Abstract
Background
Radiation-induced lymphopenia (RIL), particularly severe RIL (G3+), significantly impacts cancer outcomes. This retrospective study aimed to determine the role of irradiation dose to the thymus on RIL in breast cancer (BC) patients who received adjuvant radiotherapy (RT).
Methods
BC patients who received adjuvant RT in 2019 and had available complete blood count data were included. RIL was defined according to CTCAE v5.0 criteria. Logistic regression identified thymic dosimetric and clinical parameters linked to G3+ RIL, ROC analysis determined optimal protective thresholds, and Delong test compared model performance.
Results
The data of a total of 93 consecutive patients were retrospectively retrieved, with 37.6% (35/93) experienced G3+ RIL. Logistic regression analysis identified thymic dosimetric factors (mean dose, V5, V10), baseline ALC, RT technique and IMNI were associated with G3+ RIL. ROC analysis showed that a combined model of baseline clinical and thymic dosimetry parameters had the highest AUC (0.869). The optimal cutoffs for avoiding G3+ RIL were baseline ALC
Conclusions
Thymic radiation dose correlated with severe lymphopenia in breast cancer radiotherapy. Prospective trials are needed to validate these dose constraints for clinical use.

Keywords
Introduction
As the most radiosensitive cells in the human body, circulating lymphocytes are highly vulnerable to depletion following radiation exposure. This results in radiation-induced lymphopenia (RIL), a well-recognized and frequently unavoidable complication of radiotherapy (RT).1,2 Extensive evidence indicates that RIL correlates with adverse clinical outcomes, including diminished tumor control and reduced survival rates in various cancers.3,4 Notably, severe RIL (≥ Grade 3, G3+) demands special attention, as various studies demonstrate its significant association with inferior progression-free and overall survival compared to lower-grade RIL (Grade 0-2) in RT-treated solid tumor patients.5,6 Although RIL has gained increasing research attention in recent years, critical gaps persist in understanding its pathophysiology, risk factors, and potential interventions. 7
Based on 2022 global cancer statistics, breast cancer (BC) remains the most common malignancy in women. 8 Adjuvant RT targeting the breast, chest wall, and/or regional lymph nodes significantly reduces locoregional recurrence risk and enhances survival in high-risk patients.9,10 Despite its recognized association with poor prognosis, radiation-induced lymphopenia (RIL) remains a frequent occurrence in breast cancer (BC) patients, and its severity exhibits a clear multifactorial nature.11-13 The thymus, located within the radiation field during thoracic RT, is essential for T-lymphocyte production but highly vulnerable to radiation damage.14,15 Despite this, few studies have examined the dosimetric effect of thymus on RIL in BC patients who received adjuvant RT.16-19
Thus, this current retrospective study aimed to explore the associations between thymic dose-volume metrics and RIL, while identifying thymic dose-volume constraints for preventing RIL of BC patients undergoing adjuvant RT.
Methods and Materials
Patient Population
This retrospective study included consecutive BC patients who had undergone postoperative RT at our hospital in 2019. This study was approved and conducted according to the ethical principles established by our hospital review board (No. 2023-052). The eligibility criteria were as follows: histopathologically proven BC; age ≥18 years; female sex; Karnofsky performance status score ≥70; and available medical records and laboratory test results. The exclusion criteria were local-regional recurrent BC disease; receipt of endocrine or targeted therapy or immunotherapy administered before or concurrent with RT; and a history of thoracic RT or surgery and immune- or haematological-related diseases. The routine clinical procedure for treating BC patients at our hospital was developed according to the NCCN guidelines (Version 3. 2018). 20 The operations included breast-conserving therapy or mastectomy, and the axillary lymph nodes surgeries included typical dissection, sampling at levels I and II, or the sentinel lymph node biopsy. The typical chemotherapy regimen consisted of anthracycline and/or taxane prior to RT.
