Abstract
Objective
Method
Pharmacologically active components of aqueous ethanolic extract of
Result
Phytochemical analysis indicated the presence of chlorogenic acid, gallic acid, P-coumaric acid, kaempferol, salicylic acid, sinapic acid and benzoic acid. Aqueous ethanolic extract of plant relaxed the contractions induced by high K+ (80 mm) and carbachol (1 µM) in isolated urinary bladder strips of rat. In-vivo experiments revealed the nephroprotective and diuretic potential.
Conclusion
So it has been proved via scientific evidence that the concerned plant possesses spasmolytic, nephroprotective and diuretic potential and may be an important therapeutic agent against urinary problems in the near future.

Introduction
Materials and Methods
Plant Extract Preparation
Needles and stem of the plant were obtained from Bahauddin-Zakariya University, Multan Pakistan. The plant (card number https://www.theplantlist.org/tpl1.1/record/kew2562199) was identified by Professor Zafar Ullah Zafar, Department of Botany, Bahauddin Zakariya University, Multan. Needles and stem were dried in shade for 2 months. Dried needles and stem were ground into powders by using herbal grinder. One kilogram of this powder was soaked in a solution containing 80% ethanol and 20% distilled water. The mixture underwent triple maceration with intermittent shaking. Wet part of the plant was separated by using muslin cloth for filtration. After this, it was filtered byWhatman filterpaper. The filtrate was further treated at a temperature of 30 to 40°C using a rotary evaporator (BUCHI) with reduced pressure. Obtained extract was stored at 20°C for future use. 11 It took about 1 year and 3 months to complete the whole research.
Chemicals
Chemicals used in the study were of a purified level including solutions such as 0.9% Sodium chloride, Sodium bicarbonate,formalin,KCl,CaCl2, anhydrous Magnesium sulphate,C2H5OH,ethyl acetate and CHCl3.The above mentioned chemicals were purchased from Jiangyin Lanyu chemical Co, China. Carbachol and verapamil HCl were purchased from Sigma Chemicals Co St. Louis, MO.
Screening of Phytoconstituents
The ethanolic plant extract was analyzed in the laboratory to determine the existence and absence of various secondary metabolites of plants. The extract was analyzed with Mayer’s reagent, Wagner reagent and Dragendroff reagent to determine if alkaloid were present. 12 An alkaline reagent test was conducted to confirm the existence of flavonoids. The ferric chloride test was conducted to find out the presence of tannins and phenolic compounds. 13 To identify the presence of glycosides, Keller Killiani test was performed. 14 The froth test was conducted to detect the presence of saponins. 15
Screening for Bioactive Compounds Using HPLC
Different standards were utilized for HPLC analysis. Chlorogenic acid, kaempferol, para coumaric acid, salicylic acid, benzoic acid, sinapic acid and gallic acid were utilized as reference. A mobile phase was prepared using 0.1% acidified water and acetonitrile. A 5 µm Eclipse C18-Zorbax plus C18 column, measuring 4.6 × 150 mm was used for the process of separation. The rate of flow was 1 ml per min.
Ethical Statement and Experimental Animals
All animal experiments complied with the ARRIVE guidelines and were carried out according to the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments, or the National Institutes of Health guide for the care and use of Laboratory Animals (NIH Publications No. 8023, revised 1978). Animals were handled with great care while experimentations were executed according to drafted rules of life sciences of animal resources commissioned by the Laboratory of Bahauddin Zakariya University as authorization committee to the ethical institution of Multan (EC/06 PhL-2019-2021). BALB/c mice weighing between 15-30 g and Sprague-Dawley rats of either sex weighing between 155-200 g were utilized for both in vitro and in vivo studies. At the end of in vivo experiments, both mice and rats were euthanized using cervical dislocation. The animals were housed in a standard environment and given the controlled diet.
Inclusion/Exclusion Criteria
Inclusion criteria of the animals include: (1) weight of animals (mice) was in the range of 15-30 g and strain was BALB/c. (2) Weight of animals (rats) was in the range of 155-200 g and the strain was Sprague-Dawley. (3) Animals were healthy and free of any underlying diseases.
Exlusion criteria of the animals include: (4) Animals with pre-existing kidney disease or renal impairment. (5) Animals with signs of inflammation or infection. (6) Animals with tumors or cancer.
