Abstract
Moscatilin, a natural compound isolated from the orchid Dendrobium moscatum, has multiple pharmacological actions. The present study investigated the anti-tumor role of moscatilin in breast cancer and elucidated the underlying mechanisms. Cell proliferation, viability, and apoptosis of moscatilin treated MDA-MB-231 cells were determined by CCK-8 assay and flow cytometry. Histone deacetylases (HDACs) expression levels and global acetylated status of breast cancer cells were detected by Western blot and qPCR. Mouse xenograft model was established to evaluate the anti-cancer effects of moscatilin. Moscatilin treatment dose dependently suppressed proliferation and increased apoptosis of breast cancer cells. Moreover, moscatilin administration dramatically repressed tumor growth and extended survival time of mouse model. Mechanistically, moscatilin down-regulated HDAC3 expression, and then enhanced the global acetylated status of histone H3 (H3K9Ac) and H4 (H4K16Ac). Our findings indicate that moscatilin can inhibit the proliferation and promote apoptosis of breast cancer in vitro and in vivo, which suggests that moscatilin can be used as a potential therapeutic agent for the treatment of breast cancer.
Introduction
Breast cancer, the most commonly occurring cancer in females, is a malignant neoplasm with epithelial origin, which caused more than 2 million new cases and about 630,000 deaths in 2018 worldwide. 1,2 One in 8 of women will develop breast cancer during their lifetime in the United States, which becomes a huge public health issue in today’s society. 3 Benifitting from the deepening understanding of the biological behavior of breast cancer, and renewal of treatment concept, comprehensive treatment becomes the major therapy of breast cancer, including both local and systemic treatment. 2,4 A variety of treatments, such as surgery, radiotherapy, chemotherapy, endocrine therapy, and traditional Chinese medical (TCM) adjuvant therapy and so on, are applied in breast cancer treatment basing the tumor stage and Kanofsky performance. 2,3 Recently, the TCM therapy of cancer has emerged as a promising alternative for breast cancer treatment. 5 -7
The abnormal epigenetic regulation of gene expression is one of the major features of cancer. Histone acetylation is a classic posttranscriptional modification, which is regulated by 2 opposite enzymes: histone acetyltransferases (HATs) and histone deacetylases (HDACs). 8 HDACs change chromatin acetylation by removing acetyl group and influence the expression of oncogenes and tumor suppressors, and then alter a lot of biological events including cancer development and progression. 9 HDAC1, 2, and 3, the ubiquitously expressed members of this family, localizing in nuclear, are the well-studied HDACs and sensitive to the common HDAV inhibitors. 10 Recent studies demonstrated that high expression of HDAC1-3 positively correlate with the poor prognosis of ovarian and gastric carcinomas. 11,12 Therefore, developing new drugs to target HDACs is a rapidly emerging strategy of cancer treatment.
Moscatilin, a bibenzyl derivative isolated from the orchid Dendrobium moscatum, has been used in TCM for fever treatment. 13 Multiple pharmacological actions of moscatilin has been reported, such as repressing lipopolysaccharide induced inflammation, inhibition the production of oxidative reagents, and antiplatelet aggregation. 14 -16 Recent studies demonstrated the anti-tumor effects of moscatilin on several types of cancers. Moscatilin induced arrest of G2-M cell cycle and apoptosis in colorectal cancer through up-regulating the JNK and mitochondria associated apoptosis pathway. 17 Jin et al also demonstrated the pro-apoptotic action of moscatilin in pancreatic cancer via activation of JNK/SAPK signaling pathway. 18 Kowitdamrong and colleagues found that moscatilin is able to inhibit the migration and invasion of human non-small cell lung cancer cells by suppressing cellular production of reactive oxygen species (ROS). 19 HDACs play an important role in the occurrence and development of breast cancer, HDAC inhibition potentiates immunotherapy in triple negative breast cancer, and HDAC inhibitor suppresses proliferation and invasion of breast cancer cells. 20,21 So far, the function and molecular mechanisms of moscatilin in breast cancer development and progression remain elusive. In this study, we investigated the anti-tumor role of moscatilin and determined the underlying mechanisms of moscatilin induced inhibition of HDACs in breast cancer cell and animal models.