RT Planning and Dosimetric Analysis
Following their initial evaluation, patients underwent computed tomography scanning (GE, Discovery, CT 590-RT) with axial image thicknesses of 2-5 mm. These CT images were subsequently sent to a treatment planning system (Elekta, Pinnacle RTP System 9.0. or Varian, Eclipse Version 15.6.). The RT techniques employed included three-dimensional conformal RT (3D-CRT), intensity-modulated RT (IMRT) or 3D-CRT hybrid IMRT (Hy-IMRT) with 6-MV X-ray, with protocols developed according to the guidelines for RT for BC.
21
The delineated clinical target volumes including for breast or chest wall and the locoregional lymphatics (supraclavicular area, axillary fossa and/or internal mammary chain), were delineated according to the surgical plan and pathological examination results. The radiation dose prescribed for the planning target volume was 45-50 Gy, and most patients who underwent breast-conserving operations received an additional tumour bed boost of 10-15 Gy, all with conventional fractionation. The thymus of all eligible patients were retrospectively contoured on the CT simulation images using the aforementioned planning system. The thymus was delineated along its anatomic borders located on the mediastinal window by a single investigator for consistency across patients (Figure 1).
22
Without changing any of the RT dosimetric parameters in the original treatment plan, the thymic dose-volume histograms (DVH) parameters of each patient was calculated by a medical physicist. Contour atlas of the thymus in axial coronal and sagittal section. (A) Thymus in coronal section. (B) Thymus in sagittal section. (C and D) Thymus behind manubrium, just under the left brachiocephalic vein. (E and F) Thymus lies behind sternum, anterior to the aortic arch and its branches, and rests on the top of pericardium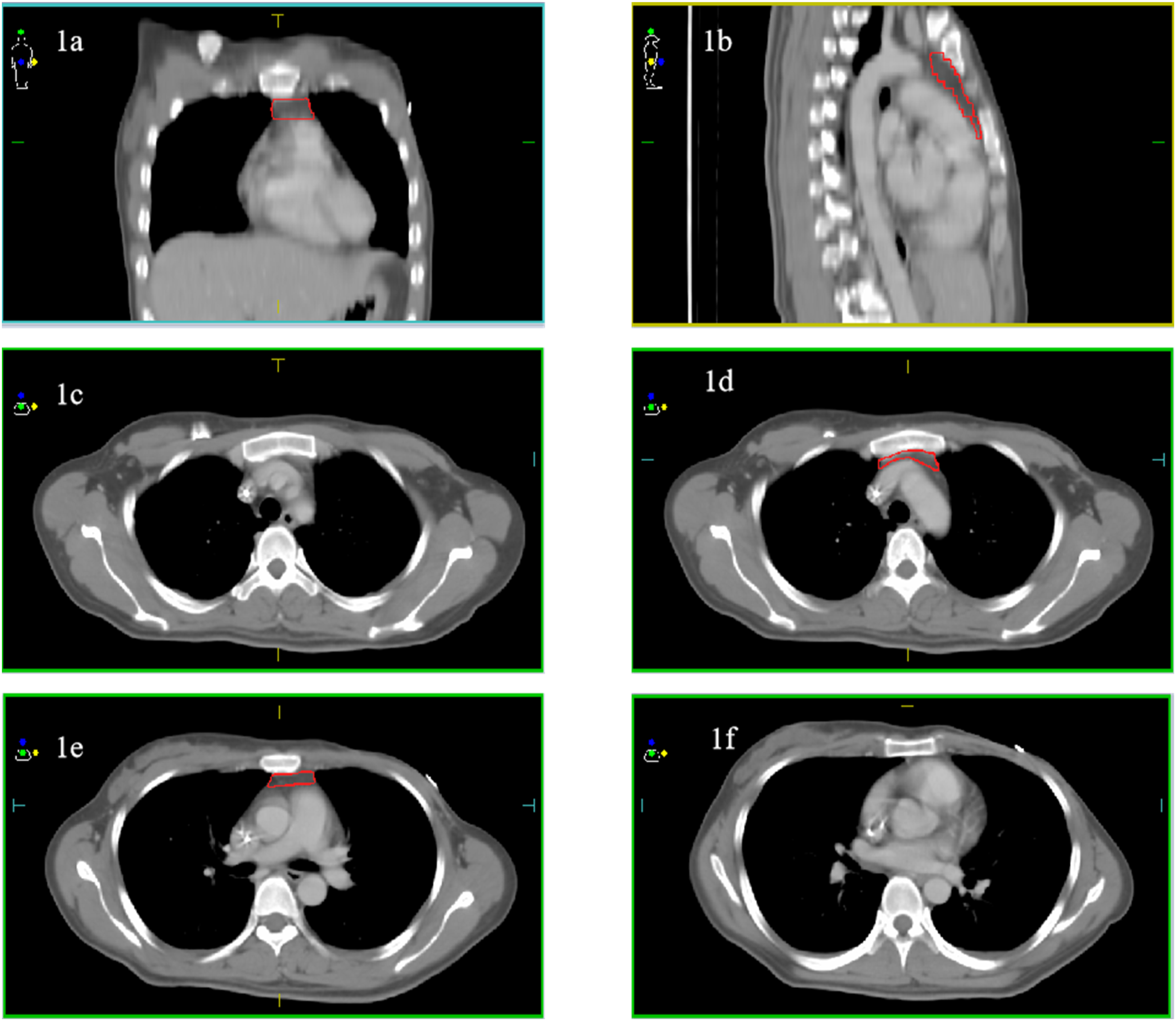
Lymphopenia Diagnosis and Severity Rating
