysis employed a C18 column and gradient elution using acetonitrile and 0.3% phosphoric acid as the mobile phase. Modeled rats were randomly grouped, given corresponding interventions for 14 days, and evaluated for neurological function, cerebral infarct ratio, vascular density, brain histological changes (hematoxylin-eosin (HE) staining), and Angiopoietin-1/Tyrosine Kinase Receptor-2(Ang-1/Tie-2) protein expressions (Western Blotting).
Results
All six target components exhibited excellent linear relationships (r ≥ 0.9994). The average recoveries ranged from 97.1% to 99.5%. Both Zhuqi Piantan Granules alone and their combination with electroacupuncture improved neurological function, reduced infarct ratio, increased vascular density, alleviated brain pathological damage, and upregulated Ang-1/Tie-2 expressions—with the combined treatment showing superior efficacy.
Conclusion
The established HPLC method is reliable and suitable for Zhuqi Piantan Granules quality control. Zhuqi Piantan Granules alone or combined with electroacupuncture alleviates rat cerebral injury by activating the Ang-1/Tie-2 pathway to promote angiogenesis.
Introduction
Ischemic stroke represents one of the neurological diseases most responsible for death and permanent disability in the world. 1 Currently, intravenous thrombolysis is the primary treatment; however, this approach often leaves patients with varying degrees of disability, severely impairing their post-recovery quality of life. Therefore, exploring safe and effective therapeutic drugs and combined treatment strategies is of great clinical significance.
Zhuqi Piantan Granules are a refined herbal formulation developed from a clinical prescription used for over a decade at Hangu Hospital of Traditional Chinese Medicine (Binhai New Area, Tianjin, China). It comprises 9 Chinese medicinal herbs: Cornus officinalis Sieb.et Zucc., Lycium barbarum L., Pheretima aspergillum (E.Perrier), Angelica sinensis (Oliv.) Diels, Chaenomeles speciose (Sweet) Nakai, Spatholobus suberectus Dunn, Paeonia lactiflora Pall., Achyranthes bidentate Bl., and Glycyrrhiza uralensis Fisch. Pharmacologically, Zhuqi Piantan Granules nourish the liver and kidneys, dredges meridians and collaterals, nourishes yin to calm wind, strengthens muscles and bones, and promotes joint function. Clinically, it is used for the recovery period and sequelae of meridian-involved stroke.
Vascular recovery in the damaged brain region after ischemic stroke is critical for achieving optimal rehabilitation outcomes. Electroacupuncture intervention has been shown to promote angiogenesis, thereby enhancing blood and oxygen supply.2–6 Clinically, combined EA-medication therapy is frequently adopted for stroke management.7,8 The Angiopoietin-1/Tyrosine Kinase Receptor-2 (Ang-1/Tie-2) system, a vascular-specific ligand-receptor complex, regulates endothelial cell survival and vascular maturation, 9 promotes angiogenesis in ischemic regions, and ameliorates neurological deficits following cerebral ischemia. 10
Our previous studies have confirmed the pharmacodynamic effects of Zhuqi Piantan Granules. 11 However, the mechanism underlying its protective and restorative effects on damaged cerebral blood vessels, as well as the efficacy of combined electroacupuncture therapy, remain underexplored. This study aimed to determine the content of six main components in Zhuqi Piantan Granules, investigate the therapeutic effects of Zhuqi Piantan Granules combined with electroacupuncture therapy, and explore the underlying mechanism via the Ang-1/Tie-2 signaling pathway, thereby providing a scientific basis for the extensive clinical application of this formulation.
Material and Methods
All experiments were performed from January 2024 to May 2025.
Source of Drugs and Reagents
Zhuqi Piantan Granules (Batch No. 240013, 240014, 240015, Specification: 6 g/bag) were provided by Hangu Hospital of Traditional Chinese Medicine (Binhai New Area, Tianjin, China). Gallic acid reference substance (Lot No.: 110831 - 201204, purity 98%) was purchased from the National Institutes for Food and Drug (Beijing, China). Morroniside reference substance (Lot No.: M14IB209606, purity 99.2%), Loganin reference substance (Lot No.: M25IB210623, purity 98.1%), Paeoniflorin reference substance (Lot No.: M28GB143089, purity 98.1%) and Ferulic acid reference substance (Lot No.: G13S11L124423, purity 99.9%) were all purchased from Shanghai Yuanye Bio - Technology Co., Ltd. (Shanghai, China). Chlorogenic acid standard substance (Lot No.: C23PC489C, purity 98%) was purchased from Beijing Jinning Bio - Technology Co., Ltd. (Beijing, China). Nimodipine Tablets (positive control, Batch No. H20043915, Specification: 30 mg/tablet) were purchased from Tianjin Central Pharmaceutical Co., Ltd. (Tianjin, China). Primary antibodies against Ang-1 and Tie-2 were obtained from Proteintech Group, Inc. (Rosemont, IL, USA).
