Abstract
Objectives
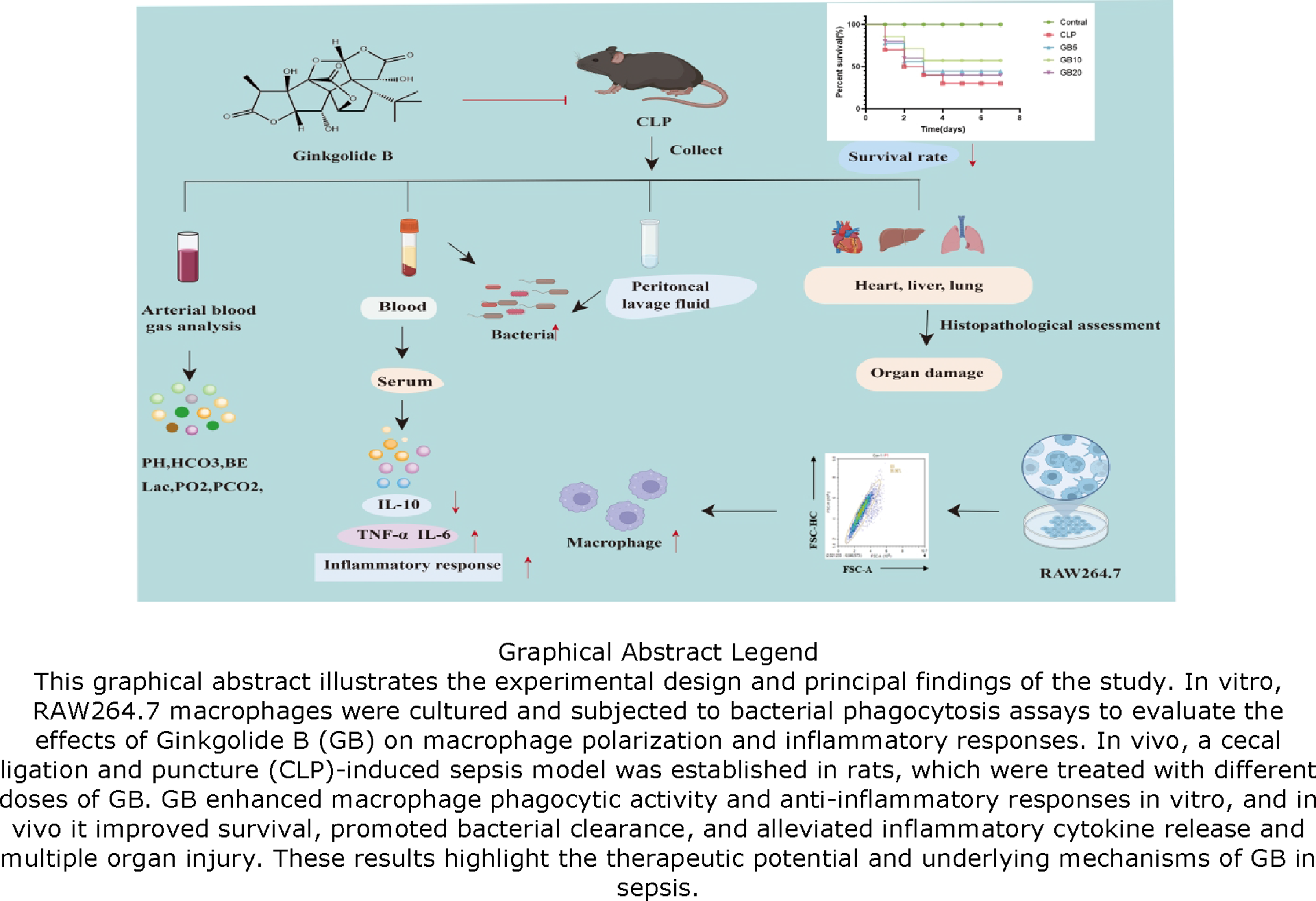
This study delineates the U-Shaped Dose-Dependent Modulation of Ginkgolide B (GB) on macrophage function, highlighting its capacity to enhance bacterial phagocytosis and attenuate systemic inflammation in sepsis.
Methods
RAW264.7 cells were co-cultured with GB, and flow cytometry was used to assess macrophage phagocytosis and polarization. Rats were divided into five groups: Healthy, cecal ligation and puncture (CLP), and three GB treatment groups. Survival was monitored over 7 days, and arterial blood was analyzed to assess acidosis. Histological examination was performed to evaluate organ damage, while ELISA measured inflammatory factors in blood and peritoneal lavage fluid (PLF). Finally, bacterial colony counts in the PLF were analyzed to assess peritoneal macrophage bacterial clearance capacity.
Results
GB co-culture enhanced macrophage bacterial phagocytosis. GB treatment alleviated acidosis and improved survival in septic rats. It also reduced pro-inflammatory cytokines TNF-α and IL-6 while increasing anti-inflammatory IL-10 levels. GB injection decreased bacterial load in the PLF, indicating enhanced macrophage bacterial clearance.
Conclusion
GB enhances macrophage phagocytic activity, improves bacterial clearance, increases the survival rate of septic rats, and reduces systemic inflammation.