Animal and Sample Size Calculation
Number of animals were calculated on the basis of reported literature and already reported activities(16-19).Total 70 animals were used in 14 groups. .For Toxicological studies(acute + subacute), 6 groups were made wheras 4 groups were made for Nephroprotective as well as Diuretic activity(each group = 5 animals)
In-Vitro Experiment on Isolated Tissue
In the experiments of isolated tissues, we followed our initially developed protocols. 16
Preparation of urinary bladder strips and experimental set-up
Krebs solution was used to fill aerated organ baths. The water pump was turned on to circulate and heat the chambers to 37 degrees Celsius. 1 g weight was used to calibrate the transducers. The dissection area was set up, and the required tools were employed, such as 2 dissecting forceps, a scalpel blade, micro scissors, electric razors, and forceps with teeth. In the induction chamber, chloroform was used to anesthetize the animal. The lower abdomen of the animal was shaved after it was put to sleep. An abdominal incision made in the midline revealed the pelvic organs. By making a cut at the neck of the bladder near the proximal urethra, the bladder was located and removed. The tissue was submerged right away in an aerated Krebs solution, and the connective tissues and lipids were extracted. To make a flat sheet, the urinary bladder was opened from the base to the dome. The tissue was divided longitudinally into 2-3 mm strips. The strips were placed in the aerated Krebs solution-filled experimental organ baths. A force transducer was attached to 1 end of the strip. A fixed metal rod was attached to the opposite end. Each strip received a certain amount of force by gradually stretching the tissue until the baseline tension reached 1 g.
The aerated Krebs solution was used to wash the tissue strips every 30 min. The tissue was given 2 h to acclimate until no more tissue relaxation was noticed. The signals were intensified by Power Lab data acquisition system (AD Instrument, Bella Vista, NSW, Australia) and shown using lab chart pro (Version 7). In order to cause contractions in bladder strips, both K+ (80mm) and carbachol (1 µm) have been utilized. It was found that the
In Vivo Experiments
Sub-acute Toxicity Studies
Three groups of Swiss albino mice had been formed. There were 5 mice in the first group. The negative control group was the first group. It was given a standard saline treatment. Doses of 250 mg/kg were given to the second group, and 500 mg/kg were given to the third 1. Prior to the trial commencing, these animals were fasted for a whole night. Over the course of 28 days, doses were given every day. Every alteration in the experimental animals’ skin tone, gait, weight, food consumption, behavior, and mortality was noted. Hematological examination was also performed. Organs were taken out at the end of experiment so they could be examined histologically. 17
Acute Toxicity Studies
Acute toxicity analysis: Swiss Albino mice (25-30 g) were used to test the acute toxicity profile of the plant extract under the necessary and standard conditions. Prior to the trial, they were fasted for the entire night. The animals were divided into 3 groups. There were 5 creatures in 1 group. Normal saline was administered to the first group. A single intraperitoneal dosage of 750 mg/kg for Group 2 and 1000 mg/kg for Group 3 had been administered, and the mortality and behavioral changes were tracked for 4 h. The animals were weighed before receiving normal saline and plant extract for the course of the 14 day study period. By the time the trial was over, the animals’ weight was also noted. Every day, the animals were observed to look for any changes in behavior. On the final day of the experiment, organs were removed for histological examination and dissection was completed. 17
Protocol of Nephroprotective Effects
Both male and female rats weighing between 150 and 200 g were utilized to assess the renal protective effect of
Protocol for the Estimation of Diuretic Activity
Twenty albino rats weighing 140-200 g were used in this investigation. The animals underwent a 15 day acclimation period in regular housing prior to the trial. Four groupings were used to classify the animals. As the negative control group, Group 1 was given regular saline. Furosemide injections (10 mg/kg) were given intraperitoneal to Group 2, which served as the positive control group. Group 3 received a low dose of extract orally, while Group 4 received a large dose of ethanolic extract orally. Following treatment, each animal received a regular saline hydration and was housed in a single metabolic cage that was specifically constructed to keep excrement and urine apart. The collection of urine lasted for 5 h. During the diuretic procedure, food or water were not supplied to the animals. Upon completion of the study, a number of parameters were assessed, including the amount of Na+, K+ and Cl- excreted in urine of animals, as well as the total amount of urine produced. Diuretic activity and impact were also ascertained. 19
Histological Examination
After the in-vivo trials concluded, the target animals were sedated, and the right and left kidneys and liver for the toxicological and nephroprotective studies were removed. Blood had been cleaned off with regular saline. Organs were kept for histological analysis in formalin. Utilizing an Olympus digital camera and Olympus light microscope ( × 400), the histopathology examination was completed.
Network Pharmacology
Phytoactive components of
Statistical Studies
The complete set of data (mean ± S.E.M., n = 5) was subjected to one-way analysis of variance, or ANOVA. In order to ascertain the methodological significance between various experimental groups, Dunnett test was conducted in conjunction with it. Data was deemed statistically significant when
Results
Pilot Phytochemical Analysis
The results of various phytoanalysis tests showed that the plant’s ethanoic extract included carbohydrates, alkaloids, flavonoids, saponins, and tannins.