Routine blood tests were performed 1 week before, each week during and at the end of RT. The complete blood cell count, including the absolute lymphocyte count (ALC), was measured with an automated cell counting machine (BC 6800, Sysmex XN3000, China). In our hospital, the cut-off value for the lower limit of normal for ALC was 1.1 × 109/L. Lymphopenia was determined according to the lowest lymphocyte count over the entire RT course and classified as grade 1 to 4 according to the common terminology criteria of adverse events (CTCAE) (version 5.0). 23 According to the CTCAE, a value of 0.5 × 109/L as the cut-off for categorizing patients as having either none/mild (normal ALC and grade 1-2, eg, ≥0.5 × 109/L) or severe (grade 3-4, eg, <0.5 × 109/L) lymphopenia. The endpoint of this study was ≥ Grade 3 (G3+) RIL, which was defiined as ALC <0.5 × 109/L.
Data Collection and Statistical Analysis
Baseline clinical and dosimetry parameters were collected from medical records, including tumour laterality, tumor stage, surgical approaches, chemotherapy (strategy, regimens and number of cycles), the RT technique and the DVH parameters of thymus, the baseline ALC before RT.
Descriptive statistics included the medians and interquartile ranges (IQR) for continuous variables and frequencies and percentages for categorical variables. The paired-sample t test was used to compare the differences in continuous variables between groups. The independent samples t-test or Mann–Whitney U-test was used for intergroup comparisons of continuous variables. Logistic regression models were employing G3+ RIL as the dependent variable were developed to evaluate the associations of G3+ RIL with patient characteristics and radiation parameters. Before the multivariable logistic stepwise regression analysis, a linear regression model was used to examine the collinearity among thymic dosimetric variables factors that showed significant differences (P < 0.05) in the univariate analysis. The degree of collinearity was quantified using the variance inflation factor (VIF), with a threshold of VIF <10 signifying acceptable collinearity levels. Receiver operating characteristic (ROC) curves were used to evaluate cutoff values of parameters to avoid G3+ RIL. The Delong test was performed to compare differences in the area under the ROC curves (AUCs) between different models. Statistical analyses were performed with SPSS Statistics (version 25). A 2-sided P value <0.05 was used in all the statistical tests.