Introduction
Sepsis is a life-threatening condition characterized by multiorgan dysfunction resulting from a dysregulated response to infection. 1 It affects millions globally each year, with mortality rates ranging from 16.7% to 33.3%. 2 The disease triggers a complex interplay of pro- and anti-inflammatory pathways, with the balance between these responses determining the patient’s prognosis. 3 In sepsis, prolonged over-inflammation coupled with immunosuppression prevents the body from returning to homeostasis, leading to multiple organ failure. 4 Early in the disease, both inflammatory responses are activated simultaneously, and this immune reaction is further amplified by endogenous factors. As sepsis progresses, pathogenic microorganisms and their metabolites colonize the gastrointestinal tract and enter the peritoneal cavity, triggering an immune-inflammatory response to clear the infection. 5 However, if the infection is not controlled, the condition worsens, leading to severe complications.
An appropriate inflammatory response is essential for a favorable prognosis in sepsis, and the main purpose of activating the inflammatory response is to clear the bacterial load and prevent the spread of pathogens. 6 With regard to the course of the inflammatory response in sepsis, it has been determined that death in cecal ligation puncture method (CLP)-induced sepsis may be due to prolonged hyperinflammation, whereas death in the later stages of the disease may be due to immune-suppression resulting in bacterial overgrowth that is not capable of clearing the existing infection. Therefore, effective control of infection and prevention of propagation and spread of pathogenic bacteria is undoubtedly essential to stop the progression of the disease. In experimental sepsis and human sepsis, complications may be mitigated by therapeutic interventions that reduce the levels of proinflammatory mediators or restore degenerative adaptive and innate immune responses. 7 Macrophages play an important role in sepsis in regulating host immune homeostasis and inflammatory responses. 8 Macrophage polarization is a dynamic process in which macrophages are stimulated by changes in the specific tissue microenvironment and develop different functional phenotypes. 9 The main phenotypes known to date are inflammatory or classically activated (M1) macrophages and healing or selectively activated (M2) macrophages. Targeted modulation of macrophage polarization and phenotypic alterations to adapt to the microenvironment may be an effective therapeutic approach for the treatment of sepsis. 10 In order to ameliorate the systemic inflammatory response in sepsis, it is particularly important to explore pharmacological therapies that enhance the body’s bacterial clearance capacity by modulating macrophage phagocytosis polarization and thereby improve sepsis outcomes.
The development of targeted therapies aimed at regulating immune balance has emerged as a pivotal focus in sepsis research. Ginkgolide B (GB), a terpenoid compound extracted from the leaves of Ginkgo biloba, is a naturally occurring bioactive ingredient in ginkgo leaf extracts. It exhibits a broad range of pharmacological activities, including anti-platelet aggregation, anti-inflammatory, antioxidant, anti-shock properties, and free radical scavenging effects.11,12 Previous studies have indicated that GB exerts protective effects on the body’s inflammatory response; however, the precise mechanisms through which it alleviates inflammation induced by sepsis remain unclear. In this study, we investigated the U-shaped dose-dependent modulation of macrophage function by GB, focusing on its effects on bacterial phagocytosis and inflammatory responses. We further examined how GB influences systemic inflammation and multi-organ injury in septic rats, providing mechanistic insight into its potential therapeutic value for sepsis management.
Material and Methods
Cultivation of RAW264.7 Cells
To investigate the effect of GB on macrophage function, in vitro experiments were initially conducted using RAW264.7 cells. These cells were obtained from Bide Biotechnology Co., Ltd. and cultured in RAW264.7-specific medium, which consisted of DMEM-H, supplemented with 10% fetal bovine serum (FBS), 1% Glutamax, 1% Sodium Pyruvate, and 1% Penicillin-Streptomycin (P/S). Cells were passaged every 2-3 days, selecting those in optimal growth conditions for experimentation. For the experiments, cells were seeded in 6-well plates, with a density of 1 × 106 cells per well. The cells were then divided into two experimental groups: the non-treated group and the GB group (cultured with 40 μM GB). The RAW cells are semi-adherent and, therefore, do not require trypsin digestion. Instead, we used a pipette to gently aspirate and blow across the bottom of the culture dish, which effectively detaches the cells from the substrate without enzymatic treatment.
Macrophage Bacterial Phagocytosis Assay
For the macrophage phagocytosis assay, RAW264.7 cells from each group were seeded in 6-well plates at a density of 1 × 10^6 cells per well and cultured under standard conditions until they reached 70-80% confluence. The cells were then incubated with 1 × 10^6 colony-forming units (CFUs) of fluorescently labeled Escherichia coli for 2 h at 37°C in a 5% CO2 incubator. The bacteria were pre-labeled with FITC to allow for easy detection via flow cytometry.
After the incubation period, the cells were harvested and washed three times with phosphate-buffered saline (PBS) to remove any non-phagocytosed bacteria. The macrophages were then detached using cell scrapers to avoid disruption of cell membranes. Following detachment, the cells were resuspended in PBS and immediately analyzed by flow cytometry to quantify the percentage of cells exhibiting phagocytosis, as indicated by the uptake of FITC-labeled E. coli. The phagocytic ability of each group was assessed based on the percentage of cells positive for bacterial uptake.
