Abstract
Obesity is associated with high risk and poor prognosis of breast cancer (BC). Obesity promotes BC cells proliferation via modulating the production of adipokines, including adiponectin (anti-neoplastic adipokine), leptin (carcinogenic adipokine) and inflammatory mediators. In the present study we investigated the anti-proliferative effects of liraglutide (LG; anti-diabetic and weight reducing drug) on MCF-7 human BC cells cultured in obese adipose tissue-derived stem cells-conditioned medium (ADSCs-CM) and whether this effect is mediated via modulating the adipokines in ADSCs and cancer cells. Proliferation was investigated using AlamarBlue viability test, colony forming assay and cell cycle analysis. Levels and expression of adipokines and their receptors were assayed using ELISA and RT-PCR. LG caused 48% inhibition of MCF-7 proliferation in obese ADSCs-CM, reduced the colony formation and induced G0/G1 phase arrest. LG also decreased the levels of inflammatory mediators, suppressed the expression of leptin, while increased mRNA levels of adiponectin and their receptors in obese ADSCs and cancer cells cultured in obese ADCSs-CM. In conclusion, LG could mitigate BC cell growth in obese subjects; therefore it could be used for clinical prevention and/or treatment of BC in obese subjects. It may assist to improve treatment outcomes and, reduce the mortality rate in obese patients with BC.
Introduction
Breast cancer (BC) is the second most common cancer worldwide. 1 Obesity has been associated with increased BC risk, 2,3 especially in postmenopausal women with estrogen receptor positive. Obesity results in increased breast tumor size, increased rate of metastasis, lower treatment outcome and increased mortality rate. 2,4 One mechanism that underpins the relation between obesity and the development of BC is the accumulation of adipose tissue which results in increased production of pro-inflammatory cytokines contributing to a state of chronic inflammation. 3,5,6 This state of chronic inflammation can promote BC growth via altering the biology and secretory profile of the cells of the adipose tissue including adipose-derived stem cells (ADSCs). 7,8
ADSCs are multipotent mesenchymal lineage that is significantly implicated in BC development, invasion and metastasis. 9,10 ADSCs enhance tumor growth through the secretion of several cytokines and growth factors that strongly promote the growth, migration and invasion of BC cells. 8,11 -13 Among these cytokines, ADSCs secrete leptin, adiponectin, interleukin (IL)-6 and tumor necrosis factor-alpha (TNF-α). 8,11 -16
Studies have shown that obesity significantly increases the number of ADSCs within the adipose tissue and this increase enhances the vascularization and proliferation of malignant cells. 12,17 ADSCs from obese women enhanced the proliferation of BC cells via different mechanisms. First, obesity alters the biology of ADSCs to favor their rapid conversion into carcinoma-associated fibroblast-like cells, which in turn enhances the proliferative rate, the phenotype, and gene expression profile of BC cells. 18 -20 Second, obesity alters the secretory profile of ADSCs to enhance cancer growth. 8,11,12,15,17 Compared to ADSCs isolated from lean women, ADSCs from obese subjects expressed significantly higher levels of leptin 8,12,15 and pro-inflammatory cytokines including IL-6, TNF-α, IL-12 and adhesion molecules. 11,21
Adiponectin and leptin are polypeptides that have critical and antagonistic effects on BC development. 3,22 -25 Adiponectin acts via its receptors; ADR1 and ADR2, on BC cells resulting in anti-proliferative effects through apoptotic, anti-inflammatory, and anti-angiogenic mechanisms. 6,24,25 Adiponectin is strongly down-regulated in obesity 5,26 which is significantly correlated with the development of BC. 3,5,6,24 On the other hand, leptin exerts its pro-proliferative effects through binding and activating its receptors ObR1 and Ob-Rt. 23 The increased secretion of leptin from obese ADSCs plays an essential role in the development and progression of BC by enhancing tumor growth, angiogenesis, and inflammation. 6,27,28 Delivery of leptin to cancer cells also results in increased proliferation, migration, invasion, and metastasis of the cells. 29,30 Therefore, inhibiting the expression of leptin and its receptors in obese ADSCs and BC cells could reduce the impact of obese ADSCs on BC cells proliferation and migration.
