Abstract
Objectives
This study investigated the cardioprotective effects of stachydrine (STA) in lipopolysaccharide (LPS)-induced septic mice and H9c2 cardiomyocytes, focusing on its anti-apoptotic, anti-inflammatory, and anti-ferroptotic actions.
Methods
We established an LPS-induced sepsis model in mice and an LPS-stimulated H9c2 cardiomyocyte model in vitro.
Results
STA markedly reduced LPS-induced myocardial apoptosis, as demonstrated by decreased TUNEL-positive cells, and attenuated the elevation of serum cardiac injury markers, including creatine kinase-MB (CK-MB), lactate dehydrogenase (LDH), brain natriuretic peptide (BNP), cardiac troponin I (cTnI), and cardiac troponin T (cTnT) levels. STA also suppressed systemic inflammation, significantly reducing interleukin-1 beta (IL-1β), IL-6, and tumor necrosis factor-alpha (TNF-α) levels at both mRNA and protein levels. Additionally, STA significantly inhibited LPS-induced production of pro-inflammatory cytokines in H9c2 cardiomyocytes. Mechanistically, STA activated the SIRT1/Nrf2 signaling axis and enhanced the expression of ferroptosis-related proteins, solute carrier family 7 member 11 (SLC7A11) and glutathione peroxidase 4 (GPX4). Additionally, STA reduced oxidative stress and iron accumulation by decreasing malondialdehyde (MDA), Total Fe, and Fe2+ levels, while increasing glutathione (GSH) content in cardiomyocytes.
Conclusion
Our results suggest that STA confers robust cardioprotective effects in LPS-induced models by mitigating apoptosis, inflammation, and ferroptosis, partly via SIRT1/GPX4 pathway activation.

Introduction
Sepsis is a life-threatening condition characterized by dysregulated host immune responses to infection, often resulting in multiple organ dysfunction and high mortality rates. 1 During sepsis, the innate immune system serves as the first line of defense, with monocytes and macrophages playing pivotal roles. These immune cells become excessively activated, leading to the overproduction of pro-inflammatory cytokines such as interleukin (IL)-1β, IL-6, and interferon-gamma (IFN-γ). 2 This excessive and uncontrolled cytokine release, commonly referred to as a “cytokine storm,” contributes significantly to systemic inflammation and the progression of multi-organ failure. 3
The cardiovascular system is particularly susceptible to the detrimental effects of systemic inflammation during sepsis. Sepsis-induced cardiomyopathy (SIC), characterized by transient myocardial dysfunction, is a frequently observed complication, often presenting with both systolic and diastolic impairments that lead to reduced cardiac output and inadequate tissue perfusion.4-6 Patients with SIC have a two- to three-fold higher mortality rate than those without cardiac involvement. 4 The pathophysiology of SIC is complex and multifactorial, involving dysregulated pro- and anti-inflammatory cytokine signaling, excessive production of nitric oxide and reactive oxygen species (ROS), mitochondrial and sarcomere protein downregulation, impaired coronary microvascular perfusion, and disrupted calcium homeostasis.5,6 Despite growing insights into its underlying mechanisms, effective therapeutic strategies and prognostic markers for SIC remain limited and poorly defined.
The SIRT1/GPX4 signaling pathway plays a pivotal role in regulating cellular responses to oxidative stress and ferroptosis, a distinct form of regulated cell death characterized by iron-dependent lipid peroxidation. Sirtuin 1 (SIRT1), a NAD+-dependent deacetylase, is involved in a wide range of cellular functions, including aging, transcriptional regulation, and resistance to oxidative stress and inflammation. 7 Glutathione peroxidase 4 (GPX4), on the other hand, is a key antioxidant enzyme that directly reduces lipid hydroperoxides to non-toxic lipid alcohols, thereby preventing the accumulation of lipid peroxides and protecting cells from ferroptotic death.6 The SIRT1/GPX4 axis has been implicated in various pathological conditions such as depression, anxiety, and rheumatoid arthritis, where oxidative stress and inflammation are central to disease progression.7,8 Activation of this pathway has shown therapeutic potential in mitigating oxidative damage and restoring cellular homeostasis. However, the role of stachydrine (STA) in modulating ferroptosis via the SIRT1/GPX4 pathway in SIC remains largely unexplored.
Motherwort, known as Yi Mu Cao in traditional Chinese medicine, has long been valued for its therapeutic effects in gynecological and obstetric disorders, as well as cardiovascular diseases.9,10 One of its primary bioactive alkaloids, STA has been widely investigated for its diverse pharmacological properties, including anti-inflammatory, anti-fibrotic, anti-tumor, and antioxidant activities.11-14 The chemical structure of STA is presented in Figure 1A. Numerous studies have demonstrated the ability of STA to attenuate inflammatory responses in various pathological contexts, such as liver fibrosis, osteolytic bone diseases, and endothelial injury caused by lipopolysaccharide (LPS) exposure or anoxia-reoxygenation injury.13,15,16 Notably, STA exerts strong anti-inflammatory effects by suppressing cytokine release and inhibiting p65 phosphorylation in models of cerebral ischemia-reperfusion injury.
17
Furthermore, STA has been shown to protect chondrocytes in osteoarthritis by downregulating IL-1β-induced inflammatory signaling.