Materials and Methods
Cell Culture and Treatment
The human breast cancer cell line MDA-MB-231 (ATCC® HTB-26™) and MDA-MB-453 (ATCC® HTB-131™) were obtained from American Type Culture Collection (ATCC, Manassas, USA). The cells were cultured in Leibovitz’s L-15 complete growth medium (ATCC, Manassas, USA), containing 10% fetal bovine serum (FBS, Gibco), 2 mM L-glutamine, and penicillin-streptomycin antibiotics, and placed in 37°C with 5% CO2 humidified incubator. All animal studies were approved by Yuhuangding Hospital Affiliated to Qingdao University.
Drug Treatment
The moscatilin was isolated from Denfrobium oulchellum, extracted and quantitated by using the protocols created by Chanvorachote et al 22 Moscatilin was dissolved in DMSO and diluted by ddH2O or serum-free Leibovitz’s L-15 medium for the indicated concentrations. MDA-MB-231 or MDA-MB-453 cells were seeded into the 24-well or 96-well plate, 12 hours later, 1, 5, 10, and 50 μM moscatilin were added and incubated for 48 hours or indicated times, DMSO were used as negative control. Each treatment of breast cancer cells was repeated for at least 3 times.
Cell Proliferation and Viability Assays
The cell proliferation and viability of MDA-MB-231 or MDA-MB-453 after indicated moscatilin treatment were determined by Cell Counting Kit-8 (CCK-8) purchased from Beyotime (Shanghai, China), following the recommendations of the manufacturer. The OD value of each samples was tested by using the SpectraMax plus 384 microplate reader spectrophotometers (Molecular Devices, San Jose, USA) at 450 and 600 nm.
Apoptosis Analysis
Forty-eight hours after the moscatilin treatment, the MDA-MB-231 or MDA-MB-453 cells were stained by Annexin V-FITC using the FITC Annexin V Apoptosis Detection Kit with PI kit (BioLegend, Inc., San Diego, USA) following the instruction manual of the manufacturer. The suspended cells were sorted and analyzed by the Guava easyCyte 8HT Flow Cytometer HT system (Merck Millipore, Burlington, USA). Each treatment was repeated for 3 times.
RNA Extraction, Reverse Transcription, and qRT-PCR
The treated MDA-MB-231 or MDA-MB-453 cells were lysed using the TRIzol RNA isolation reagents (Invitrogen, Carlsbad, USA), and total RNA was isolated by using the TRIzol™ plus RNA purification kit (Thermo Fisher Scientific, Waltham, USA). One microgram total RNA was reversely transcribed into complementary DNA (cDNA) using the M-MLV reverse transcriptase, random primers, and dNTP. qRT-PCR was performed by using Bio-Rad CFX96 qPCR system (Bio-Rad, Hercules, USA) with KAPA SYBR® FAST qPCR Master Mix (2X) Kit (Sigma-Aldrich, St. Louis, USA). The expression level of HDACs was normalized by GAPDH, and calculated through the 2-ΔΔCT method. Primers used in this study were listed below:
HDAC1 (product length: 133 bp): Forward: TGGGGCTGGCAAAGGCAAGT; Reverse: GACCACTGCACTAGGCTGGAACA; HDAC2 (product length: 97 bp): Forward: CGTACAGTCAAGGAGGCGGCAA; Reverse: TGAGGCTTCATGGGATGACCCTGG; HDAC3 (product length: 150 bp): Forward: ACGTGCATCGTGCTCCAGTGT; Reverse: AGTGTAGCCACCACCTCCCAGT.
Western Blot
Forty-eight hours after the moscatilin treatment, MDA-MB-231 cells were lysed using the RIPA lysis buffer (50 mM Tris (pH7.4), 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, and protease inhibitor cocktails). The protein expression levels of indicated genes were detected using Western blotting as previous described. 18 The primary antibody of HDAC1 (ab53091, 1:1500 dilution), HDAC2 (ab137364, 1:1000 dilution), and GAPDH (ab181602, 1:2000 dilution) were purchased from Abcam (Cambridge, UK); HDAC3 (#3949, 1:1000 dilution), H3K9Ac (#9677, 1:1000 dilution), H3K9Ac (#13534, 1:1000 dilution), H3 (#9728, 1:2000 dilution), and H4 (#13944, 1:2000 dilution) were obtained from Cell Signaling Technology, Inc. (Danvers, USA).