The altered secretory profile of ADSCs in obese subjects highlights the potential for therapeutic strategies to reduce BC development and progression targeting these cytokines and their receptors in ADSCs and BC cells. Recently, incretin therapies, including glucagon-like peptide-1 receptor (GLP-1 R) agonists, have gained significant attention, not only due to their anti-diabetic potential, but also because of their cytoprotective effects. 31 -33
Liraglutide (LG) is a potent long acting GLP-1 R agonist that improves glucose homoeostasis and reduces the body weight. 34 The activity of LG on weight loss has been attributed to the significant reduction of circulating leptin. 35 LG causes both upregulation of adiponectin and anti-inflammatory effect in diabetic patients. 36,37 Although the anti-proliferative activity of LG has been reported on human prostate 31 and pancreatic 32 cancers as well as in hepatocellular carcinoma cells, 33 it is unknown whether LG could exert anti-proliferative effects against BC, particularly in obese subjects. According to previously mentioned data, we hypothesize that treatment with LG may mitigate the BC growth in obese subjects and this effect is mediated via modulating the secretion of leptin, adiponectin and inflammatory cytokines from ADSCs and cancer cells
Materials and Methods
Materials
Dulbecco modified Eagle’s medium (DMEM), fetal calf serum (FCS) and fetal bovine serum (FBS) were purchased from Sigma–Aldrich (St. Louis, MO). LG (Victoza®, 6mg/ml Pen fill, Novo Nordisk, USA) was obtained from a local pharmacy, Riyadh, Saudi Arabia. MCF-7 cells, a human BC cell lines which are positive for estrogen receptor were obtained from American Type Culture Collection (ATCC, USA) and were grown in DMEM supplemented with 2 mM L-glutamine, penicillin (20 units/ml), Streptomycin (20 μg/ml), and 10% (v/v) heat-inactivated FCS at 37°C in a saturated humidity atmosphere 6 containing 95% air and 5% CO2.
Preparation of Pre-Adipocytes Cells
Adipose tissues were obtained from the abdominal subcutaneous fat of healthy female subjects who are free from metabolic or endocrine diseases & with body mass index (BMI) from 18.5-25 kg/m2 and were used to prepare lean ADSCs. Another group of adipose tissues were obtained from obese female subject with BMI 30-35 kg/m2 to prepare obese ADSCs. The adipose tissues were taken from plastic surgeries (King Khalid University Hospital, Riyadh, Saudi Arabia), informed consent was taken from each patient and the protocol & all procedures were approved by the Research Ethics Committee at King Saud University (Ethics Reference No E-19-4477). The adipose tissues were cut into 1-mm pieces and digested with collagenase with shaking at 37°C for 2 hours, and then adipose-derived stromal vascular fraction cells were isolated by centrifugation. 38 The adherent adipocyte stem cells were taken as pre-adipocytes (adipose tissue)-derived stem cells (ADSCs).
Generation of the Adipose Stem Cells-Conditioned Media (ADSCs-CM)
The conditioned media were used to mimic the adipose tissue microenvironment. ADSCs-CM were generated when ADSCs from lean or obese subjects were seeded at a density of 8 × 106 in 10% FBS/DMEM using T150 flask until to reach 80% confluence. Then the media were changed to incubate cells for 48 hr. After that the supernatants were collected and used as ADSCs-CM for subsequent in vitro studies.