Animals
Seventy-two specific pathogen-free (SPF)-grade male Wistar rats (210-220 g, 7-8 weeks old) were supplied by Beijing Huafukang Biotechnology Co., Ltd. (Beijing, China). The rats were housed in an SPF facility under controlled conditions: temperature (22 ± 2°C), humidity (35%-45%), and a 12 h light/dark cycle. Standard rodent chow and water were provided ad libitum. Standard rodent chow were supplied by Beijing Keao Xieli Feed Co., Ltd. (Beijing, China). Animal feed mainly contains corn, soybean meal, fish meal and other ingredients. The rats were euthanized by carbon dioxide asphyxiation, and each animal was checked individually to confirm complete death. If any animal was found not to be deceased, cervical dislocation was supplemented as a secondary euthanasia method.
Preparation of Reference Substance Solutions
An appropriate amount of each reference substance (gallic acid, morroniside, chlorogenic acid, loganin, paeoniflorin, and ferulic acid) was accurately weighed and individually transferred to 10 mL brown volumetric flasks. Each flask was then added with 80% methanol to dissolve the reference substance completely; the volume was further adjusted to the 10 mL mark with 80% methanol. After thorough mixing, reference substance stock solutions were obtained. The concentrations of the stock solutions were as follows: 0.1827 mg/mL (gallic acid), 1.148 mg/mL (morroniside), 0.0604 mg/mL (chlorogenic acid), 0.5533 mg/mL (loganin), 1.419 mg/mL (paeoniflorin), and 0.04584 mg/mL (ferulic acid).
Subsequently, 1.0 mL of each reference substance stock solution was precisely pipetted into a new set of 10 mL brown volumetric flasks. 80% methanol was added to dilute the solutions to the 10 mL mark, followed by thorough shaking to ensure homogeneity. The concentrations of the reference substance solutions were 0.01827 mg/mL (gallic acid), 0.1148 mg/mL (morroniside), 0.00604 mg/mL (chlorogenic acid), 0.05533 mg/mL (loganin), 0.1419 mg/mL (paeoniflorin), and 0.004584 mg/mL (ferulic acid).
Preparation of Test Sample Solutions
An accurately weighed portion (2.0 g, precision: 0.0001 g) of Zhuqi Piantan Granules were transferred into a stoppered conical flask. A precise volume of 25 mL 80% methanol was added to the conical flask using a volumetric pipette, and the conical flask was immediately sealed with its stopper to prevent solvent volatilization. The total weight of the stoppered conical flask including contents was recorded. The conical flask was then placed in an ultrasonic extractor for ultrasonic-assisted extraction at room temperature for 20 min. After extraction, the conical flask was allowed to cool to room temperature. The total weight of the conical flask was re-measured, and the weight loss was replenished with 80% methanol. The mixture was thoroughly shaken to ensure homogeneity, and then filtered through a 0.45 μm filter membrane to remove insoluble particles. The subsequent filtrate was collected as the final test sample solution for a high-performance liquid chromatography (HPLC) analysis.
Preparation of Negative Control Solutions
Negative samples were prepared to verify the specificity of the analytical method, following the same formulation proportions and manufacturing procedures as the test samples but excluding specific medicinal materials known to contain the target components: Gallic acid-, morroniside-, loganin-, and paeoniflorin-free negative samples: Prepared using L. barbarum, P. aspergillum, A. sinensis, S. suberectus, C. speciose, A. bidentate, and G. uralensis. Chlorogenic acid-free negative samples: Prepared using C. officinalis, P. aspergillum, and A. bidentate. Ferulic acid-free negative samples: Prepared using C. officinalis, P. aspergillum, S. suberectus, A. bidentate, and C. speciose.
All negative samples were processed into solutions according to the above sample preparation method.
Chromatographic Conditions
Identification and quantification was performed on a Shimadzu LC-20AT HPLC system (Shimadzu Corporation, Japan), equipped with an SPD-M20A detector, and a C18 column (250 mm × 4.6 mm i.d., 5 μm Dikma). Solvent A was acetonitrile and solvent B was 0.3% phosphoric acid aqueous solution. The gradient elution program was set as follows: 0∼7 min, 3% A; 7∼8 min, 3%∼10% A; 8∼20 min, 10%∼16% A; 20∼25 mins, 16%∼19% A; 25∼30 min, 19%∼24% A; 30∼45 min, 24%∼3% A. The flow rate was 1.0 mL/min. The column oven was maintained at a temperature of 30°C. The injection volume of 5 μL was used for each sample. The detection wavelengths were monitored at 240 nm. Data acquisition and quantitative analysis were performed using LabSolutions (v 5.57 SP1). System suitability requirement: The number of theoretical plates (N) for each target component was required to be no less than 3000, ensuring adequate chromatographic separation efficiency.
Validation of the Method
Specificity Investigation
The reference substance solution, test sample solution, and negative solution were injected into the HPLC respectively. Chromatographic separation was performed under the aforementioned chromatographic conditions, and the chromatograms were recorded.
Limit of Detection (LOD) and Limit of Quantitation (LOQ)
An appropriate volume of the standard solution was taken and serially diluted in proportion. The concentration corresponding to a signal-to-noise ratio (S/N) of 3 was defined as the LOD of the method, while the concentration corresponding to an S/N of 10 was designated as the LOQ.