For the polarization analysis, each group of cells was incubated with 2 μL of the following flow cytometry antibodies: CD68 (BioLegend, PE conjugated, 1:100), CD86 (BioLegend, FITC conjugated, 1:100), and CD163 (BioLegend, APC conjugated, 1:100). The cell suspension was gently mixed to ensure proper antibody binding to the target molecules on the cell surface. The staining reaction was carried out in the dark and incubated at room temperature for 30 min. Following incubation, 1 mL of PBS was added to the cells, and the mixture was gently inverted to wash away any unbound antibodies. The cells were then centrifuged at 1800 rpm for 5 min at 4°C to collect the cell pellet. After washing, the cells were resuspended in 100 μL of PBS to obtain a single-cell suspension and analyzed using a flow cytometer.
Experimental Animals and Grouping
The experimental animals were purchased from Shanghai Bikai Keyi Biotechnology Co., Ltd. After a 1-week acclimatization period, the experiments were conducted. A total of 40 male Sprague-Dawley rats, weighing between 200-250 g, were randomly assigned to five groups: the Healthy group, the cecal ligation puncture method (CLP) model group, the GB low-dose treatment group (5 mg/kg), the GB medium-dose treatment group (10 mg/kg), and the GB high-dose treatment group (20 mg/kg). Furthermore, an additional 40 rats were treated in the same way to observe their survival over a period of 7 days. All experimental procedures were performed in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals, and the study was approved by the Ethics Committee of Laboratory Animal Use at Jinling Hospital, Nanjing University of Chinese Medicine. The animal experiments were conducted from September 2023 to June 2024, and the cell culture experiments were performed from November 2023 to December 2024. Based on prior experience with the CLP rat model, which has an expected mortality rate of 40-50%, and to ensure sufficient statistical power for one-way ANOVA analysis, eight rats were included in each group. This sample size was considered appropriate to balance scientific rigor and ethical considerations.
Sepsis Animal Model Construction of CLP
Sepsis was induced in rats using the CLP method. 13 Initially, the rats were anesthetized with isoflurane gas and placed on a surgical table. After shaving and disinfecting the surgical area, a 2 cm midline incision was made in the lower abdomen. The skin and subcutaneous tissues were incised sequentially to access the peritoneal cavity. Under sterile conditions, the cecum was exposed, and a 4-0 silk suture was used to ligate the cecum 1 cm from its distal end. A 20-gauge needle was then used to puncture the ligated cecum once, taking care not to damage the mesentery or blood vessels of the intestinal wall. Subsequently, the cecum was carefully returned to the abdominal cavity, and a gentle squeeze allowed a small amount of feces to leak into the peritoneal cavity. The incision site was disinfected, and the abdominal wall was sutured back in place. Post-surgery, the rats were placed on heating pads and treated with 1 mL of saline subcutaneously for shock management.
GB was obtained from MedChemexpress, USA (New Jersey). After CLP modeling, GB was administered via intraperitoneal injection at a volume of 1 mL at 1 h and 12 h post-surgery, with varying concentrations of GB. The control and sepsis groups received the same volume of saline at the corresponding time points. To evaluate the role of GB in bacterial clearance, all treatment groups were not given antibiotics. Survival rates were monitored for all rats, and survival curves were plotted. Additionally, peritoneal lavage fluid (PLF), blood, and tissue samples were collected 24 h post-surgery, just before euthanizing the animals.
Vital Sign Monitoring and Blood Gas Analysis
The heart rate, respiration rate, pulse, and body temperature of the rats in each experimental group were continuously monitored at 0, 1, 12, and 24 h post-treatment using a comprehensive animal monitoring system. The monitoring system was calibrated according to the manufacturer’s guidelines to ensure accurate readings. Specifically, heart rate and pulse were measured using a non-invasive pulse oximeter, respiration rate was recorded via a respiratory rate monitor, and body temperature was assessed using a rectal thermometer. All data were collected in real time and analyzed to track physiological changes over the 24-h period.
At 24 h post-modeling, the rats were deeply anesthetized using isoflurane, and the depth of anesthesia was confirmed by a lack of response to toe pinching. Arterial blood was then collected via abdominal aorta puncture, with care taken to minimize discomfort and ensure accurate sampling. The blood samples were immediately analyzed using a blood gas analyzer to assess critical parameters, including pH, lactate levels, partial pressure of oxygen (PaO2), partial pressure of carbon dioxide (PaCO2), and bicarbonate concentration (HCO3−).
The HE Staining
At the conclusion of the experiment, heart, liver, and lung tissues were harvested from the rats in each group. The tissues were carefully excised and immediately fixed in 3% formaldehyde at 4°C for 24 h to ensure adequate preservation of cellular structures. After fixation, the tissues were thoroughly washed with distilled water to remove excess fixative, followed by a series of dehydration steps. The dehydration was performed using a graded series of ethanol solutions (70%, 80%, 90%, 100%) for 1 h each, and then the tissues were cleared with xylene for 2 h to remove ethanol.