Animals and Treatment
The 8- to 10-week-old nude mice (Shanghai SLAC laboratory Animal Co., Ltd., Shanghai, China) were used in this study. Mice were maintained under the standard rearing conditions with standard chow diet feeding and water accessible at all times. Animal studies were performed following the guideline established by the Quanzhou Guangqian Hospital Animal Care and Use Committee. Then, 1 × 106 MDA-MB-231 cells were implanted subcutaneously into left flank of nude mice. Mice were intravenously injected vehicle or moscatilin (100 mg/kg, daily). The HDAC3 inhibitor, RGFP966, was used as positive control (10 mg/kg, every 5 days) in this study. 23 The tumor size of each mouse (width × length; mm2) was measured by calipers every 10 days. The survival time of each mouse was recorded to create the survival curves of different administrations.
Statistical Analysis
Statistical analyses were performed by using the GraphPad Prism package. One-way and two-way analysis of variance (ANOVA) methods followed by a post hoc test was used to analyze the differences between groups. Kaplan and Meier method were used to calculate the 95% confidence interval for fractional survival and create survival curves of different administrations. The data were represented mean ± standard deviation (SD).
Results
Moscatilin Inhibits Cell Proliferation and Viability of Breast Cancer Cells
The uncontrolled cell proliferation is a major feature and risk factor of breast cancer. In order to investigate the anti-proliferation ability of moscatilin, the human breast cancer cell line, MDA-MB-231, was used to perform the cell proliferation assay after different concertation of moscatilin treatment (1, 5, 10, and 50 μM). The cell counting result showed that there is no difference between vehicle and 1 μM of moscatilin treatment on cell numbers; higher doses (5 and 10 μM) of moscatilin treatment exhibit the trend of lowering the cell proliferation; the 50 μM of moscatilin treatment significantly reduce the cell numbers of MDA-MB-231 (Figure 1A). The cell viability was determined by CCK-8 assay after 48 hours of moscatilin treatment. As shown in Figure 1B, both 10 and 50 μM of moscatilin treatment dramatically decrease the viability of MDA-MB-231, and the more robust inhibitory effect of cell viability is observed in 50 μM group (Figure 1B). Chemical structure of moscatilin was shown in Figure 1C. Moreover, the moscatilin induced inhibition on cell proliferation and viability were also observed in MDA-MB-453 cells, another breast cancer line (Supplementary Figure 1A and 1B). These results suggested that moscatilin dose dependently suppress the cell proliferation and viability of breast cancer cells.

Moscatilin inhibited the cell proliferation of MDA-MB-231 cells in vitro. (A) Cell proliferation of indicated groups was determined by cell counting. n = 3. *** indicated P < 0.001. (B) 48 hours after moscatilin treatment, cell viability was determined by CCK-8 assay. n = 5. * indicated P < 0.05 and *** indicated P < 0.001 compared to the control group. (C) Chemical structure of moscatilin.
Moscatilin Promotes the Apoptosis of Breast Cancer Cells
To further investigate the anti-tumor effects of moscatilin on breast cancer, MDA-MB-231 cells were incubated with different dose of moscatilin for 48 hours and cell apoptosis was determined by Annexin-V-FITC staining followed by flow cytometry. As shown in Figure 2A and B, the proportion of apoptotic cells in 10 and 50 μM of moscatilin treatment groups are 10.9% and 27.8%, respectively, both are significantly higher than the vehicle control (3.62%), which is consistently with the result of cell viability assay. In addition, after the moscatilin treatment, the dose dependent induction of cell apoptosis was observed, hence, 50 μM concentration was used in the following treatment. In MDA-MB-453 cells, moscatilin promoted cell apoptosis in a dose-dependent pattern (Supplementary Figure 1C). The above data indicated that moscatilin suppresses breast cancer cells through inhibiting cell proliferation and promoting cell apoptosis.

Moscatilin promoted the apoptosis in MDA-MB-231 cells. (A) 48 hours after moscatilin treatment, the apoptosis of MDA-MB-231 cells was determined by Annexin-V-FITC staining, followed by flow cytometry. (B) Statistic of percentage of the apoptotic cells (AnnexinV positive cells) performed in panel (A). n = 6. * indicated P < 0.05 and *** indicated P < 0.001 compared to the control group.