Investigation of Linear Relationship
Accurately measured volumes of 0.1, 0.5, 1.0, 1.5, and 2.0 mL of the reference substance stock solution were respectively transferred into 10 mL brown volumetric flasks. Each flask was filled to the marked volume with water, followed by thorough mixing to prepare a series of reference substance solutions. Each solution was injected into the liquid chromatograph for analysis.
Precision Test
The test sample solution was prepared as described above and was subjected to HPLC analysis for 6 times consecutively.
Stability Test
The same sample solution stored at room temperature was injected into HPLC for determination at 2, 4, 6, 12, 18, and 24 h, respectively.
Repeatability Test
Six different samples, which were prepared with the same sample preparation procedure, were examined.
Recovery Test
Nine portions of the test product were taken, with each portion accurately weighed to 1 g. An appropriate volume of the stock solutions containing gallic acid, morroniside, chlorogenic acid, loganin, paeoniflorin, and ferulic acid reference substances was precisely added to each sample portion. This spiking process was designed to make the final concentrations of the 6 components in the samples reach 3 different levels (high, medium, and low), with 3 replicate portions prepared for each concentration level. The above samples were prepared following the method described in Section “Preparation of Test Sample Solutions”. Subsequently, the prepared test solutions were analyzed under the aforementioned chromatographic conditions, and the average recovery rate and Relative Standard Deviation (RSD) for each component were calculated.
Sample Determination
Samples from 3 different batches were collected for analysis. For each batch, test sample solutions were prepared in parallel, with 3 replicate portions prepared per batch, following the preparation method described in Section “Preparation of Test Sample Solutions”. Each prepared test sample solution was injected into the liquid chromatograph under the aforementioned chromatographic conditions for content determination.
Animal Modeling, Grouping, and Administration
Seventy-two SPF male Wistar rats were randomly divided into six groups: sham-operation group, model group, nimodipine control group, electroacupuncture group, Zhuqi Piantan Granules group, and combined treatment group. A modified Zea Longa method was used to establish the MCAO model.11–13 After 1 h of occlusion, the suture was gently withdrawn to induce reperfusion. Rats in the sham-operation group underwent the same surgical procedure without suture insertion. The model was considered successfully established when the rats exhibited obvious hemiplegic symptoms, body tilt, and crawling rotation after regaining consciousness.
After successful modeling, the following operations were performed. The Zhuqi Piantan Granules group received intragastric administration of Zhuqi Piantan Granules (5.4 g/kg) once daily for 14 consecutive days. The electroacupuncture group was treated with acupuncture at the “Dazhui” (GV14), “Baihui” (GV20), and “Renzhong” (GV26) acupoints. Acupoint localization was performed using the bone-length measurement method (simulated from human acupoint standards) in accordance with Experimental Acupuncture and Moxibustion and the experimental animal acupoint map developed by Hua Xingbang. A G6805 EA therapeutic apparatus was used with the following parameters: peak voltage 6 mV, sparse-dense wave, and frequency 1/20 Hz. “Deqi” (needle sensation) was confirmed by slight vibration of the needle body. EA treatment was administered for 30 min once daily for 14 consecutive days. The combined treatment group received intragastric Zhuqi Piantan Granules followed by electroacupuncture therapy, once daily for 14 days. The sham-operation and model groups were given normal saline via intragastric gavage and subjected to 30 min of restraint (mimicking electroacupuncture group handling) once daily for 14 days. On Day 14, after 12 h of fasting, rats in each group were sacrificed for sample collection.
Neurological Behavioral Evaluation
Neurological function scores were evaluated for rats at 6, 24, and 48 h after modeling. The scoring criteria were as follows: 0 points, no neurological deficit, normal activity; 1 point, inability to fully extend the left forepaw; 2 points, rats circling to the left; 3 points, rats leaning to the left; 4 points, inability to walk spontaneously and loss of consciousness.
Beam Walking Test
The beam walking test was conducted 1 h after the final administration. The beam was 2.5 cm wide, 120 cm long, 2.5 cm thick, 60 cm above the ground, and suspended horizontally. Preoperative training was for 2 days, twice a day. Scoring criteria: 0 points, the rat was able to cross the beam and maintain balance with all 4 paws on the surface the beam; 1 point, the rat was able to cross the beam, but its paws were grasping the edge of the balance beam; 2 points, the rat grasping the balance beam, and 1 paw hung down from the balance beam; 3 points, the rat had 2 paws hanging down from the balance beam, or rotated on balance beam (>60 s); 4 points, the rat tried to maintain its balance on the balance beam but fell (>40 s); 5 points, the tried to maintain its balance on the balance beam but fell (>20 s); 6 points, the rat fell from the balance beam (<10 s).
Forelimb Grip Strength Measurement
Grip strength was measured immediately after the beam walking test using a self-made grip strength tester. Rats were allowed to grasp the pull-up bar with their forelimbs. The tester fixed the pull-up plate with the left hand, held the rat’s tail with the right hand, and gently pulled the rat backward (after releasing the left hand) until the rat slipped. The maximum pull force at which slipping occurred was recorded. Each rat was tested twice, and the average value was calculated.