Subsequently, the dehydrated tissues were embedded in paraffin using a tissue embedding system. Paraffin infiltration was performed at 60°C for 12 h to ensure complete impregnation of the tissues. Once embedded, the tissues were sectioned into 5 µm thick slices using a rotary microtome, with each section mounted onto glass slides. The sections were then dried on a heating plate at 37°C for 1 h to remove excess paraffin.
For histopathological analysis, the tissue sections were subjected to hematoxylin and eosin (H&E) staining. The staining procedure involved the following steps: (1) the sections were first deparaffinized using xylene, followed by rehydration through decreasing concentrations of ethanol; (2) the slides were then stained with hematoxylin for 5 min, washed with water, and differentiated in acid alcohol; (3) the sections were stained with eosin for 2-3 min, rinsed in water, dehydrated with ethanol, and cleared with xylene. The stained sections were then mounted with a coverslip using a mounting medium.
The entire section was scanned for a comprehensive analysis, and representative images were captured for further documentation and comparison between the experimental groups.
Collection of Peritoneal Lavage Fluid and Bacterial Culture
Twenty-four hours after CLP modeling, the rats were deeply anesthetized, and a midline abdominal incision was made to expose the peritoneal cavity. The abdomen was irrigated with 3 mL of PBS, and the peritoneum was gently massaged to collect the PLF. Afterward, the PLF was centrifuged at 1000 rpm for 1 min to collect the supernatant.
24 h after CLP modeling, rats were deeply anesthetized, and a midline incision was made to expose the abdominal cavity. The peritoneum was gently massaged and irrigated with 3 mL of sterile PBS to collect the peritoneal lavage fluid. The collected fluid was then centrifuged at 1000 rpm for 1 min to collect the supernatant. To further assess whether GB enhances macrophage-mediated bacterial clearance, we measured bacterial colony-forming units (CFUs) in PLF. Samples were serially diluted in sterile PBS (1:10 to 1:100), and 100 μL of each dilution was plated on 5% sheep blood tryptic soy agar plates. The plates were incubated at 37°C for 24 h, and the CFUs were counted to evaluate bacterial load.
Enzyme-Linked Immunosorbent Assay (ELISA)
The arterial blood samples were promptly placed on ice, and serum was separated by centrifugation at 3000 rpm for 5 min. The collected serum and PLF were analyzed using ELISA kits following standard protocols to measure interleukin-6 (IL-6), interleukin-10 (IL-10), and tumor necrosis factor-alpha (TNF-α) levels.
Briefly, 100 µL of serum or PLF sample was added to each well of the ELISA plate pre-coated with specific antibodies against IL-6, IL-10, or TNF-α. After incubation for 2 h at room temperature, the wells were washed with PBS and incubated with a secondary antibody conjugated to an enzyme for 1 h at room temperature. Following a second wash, the enzyme substrate was added, and the color change was monitored at 450 nm using a microplate reader. The concentration of each cytokine was determined by comparing the optical density (OD) values to a standard curve generated from known cytokine standards.
Statistical Analysis
The statistical analyses and graphical presentations in this study were conducted using GraphPad Prism 10 software. For comparisons among multiple groups, one-way analysis of variance (ANOVA) was performed, followed by post-hoc testing to evaluate pairwise differences. Specifically, Tukey’s multiple comparisons test was applied to adjust for multiple testing and reduce the risk of false positives. A P-value of less than .05 was considered statistically significant. All data are presented as mean ± standard deviation (SD), and the results were considered robust only if they met the criterion for statistical significance after appropriate corrections.