18
Collectively, these findings suggest that STA functions as a potent modulator of inflammation in multiple disease models. However, despite these promising effects, its pharmacological role in SIC remains poorly understood and requires further exploration. STA Treatment Shows No Systemic Toxicity and Reduces the 72-h Mortality Rate. (A) Chemical Structure of Stachydrine (STA, C7H13NO2). Mice Were Gavaged With STA (20 mg/kg/day) for Seven Consecutively Days. Serum Hepatic and Renal Function Indicators Were Measured in Mice at 7 days after STA Administration, including (B) ALT, AST, (C) Creatinine and BUN. (D) The Representative Image of Hepatic and Renal Tissues of HE Staining (Magnification 200×). STA was Gavaged (20 mg/kg/day) in Mice for 2 hours before Intraperitoneal Injection of LPS (20 mg/kg) and for 3 Days. (E) The Survival Rate of Each Group was Measured. Kaplan-Meier Curves for the 72-h Mortality Rate Were Plotted. Data are Presented as Mean ± SD (n = 8 in Each Group). T test was Used to Compare the Differences in Hepatic and Renal Toxicity. The Log-Rank (Mantel-Cox) Test was Used to Compare the 72-h Mortality Rate. STA, Stachydrine; LPS, Lipopolysaccharide; ALT, Alanine Transaminase; AST, Aspartate Transaminase; BUN, Blood Urea Nitrogen
This study aimed to investigate the protective effects of STA in a murine model of SIC and elucidate the underlying mechanisms, with a particular focus on the suppression of ferroptosis via activation of the SIRT1/GPX4 signaling pathway. Our findings offer novel therapeutic insights into the potential of targeting ferroptosis as a strategy for mitigating SIC.
Materials and Methods
Animals and Treatment
Male C57BL/6 mice (8-10 weeks, approximately 25 g) were obtained from Shanghai SLAC Laboratory Animal Co., Ltd Shanghai, China, housed in a SPF and temperature-controlled conditions with a 12-hour light/dark cycle, and had free access to tap water and food. STA (HY-N0298, purity ≥98%) was obtained from MedChemExpress (Monmouth Junction, NJ, USA). In vivo and in vitro experiments were performed in our laboratory from March 2022 to December 2024. The Animal Ethics Committee of the Laboratory Animal Center of our hospital approved all experiments. In the first experiment, mice were randomly assigned to two groups: (a) control and (b) stachydrine (STA, 20 mg/kg/day). A sepsis model was established by intraperitoneal injection of 10 or 20 mg/kg of LPS. In the second experiment, the mice were randomly assigned to three groups: (a) control, (b) LPS, and (c) LPS + STA. Mice were gavaged with saline containing 0.1% DMSO or STA (20 mg/kg/day) 2 h before intraperitoneal injection of LPS and for three consecutive days. For the 72-h mortality rate, one dose of LPS at 20 mg/kg was used, for the other experiments, LPS at 10 mg/kg was used. Two hours after the last administration, the mice were anesthetized with pentobarbitone. Blood samples were collected from the abdominal aorta to separate the serum and stored at −20°C for testing.
Biochemical Analysis
Serum was collected from mice on day 3 following the initial infusion of LPS to analyze biochemical markers, including alanine transaminase (ALT) (C009-2-1, Nanjing Jiancheng Bioengineering Institute, Nanjing, China), aspartate aminotransferase (AST) (C010-2-1, Nanjing Jiancheng), and lactate dehydrogenase (LDH) (E-BC-K046-M, Elabscience, China) activity. An automatic biochemical analyzer was used to measure serum creatinine (Scr) and blood urea nitrogen (BUN) levels.
Enzyme-Linked Immunosorbent Assay (ELISA)
The quantification of several biomarkers, including creatine kinase MB (CK-MB) (SEKM-0152, Solarbio, China), brain natriuretic peptide (BNP) (E-EL-M0204c, Elabscience, China), cardiac troponin I (cTnI) (SEKM-0153, Solarbio, China), cardiac troponin T (cTnT) (SEKM-0150, Solarbio, China), interleukin-1β (IL-1β, MLB00 C, R&D Systems), interleukin-6 (IL-6, M6000 B, R&D Systems), and tumor necrosis factor-alpha (TNF-α, MTA00 B, R&D Systems) in blood serum samples was performed using ELISA assay kits. Samples were collected from murine subjects on the third day after the initial LPS administration. The optical density at 450 nm was measured using a microplate reader, and the concentrations of the biomarkers were determined using a standard calibration curve.
Histology
Hepatic, renal, and cardiac tissues were preserved in 4% paraformaldehyde overnight, embedded in paraffin, and sectioned to a thickness of 5 μm. The morphological characteristics of the hepatic and renal tissues were evaluated using hematoxylin and eosin (HE) staining. Masson’s trichrome staining was employed to assess the degree of collagen deposition in the left ventricle. A minimum of five randomly selected fields of view from each histological section were analyzed, and the ratio of the fibrotic area was calculated. The resulting images were analyzed using Image-Pro Plus (version 6.0).