Determination of Cerebral Infarct Ratio
After the experiment, 5 rats per group were sacrificed. Brains were harvested following decapitation, rinsed with cold normal saline, and rapidly stored at −20°C for 10 min. The olfactory bulb, pituitary gland, and lower brainstem were removed, and the remaining brain tissue was cut into five 2-mm-thick coronal slices. Slices were incubated in 1% 2, 3, 5-triphenyltetrazolium chloride solution at 37°C in the dark for 30 min with flipping every 5 min. Normal tissue stained rose red, while infarcted tissue appeared white with a clear boundary. Infarcted regions were carefully dissected, weighed, and the cerebral infarct ratio was calculated as follows:
Hematoxylin-Eosin (HE) Staining for Brain Tissue Morphology
Brain tissue samples were fixed in 10% formalin solution. Routine processing included sampling, dehydration, clearing, paraffin embedding, and sectioning (3 μm thickness). Sections were dewaxed in xylene, rehydrated with gradient ethanol, stained with hematoxylin for 4 min, differentiated with hydrochloric acid-ethanol, blued with ammonia water, stained with eosin for 1 min, dehydrated with gradient ethanol, cleared in xylene, and mounted with neutral balsam. Morphological changes in the cerebral cortex and medulla were observed under a light microscope, and images were captured using a high-resolution digital microscopic image analysis system.
Immunofluorescence Staining
Brain sections were dewaxed to water, subjected to antigen retrieval, and blocked with serum. After removing the blocking solution, appropriately diluted Factor VIII primary antibody was added to the sections, which were then incubated overnight at 4°C in a humidified chamber. Sections were washed 3 times with phosphate-buffered saline (PBS, pH 7.4) (5 min per wash). Fluorescently labeled secondary antibody (diluted in PBS) was added, and the sections were incubated at room temperature for 50 min. After three additional PBS washes (5 min per wash), the cell nuclei were counterstained with 4’, 6-diamidino-2-phenylindole (DAPI), and the sections were mounted with anti-fluorescence quenching medium. Images were acquired under a fluorescence microscope, and the vascular density was analyzed using Image J software.
Western Blot for Ang-1 and Tie-2 Protein Detection
Cerebral cortex tissue was homogenized in tissue lysis buffer, centrifuged at 12 000 r/min for 10 min, and the supernatant was collected. Total protein concentration was determined using the bicinchoninic acid method. Loading buffer was added to the protein samples, which were then denatured at 100°C for 10 min. A 30 - 50 μg sample was subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes. Membranes were blocked with 5% skimmed milk powder for 3 h, then incubated with primary antibodies (dilution 1:1000) overnight at 4°C. After 5 washes with Tris-buffered saline with TBST (5 min per wash), membranes were incubated with secondary antibody (dilution 1:2000) at room temperature for 1.5 h. Membranes were washed 5 times with TBST (5 min per wash), developed with enhanced chemiluminescence reagent, and images were analyzed using Image J software. Experiments were repeated 3 times.
Statistical Analysis
SPSS 17.0 software was used to process the experimental data. Measurement data were expressed as mean ± standard deviation. Intergroup comparisons were performed using t-test or one-way ANOVA followed by post hoc analysis with LSD test or Dunnett test. Significant differences were determined at P < 0.05.
Results
Validation of the Method
Specificity Investigation
The results demonstrated that under the established chromatographic conditions, other components in the test sample solution were well separated from gallic acid, morroniside, chlorogenic acid, loganin, paeoniflorin, and ferulic acid. Furthermore, no absorption peaks were observed at the corresponding retention positions of the aforementioned six components in the chromatogram of the negative control solution, indicating that the negative control did not interfere with the determination (Figure 1). HPLC chromatograms (A) standard solution; (B) sample solution; (C) Gallic acid-, morroniside-, loganin-, and paeoniflorin-free negative samples; (D) Chlorogenic acid-free negative samples; (E) Ferulic acid-free negative samples. 1-gallic acid; 2-morroniside; 3-chlorogenic acid; 4-loganin; 5-paeoniflorin;6-ferulic acid
LOD and LOQ
For gallic acid, 0.3654 μg/mL (LOD) and 1.096 μg/mL (LOQ); for morroniside, 0.2296 μg/mL (LOD) and 0.8036 μg/mL (LOQ); for chlorogenic acid, 0.1208 μg/mL (LOD) and 0.4228 μg/mL (LOQ); for loganin, 0.2213 μg/mL (LOD) and 0.7746 μg/mL (LOQ); for paeoniflorin, 0.5676 μg/mL (LOD) and 1.419 μg/mL (LOQ); and for ferulic acid, 0.0917 μg/mL (LOD) and 0.2750 μg/mL (LOQ).
Investigation of Linear Relationship
Linear Relationships of Various Constituents

Precision Test
The RSD values were as follows: gallic acid 0.94%, morroniside 0.30%, chlorogenic acid 0.31%, loganin 1.02%, paeoniflorin 0.42%, and ferulic acid 0.94%.
Stability Test
The RSD values of the peak areas for each component were as follows: gallic acid (0.84%), morroniside (0.16%), chlorogenic acid (0.91%), loganin (0.93%), paeoniflorin (0.46%), and ferulic acid (0.82%). These results indicate that gallic acid, morroniside, chlorogenic acid, loganin, paeoniflorin, and ferulic acid remained stable in the test sample solution within the 24 h storage period.