Culture of BC Cells in ADSCs-CM and the Treatment With LG
To investigate the effect of obesity-related mediators on the growth of MCF-7 cells, cancer cells were cultured in lean/obese ADSCs-CM mixed with basal medium (10% FBS/DMEM) in ratio 1:1 for 72 h, and cell viability was determined by AlamarBlue assay (described below). The control cell proliferation was determined by culturing BC cells for 72 h in 10% FBS/DMEM only. Both ADSCs-CM and the basal media contained 10% FBS/DMEM to provide a base level of nutrition for cellular function. To study the effect of LG on the cell proliferation of MCF-7 cells in obese ADSCs-CM, MCF-7 cells were seeded at a density of 7 × 103 /well in 96-well plates and cultured in ADSCs-CM mixed with basal medium in ratio 1:1 with different doses of LG (25,50, 100, 200 µM) for 72 h. The dose-dependent effect of LG on cell proliferation was determined using AlamarBlue assay. The 50% inhibitory concentration was used as effective anti-proliferative concentration to complete the study. In the main study, MCF-7 cells were cultured for 72 h with either lean or obese ADSCs-CM with or without the selected dose of LG. In addition, the lean/obese ADSCs were cultured in basal medium with or without LG to evaluate the effect of LG on the expression of leptin, adiponectin and their receptors in ADSCs
Evaluating the Inhibitory Effect of LG on the Growth and Proliferation of MCF-7 Cells Cultured in Lean/Obese ADSCs-CM
Cell proliferation assays
Alamar Blue cell proliferation assay
In the presence of living cells, and in response to the chemical reduction due to cell growth, the blue-colored assay reagent is reduced into red color. The inhibition of growth maintains an oxidized environment (blue), which can be detected using fluorescence detector. AlamarBlue assay was conducted according to manufacturer’s instructions using kits obtained from Thermo Fisher Scientific Inc. Briefly, 7,000 cells were cultured in a 96-well plate for 24 hrs in basal media then the media were changed by either conditioned media or basal media and using different concentrations of LG. Then cell viability was measured after 72 hours by adding 10% AlamarBlue assay reagent and measuring fluorescence with excitation wavelength at 530-560 nm and emission wavelength at 590 nm. 39 The % difference between treated (T) & untreated (UT) cells based on fluorescence (RFU values) was calculated by dividing Experimental RFU value with Untreated Cell Control RFU value as below:
Colony forming assay
MCF-7 cells were treated by 50 µM LG and cultured in different media for 72 hrs then these cells were trypsinized and seeded again in 6-well plates at density 0.02 × 106, 0.01 × 106, 0.005 × 106 cells per well, and incubated at 37°C under 5% CO2 for about 14 days using different media until cells being confluent by at least 80% in control groups. 40 The formed colonies were washed and stained with Diff-Quik stain set (Siemens Healthcare Diagnostics) according to the manufacturer’s protocol (The media were removed then cells were fixed by fixation solution in the kit then staining solution was added for seconds and finally washed by distilled water). The colonies were observed under microscope and captured using a Nikon digital photo camera (Tokyo, Japan).
Cell cycle analysis using flow cytometry
Cells were trypsinized and suspended in 1 ml of PBS/0.5% BSA (2 × 106 cells), and then were fixed using ice-cold 70% alcohol for overnight. Next, the cells were centrifuged and washed in PBS, re-suspended in 500 µL of PBS supplemented with 40 µg/mL RNAse A and 50 µg/mL propidium iodine, before being analyzed using a flow cytometer (Beckman Coulter, Miami, FL, USA). 39 Staining was detected in the green fluorescence channel (FL1) and the data was analyzed by Kaluza software (Beckman Coulter).
Investigating the Effect of LG on Inflammatory Cytokines, Leptin, Adiponectin, ADR1, ADR2, ObR1 and Ob-Rt in ADSCs and MCF-7 cells
Assessment of the levels of adipokines in ADSCs-CM
The effect of LG on the production of adipokines from ADSCs was determined by assessing the levels of TNF-α, IL-6, leptin and adiponectin in the lean and obese ADSCs-CM with or without LG using ELISA kits purchased from Abcam Co. (Abcam Inc, Cambridge, UK, C) according to the manufacturers’ instructions
Determination of the mRNA levels of adiponectin, leptin and their receptors in MCF-7 cells and ADSCs
The expression of adiponectin, leptin and their receptors (ADR1, ADR2, ObR1, and Ob-Rt) in treated MCF-7 cells and ADSCs was analyzed by qRT-PCR. Briefly, total RNA was isolated from cells using innuPREP RNA Mini Kit (Life Science Inc., Germany) according to the manufacturer’s instructions. The cDNA was synthesized using Applied Biosystems™ High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific Inc., MA). RT-PCR amplification and analysis was performed using an Applied Biosystem StepOne™ (USA) using gene-specific primer sequences for adiponectin, leptin, ADR1, ADR2 and Ob-R (Table 1) by Syber green. Gene expression was calculated using the relative gene expression method (Livak; 2–ΔΔCT). All values were normalized to GAPDH which was used as the control housekeeping gene.
Gene-Specific Primer Sequences for Adiponectin, Leptin, ADR1, ADR2, ObR1 and ObRt.