Results
GB Enhances Bacterial Phagocytic Ability of Macrophages
First, we explored the effect of GB on the bacterial clearance ability of macrophages through in vitro experiments. The proportion of RAW264.7 cells that had phagocytosed fluorescently labeled bacteria was measured by flow cytometry (Figure 1A). The results indicated that GB co-culture significantly enhanced the bacterial phagocytic ability of macrophages (P = .00198). Furthermore, we assessed the polarization status of macrophages in each group by flow cytometry. GB treatment significantly increased the proportion of M1 macrophages (P = .00431 and P = .00780) (Figures 1BandC). MTT assay results demonstrated that within the concentration range of 0 μM-40 μM, GB alone did not significantly affect the viability of RAW264.7 cells. The highest cell viability was observed at a concentration of 40 μM (Figure 1D). Effects of GB on bacterial phagocytosis and polarization characteristics in RAW264.7 cells. (A) Phagocytic ability of macrophages in each group, with the positive rate representing the bacterial phagocytosis capacity. (B) M1 polarization ratio of cells in each group, with CD86 as a marker for M1 polarization. (C) M2 polarization ratio of cells in each group, with CD206 as a marker for M2 polarization. (D) Cell survival was assessed following treatment with varying concentrations of GB. n = 3, *P < .05
GB Reduces Mortality in Septic Rats
We successfully established a septic rat model using the CLP method. The effect of GB on intra-abdominal infection in septic rats was first evaluated by recording the 7-day survival rates in different treatment groups. We found that CLP significantly increased the 7-day mortality in septic rats, with the majority of deaths occurring within the first 3 days. Interestingly, all three doses of GB (5 mg/kg, 10 mg/kg, and 20 mg/kg) improved the 7-day survival rate of septic rats. Specifically, the survival rates were 62.5% in both the GB 5 mg/kg and GB 20 mg/kg groups, and 75% in the GB 10 mg/kg group, all of which were significantly higher than the 50% survival rate observed in the CLP group (Figure 2A). GB modulates systemic and peritoneal inflammatory responses and improves survival in CLP-induced septic rats. (A) Survival curves of rats in each group. (B and C) GB downregulates the expression levels of TNF- α, and IL-6 in the PLF and peripheral serum of rats with sepsis induced by CLP. Additionally, the GB5 group significantly increased the serum IL-10 levels. The levels of TNF- α, IL-6, and IL-10 in both peritoneal lavage fluid and peripheral blood were measured using ELISA. n = 5, *P < .05
GB Treatment Alleviates Pro-inflammatory Response and Enhances Anti-inflammatory Response in Septic Rats
We measured the levels of pro-inflammatory cytokines TNF-α and IL-6, as well as the anti-inflammatory cytokine IL-10, in the PLF and peripheral blood of septic rats 24 h after CLP surgery (Figure 2B and C). Compared to the CLP group, the levels of TNF-α and IL-6 in both the PLF and Serum of the GB5 group significantly decreased 24 h post-surgery. However, the IL-10 levels in the PLF of the GB5 group markedly increased, and the GB medium-dose group exhibited a more pronounced anti-inflammatory effect (P < .001). The effects of different concentrations of GB on inflammatory cytokines varied. Notably, the GB10 group exhibited the lowest levels of pro-inflammatory factors among all GB-treated groups, while anti-inflammatory factors were expressed at the highest levels in this group. These differences were statistically significant.