Tissue Immunofluorescence
Immunofluorescence was performed following a previously established protocol. 19 Frozen sections of murine atrial tissues were collected for analysis. After three washes with phosphate-buffered saline (PBS), terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL, C1086; Beyotime) was applied. The specimens were stained with 4′, 6-diamidino-2-phenylindole (DAPI) and examined using an inverted microscope (IX51, Olympus, Japan). TUNEL procedures were conducted according to the manufacturer’s guidelines.
Cell Culture and Treatment
The rat cardiac muscle cell line H9c2 was purchased from Cell Biotech Co., Ltd (Shanghai, China). H9c2 cells were cultivated in complete medium supplemented with 10% FBS (Gibco, USA), and 100 U/mL penicillin/streptomycin and maintained at 37°C in 5% CO2. To construct an in vitro septic cardiomyopathy model, H9c2 cells were treated with different concentrations of LPS for 24 h. Further analysis was conducted by treating H9c2 cells with 10 μg/mL LPS for 24 h, and PBS treatment was used as a control. To explore the protective effect of STA, 5, 10, 20, 50, and 100 μM STA were used to treat LPS-induced H9c2 cells for 24 h.
Cell Viability
H9c2 cells (1 × 104 cells per well) were inoculated into 96-well plates and treated with 0, 5, 10, 20, 50, and 100 μM STA for 24 h. H9c2 cells were treated with 0, 1, 2, 5, 10, and 20 μg/mL LPS for 24 h. In addition, H9c2 cells were pretreated with STA at a concentration of 50 μM for 2 h before incubation with 10 μg/mL LPS for 24 h. After the treatments were completed, 10 μL of CCK-8 solution (Beyotime) was added to each well. Following a 3-hour incubation period, a microplate reader was used to quantify the cell absorbance at a wavelength of 450 nm.
Apoptosis
H9c2 cells were cultured in a 24-well plate at a density of 5 × 104 cells/well and treated for 48 h. After treatment, the cells were dissociated using trypsin and stained with Annexin V-FITC and propidium iodide (PI) for 15 min at room temperature. The rate of cell apoptosis was determined using flow cytometry by analyzing the positive cells for Annexin V-FITC and the negative cells for PI. To quantify apoptotic cells, we used an Annexin V-FITC assay kit (Solarbio, Beijing, China), ensuring the validity and reliability of our results.
ELISA
ELISA was used to measure the levels of inflammatory cytokines in the culture medium of H9c2 cells. Following various treatments, supernatants were collected, and the levels of interleukin-1β (IL-1β, MLB00 C, R&D Systems), interleukin-6 (IL-6, M6000 B, R&D Systems), and tumor necrosis factor-alpha (TNF-α, MTA00 B, R&D Systems) were assessed using ELISA kits (R&D Systems, Minneapolis, MN, USA). Cytokines concentrations were expressed in picograms per milliliter (pg/mL).
Measurement of Oxidative Stress Indicators
H9c2 cells were homogenized at a concentration of 10% (w/v) to measure oxidative stress markers. This was conducted before assessing oxidative stress markers, such as malondialdehyde (MDA) (S0131S, Beyotime, Shanghai, China) and glutathione (GSH) (S0051, Beyotime), using commercially available kits.
Iron Quantification
The concentrations of total iron (A039-2-1, Nanjing Jiancheng Bioengineering Institute) and ferrous ions (Fe2+: ab83366, Abcam) in H9c2 cells were analyzed using spectrophotometry. Iron was carefully added to 96-well plates, including a blank (ddH2O), iron standard solution, and test samples for analysis. Subsequently, Fe assay buffer and ferrous chromogenic solution were added to each well and incubated. The samples were then dissolved in 2 M HCl, followed by spectrophotometric measurement at 560 nm. 20
Quantitative Reverse Transcription Polymerase Chain Reaction (RT-qPCR)
List of Primer Sequences Used in This Investigation

Western Blotting
Total protein isolation was performed using RIPA lysis buffer. Protein concentration was measured using a BCA protein assay kit (Beyotime Biotechnology, China). Protein samples (50 μg each) were separated using 10% SDS-PAGE and transferred to PVDF membranes. The membranes were blocked with 5% low-fat milk and incubated with primary antibodies: • SIRT1 (1:500, sc-74465, mouse monoclonal, Santa Cruz) • Nrf2 (1:500, ab313825, rabbit monoclonal, Abcam) • SLC7A11 (1:500, ab175186, rabbit monoclonal, Abcam) • GPX4 (1:500, sc-166570, mouse monoclonal, Santa Cruz)
Subsequently, HRP-linked secondary antibodies were applied to the membranes. GAPDH (1:2000, ab9485, rabbit polyclonal, Abcam) and PCNA (1:2000, ab92552, rabbit monoclonal, Abcam) were used as internal controls. Finally, ECL (Thermo, Waltham, MA, USA) was used to visualize the bands, and ImageJ was used for analysis.
Statistical Analysis
All data are presented as mean values with standard deviation (SD) calculated from at least three independent experiments. Statistical analyses were performed using SPSS 20.0 (SPSS, Chicago, IL, USA) or GraphPad Prism 9.0 software. Differences between groups were assessed using t test or one-way ANOVA with post hoc Tukey’s test. The log-rank (Mantel-Cox) test was used to compare the 72-h mortality rates. Statistical significance was set at P value of less than 0.05.