Results
Patient Characteristics
Patient, Tumour and Treatment Characteristics (N = 93)

Abbreviations: SLNB, Sentinel lymph node biopsy; ALND, Axillary lymph node dissection; 3D-CRT, Three-dimensional conformal radiation therapy; IMRT, Intensity-modulated radiationtherapy; Hy-IMRT, Three-dimensional conformal radiation therapy hybrid intensity-modulatedradiation therapy; ALC, Absolute lymphocyte count; RNI (included axillary lymph node, supraclavicular lymph node, and internal mammary lymph node), Regional lymph nodes; IMNI, Internal mammary node irradiation.
Lymphopenia during RT
Generally, the median baseline ALC prior to RT was 1.3 × 109/L, which significantly declined to a nadir of 0.5 × 109/L during treatment (P < 0.001). RIL developed in 95.7% (89/93) of BC patients, with grade distribution as follows: G1 (15.1%), G2 (43.0%), G3 (37.6%), and G4 (0.0%). Notably, 37.6% (35/93) of patients developed severe (G3+) RIL.
Risk Factors Associated with G3+ RIL
The mean thymus volume of the whole group of patients was 14.94 cm3 (IQR: 9.93). The mild and severe (G3+) RIL groups had comparable thymus volumes (mean: 15.47 cm3 vs 14.15 cm3; P = 0.668), with no significant difference observed between them. In terms of the dosimetry of thymus, the median mean thymus dose (MTD) was 6.01 Gy (IQR: 9.13). The V5, V10 and V20 of the thymus were 46.75% (IQR: 81.82), 8.76% (IQR: 56.02), and 0.37% (IQR: 10.58) respectively. Figure S1 demonstrated that patients with G3+ RIL had significantly higher MTD, thymic V5, and thymic V10 compared to those who did not develop G3+ RIL (All P < 0.001).
The Univariable and Multivariable Analyses of the Clinical and Dosimetric Variables Associated With G3+ RIL