Repeatability Test
For gallic acid, the average content was 0.216 mg/g with RSD of 1.14%; for morroniside, the average content was 1.418 mg/g with RSD of 0.57%; for chlorogenic acid, the average content was 0.075 mg/g with RSD of 1.08%; for loganin, the average content was 0.709 mg/g with RSD of 1.08%; for paeoniflorin, the average content was 1.908 mg/g with RSD of 1.04%; and for ferulic acid, the average content was 0.052 mg/g with RSD of 1.09%. All RSD values were within the acceptable range for analytical repeatability, confirming that the method exhibits good repeatability for the determination of the 6 target components.
Recovery Test
The average spiked recovery rates of the 6 components were as follows: gallic acid 99.2% with RSD of 2.59%, morroniside 98.1% with RSD of 1.44%, chlorogenic acid 99.4% with RSD of 1.75%, loganin 97.1% with RSD of 2.03%, paeoniflorin 99.5% with RSD of 2.31%, and ferulic acid 97.8% with RSD of 1.24%. All average recovery rates fall within the typical acceptable range (95%-105%) for analytical methods.
Sample Content
Results of Content Determination of Various Constituents (n = 3)

Neurological Function Scores
Neurological Function Scores of Rats (
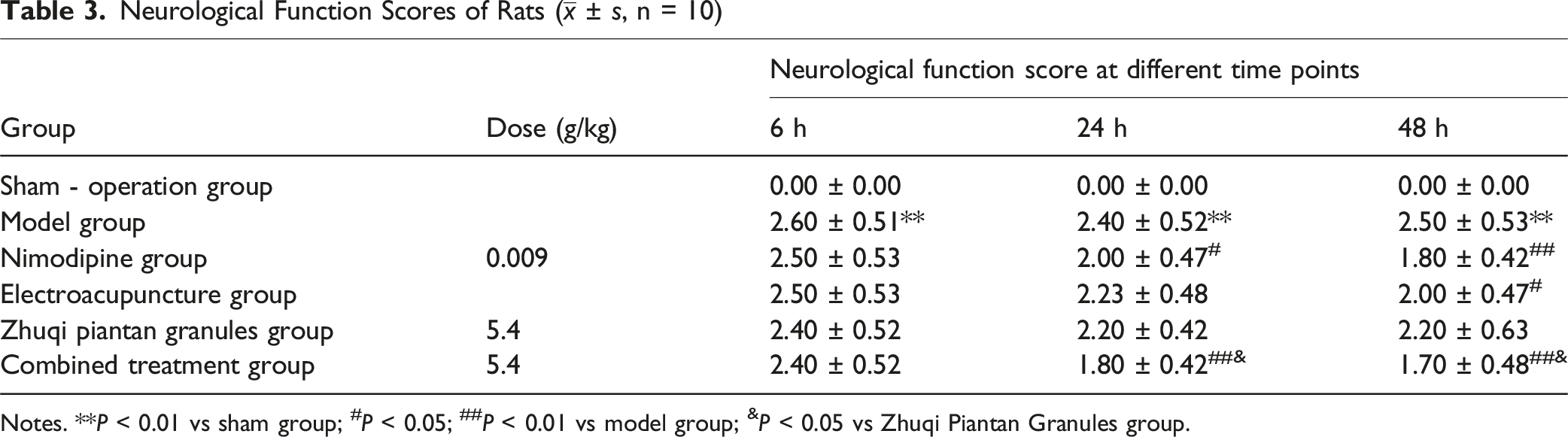
Notes. **P < 0.01 vs sham group; #P < 0.05; ##P < 0.01 vs model group; &P < 0.05 vs Zhuqi Piantan Granules group.
Beam Walking Test Scores and Forelimb Grip Strength
Determination Results of Beamwalking Test Scores and Grip Strength of the Forelegs in all Group (

Note. *P < 0.01 vs sham group; #P < 0.01 vs model group; &P < 0.05, &&P < 0.01 vs Zhuqi Piantan Granules group.
Cerebral Infarct Ratio
Determination of Cerebral Infarct Ratio in Rats of Each Group (

Notes. **P < 0.01 vs the sham - operation group; ##P < 0.01 vs the model group.
HE Staining for Brain Tissue Morphology
HE staining was performed to observe the pathological changes in cell morphology of the cerebral cortex and cerebral medulla across all groups.
In the sham-operation group: The cerebral cortex and medulla exhibited an intact structure. Neurons, glial cells, and capillaries showed normal morphology and complete structural integrity, with clear nucleoli, no eosinophilic staining of the cytoplasm, and dense, orderly arrangement of nerve fibers.
In the model group: Extensive liquefactive necrosis was observed in the cerebral cortex, accompanied by a reduction in capillary count. Neurons were significantly decreased in number and arranged in disarray; pyramidal cell bodies were shrunken, Nissl bodies had disappeared, and cell nuclei showed pyknosis and hyperchromatism. The cytoplasm exhibited eosinophilic staining, and neurons presented with obvious vacuolation. Additionally, the medullary nerve fibers showed large-scale degeneration and necrosis with vacuolation, along with significant glial cell proliferation, cerebral edema, widened intercellular spaces, and occasional hemorrhage.