Statistical Analysis
Data was expressed as means ± SEM. Statistical comparison between different groups was performed using 2-way analysis of variance (ANOVA) followed by Tukey-Kramer multiple comparisons test. Paired t-test was used to compare the data from the same group before and after treatment. All statistical analysis was performed using Prism GraphPad Program (version 8). Differences were considered significant at P < 0.05.
Results
Results of the Proliferation and Growth Studies
Effect of obese ADSCs-CM on the proliferation of MCF-7. CM from obese ADSCs revealed significant increase in MCF-7 cell proliferation by 1.46 fold compared to both basal medium (P < 0. 001) and lean ADSCs-CM (P < 0.001) supporting the role of obesity as a risk factor in the growth and progression of BC (Figure 1).

The percent of MCF-7 cells proliferation in lean and obese adipose-derived stem cells-conditioned medium (ADSCs-CM) compared to basal medium. *** P < 0.001.
Effect of different doses of LG on the proliferation of MCF-7 in obese ADSCs-CM
Treatment with 25, 50,100 and 200 µM of LG significantly reduced the proliferation of MCF-7 cells in obese ADSCs-CM by 30, 48, 70 and 82%, respectively (Figure 2A). The dose 50 µM was selected to complete the study because it shows the range of 50% inhibitory concentration for the proliferation of MCF-7 cells in obese ADSCs-CM.
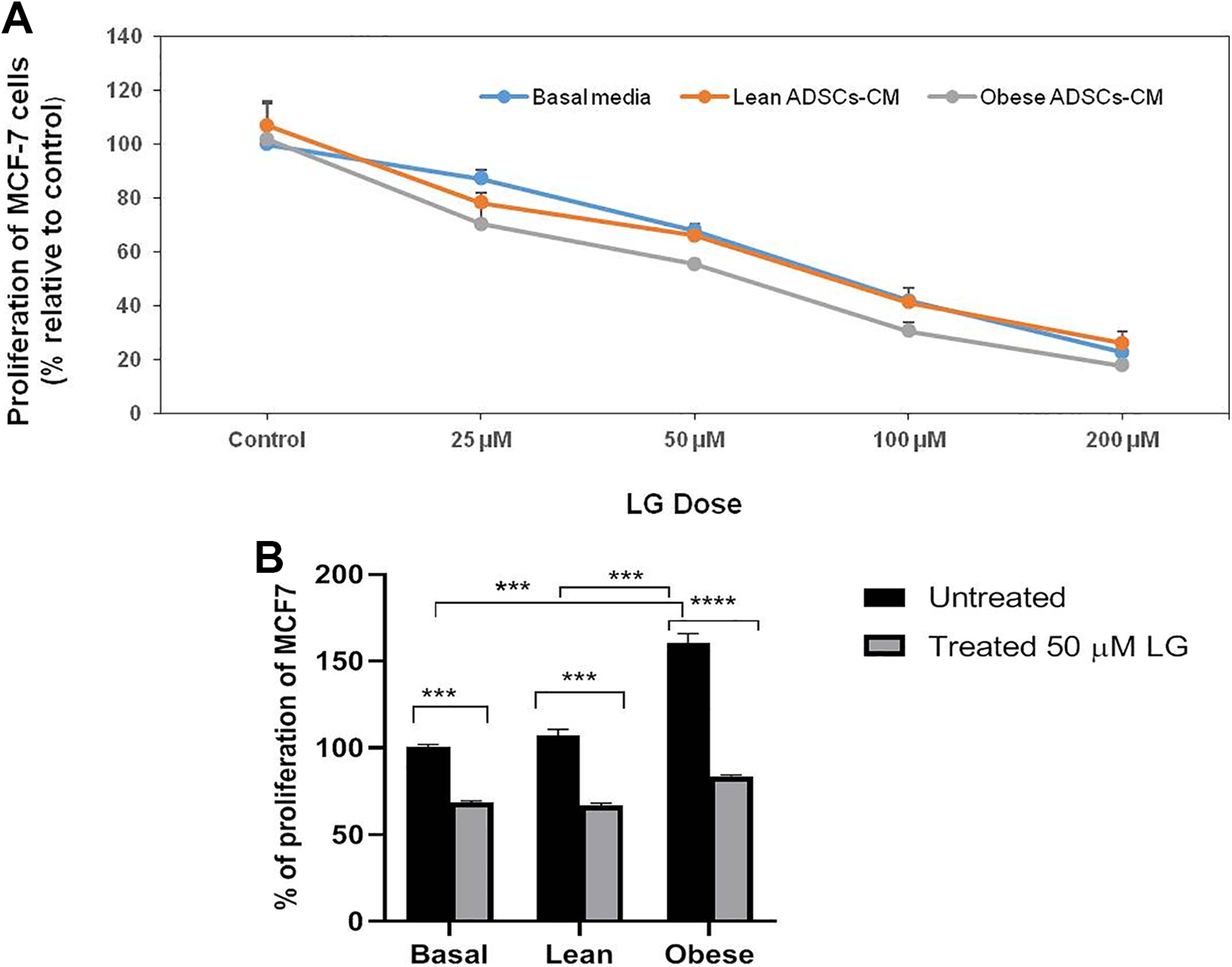
A, Effect of different doses of liraglutide (LG) on the proliferation of MCF-7 in different media using AlamarBlue test. B, Effect of the selected dose of LG (50 µM) on the proliferation of MCF-7 in different media using AlamarBlue test. *** P < 0.001, **** P < 0.0001.