Moscatilin Specifically Reduces the Expression of HDAC3 in Breast Cancer Cells
HDACs closely implicate in the development and progression of breast cancer. Next, we examined the expression levels of HDAC family members upon moscatilin treatment. The MDA-MB-231 breast cancer cells were treated by moscatilin for 48 hours, and cell lysates were analyzed by western blot to determine the protein level of HDAC1, HDAC2, and HDAC3. As shown in Figure 3, in comparison with vehicle control, moscatilin treatment does not affect the protein level of HDAC1 and HDAC2 (Figure 3A-C), however, there is a dramatically reduction of HDAC3 expression, more than 2-fold decrease (Figure 3A and D). Moreover, the similar expressional changes of HDAC family members were observed under the transcriptional level upon moscatilin treatment. The qPCR results showed that the mRNA levels of both HDAC1 and HDAC2 are comparable among all treatment (Figure 4A and B), however, HDAC3 mRNA level is significantly decreased after 48 hours of moscatilin treatment (Figure 4C). Similarly, moscatilin treatment significantly reduced the expression of HDAC3 instead of HDAC1 and HDAC2 in another breast cancer line (Supplementary Figure 1D). All these data indicated that moscatilin inhibits the expression of HDAC3 in both protein and mRNA levels in breast cancer cells.
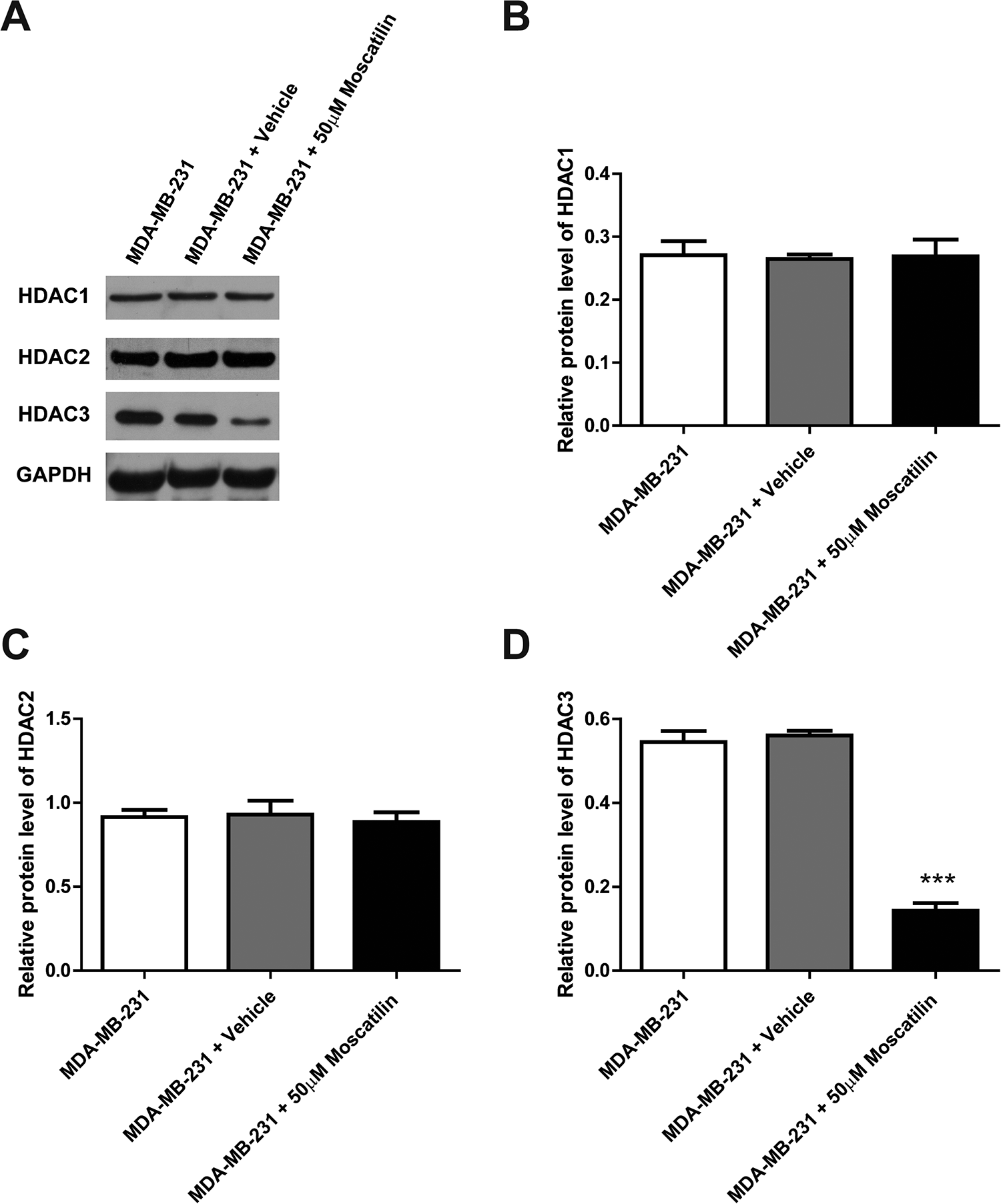
Moscatilin reduced the protein level of HDAC3. (A) Protein levels of HDAC1, HDAC2 and HDAC3 were determined by western blot after the treatment of moscatilin for 48 h. (B-D) Statistic analysis of protein levels of HDAC1, HDAC2, and HDAC3 performed in panel (A). n = 3. *** indicated P < 0.001 compared to the control group.