Compared with the model group: In the nimodipine group, Zhuqi Piantan Granules group, and combined treatment group, the pathological changes in most animals were alleviated. The number of capillaries increased slightly, necrosis of cerebral cortical neurons was reduced, the number of pyramidal cells increased, and the morphological structure tended to be normal with relatively regular cell nuclei. Only a small number of neurons showed shrunken cell bodies, nuclear pyknosis and hyperchromatism, vacuolation, medullary nerve fiber degeneration, local vacuolation, and a small amount of glial cell proliferation. Among these 3 groups, the reduction in cerebral cortical neuron necrosis was more significant in the Zhuqi Piantan Granules group and combined treatment group. In the electroacupuncture group, the pathological changes in some animals were alleviated, with reduced necrosis of cerebral cortical neurons, increased number of pyramidal cells, and morphological structure tending to be normal, cell nuclei were relatively regular. However, some neurons still showed shrunken cell bodies, nuclear pyknosis and hyperchromatism, vacuolation, medullary nerve fiber degeneration and necrosis, local vacuolation, and a small amount of glial cell proliferation; obvious hemorrhage was also observed in some animals (Figures 2 and 3). Pathological morphological changes of rats cerebral cortex in each group (HE, × 100) (A) Sham – operation group; (B) Model group; (C) Nimodipine group; (D) Electroacupuncture group; (E) Zhuqi Piantan Granules group; (F) Combined treatment group. Red arrow:cortical liquefactive necrosis; green arrow: cone cell atrophy, disappearance of Nissl bodies; blue arrow: neuronal vacuolation Pathological morphological changes of rats cerebral medulla in each group (HE, × 100) (A) Sham – operation group; (B) Model group; (C) Nimodipine group; (D) Electroacupuncture group; (E) Zhuqi Piantan Granules group; (F) Combined treatment group. Red arrow: degeneration, necrosis of medullary nerve fibers; blue arrow: neuronal vacuolation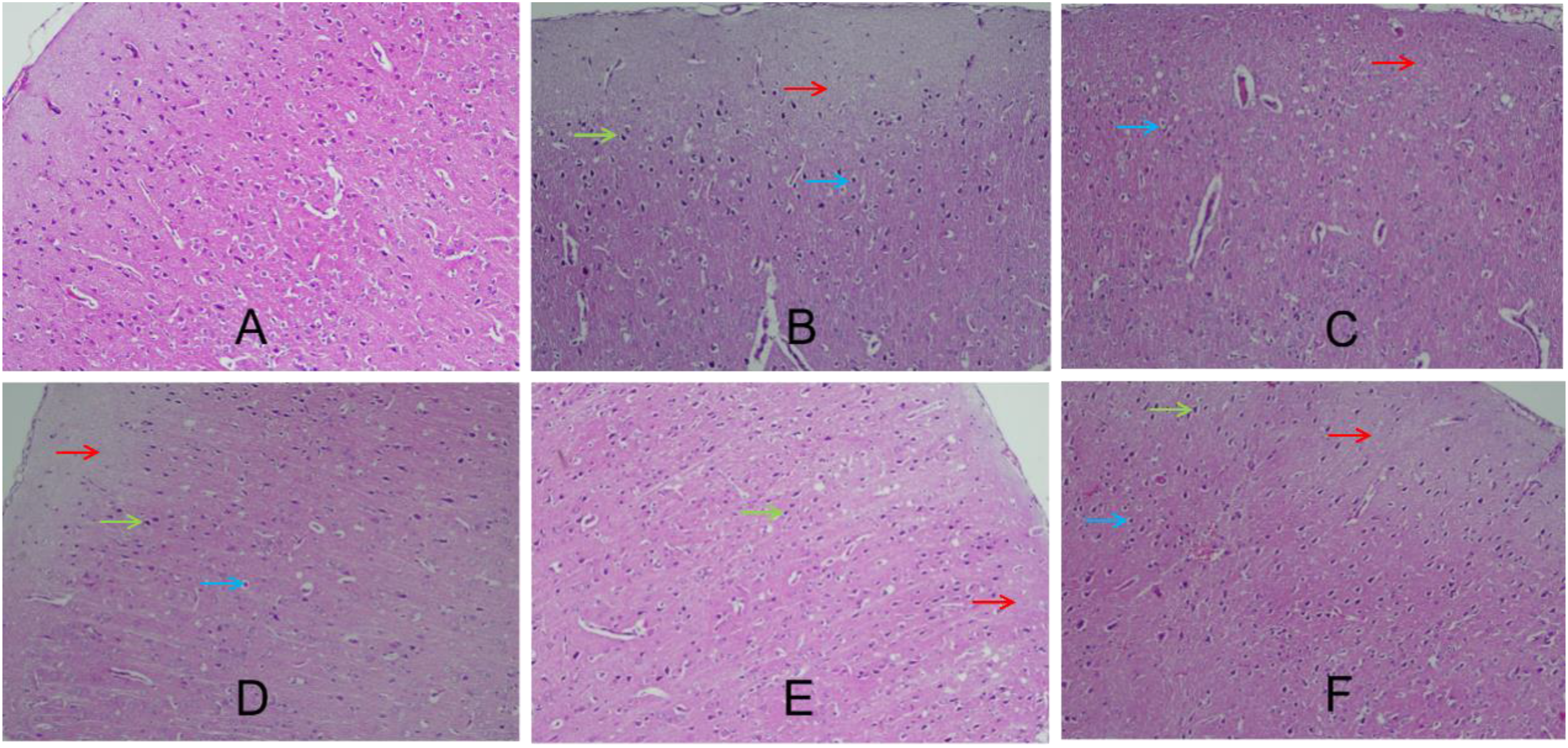

Comparison of Vascular Density
Compared with the model group, the vascular density in the electroacupuncture group was significantly increased (P < 0.05), while the vascular densities in the Zhuqi Piantan Granules group and combined treatment group showed a more significant increase (P < 0.01). Furthermore, when compared with the Zhuqi Piantan Granules group, the combined treatment group exhibited a significantly higher vascular density (P < 0.01) (Figure 4). Factor VIII staining images and statistical results of each group. (A) Factor VIII staining images of each group (scale bar = 50 μm); (B) statistical results of each group, *P < 0.05, **P < 0.01 vs the model group, &&P < 0.01 vs the Zhuqi Piantan Granules group
Protein Expressions of the Ang-1/Tie-2 in Each Group
Detection of protein expressions related to the Ang-1/Tie-2 signaling pathway in rat brain tissues revealed the following results: Compared with the sham-operation group, the relative expression levels of Ang-1 and Tie-2 proteins in the brain tissues of the model group were significantly increased (P < 0.05). In contrast, when compared with the model group, the Zhuqi Piantan Granules group and the combined treatment group both exhibited significantly higher relative expression levels of Ang-1 and Tie-2 proteins in brain tissues (P < 0.05). Additionally, the expression levels of Ang-1 and Tie-2 proteins in the brain tissues of the combined treatment group were significantly higher than those in the Zhuqi Piantan Granules group (P < 0.05) (Figure 5). Effects on Ang - 1 and Tie - 2 in rat brain tissues of each group. (A) The protein expression was measured by Western blot, and the representative bands are shown. (B, C) Quantification of protein bands of Ang-1 and Tie-2. Actin expression was used to normalize protein expression. #P < 0.05, ##P < 0.01 vs the sham - operation group; *P < 0.05, **P < 0.01 vs the model group; &&P < 0.01 vs the Zhuqi Piantan Granules group