Effect of the selected dose of LG (50 µM) on the proliferation of MCF-7
As shown in Figure 2B, the proliferation of untreated MCF-7 cells in obese ADSCs-CM was significantly increased compared to basal medium (P < 0.001) (Figure 2B). Treatment with 50 µM of LG significantly reduced the growth of MCF-7 in obese ADSCs-CM by 48% compared to before treatment. However, the same dose inhibits MCF-7 proliferation by only 38% and 32% in lean ADSCs-CM and basal medium, respectively reflecting the prominent anti-proliferative effect of LG on obese medium than in lean CM or basal medium.
Effect of LG (50 µM) on MCF-7 cell growth in different media using colony forming assay. Colony formation assay revealed that the number of colonies formed in obese ADSCs-CM was much more compared to basal medium and lean ADSCs-CM (Figure 3). Treatment with LG (50 µM) inhibited the colony formation ability. This inhibition was stronger in obese ADSCs-CM compared to basal media and lean ADSCs-CM.

Effect of liraglutide (LG) on MCF-7 cell growth in different media using colony formation assay.
Effect of LG on cell cycle in different media using flow cytometry
We utilized flow cytometry to assess changes in cell cycle progression under different conditioned media (Figure 4). Cell cycle analysis revealed 3 distinct cell populations in the G0/G1, S and G2/M phases of the cell cycle (Figure 4A). Accumulation of G0 phase or resting phase in the cell cycle means that cells exist in a quiescent state. G0 phase is viewed as an extended G1 phase, where the cell is neither dividing nor preparing to divide. As shown in Figure 4B, the percent of cell population in G0/G1 phase before LG treatment was significantly lower in obese ADSCs-CM compared to both basal medium and lean ADSCs-CM (P < 0.05) reflecting higher cell growth in obese conditions. As revealed in Figure 4A, treatment with 50 µM LG resulted in G0/G1 cell cycle arrest in all different media where the cell population in LG treated groups showed more accumulation of the cells in G0/G1 with more effect in obese conditions. As shown in Figure 4B, LG treatment significantly increased the percent of cell population in this phase by 14% in obese ADSCs-CM and by 6% in basal and lean conditioned media compared to the percent before treatment.

Cell cycle analysis using propidium iodide (PI) staining and flow cytometry. MCF-7 cells were treated for 72 h with liraglutide (LG). A, Examples of G0/G1 peaks for MCF-7 treated with 50 µM LG in different media. B, Average percent of MCF-7cells in G0/G1 phase using different media, * P < 0.05; *** P < 0.001.
The Effect of LG on Different Adipokines
Next, we investigated whether the anti-proliferative effect of LG on the MCF-7 cells cultured in obese ADSCs-CM could be related to modulating levels of adipokines—including adiponectin, leptin, TNF-α and IL-6.