Ginkgolide B Alleviates Multiple Organ Dysfunction Syndrome (MODS) in Septic Rats
Heart, liver, and lung tissues from all five groups of rats were collected 24 h after CLP surgery. As shown in Figure 3, the organ structures of rats in the Healthy group appeared normal with no histopathological changes. In contrast, after CLP, the heart, liver, and lung tissues of sepsis rats exhibited significant histopathological alterations. Myocardial cells showed disorganized and irregular alignment, with interstitial congestion and edema. Hepatocytes were swollen, with enlarged nuclei, pigmentation, and extensive infiltration of inflammatory cells in the hepatic portal area. The alveolar spaces were markedly widened, accompanied by inflammatory cell infiltration. After GB treatment, the myocardial interstitial congestion and edema were alleviated, hepatocyte swelling and inflammatory cell infiltration in the hepatic portal area were reduced, and lung tissue edema improved with a significant reduction in cellular infiltration. The GB 10 mg/kg group exhibited the most pronounced therapeutic effect. These results suggest that GB treatment significantly alleviated the MODS induced by CLP-induced sepsis in rats. GB mitigates histopathological injury in heart, liver, and lung tissues of CLP-induced septic rats. (A) GB alleviates pathological damage in the heart, liver, and lung tissues of septic rats induced by CLP (n = 5). Tissue samples from the heart, liver, and lungs of five groups of rats were collected 24 h after CLP surgery. Pathological sections stained with hematoxylin and eosin were compared under a microscope, with a scale bar of 100 μm. Original magnification × 200. Red arrow, hemorrhagic foci. Black arrow, necrotic cells. Blue arrow, edema. Five rats per group
GB Improves Acid-Base Imbalance in Septic Rats
After treatment with varying doses of Ginkgolide B (GB), the acidotic condition in septic rats was notably improved. Specifically, GB treatment led to a dose-dependent reduction in blood lactate levels, restoration of PaO2, and normalization of PaCO2 (Figure 4A-C). Furthermore, the pH, HCO3−, and BE levels were significantly improved, demonstrating GB’s potential to ameliorate the metabolic disturbances associated with sepsis (Figure 4D-F). These findings suggest that GB not only helps correct the acid-base imbalance but also enhances oxygenation and reduces systemic metabolic stress in septic rats. GB modulates arterial blood gas parameters and metabolic status in CLP-induced septic rats. (A, B, and C) Effects of Ginkgolide B on blood lactate levels, partial pressure of oxygen (PaO2), and partial pressure of carbon dioxide (PaCO2) levels in the arterial blood of septic rats. (D, E, and F) Effects of Ginkgolide B on pH, HCO3 and BE levels in the arterial blood of septic rats. The changes in arterial blood gas during the experimental period following the injection of saline or different doses of Ginkgolide B after sepsis induction. n = 5, *P < .05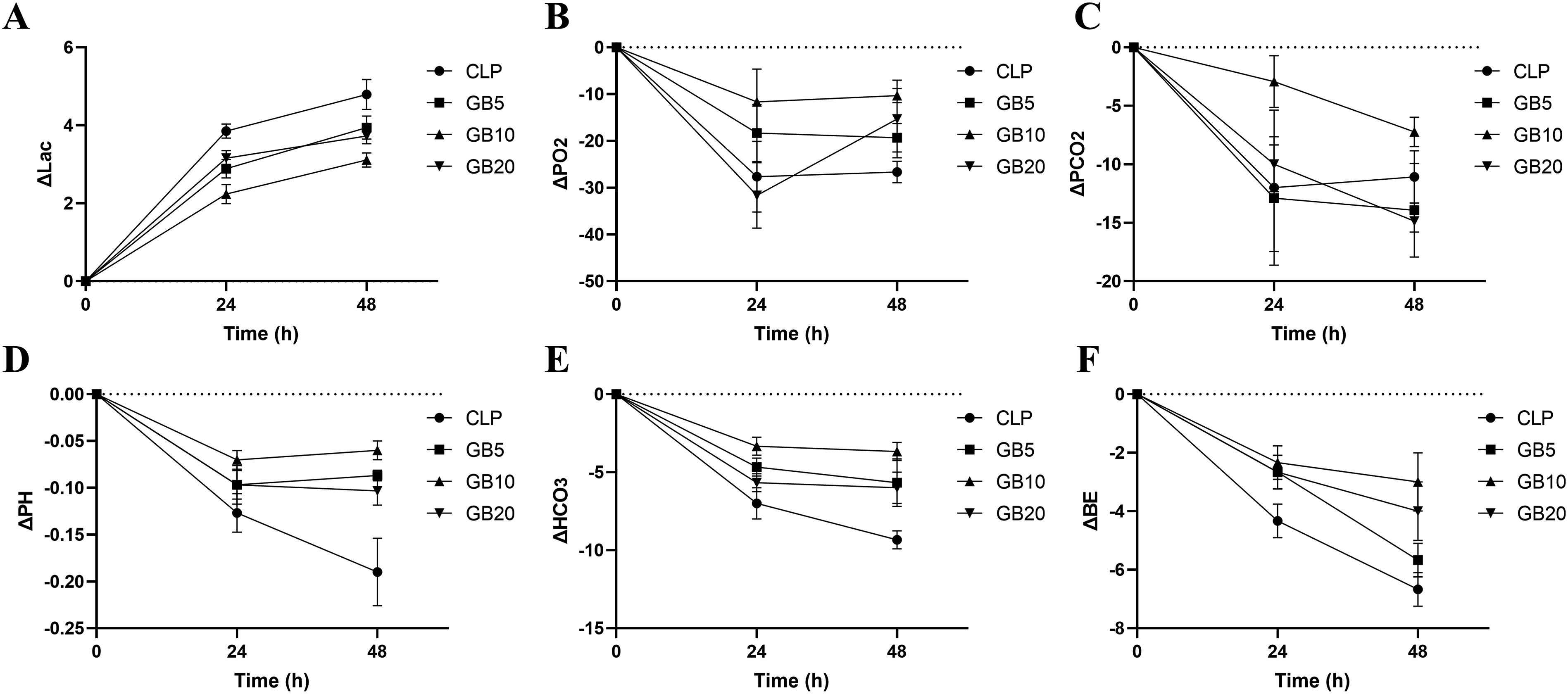
GB Improves Bacterial Clearance in Intra-Abdominal Sepsis
We measured the colony count in the PLF of rats in each group to verify whether GB enhances macrophage-mediated clearance of intra-abdominal bacteria, thereby improving survival in septic rats. Compared to the CLP group, the colony counts in the PLF and peripheral blood of the GB-treated rats decreased 24 h after surgery, with the GB5 group showing the most significant reduction (P < .001) (Figure 5AandB). GB enhances bacterial clearance in CLP-induced septic rats. (A) GB improved bacterial clearance in the PLF of rat with CLP-induced sepsis (n = 5). (B) GB improved bacterial clearance in the blood of rat with CLP-induced sepsis (n = 5). The number of colonies formed in each plate was measured in CFUs. n = 5, *P < .05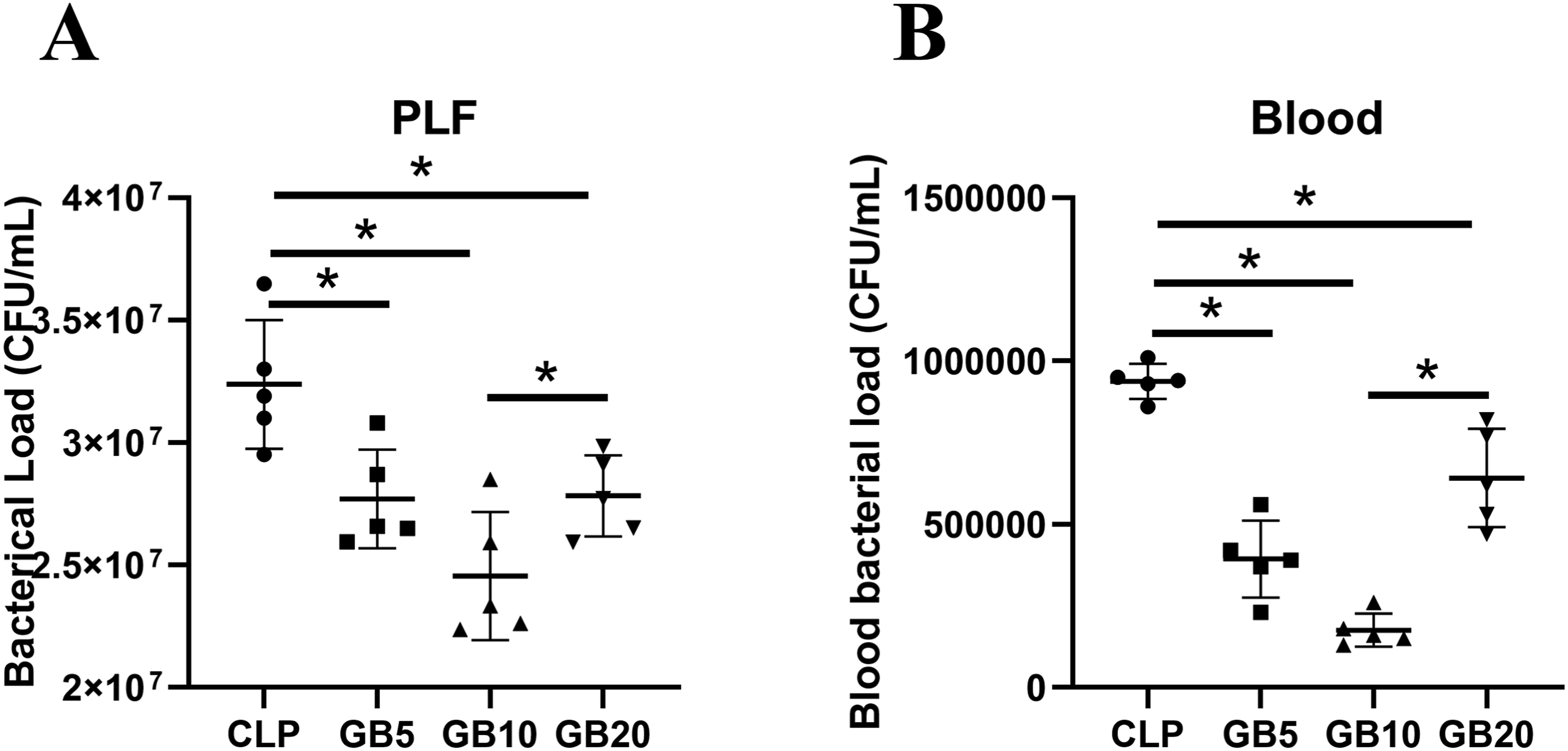