Discussion
In determining the detection wavelength standard solutions of gallic acid, chlorogenic acid, morroniside, loganin, paeoniflorin, and ferulic acid were subjected to full-wavelength scanning within the range of 190-400 nm. The results revealed that gallic acid exhibited maximum absorption at 271 nm, chlorogenic acid at 211 nm, morroniside and loganin both at 240 nm, paeoniflorin at 231 nm, and ferulic acid at 323 nm. However, at 211 nm (the maximum absorption wavelength of chlorogenic acid), significant interference from the mobile phase was observed, leading to an unstable baseline. For ferulic acid, no obvious difference in peak area was detected between 323 nm and 240 nm. Furthermore, all target components could be completely separated at 240 nm, with well-formed peaks and no interference from the negative control. Therefore, 240 nm was selected as the detection wavelength.
For the selection of the sample preparation method for content determination, different extraction solvents (water, 100% methanol, 80% methanol, and 50% methanol) were evaluated. The use of 80% methanol resulted in a relatively higher content of target components, and the solution was easy to filter. In addition, a comparison was conducted between ultrasonic extraction and reflux extraction, the results showed no significant difference in the content of target components obtained by the 2 methods. Thus, ultrasonic extraction was selected due to its simpler operation.The effect of ultrasonic extraction time was further investigated at 3 levels: 10 min, 20 min, and 30 min. The results indicated that the content of target components after 20 min and 30 min of ultrasonic extraction was significantly higher than that after 10 min, while no difference was observed between 20 min and 30 min. Therefore, 20 min was determined as the optimal ultrasonic extraction time. Finally, the extraction method for this product was confirmed.
Stroke is one of the major diseases that seriously threaten global human health. Particularly in low- and middle-income countries, the incidence, prevalence, and mortality of stroke remain persistently high, with ischemic stroke being the most common subtype. 14 Traditional Chinese medicine (TCM) holds unique advantages in the prevention and treatment of stroke. Zhuqi Piantan Granules, a TCM formulation, exerts effects including nourishing the liver and kidneys, dredging meridians and collaterals, nourishing yin to calm wind, strengthening muscles and bones, and improving joint function.
Electroacupuncture is a therapeutic technique that integrates traditional acupuncture with electrical pulse stimulation. It enables objective quantification of acupuncture intervention and is more suitable for basic research due to its controllable parameters. Currently, electroacupuncture has been widely applied in the rehabilitation of stroke and post-stroke sequelae, achieving favorable clinical efficacy.15,16 Combined electroacupuncture-medication therapy refers to a scientific process: under the guidance of TCM theory and/or modern medical theory, and based on the characteristics and respective advantages of electroacupuncture and drugs, a clinical protocol is formulated to treat the same disease using both interventions simultaneously. This process is not a simple superposition of their individual effects, but rather a relationship of mutual complementation and synergy.