Effect of LG on the levels of adipokines in lean/obese ADSCs
As shown in Figure 5, the untreated obese ADSCs-CM showed a significant lower concentration of adiponectin (10.86 ± 2.4 pg/ml vs 13 ± 2.1, P < 0.05), together with significant higher concentrations of leptin (29.23 ± 1.5 pg/ml vs 24.37 ± 0.84, P < 0.05), IL-6 (42.62 ± 3.3 pg/ml vs 32.27 ± 1.2, P < 0.05) and TNF-α (10.07 ± 0.83 pg/ml, vs 7.46 ± 0.68, P < 0.05) compared to untreated lean ADSCs-CM (Figure 5). Treatment of obese ADSCs-CM with LG leads to significant increase in adiponectin concentration (19 ± 1.12 vs 13 ± 2.1, P < 0.01) along with significant decreases in the concentrations of leptin (26.32 ± 1.24 vs 29.23 ± 1.5, P < 0.05), IL-6 (30.54 ± 3.36 vs 42.62 ± 3.3, P < 0.01) and TNF-α (6.88 ± 0.5 vs 10.07 ± 0.83, P < 0.01) compared to the levels in untreated medium.

Effect of liraglutide (LG) on the levels of adiponectin, leptin and inflammatory cytokines including tumor necrosis factor–α (TNF-α) and interleukin-6 (IL-6), in lean and obese adipose-derived stem cells (ADSCs). ** P < 0. 01; * P < 0.05.
Effect of LG on the expression of adiponectin, leptin and their receptors in ADSCs and MCF-7 cells
After treatment with LG, the expression of adiponectin and its receptors (ADR1 and ADR2) were significantly up-regulated in lean and obese ADSCs (P < 0.001, Figure 6A) with prominent improvement in obese conditions. The mRNA levels of adiponectin, ADR1, ADR2 in obese ADSCs cells were up-regulated by 1.6-fold, 1.7-fold and 1.3-fold, respectively. The expression of adiponectin and ADR1 in obese ADSCs cells was significantly higher after LG treatment compared to their expression in lean cells (P < 0.001, P < 0.05, respectively). In addition, mRNA levels of adiponectin, ADR1, ADR2 were significantly up-regulated in MCF-7 cells cultured in lean (P < 0.05, 0.001) and obese ADSCs-CM (P < 0.01, 0.001), Figure 6B. The expression of ADR1 and ADR2 in MCF-7 cells cultured in obese ADSCs-CM cells was significantly higher compared to the expression in MCF-7 cells cultured in lean ADSCs-CM (P < 0.05), (P < 0.001), respectively.

A, Effect of liraglutide (LG) on the mRNA levels of adiponectin and their receptors in lean and obese adipose-derived stem cells (ADSCs). * Difference in the same group before and after treatment with LG; *** P < 0.001. # Significance of treated obese ADSCs vs treated lean ADSCs; # P < 0.05; ###P < 0.001. B, Effect of LG on the mRNA levels of adiponectin and their receptors in MCF-7 cells incubated in lean/obese adipose-derived stem cells-conditioned medium (ADSCs–CM). * Difference in the same group before and after treatment with LG; *** P < 0.001;** P < 0. 01, * P < 0.05. # Significance of treated MCF-7 in obese ADSCs-CM vs treated MCF-7 in lean conditions; ###P < 0.001; # P < 0.05. C, Effect of LG on the mRNA levels of leptin and their receptors in lean and obese ADSCs. * Difference in the same group before and after treatment with LG. ** P < 0. 01, * P < 0.05. # Significance of treated obese ADSCs vs treated lean ADSCs; # P < 0.05. D, Effect of LG on the mRNA levels of leptin and their receptors in MCF-7 cells incubated in lean/obese ADSCs–CM. * Difference in the same group before and after treatment with LG; *** P < 0.001, * P < 0.05. # Significance of treated MCF-7 in obese ADSCs-CM vs treated MCF-7 in lean conditions; # P < 0.05.

(continued).
Concerning leptin and their receptors (ObR1 and ObRt), their mRNA levels were significantly down-regulated in obese ADSCs after treatment with LG (P < 0.01, P < 0.01, P < 0.05, respectively) (Figure 6C) and these levels were significantly lower compared to the levels in lean ADSCs (P < 0.05). In MCF-7 cells incubated in obese ADSCs-CM (Figure 6D), the mRNA levels of leptin, ObR1, ObRt were significantly down-regulated in (P < 0.001). The expression of OBR1 and ObRt in MCF-7 cells cultured in obese ADSCs -CM was significantly lower compared to their expression MCF-7 cells cultured in lean ADSCs-CM (P < 0.05).