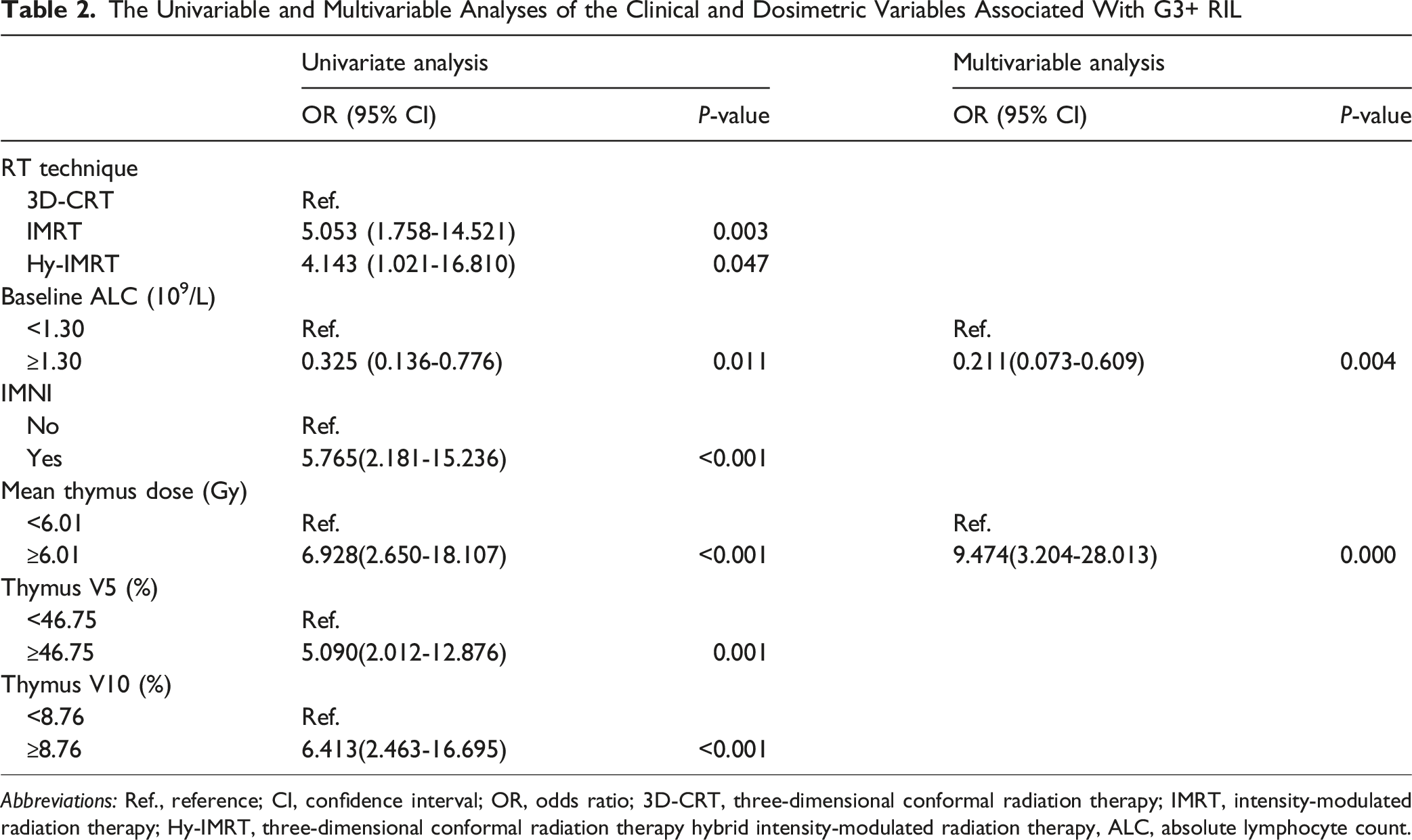
Abbreviations: Ref., reference; CI, confidence interval; OR, odds ratio; 3D-CRT, three-dimensional conformal radiation therapy; IMRT, intensity-modulated radiation therapy; Hy-IMRT, three-dimensional conformal radiation therapy hybrid intensity-modulated radiation therapy, ALC, absolute lymphocyte count.
Predictive Performance of Dosimetric and Clinical Variables for G3+RIL
As shown in Table 2, the univariate analysis showed that baseline ALC, RT technique involving IMRT, IMNI inclusion and higher thymic dose (MTD, V5, V10) were significantly associated with an increased risk of G3+ RIL, while further multivariable analysis demonstrated that baseline ALC and MTD were independent predictors.
ROC Analysis for the Prediction of G3+ RIL

Abbreviations: CI, confidence interval; AUC, area under the ROC curve; MTD, Mean thymus dose.

Receiver operating characteristic curves of the different single parameters and their combinations for G3+ RIL. (A) Single parameter. (B) Combination of different parameters. Model 1: Baseline ALC+MTD; Model 2: MTD+Thymus V5+Thymus V10+ Baseline ALC+IMNI+IMRT
Pairwise DeLong’s Test of ROC Curves

Abbreviations: CI, confidence interval; AUC, area under the ROC curve; MTD, Mean thymus dose; SE, Standard Error.
Discussion
This study demonstrated a significant correlation between the dose-volume parameters of the thymus and RIL. Combined model including thymic dosimetric and clinical parameters showed the most superior prediction performance in predicting G3 + RIL. Our findings emphasize that, in addition to known clinical factors, thymus irradiation should be considered a modifiable factor to better prevent RIL in BC patients receiving RT.