Numerous studies have emphasized the critical role of angiogenesis in alleviating ischemic stroke-induced injury.17,18 The Ang-1/Tie-2 pathway is a vascular-specific signaling pathway identified in recent years that mediates angiogenesis.19,20 During vascular regeneration after cerebral ischemia, Ang-1 binds to its receptor Tie-2, triggering Tie-2 phosphorylation and subsequent activation of the downstream PI3K/Akt signaling pathway. This cascade promotes endothelial cells to form vascular-like structures and recruits perivascular supporting cells, thereby completing vascular remodeling and maturation. A large body of evidence has demonstrated that many TCM herbs can enhance vascular regeneration and remodeling via the Ang-1/Tie-2 signaling pathway, ultimately mitigating cerebral injury.9,21–25 Additionally, acupuncture therapy has also been shown to regulate angiogenesis through this pathway. 26
The findings of this study are summarized as follows: ① All treatment groups improved the neurological function scores of model rats, indicating their ability to alleviate neurological injury. Notably, the combined treatment group exhibited a more pronounced improvement compared to the Zhuqi Piantan Granules group. ② Each treatment group reduced the cerebral infarct ratio, exerting a protective effect against cerebral injury, with the combined treatment group showing the lowest infarct ratio. ③ HE staining of brain tissue revealed that, in the Zhuqi Piantan Granules and combined treatment groups, most rats displayed alleviated pathological changes, including a slight increase in capillary count, a significant reduction in cerebral cortical neuron necrosis, an elevated number of pyramidal cells, and a tendency toward normal tissue morphology. ④ Regarding cerebral vascular density, all treatment groups increased vascular density in the brain, and the combined treatment group had a significantly higher vascular density than the Zhuqi Piantan Granules group. ⑤ Each treatment group upregulated the relative expression levels of Ang-1 and Tie-2 proteins in rat brain tissue, with the combined treatment group showing significantly higher expression than the Zhuqi Piantan Granules group. These results suggest that the combination of Zhuqi Piantan Granules and electroacupuncture can more effectively upregulate the expression of Ang-1 and Tie-2 proteins, promote angiogenesis, and improve tissue blood circulation.
Limitations of the Study
This experiment still has several limitations that require further improvement. Firstly, the sample size of experimental animals was not determined through systematic statistical calculation but only based on relevant literature references or previous experimental experience; a statistical method should be adopted to standardize the sample size design in subsequent studies. Secondly, in the research on the Ang-1/Tie-2 signaling pathway, specific inhibitors were not used for intervention verification, making it difficult to clarify the core regulatory role of this pathway in the pharmacological effects of the drug. Thirdly, due to the high complexity of the angiogenesis mechanism, whether Zhuqi Piantan Granules can exert vascular regulatory effects through other molecular signaling pathways remains to be further explored and verified in follow-up studies.
Conclusions
The Zhuqi Piantan Granules group, electroacupuncture group, and combined treatment group all alleviated cerebral ischemic injury in model rats,and the experimental results suggested that it was associated with the Ang-1/Tie-2 signaling pathway. Among these groups, the combined electroacupuncture-medication group exhibited a superior therapeutic effect compared to the Zhuqi Piantan Granules group and the electroacupuncture group alone. This indicates that combined electroacupuncture-medication therapy effectively integrates the dual advantages of electroacupuncture and drugs. And it can promote angiogenesis and improve tissue blood circulation, thereby reducing cell necrosis induced by ischemia and hypoxia and mitigating cerebral injury.
This study not only provides evidence-based medical support for the clinical application of combined electroacupuncture and Zhuqi Piantan Granules therapy in stroke treatment but also offers valuable reference for the development of therapeutic strategies for stroke.
Footnotes
Acknowledgments
The authors would like to express their sincere gratitude to the institutions and laboratories that supported this research. We are also grateful to the Hangu Hospital of Traditional Chinese Medicine (Binhai New Area, Tianjin, China) for providing prescriptions and support.
Ethical Considerations
All experimental protocols were approved by the Experimental Animal Welfare and Ethics Committee of Tianjin Institute of Medical and Pharmaceutical Sciences (Approval Code: IMPS-EAEP-Z-2023103-01).
Author Contributions
Lingyu Kong designed the study and wrote the manuscript. Ping Yang, Nan Li, Ting Zhang and Xiaolin Wang performed the content determination. Zi Wang and Di Hao conducted the animal experiments. Weiwei Liu was responsible for the pathological experiments. Na Gu was in charge of the Western Blot experiments. Ying Xu and Yedong Bi provided the prescription.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tianjin Municipal Health Commission Traditional Chinese Medicine and Integrated Chinese and Western Medicine Project General Project (2023103); Tianjin Binhai New Area Health and Health Commission Science and Technology Project General Support Project (2022BWKY018); Natural Science Foundation of Tianjin General Project (23JCYBJC00410)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.