Discussion
In the case of severe abdominal infections, factors such as endotoxins, inflammatory mediators, and free radical damage work synergistically to impair the intestinal mucosal barrier, leading to bacterial translocation and endotoxin release, which further contributes to the development of sepsis. 14 Currently, clinical management of abdominal infections and sepsis often involves a combination of strategies, including fluid resuscitation, organ function support, aggressive management of the infection source, appropriate use of antibiotics, prevention of nutritional and metabolic complications, and immunosuppressive treatment. 15 However, the increasing antibiotic resistance of bacteria poses a significant challenge in the treatment of abdominal infection-induced sepsis. 16 The development of effective traditional Chinese medicine components for anti-infection therapy may help combat antibiotic-resistant bacteria and improve survival rates in sepsis patients. In this study, we found that GB exerts a U-shaped dose-dependent modulation of macrophages, which mitigated systemic inflammation in septic rats, reduced multi-organ tissue damage, and improved survival. These protective effects were associated with a decrease in pro-inflammatory cytokines (TNF-α and IL-6) and an elevation of the anti-inflammatory cytokine IL-10. These findings provide evidence for the clinical potential of TCM in treating sepsis and highlight its potential therapeutic targets.
GB, an active bioactive component of Ginkgo biloba, is widely used as a cardiovascular treatment in Asian countries. 17 Existing studies have reported the anti-inflammatory value of GB. In Alzheimer’s disease, GB has been found to prevent Aβ-induced neuroinflammation and neurotoxicity in various neurodegenerative diseases by inhibiting the activation of the NLRP3 inflammasome, thereby suppressing the progression of Alzheimer’s disease. 18 In pulmonary infections, GB reduces TRIM37 expression by antagonizing the PAF-R pathway, which in turn inhibits the activation of NF-κB and alleviates the inflammatory response in alveolar epithelial cells. 19 Consistent with these studies, our research demonstrates that GB can alleviate systemic inflammation in septic rats and improve the 7-day survival rate of these rats.
Sepsis is characterized by elevated levels of inflammatory cytokines such as TNF-α, IL-1β, IL-6, and nitric oxide (NO). 20 IL-10, an important anti-inflammatory mediator, plays a crucial role in protecting the host from excessive immune responses to pathogens and the microbiota. 21 The immune system has evolved to combat infections while minimizing harm to the host, and IL-10 has been shown to regulate the delicate balance between effective immune responses and tissue damage. 22 Excessive production of pro-inflammatory cytokines significantly impacts the treatment of sepsis. Therefore, inhibiting inflammatory cytokines and enhancing anti-inflammatory cytokines may represent a promising strategy for treating sepsis. Recent studies indicate that GB can exert anti-inflammatory effects by modulating the levels of TNF-α, IL-6, and other inflammatory factors, protecting the nervous system, and alleviating neuropathic pain. 23 In this study, GB was shown to reduce serum levels of TNF-α and IL-6 while increasing IL-10 levels. These results suggest that GB can regulate the inflammatory cytokine network to maintain the balance between pro-inflammatory and anti-inflammatory responses, thereby alleviating visceral inflammation.