Numerous studies and reviews have revealed that RIL is associated with relatively low progression-free survival and overall survival rates in patients with a variety of solid tumours.2-4,24 In a meta-analysis, 56 cohorts with a total of 13 223 patients with 11 different types of cancer were treated with RT, and more than one-third (37.25%) of the patients experienced RIL; notably, RIL had a negative impact on overall survival across all cancer types, with an even greater impact observed when the sites treated with RT were located in the brain, thorax, and upper abdomen. 5 Similarly, RIL has a significant impact on local tumour control and survival in BC patients. Cho et al and Yoon et al reported that RIL was a potential predictor of decreased recurrence-free survival rates in patients with early BC who were treated with breast-conserving therapy.12,13 Another post hoc analysis of 598 BC patients from a phase III randomized clinical trial reported that patients with a minimum ALC/baseline ALC ratio <0.8 had a lower 5-year disease-free survival (71.8% vs 82.6%; P = 0.01). 6 Consequently, it is essential to identify the factors that can predict RIL risk and take effective measures to improve the prognosis of BC patients.
The development and severity of RIL are influenced by a variety of contributing factors, factors such as baseline ALC, concurrent chemotherapy, tumour stage, gross tumour volume or planning target volume, radiation dose, radiation fractions, and body dose.2,25 Our study focused particularly on baseline ALC and treatment factors such as chemotherapy, RT technique and dose-volume parameters. The impact of baseline ALC on RIL shown in this study was consistent with the findings of a study involving patients with prostate cancer by Pavarini M, et al 26 and a study involving patients with lung cancer by Jing et al 24 However, evaluating lymphopenia solely based on the total lymphocyte counts may not be comprehensive, as the functions and radiosensitivity of different lymphocyte subsets vary significantly. Belka et al 27 found that in patients with seminoma, RT caused a significant reduction in the number of B cells and T cells, with B cells being the most significantly affected. However, the decline in T-cell subsets (particularly CD4 and CD8 T cells) should also not be overlooked, because cellular immunity plays an important part in the body’s anti-tumor immune response, and T lymphocyte subsets have a leading role in the cellular immune response. For example, The CD4 T lymphopenia and CD8 T cells not expressing CD28 lymphopenia were related to worse prognosis in lung cancer.28,29 Prior chemotherapy seems to be a predictor of lymphopenia before adjuvant RT, which may adversely affect the long-term prognosis. Nevertheless, its impact on reducing lymphocyte counts during RT appears to be minimal. 30 This finding is consistent with findings from a study by Tang C et al, who reported that RIL was independent of concurrent chemotherapy use in patients with lung cancer. 31 Similarly, Chen F et al reported that chemotherapy was not a significant independent risk factor for the minimum-ALC/baseline-ALC ratio in a cohort of 735 BC patients. 32 Moreover, studies by Yoon et al and Cho et al included early breast cancer patients who received adjuvant RT without prior (neo)adjuvant chemotherapy and reported that RT alone can substantially reduce lymphocyte counts, independent of the effects of chemotherapy.12,13
Lymphocytes are extremely radiosensitive and can be wiped out at even low radiation doses (<1 Gy) because of radiation-induced DNA fragmentation. 7 Importantly, the RT technique emerged as a significant predictor of RIL. Previous studies have indicated that employing more conformal radiation techniques, such as proton therapy compared to IMRT, or reducing treatment duration, such as stereotactic body radiation therapy compared to conventionally fractionated RT, can effectively reduce the risk of lymphopenia. 33 In this study, we found that compared with 3D-CRT, IMRT was associated with a higher risk of G3+ RIL. A key consideration when interpreting our findings is the intrinsic characteristic of IMRT. Compared to 3D-CRT, IMRT typically produces a more extensive low-dose “bath” to normal tissues, a well-established risk factor for RIL.34-36 It could therefore be hypothesized that a high thymus dose is not directly pathogenic but merely a proxy for this generalized low-dose exposure. To investigate this possibility, we first performed a Spearman correlation analysis, which confirmed positive correlations between thymic dosimetric parameters (MTD, V5, V10) and both whole-body V5 and lung V5 (Table S1). We then constructed a multivariable logistic regression model to predict G3+ RIL, incorporating thymus parameters together with whole-body V5 and lung V5. After adjusting for these covariates, MTD remained an independent and statistically significant predictor (OR = 3.258, 95% CI: 1.067-9.947, P = 0.038, Table S2). This indicates that, despite its correlation with low-dose volumes, the thymus likely plays a unique and independent role in the development of RIL, a finding that warrants further investigation. Meanwhile, these findings highlight that although advanced RT techniques are utilized to increase target dose conformity and adherence to dose constraints for OARs, there is a need for radiation therapists must commit to incorporating strategies in clinical practice that aim to reduce low-dose volumes of critical organs during BC treatment.