Discussion
Growing evidence revealed that obesity promotes BC cells proliferation, migration, and invasion via modulating the levels of the adipokines secreted from obese ADSCs. 8,11,12,15 In the current study, we investigated the anti-proliferative activity of LG on obesity-associated BC. The impact of LG on modulating the adipokines in ADSCs and BC cells was also studied. We used MCF-7 cells which are the most frequently hired BC cell line in research and are estrogen receptor-positive with a high proliferation rate.
First, we investigated the effect of obese ADSCs on the proliferation of MCF-7 cells by using conditioned media. We found that the proliferation of MCF-7 BC cells significantly increased in obese ADSCs-CM compared to lean ADSCs-CM as well as to basal media. In their work, and similar to our data, Kucerova et al. 41 and Strong et al. 12 found that CM collected from obese ADSCs increased BC cells proliferation suggesting a cell-to-cell contact-independent mechanism, and that the high levels of cytokines and growth factors in obese ADSCs are released into CM promoting BC cells growth. Also, and in agreement with our data, Strong et al. 8 found that ADSCs-CM from lean people had less impact on BC cells proliferation.
Collective data from our study revealed—for the first time—the effective anti-proliferative activity of LG on BC cells isolated from obese women. LG markedly reduced the proliferation of MCF-7 cells in obese ADSCs-CM in a dose-dependent manner. The selected dose (50 µM) showed nearly 50% proliferation inhibition in obese ADSCs-CM, diminished colony formation and led to G0/G1 phase arrest in cell cycle progression. Interestingly, the anti-proliferative effects of LG were higher in obese population compared to lean one which strongly implies the potent anti-neoplastic activity of LG in obese subjects.
To understand the mechanism that underlies the anti-proliferative effects LG on BC cells in obese ADSCs-CM, we investigated its effect on adipokines that affect BC cell proliferation in obesity—including IL-6, TNF-α, leptin and adiponectin—in both obese ADSCs and MCF-7 cancer cells.
As mentioned before, ADSCs from obese women secreted higher levels of pro-inflammatory cytokines including IL-6 and TNF-α 11 -14,16 and this inflammatory microenvironment triggers cancer development. 3,5 TNF-α has been shown to be involved in all stages of carcinogenesis including survival, proliferation, invasion, angiogenesis and metastasis. 3,5 IL-6 is a key modulator in inflammation-associated tumorgenesis in obesity 42 via regulating the expression of genes involved in the different steps of tumor growth and progression. 5 Concerning BC in particular, IL-6 also plays a role in ADSCs-related BC progression and induces proliferation and a more aggressive phenotype in ER+ BC cells occurrence. 12,16,41 Our study showed that LG ameliorated the significant high levels of IL-6 and TNF-α produced from obese ADSCs suggesting the association of anti-inflammatory effects with its anti-proliferative efficacy in obese conditions. Recent studies proved the potent anti-inflammatory effect of LG in obese diabetic patients 37 and in obesity-related glomerulopathy 43 via down-regulation of pro-inflammatory factors including TNF-α and IL-6.