Results
STA Treatment Exhibited No Systemic Toxicity and Improved Survival Outcomes in Mice
To assess systemic toxicity, mice were administered STA (20 mg/kg/day) via oral gavage for 7 days. Serum analysis revealed no significant alterations in hepatic or renal function markers, including alanine aminotransferase (ALT), aspartate aminotransferase (AST) (Figure 1B), creatinine, and blood urea nitrogen (BUN) (Figure 1C), indicating no adverse effects on liver or kidney function. Furthermore, histopathological examination of hepatic and renal tissues via hematoxylin and eosin (H&E) staining (200× magnification) confirmed the absence of STA-induced tissue damage (Figure 1D).
In a separate survival study, mice pretreated with STA (20 mg/kg/day) for 2 h prior to LPS (20 mg/kg) challenge and continued for 3 days exhibited a significant reduction in 72-hour mortality compared to untreated controls, as demonstrated by the survival curve in Figure 1E. These findings suggest that STA administration is well tolerated and confers a protective effect against LPS-induced lethality.
STA Suppressed Myocardial Apoptosis and Injury in LPS-Treated Mice
To evaluate the protective effects of STA against LPS-induced myocardial apoptosis and injury, we first assessed apoptosis levels in the myocardium using TUNEL staining. As shown in Figure 2A, LPS treatment significantly increased the number of TUNEL-positive cells compared to the control group, whereas STA administration markedly reduced myocardial apoptosis. Quantitative analysis confirmed that STA significantly decreased the percentage of TUNEL-positive cells in LPS-treated mice (P < 0.05) (Figure 2B). STA Suppresses Myocardium Apoptosis and Injury in LPS-Treated Mice. (A) Apoptosis was Evaluated by TUNEL Staining in the Myocardium (Magnification 200×). (B) Quantification of TUNEL-Positive Cells. Spectrophotometer and ELISA Were Used to Measure Serum Levels of Myocardial Injury-Related Molecules, including (C) CK-MB, (D) LDH, (E) BNP, (F) cTnI, and (G) cTnT. Data are Presented as Mean ± SD (n = 8 Mice in Each Group) and Analyzed Using One-Way ANOVA, With the Bonferroni Test Employed for the Post Hoc Analysis. ***P < 0.001 vs Control Group; ###P < 0.001 vs LPS Group. CK-MB, Creatine Kinase Isoenzyme MB; LDH, Lactate Dehydrogenase; BNP, Brain Natriuretic Peptide; cTnI, Cardiac Troponin I; cTnT, Cardiac Troponin T
Next, we measured the serum levels of biomarkers related to myocardial injury. LPS challenge caused a substantial increase in the release of creatine kinase-MB (CK-MB), lactate dehydrogenase (LDH), brain natriuretic peptide (BNP), cardiac troponin I (cTnI), and cardiac troponin T (cTnT) (Figure 2C-G). STA treatment significantly attenuated these elevations, indicating a protective effect against LPS-induced myocardial injury (P < 0.05). Collectively, these results demonstrate that STA effectively suppresses myocardial apoptosis and mitigates cardiac injury in LPS-treated mice.
STA Inhibited LPS-Induced Inflammation in Mice