In addition to optimizing the RT technique, the LymphoTEC proposes that sparing immune organs at risk (iOARs) and setting dosimetric constraints is another promising approach for preventing RIL during RT planning for solid tumours. The lymphocyte-rich regions mostly include primary and secondary lymphoid organs and circulating blood pool.2,7 To date, many dose-volume parameters of iOARs (eg, diploe of the skull, heart, lungs, vertebrae, ribs, sternum, the thymus, the spleen and bones of the pelvis) have been shown to be strongly correlated with RIL. 1 For thoracic RT, previous studies mostly reported that increased radiation doses in bone marrow (located in the vertebrae, ribs, and sternum), heart, lungs and prominent blood vessels associated with RIL in patients with NSCLC, esophageal cancer and BC.3,34,37 However, the significance of thymus protection in maintaining lymphocyte count levels during RT has also been increasingly recognized.
The thymus is crucial in adaptive immunity, as it produces a diverse array of T cells that safeguard the body against pathogens and neoplastic diseases.14,15 A study involving 1420 patients who had undergone thymectomy and 6021 controls revealed that the rate of all-cause mortality (8.1% vs 2.8%) and the risk of cancer (7.4% vs 3.7%) were greater in the thymectomy group than in the control group. 38 Monti et al used a Voxel-Based Analysis to examine the correlation between dose patterns and genomic factors with RIL in 186 patients with NSCLC. This analysis generated three-dimensional maps showing strong correlation between RIL and radiation dose in thymus, sternum, and vertebrae. 39 Zhang JL et al reported that the radiation dose to the thymus and thoracic duct was strongly associated with the probability of RIL in patients with lung cancer. 19 As a type of thoracic cancer, BC is similar to lung cancer in terms of its main irradiated iOARs. It is also impracticable to entirely evade thymic exposure during RT. However, the thymus has not been identified as an iOAR, and there are no recommended radiation dose limits for it in the treatment protocols for BC.
Over the years, several studies have investigated the impact of thymic radiation on RIL in patients with BC. In 1970, Meyer et al hypothesized that direct radiation damage to the thymus hinders the recovery of normal levels of lymphocytes. 16 Stjernsward et al reported that the duration of RIL in BC patients cannot be explained solely by the irradiation of lymphocytes that flow through large vessels. The thymus is included in the irradiated volume, which may affect its function, thereby also affecting the production and maintenance of T cells. 18 However, these viewpoints were opposed by Stratton JA et al, who reported that patients receiving pelvic and mediastinal (including thymic) RT presented a similar degree of lymphopenia and decreased lymphocyte responsiveness. Therefore, they ruled out thymic irradiation as a cause of RIL and concluded that the reduction in lymphocyte counts after RT was due to irradiation of lymphocytes in the recirculating pool rather than thymic irradiation. 17 However, there may be some limitations to the above studies, as they were conducted during the era of two-dimensional RT, lacking quantitative evidence to establish the relationship between thymic irradiation and lymphopenia. Our study overcomes this deficiency because the thymus was contoured in individuals and the relationship between dose-volume parameters of the thymus and the development of RIL was quantified, not only on the basis of comparisons of different irradiation sites that include or exclude the thymus. Our findings demonstrated that thymus irradiation plays a crucial role in the incidence of RIL and that the thymus can be considered as a critical immune organ to minimize the damage to the immune system during RT planning for BC patients.