Moscatilin inhibited the mRNA expression level of HDAC3. The mRNA levels of HDAC1 (A), HDAC2 (B), and HDAC3 (C) were determined by Q-PCR after the treatment of moscatilin for 48 h. n = 3. *** indicated P < 0.001 compared to the control group.
Moscatilin Enhances the Expression of H3K9Ac and H4K16Ac
The marks of histone lysine acetylation, H3K9Ac and H4K16Ac, are regulated by HDAC3. To further confirm the moscatilin-induced reduction of HDAC3, we detected the expression of H3K9Ac and H4K16Ac in breast cancer cells. Western blot results showed that the protein level of H3K9Ac is significantly up-regulated by moscatilin relative to the vehicle control group (Figure 5A and B). Similarly, H4K16Ac protein level also dramatically increased after 48 hours of moscatilin incubation (Figure 5A and C). The up-regulation of H3K9Ac and H4K16Ac induced by moscatilin treatment further indicated that moscatilin can suppress the expression of histone deacetylase HDAC3.
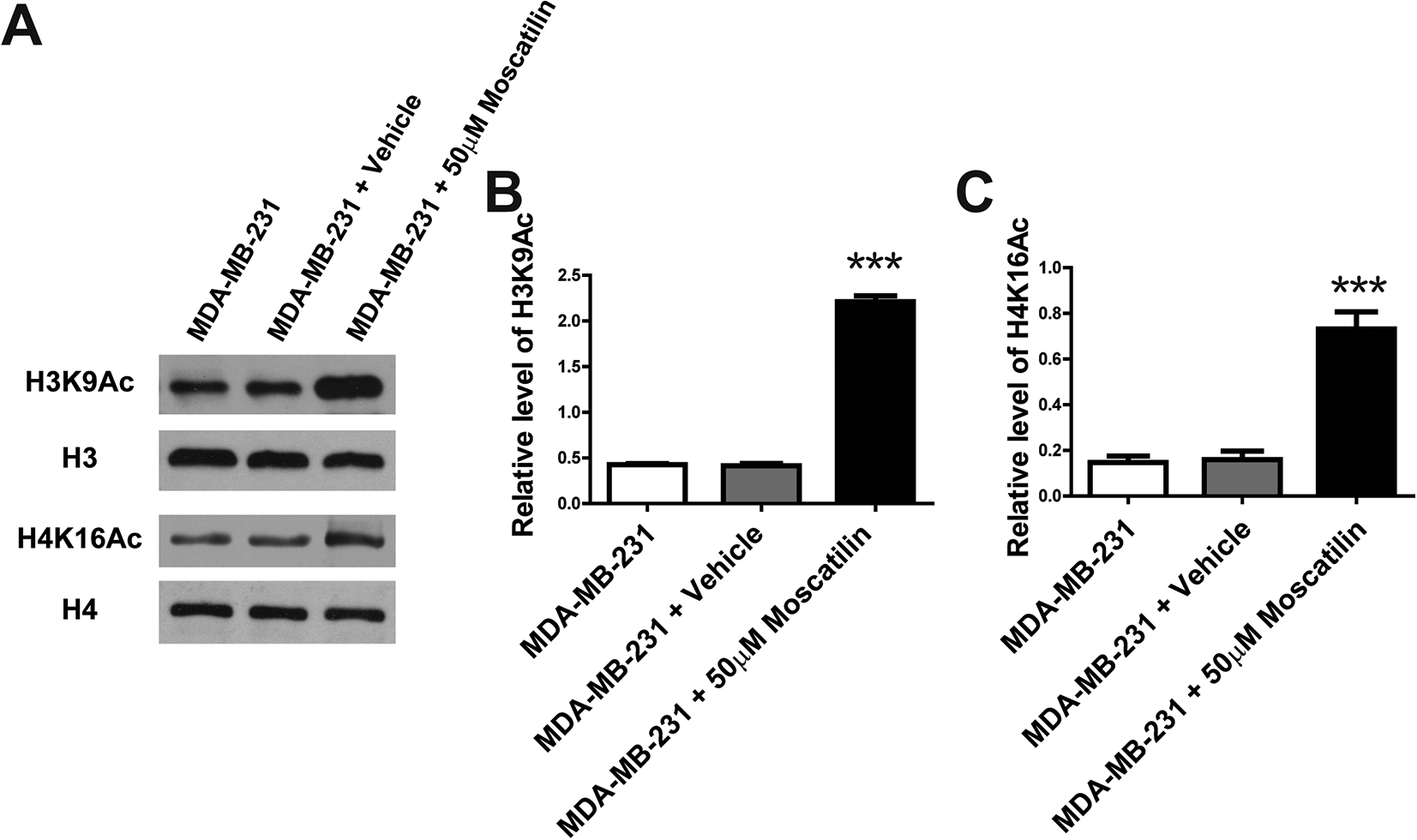
Moscatilin increased the global H3K9Ac and H4K16Ac. (A) The levels of H3K9Ac and H4K16Ac were determined by western blot after the treatment of moscatilin for 48 h. n = 3 for independent experiments, and this panel presented one of these repeats. (B-C) Statistic analysis of protein levels of H3K9Ac and H4K16Ac relative to H3 and H4 respectively in panel (A). n = 3. *** indicated P < 0.001 compared to the control group.
Moscatilin Suppresses Breast Cancer Growth and Progression in Vivo
Next, we evaluated the anti-tumor effects of moscatilin in the mouse xenograft model. MDA-MB-231 cells were axillary injected into nude mice to establish the mouse model of breast cancer. The mice were intravenously injected vehicle or moscatilin (100 mg/kg, daily), and RGFP966 (HDAC3 inhibitor) was used as positive control in this study. The survival curves showed that RGFP966 administration extends the survival of the treated mice compared to mice of vehicle group, importantly, mice administrated by moscatilin have the longest survival time among the 3 groups (Figure 6A). The tumor volume of treated mice was measured every 10 days. As shown in Figure 6B, both moscatilin and RGFP966 administration significantly reduce the tumor size, and moscatilin treatment