To investigate the effects of STA on LPS-induced inflammation in mice, several inflammatory biomarkers, including IL-1β, IL-6, and TNF-α, were quantified in serum samples using ELISA. LPS treatment resulted in elevated IL-1β, IL-6, and TNF-α levels in the LPS group. In contrast, STA treatment significantly reduced the levels of these cytokines in the LPS + STA group (Figure 3A-C). Furthermore, RT-qPCR was performed to corroborate the ELISA results. The mRNA expression levels of IL-1β, IL-6, and TNF-α were increased by LPS treatment and subsequently decreased by STA treatment (Figure 3D-F). These findings suggest that STA effectively attenuates LPS-induced inflammation in mouse models. STA Suppresses LPS-Induced Inflammation in Mice. (A, B, C) the Levels of Inflammatory Factors IL-1β, IL-6, and TNF-α Were Measured in the Serum of LPS-Treated Mice by ELISA. (D, E, F) the mRNA Levels of IL-1β, IL-6, and TNF-α Were Measured in the Myocardium of LPS-Treated Mice by RT-qPCR. Data are Presented as Mean ± SD in Triplicates and Analyzed Using One-Way ANOVA, With the Bonferroni Test Employed for the Post Hoc Analysis. ***P < 0.001 vs Control Group; ###P < 0.001 vs LPS Group
STA Suppressed Viability Reduction and Apoptosis in LPS-Treated H9c2 Cells
In this study, we evaluated the possible effects of STA on LPS-induced cell viability and apoptosis in H9c2 cells. H9c2 cells were treated with 0, 5, 10, 20, 50, and 100 μM STA for 24 h. The CCK-8 assay was used to determine the viability of H9c2 cells. The results showed no adverse effects of STA on the cells (Figure 4A). In addition, H9c2 cells were treated with 0, 1, 2, 5, 10, and 20 μg/mL LPS for 24 h. Cell viability was determined using a CCK-8 assay, and it was found that cell viability was significantly reduced at 5 and 10 μg/mL dosages of LPS (Figure 4B). We pretreated H9c2 cells with 50 μM STA for 2 h and incubated them with 10 μg/mL LPS for 24 h. The results showed that LPS treatment reduced cell viability, which was improved by STA treatment (Figure 4C). Cell apoptosis was evaluated using Annexin V-FITC and PI double staining and analyzed by flow cytometry. The results indicated that LPS treatment elevated the apoptosis rate in the LPS group, which was reduced by STA treatment in the LPS + STA group (Figure 4D). After quantification analysis, we observed consistent results (Figure 4E). These results demonstrate that STA inhibited the reduction in cell viability and apoptosis in LPS-induced H9c2 cells. STA Suppresses Viability Reduction and Apoptosis in LPS-Treated Cells. (A) H9c2 Cells Were Treated With 0, 5, 10, 20, 50 and 100 μM STA for 24 h. The Cell Viability of H9c2 Cells was Determined by CCK-8 Assay. (B) H9c2 Cells Were Treated With 0, 1, 2, 5, 10 and 20 μg/mL LPS for 24 h. Cell Viability was Determined by Using a CCK-8 Assay. (C) H9c2 Cells Were Pretreated With STA at 50 μM for 2 h and Incubated With 10 μg/mL LPS for 24 h. (D) Cell Apoptosis was Evaluated by Annexin V-FITC and PI Double Staining and Analyzed by Flow Cytometry. (E) Quantification of Apoptotic Rate of H9c2 Cells. Data are Presented as Mean ± SD in Triplicates and Analyzed Using One-Way ANOVA, With the Bonferroni Test Employed for the Post Hoc Analysis. ***P < 0.001 vs Control Group; ###P < 0.001 vs LPS Group
STA Attenuated LPS-Induced Inflammation in Cardiomyocytes