This study has several limitations. First, as a single-center retrospective analysis with a relatively small sample size, it is susceptible to selection and information biases. The inclusion of patients treated solely at our institution may not fully represent the broader breast cancer population, and the reliance on historical medical records and laboratory data may introduce heterogeneity and incompleteness. Nevertheless, the results indicate a notable trend that merits further investigation. Second, no a priori sample size or power calculation was performed. Although the current cohort allowed for the detection of statistically significant associations between thymic dosimetric parameters and severe radiation-induced lymphopenia (RIL), the estimates may lack precision. Third, thymus contouring was performed by a single researcher without objective validation of consistency. Future studies should involve multiple independent observers contouring a subset of cases, with inter-observer agreement quantified using standardized metrics such as the Dice similarity coefficient or intraclass correlation coefficient. Fourth, our analysis was restricted to absolute lymphocyte counts without further stratification into specific lymphocyte subpopulations. However, an ongoing prospective study led by our team is addressing this limitation. Finally, the impact of thymic irradiation on prognosis in breast cancer patients remains inconclusive, underscoring the need for further validation in larger, multi-center cohorts. Despite these limitations, this study establishes a compelling link between thymus irradiation and lymphocyte depletion in radiotherapy-treated BC patients. The findings offer potential guidance for optimizing RT planning to mitigate radiation-induced immune suppression. To rigorously validate these findings, a randomized controlled trial derived from these parameters is now underway at our institution.
Conclusion
This study demonstrates that RIL commonly occurs in BC patients after adjuvant radiotherapy, with thymus irradiation identified as a significant risk factor. Optimizing radiotherapy plans to limit thymic exposure—particularly by reducing the mean dose, V5, and V10—may help decrease the risk of RIL, especially in patients with unfavorable prognostic features such as low ALC, receipt of IMRT, and inclusion of IMNI. Although these results may offer useful insights for clinical practice, they require further validation in large-scale prospective trials.
Supplemental Material
Supplemental Material - Dosimetric Effect of Thymus on Radiation-Induced Lymphopenia in Breast Cancer Patients Who Received Adjuvant Radiotherapy
Supplemental Material for Dosimetric Effect of Thymus on Radiation-Induced Lymphopenia in Breast Cancer Patients Who Received Adjuvant Radiotherapy by Tong Xia, Sisi Zheng, Jianjun Qian, Yinyin Yang, Liyan Jin, Guoqin Jiang, Peifeng Zhao, Yijia Hu, Shicheng Li, Liyuan Zhang, Ye Tian, and Shang Cai in Dose-Response
Footnotes
Acknowledgments
We would like to thank our medical physicists Xi Zhang and Li Chen for offering their help in data collection.
Ethical Considerations
Our study adhered to Good Clinical Practice guidelines and the principles of the Declaration of Helsinki. The institutional review boards approved the study protocol. The retrospective study design warranted a waiver of the institutional review boards’ written informed consent requirement.
Author Contributions
YT, LZ, SC and JQ conceptualized and designed the study. TX, SZ and YH performed the data collection. TX and SL carried out statistical analysis and data interpretation. TX drafted the manuscript. YT, SC and YY critically revised the manuscript. PZ provided technical support. LJ and GJ provided study materials or patients. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by grants from the Young Physician Scientist Project at Suzhou Medical College of Soochow University (ML12300723), 333 Training Project (Shang Cai), National Natural Science Foundation of China (NSFC82171828), the research projects of the Jiangsu Province (JSTJ-2024-113, JSDW202236), the Soochow University (GZK1202405, MA12300123), Suzhou (GSWS2021025, Szlcyxzx202103, SKY2022167, DZXYJ202304), and the Second Affiliated Hospital of Soochow University (XKTJ-RC202407)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