Among the cytokines that are abundantly secreted by obese ADSCs, leptin plays a significant role in stimulating BC cells proliferation. 8,12,18 In the current study, we revealed augmented levels of leptin produced from obese ADSCs, which are significantly higher than those produced from lean ADSCs. Consistent with our results, Strong et al. 12 revealed that leptin expression significantly increased in obese ADSCs compared with lean ADSCs. Orecchioni et al. 3 and Strong et al. 12 revealed that leptin produced by obese adipose tissue enhances the proliferation and metastasis of ER+ breast cancers. Leptin delivers into cancer cells, binds to its receptors (OBRt and OBR1), and favors cancer cells proliferation and survival via its anti-apoptotic and proangiogenic properties 6,25 as well as triggering the release of pro-inflammatory cytokines favoring the state of obesity-associated chronic inflammation 27,28 which in turn triggers tumor growth. 3 In addition we reported significant higher expression of leptin, OBRt and OBR1 in MCF-7 cells cultured in obese ADSCs-CM compared to cancer cells incubated in lean CM which could also explain the higher proliferative activities of MCF-7 cells in obese CM. Strong et al. 18 reported that obese ADSCs alters the secretory profile of MCF-7 cells, they detected 102 cytokines released from MCF-7 cells co-cultured with lean or obese ADSCs including leptin with significant higher expression of leptin from MCF-7 cells cultured in obese ADSCs compared to those cultured in lean ADSCS. Our data revealed that also CM from obese ADSCs resulted in higher expression of leptin and its receptor in MCF-7 cells. It has been demonstrated that inhibiting leptin expression in obese ADSCs reduced its ability to induce BC proliferation, reduced tumor volume and metastasis to distant organs, reducing the burden of obesity-associated breast cancers. 12
In contrast to leptin, the levels of adiponectin are significantly lower in obese ADSCs compared to lean one and the expression of adiponectin and its receptors are also significantly depleted in both obese ADSCs and MCF-7 cultured in obese ADSCs. Since, adiponectin is an anti-proliferative adipokine via pro-apoptotic and anti-inflammatory effects in addition to inducing cell cycle arrest 22,25,44,45 therefore these reduced levels in obese subjects may participate in the increased proliferation of MCF-7 cells. Adiponectin has the ability to reduce the production of TNF-α, 5 and was shown to reduce TNF-α induced effect on cell proliferation and migration 44,45 ; therefore the reduced levels of adiponectin may participate in the elevated levels of TNF-α in obese condition.
Our study showed that treatment with LG successfully reduced the levels and expression of leptin & its receptors, while enhancing the expression of adiponectin & its receptors both in obese ADSCs and MCF-7 cancer cells incubated in obese ADSCs-CM. Reduction in leptin expression along with increased adiponectin by LG limits the viability of MCF-7 cells and may participate to the decreased production of inflammatory cytokines and hence block their impact on tumor growth. Few reports are available about the effect of LG on adipokines production from adipose tissue. Only recently, LG treatment resulted in up-regulation of adiponectin along with down-regulation of leptin in differentiated 3T3-L1 adipocytes, 46 and as well it increased the expression of adiponectin and reduced the body weight in polycystic ovary in mice. 46 Previously, LG causes upregulation of serum adiponectin in type 2 diabetic patients. 36 Moreover, it has been demonstrated that the activity of LG to reduce body weight is due to reducing leptin plasma level 35 and that, GLP-1 analogue, NN2211, induced weight loss and reduced adiposity as indicated by lowered plasma levels of leptin in both normal lean and obese rats. 47
Reduction in leptin expression along with enhanced expression of adiponectin by LG may participate to the decreased production of inflammatory cytokines and hence block their impact on tumor growth.
In the current study, the beneficial effects of LG on leptin, adiponectin and their receptors were higher and more obvious in obese cases compared to lean ones which is parallel to the higher anti-proliferative activity on obese conditions compared to lean ones. Interestingly, Kucerova et al. 48 revealed that chemosensitivity of BC cells to anticancer drugs was increased by ADSCs-secreted cytokines, this may be an explanation of the higher efficacy of LG on MCF-7 cells cultured in obese ADSCs-CM compared to cancer cells incubated in lean CM, where obese ADSCs secrete more inflammatory and growth factors compared to lean ADSCs. According to our data, the ability of LG to suppress the proliferation and survival of MCF-7 cells cultured in obese ADSCs-CM may be via modulating the production of leptin, adiponectin and their receptors from both ADSCs and BC cells.
Conclusion
Our results demonstrated, for the first time, that LG could suppress BC growth and induce cell cycle arrest in obese subjects, via correcting the levels adipokines and their receptors in both ADSCs and cancer cells. Our results may provide a novel strategy for the clinical prevention and treatment of breast cancer in obese population. This study may also provide a basis for the selection of hypoglycemic agents in obese patients with type 2 diabetes, particularly with BC. In order to understand how the modulating effect of LG on leptin/adiponectin could suppress the BC cell growth, further study is recommended to investigate the effect of LG on leptin/adiponectin pathways in ADSCs.
Footnotes
Acknowledgment
This research project was supported by a grant from the “Research Center of the Female Scientific and Medical Colleges,” Deanship of Scientific Research, King Saud University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Received financial support from the “Research Center of the Female Scientific and Medical Colleges,” Deanship of Scientific Research, King Saud University.