ELISA was performed to measure the levels of inflammatory cytokines in the culture medium of H9c2 cells. Different pro-inflammatory cytokines, including IL-1β, IL-6, and TNF-α, were measured in the cell culture supernatants. The results showed that IL-1β, IL-6, and TNF-α levels were elevated by LPS treatment, whereas STA treatment remarkably reduced IL-1β, IL-6, and TNF-α levels in the supernatants of the cells (Figure 5A-C). Additionally, the mRNA levels of IL-1β, IL-6, and TNF-α were measured in H9c2 cells using RT-qPCR. We observed that LPS treatment stimulated the mRNA levels of IL-1β, IL-6, and TNF-α, which decreased after STA treatment (Figure 5D-F). The results demonstrated that STA suppressed LPS-induced inflammation in cardiomyocytes. STA Suppresses LPS-Induced Inflammation in Cardiomyocytes. (A, B, C) the Levels of Pro-inflammatory Cytokines IL-1β, IL-6 and TNF-α in the Culture Media of H9c2 Cells Were Measured by ELISA. (D, E, F) the mRNA Levels of IL-1β, IL-6 and TNF-α Were Measured in H9c2 Cells by RT-qPCR. Data are Presented as Mean ± SD in Triplicates and Analyzed Using One-Way ANOVA and the Bonferroni Test for the Post Hoc Test. ***P < 0.001 vs Control Group; ###P < 0.001 vs LPS Group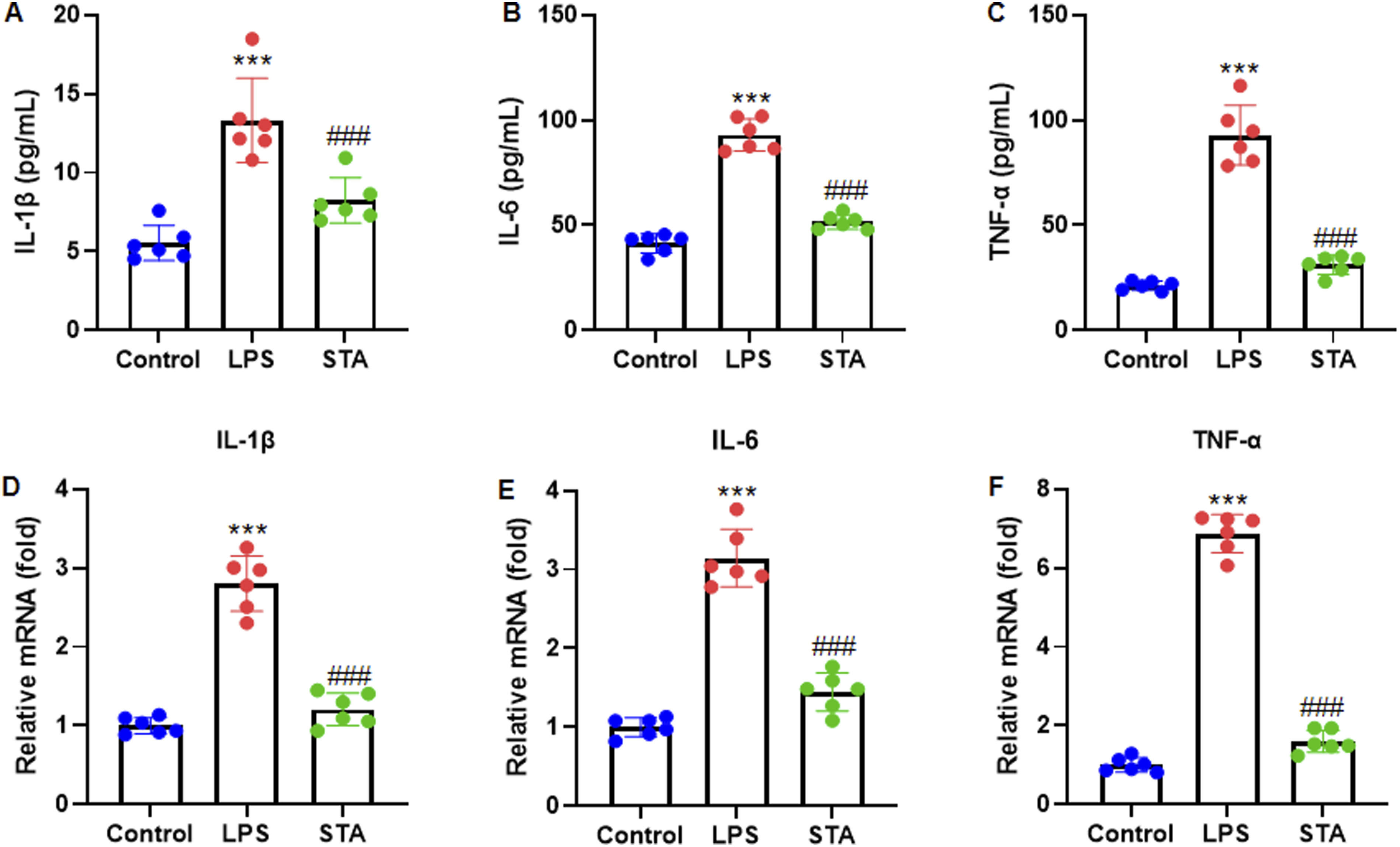
STA Activated SIRT1 and Inhibited Ferroptosis in Cardiomyocytes Treated with LPS
To investigate the impact of STA on the SIRT1/GPX4 pathway and ferroptosis in LPS-treated cardiomyocytes, we performed western blot analysis to determine the expression of SIRT1, Nrf2 and two ferroptosis-related proteins, SLC7A11 and GPX4. The results showed that LPS treatment decreased SIRT1, Nrf2, SLC7A11, and GPX4 expression levels, which were elevated by STA treatment (Figure 6A). After quantification analysis, we observed the same trends in SIRT1, Nrf2, SLC7A11, and GPX4 expression levels (Figure 6B-E). Additionally, the cell lysate of H9c2 cells was used to measure the oxidative stress and ferroptosis markers, including MDA, GSH, Total Fe and Fe2+. The results indicated that LPS treatment elevated the MDA, Total Fe and Fe2+ levels but reduced the GSH level. In contrast, STA treatment reduced MDA, total Fe, and Fe2+ levels, whereas GSH levels increased (Figure 6F-I). These results demonstrate that STA activated the SIRT1/ GPX4 pathway and inhibits ferroptosis in LPS-treated cardiomyocytes. STA Activates SIRT1 and Inhibits Ferroptosis in LPS-Treated Cardiomyocytes. (A) Western Blot was Performed to Determine SIRT1, Nrf2 and Two Ferroptosis-Related Proteins, SLC7A11 and GPX4. (B, C, D, E) Quantification of SIRT1, Nrf2, SLC7A11 and GPX4 Protein. SIRT1, SLC7A11 and GPX4 Were Normalized to GAPDH, and Nrf2 was Normalized to PCNA. The Cell Lysate of H9c2 Cells was Used to Measure the Oxidative Stress and Ferroptosis Markers for (F) MDA, (G) GSH, (H) Total Fe and (I) Fe2+. Data are Presented as Mean ± SD in Triplicates and Analyzed by ANOVA. ***P < 0.001 vs Control Group; ###P < 0.001 vs LPS Group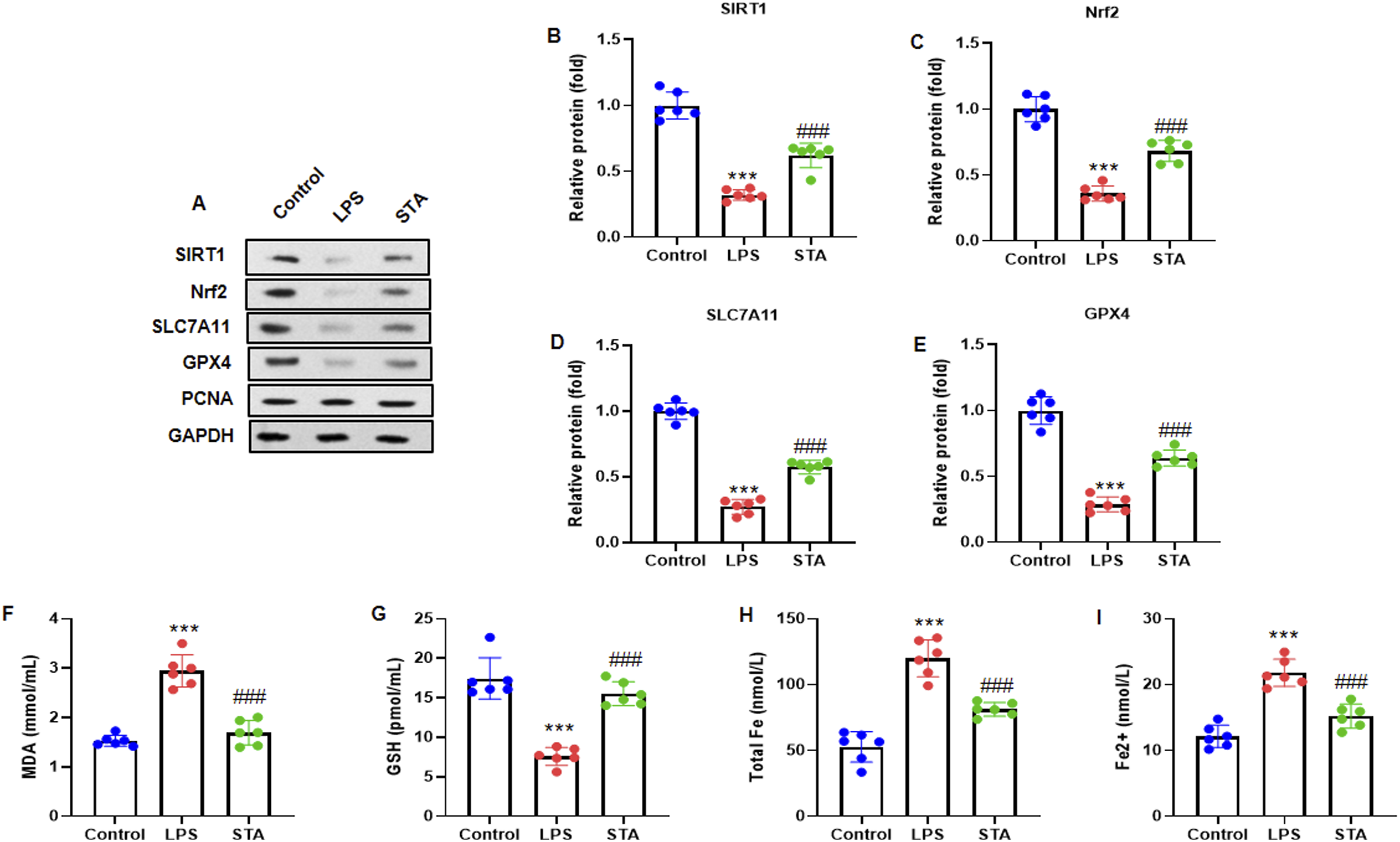
Discussion
In this study, we demonstrated that STA confers significant cardioprotective effects in both in vivo and in vitro models of sepsis-induced cardiomyopathy (SIC). STA treatment not only improved survival outcomes and attenuated systemic toxicity in LPS-challenged mice but also reduced myocardial apoptosis, inflammation, and injury in the heart. Mechanistically, our results show that STA activates the SIRT1/Nrf2/GPX4 axis, thereby suppressing ferroptosis, a form of regulated cell death driven by iron-dependent lipid peroxidation. These findings are consistent with recent studies suggesting that inhibition of ferroptosis is a promising therapeutic strategy for septic organ dysfunction, including the heart.22,23