has the more robust inhibitory effect on tumor growth compared to RGFP966 treatment (Figure 6B). Similar findings could be observed in the tumor weight. Moscatilin exerted the most effective power in inhibiting the tumor weight (Figure 6C). In line with the in vitro result, moscatilin administration dramatically decreased the protein level of HDAC3 compared to that in vehicle treated mice (Figure 6D). These data suggested that moscatilin suppresses breast cancer growth and progression in vivo.

Moscatilin suppressed the growth and progression breast cancer in vivo. (A) Survival curve of the mice treated moscatilin or RGFP966. (B) Tumor volume of the mice with different treatment as indicated was measured at different time points. Each group contains 20 mice. (C) Tumor weight was showed for each group. (D) Western blot showing the HDAC3 protein level in moscatilin treated or untreated mice tumor tissues. n = 5. *** indicated P < 0.001 compared to the control group.
Discussion
Breast cancer is the second leading cause of cancer-associated mortality in women worldwide, which indicate the current treatments for breast cancer are ineffective. 2,24 Hence, it is very necessary to develop new anti-tumor drugs that can inhibit proliferation and promote apoptosis of breast cancer. The TCM therapy has emerged as a promising alternative for cancer treatment. 5 -7 In this study, we investigated the anti-breast cancer effects of moscatilin, a bibenzyl derivative isolated from the orchid Dendrobium moscatum. 13 We found that moscatilin treatment significantly suppress proliferation and increase apoptosis of different breast cancer cells in a dose-dependent manner. Moreover, moscatilin administration dramatically repress tumor growth and extend survival time for mouse xenograft model of breast cancer. Mechanistically, moscatilin down-regulates the expression of HDAC3, a ubiquitously expressed member of the histone deacetylase family, 8 on both mRNA and protein levels, and then increase the global acetylated status of histone H3 (H3K9Ac) and H4 (H4K16Ac). Our findings confirm that moscatilin can inhibit the proliferation and promote apoptosis of breast cancer in vitro and in vivo, which providing new theoretical basis for clinical medication.