Sepsis causes systemic damage, leading to multi-organ dysfunction, including in the cardiovascular, hepatobiliary, and respiratory systems. 24 Therefore, this study comprehensively assessed the pathological changes in the function of multiple organs, such as the heart, liver, and lungs, following sepsis, and suggested a protective role of GB against organ damage. As the main component of Ginkgolide, GB has been shown to play a role in alleviating lipopolysaccharide-induced lung injury. 25 In an acetaminophen-induced acute liver injury model in rats, GB attenuated acute liver damage by antagonizing platelet-activating factor. 26 Similarly, studies have demonstrated that GB protects against myocardial inflammatory damage induced by ischemia/reperfusion through modulation of the A20-NF-κB pathway. 27 These studies collectively highlight the protective effects of GB in various organ injuries.
Sepsis causes rapid damage to the body’s vital signs. In this study, septic rats showed a decrease in pH, HCO3−, and BE levels in arterial blood gases 24 h after surgery. After GB intervention, the acidotic levels were partially alleviated within 24 h, and by 48 h, a significant improvement in acidosis was observed, with arterial blood gases approaching balance. This improvement may be due to the adjustment and balance of inflammatory cytokine levels in septic rats, especially the modulation of IL-6 and IL-10 levels. These findings are consistent with previous reports that highlight the correlation between acidosis and inflammatory cytokines.28,29
The CLP-induced sepsis model is initiated by abdominal infection, making innate immunity in the peritoneum crucial. Macrophages are a key component of the innate immune system, playing a broad range of biological roles, including phagocytosis of dead cells, invading pathogens, and virus-infected cells. 30 Under stable conditions, peritoneal macrophages exist in a free form within the peritoneal fluid. 31 Early in sepsis, macrophages serve as the first line of defense. 8 After infection, macrophages can differentiate into two types under different environmental stimuli: pro-inflammatory M1 macrophages and anti-inflammatory M2 macrophages. Maintaining an appropriate balance between these types is critical for preserving homeostasis during inflammation. 32 During the early stages of infection, macrophages play an essential role in bacterial phagocytosis. Our study showed that after GB treatment, the CFU levels in the PLF of septic rats significantly decreased, suggesting that GB enhances macrophage bacterial phagocytosis. Effective suppression of early peritoneal infection can directly reduce the systemic inflammatory response and improve survival rates in experimental animals. Previous studies have suggested that sepsis prognosis may be related to the recruitment and proper activation of phagocytes in injured tissues. 33 During the early stages of infection, macrophages play an essential role in bacterial phagocytosis. Our study showed that GB treatment significantly reduced CFU levels in the PLF of septic rats, suggesting that GB enhances macrophage bacterial phagocytosis. Importantly, only appropriate doses of GB resulting in greater reductions in bacterial burden. Effective suppression of early peritoneal infection can directly reduce the systemic inflammatory response and improve survival in experimental animals. Therefore, we hypothesize that following sepsis, GB improves sepsis prognosis by enhancing the bacterial phagocytosis function of macrophages. Enhancing macrophage phagocytic capacity may contribute to the recovery after sepsis.
This study has several limitations. First, although we observed that GB enhances the bacterial phagocytosis function of macrophages, the detailed mechanisms underlying this effect remain to be further elucidated. Additionally, while GB effectively alleviated the systemic inflammatory response in septic rats, its effects on other immune cells are not yet clear. Finally, due to the high mortality rate in the CLP-induced sepsis model, the long-term effects of GB on sepsis could not be observed.
Conclusion
In summary, our study demonstrates that GB exerts a U-shaped dose-dependent modulation of macrophage function, enhancing bacterial phagocytosis and restoring immune balance. In septic rats, early peritoneal administration of GB effectively mitigated systemic inflammation, alleviated multi-organ injury, and improved survival. These findings highlight the mechanistic basis and therapeutic potential of GB as a natural agent for sepsis management.
Supplemental Material
Supplemental Material - U-Shaped Dose-Dependent Modulation of Macrophages by Ginkgolide B Mitigates Systemic Inflammation in Septic Rats
Supplemental Material for U-Shaped Dose-Dependent Modulation of Macrophages by Ginkgolide B Mitigates Systemic Inflammation in Septic Rats by Zhaofeng Luo, Lan Ming, Jiamin Ji, and Qian Huang in Dose-Response
Footnotes
Ethical Considerations
Animal protocols were approved by Eastern Theater Command General Hospital Experimental Animal Ethics Committee (2023JLHGZRDWLS-00087), Nanjing, China, and complied with the revised Animals (Scientific Procedures) Act 1986 in the UK and Directive 2010/63/EU in Europe.
Author Contributions
Zhaofeng Luo: Conceptualization, Methodology, Writing- Original draft preparation. Lan Ming: Visualization, Investigation. Jiamin Ji: Software, Validation. Qian Huang: Writing- Reviewing and Editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Grant No. 82070579 to Qian Huang).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