The absence of toxicity following STA administration is a crucial finding, particularly for translational applications. STA treatment (20 mg/kg/day) did not alter the serum levels of liver enzymes (ALT and AST) or kidney function markers (creatinine and BUN), and histopathological analysis confirmed intact tissue architecture, suggesting excellent systemic tolerance. These results are consistent with prior safety reports of STA in cardiovascular models, 24 supporting its use as a therapeutic candidate for treating cardiac hypertrophy. Moreover, STA significantly reduced LPS-induced mortality, highlighting its potential to counteract sepsis-associated mortality. In sepsis models, ferroptosis and excessive inflammation play central roles in organ failure and death. 24 The observed survival advantage aligns with the notion that suppressing these pathophysiological processes can meaningfully impact outcomes.
STA markedly reduced myocardial apoptosis in vivo, as shown by a decrease in TUNEL-positive nuclei in heart tissue, and attenuated LPS-induced elevations in cardiac injury markers, such as CK-MB, LDH, BNP, cTnI, and cTnT. These findings demonstrate that STA protects cardiomyocytes from LPS-induced injury, likely through the inhibition of apoptosis. Similar cardioprotective effects of STA have been reported in ischemia-reperfusion injury, where STA was shown to reduce oxidative damage and cell death. 17
The anti-inflammatory effects of STA were evident in both in vivo and in vitro studies. In LPS-induced mice, STA significantly lowered the serum levels and mRNA expression of pro-inflammatory cytokines IL-1β, IL-6, and TNF-α. Similarly, in H9c2 cardiomyocytes, STA reduced cytokine secretion and mRNA expression in response to LPS. This is particularly relevant, as cytokine storms contribute to myocardial dysfunction and mortality in sepsis. 25
Several pharmacological agents, including Ferrostatin-1 (Fer-1), resveratrol, and dexmedetomidine, have been investigated for SIC. Fer-1, a potent ferroptosis inhibitor, improves cardiac function and reduces lipid peroxidation in septic mice. 22 Similarly, resveratrol, through activation of SIRT1 and suppression of oxidative stress, has demonstrated anti-inflammatory and anti-apoptotic effects in SIC. 26 The mechanism of action of STA appears to overlap with that of both agents, as it activates SIRT1 and suppresses ferroptosis while also providing anti-inflammatory benefits. This suggests that STA integrates the beneficial properties of multiple agents, potentially offering a broader therapeutic profile. Notably, STA-induced SIRT1 upregulation and consequent activation of Nrf2 and GPX4, along with elevated SLC7A11 levels, established its role in mitigating ferroptosis. These findings are in line with those of a recent study that reported that STA alleviates myocardial ferroptosis in heart failure by modulating the SIRT1/GPX4 axis.27 The present data extend these findings to an acute sepsis model, reinforcing the role of STA in regulating ferroptosis beyond chronic heart conditions.