HDACs induced deregulation of histone acetylation has serious consequence of gene transcription related to cancer initiation and progression. For example, histone H3 and H4 are important substrates of HDACs, loss of mono-acetylated lysine 9 of histone H3 (H3K9Ac) and lysine 16 of H4 (H4K16Ac) are reported to be the predictor of poor survival in lung, prostate, breast, and pancreatic cancers. 25 HDAC3 is aberrantly recruited by oncogenic fusion proteins (AML1-ETO and PML-RAR), which act as the genetic drivers of hematologic malignancies, and result in the induction of acute myeloid leukemia. 26 Moreover, Barberio et al reported that HDAC inhibitor (vorinostat) enhances in vivo immune response to PD-1/CTLA-4 blockade and decrease tumor growth in triple-negative breast cancer model. 21 Hsieh et al found that HDAC3 mechanistically links to the homeostasis of breast cancer stem cells by regulating β-catenin signaling pathway. 27 Here, we revealed that the anti-tumor effects of moscatilin are through down-regulating of HDAC3 instead of HDAC1 and HDAC2 to inhibit proliferation and promote apoptosis of breast cancer cells. HDAC3 is most commonly found in transcription co-repressor complexes, unlike HDAC1 and HDAC2, HDAC3 uniquely associate with the regulatory and catalytic subunits of PP4 complex, and strong expression of HDAC3 closely correlate with poor prognosis in lung cancer patients. 28,29 The moscatilin induced down-regulation of HDAC3 might be mediated by estrogen, steroid hormone and progesterone receptor, which is interesting to be addressed in future study. The protein levels of acetylated histone H3 (H3K9Ac) and H4 (H4K16Ac) are dramatically increased after moscatilin treatment, which is in line with the above findings that global acetylated status closely correlate to cancer progression. 11,12,25
Triple negative breast cancer is a more aggressive and difficult subtype of breast cancer, and loss of estrogen receptor alpha (ERα) induced endocrine resistant is one of the major features of it. 30 Recent study reported that breast cancer cells without ERα expression can keep the ability of proliferation by elevating the expression of HDAC3, and further study indicated that pan-HDAC inhibitors mediated down-regulation of HDAC3 effectively reverse this endocrine resistance. 31 Barberio and colleagues also demonstrated that HDAC inhibition is able to potentiate immunotherapy through increasing the immune response to PD-1/CTLA-4 blockade in triple negative breast cancer models. 21 The MDA-MB-231 cells were used in current study, which is ERα negative breast cancer cell line. 32 Moscatilin treatment significantly suppress the proliferation of MDA-MB-231, moreover, the tumor growth and survival time of xenograft mouse model established by MDA-MB-231cell injection are remarkably improved after moscatilin administration. Our findings indicated that moscatilin is a valuable candidate for triple negative breast cancer treatment. It is worth to note that the anti-tumor effects of moscatilin is as potent as the well-known HDAC inhibitor, RGFP966, 27 in the in vivo testing of this study. In addition, there might be the extra benefits of moscatilin administration, such as anti-inflammation and anti-oxidation, in addition to the inhibition on HDAC3, which need to be further addressed in the future studies.
Conclusion
This study demonstrated that moscatilin suppresses tumor growth by inhibiting proliferation and increasing apoptosis of breast cancer cells. Mechanistically, moscatilin down-regulates the expression of HDAC3 and enchases the global acetylated status. Our findings indicate that moscatilin, a natural compound, can be used as potential therapeutic agents for the treatment of breast cancer.
Supplemental Material
Supplemental Material, sj-pdf-1-dos-10.1177_15593258211001251 - Moscatilin Suppresses the Breast Cancer Both In Vitro and In Vivo by Inhibiting HDAC3
Supplemental Material, sj-pdf-1-dos-10.1177_15593258211001251 for Moscatilin Suppresses the Breast Cancer Both In Vitro and In Vivo by Inhibiting HDAC3 by Wenjie Su, Lianfu Zeng and Weida Chen in Dose-Response
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