Ferroptosis, characterized by iron accumulation and lipid peroxidation, is a major contributor to cardiomyocyte death during sepsis. Our data show that STA decreased intracellular levels of MDA, Fe2+, and total iron, while increasing GSH levels, a key antioxidant. These biochemical changes, along with the restored expression of SIRT1, GPX4, and SLC7A11, indicate that ferroptosis inhibition is a critical mechanism underlying cardioprotective effects of STA. This is especially important because ferroptosis has emerged as a therapeutic target for SIC. Prior studies have highlighted the benefits of targeting ferroptosis to protect against septic myocardial injury,23,28 but many such agents lack clinical viability owing to poor safety profiles or limited bioavailability. With its favorable toxicity profile and efficacy in both cell and animal models, STA represents a compelling alternative.
It is important to acknowledge that while our findings support the role of ferroptosis in the pathogenesis of SIC and its modulation by STA, other forms of programmed cell death, such as autophagy, necroptosis, and pyroptosis, have also been implicated in septic cardiomyopathy and were not assessed in this study. For instance, autophagy plays a dual role in cardiac homeostasis during sepsis and has been shown to be either protective or deleterious depending on its regulation and context.29,30 Necroptosis, a regulated form of necrotic cell death, has also been identified as a contributor to cardiac dysfunction in LPS-induced models through the RIPK1/RIPK3/MLKL axis. 31 Additionally, pyroptosis, a caspase-1-dependent pro-inflammatory cell death process, is known to mediate myocardial injury in sepsis via the NLRP3 inflammasome pathway. 32 Therefore, the exclusive focus on apoptosis, inflammation, and ferroptosis in this study represents a limitation in fully elucidating the spectrum of cell death mechanisms involved in SIC.
Limitations
Although this study provides important insights, several limitations should be acknowledged. First, power analysis was not performed to determine the appropriate sample size in the current study. In future investigations, we will incorporate power calculations to ensure an adequate sample size. Second, we did not evaluate the dose-dependent efficacy of STA in this study. Future research should investigate the effects of STA across different doses. Third, our investigation primarily focused on the anti-ferroptotic effects of stachydrine without concurrently evaluating its potential influence on other forms of regulated cell death, such as autophagy and necroptosis. Further studies are needed to determine whether these additional pathways contribute to the observed cardioprotective effects. Fourth, the external validity of our findings is limited by the use of a single animal model, which may not fully replicate the complexity and heterogeneity of human sepsis-induced cardiomyopathy. Fifth, the absence of gene knock-out or knock-down models restricts our ability to definitively identify and validate the molecular targets and signaling pathways involved in the protective effects of stachydrine. Finally, as in all preclinical studies, caution must be exercised when extrapolating these results to humans. Further research involving multiple animal models and clinical investigations is essential to confirm the translational potential of stachydrine in the treatment of SIC.
Conclusions
In summary, our data suggest that STA offers comprehensive protection against SIC in a murine model by targeting multiple pathological processes, including apoptosis, inflammation, and ferroptosis. Compared to existing candidates like Ferrostatin-1 and resveratrol, STA presents a more holistic therapeutic profile, supported by both in vivo and in vitro evidence. The absence of systemic toxicity, coupled with its multimodal protective mechanisms, highlights the potential of STA for clinical translation in sepsis-associated cardiac dysfunction (Supplement Figure S1).
Supplemental Material
Supplemental material - Stachydrine Alleviates Sepsis-Induced Cardiomyopathy by Inhibiting Ferroptosis via Regulating SIRT1/GPX4 Pathway
Supplemental material for Stachydrine Alleviates Sepsis-Induced Cardiomyopathy by Inhibiting Ferroptosis via Regulating SIRT1/GPX4 Pathway by Linbo Tao, Xiaoquan Huang, Feng Zhao, Xiude Wang in Dose-Response
Footnotes
Ethical Considerations
This study was approved (2024-029) by Nantong Second People’s Hospital Ethics Committee. The authors envisaged all standard protocols in accordance with the 1964 Declaration of Helsinki. The animal experiments in this study were carried out in accordance with the ARRIVE guidelines.
Author contributions
Linbo Tao and Xiaoquan Huang: Conceptualization, Methodology, Data curation, Visualization, Investigation, Writing-original draft. Feng Zhao: Data curation, Visualization, Investigation. Xiude Wang: Project administration, Supervision, Funding acquisition, Resources, Writing-review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding of this research by the Nantong Second People’s Hospital Surface Project (MSZ2024054).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The results presented in the study are included in the article; further inquiries can be directed to the corresponding authors.
Supplemental Material
Supplemental material is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