Analysis of Ethanoic Extract of Pinus roxburghii by HPLC
Through HPLC examination, many chemicals were identified. The chromatogram showed the substances that were detected, including benzoic acid, salicylic acid, kaempferol, sinapic acid, P-coumaric acid, and chlorogenic acid. Figure 1A and B (A) Graphical Results of HPLC Analysis. (B) HPLC Chromatogram of Ethanolic Extract of P.Roxburgh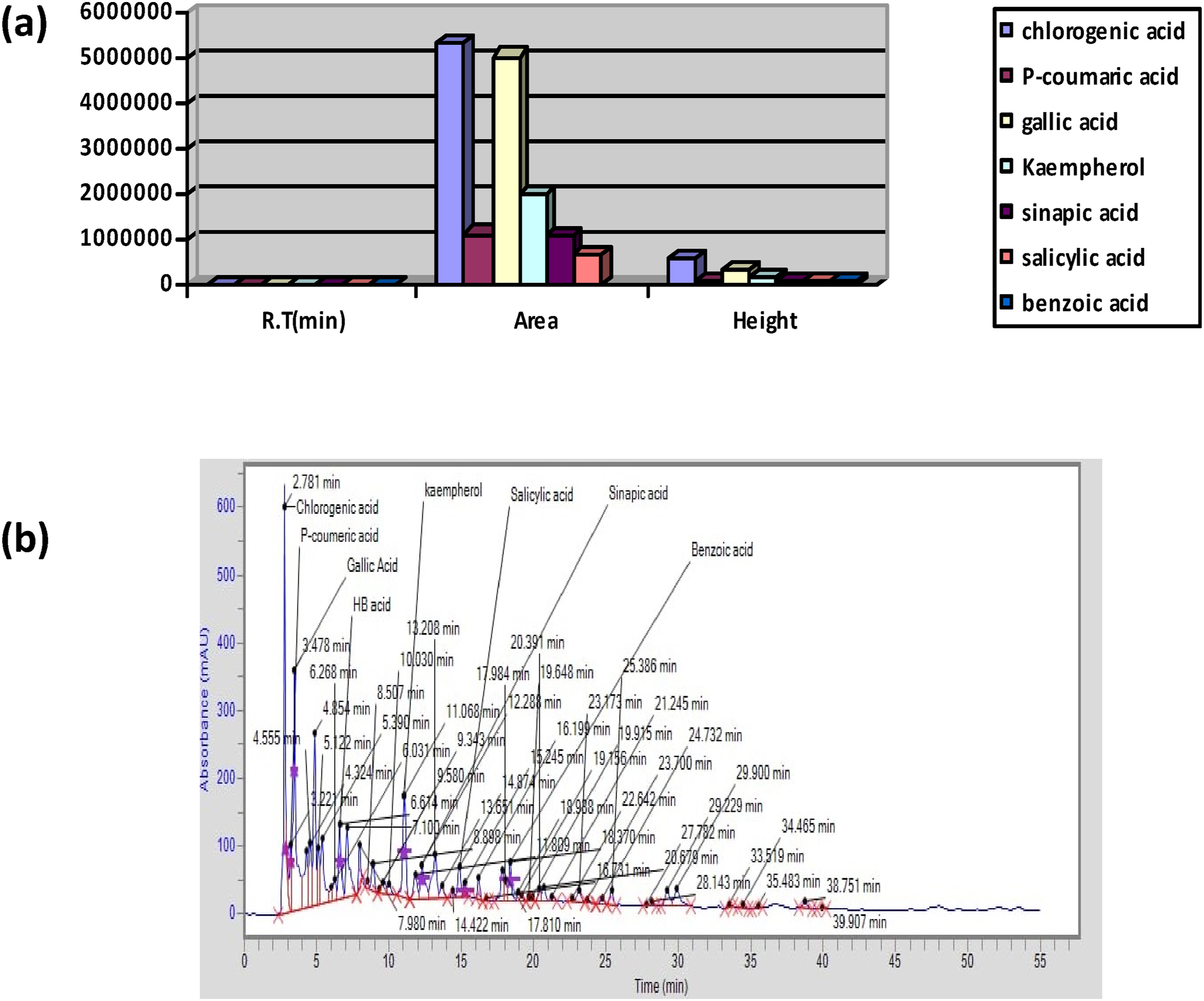
Findings of Toxicological Research
Acute Toxicity Studies
Results of Acute Toxicity Studies

The values are represented as Mean ± S.E.M. To interpret data by comparing with the control group,
#: data is not significant as compare to control group.
Influence of Plant Extract on Behavior of Animals
There were no discernible behavioral changes at the end of the studies. No mortality was observed. Although the test animals were hyperactive during the trial but this behavior was not observed in the control group.
Findings of Histopathology
The experimental animals’ slides resembled almost exactly with those of the control group. There was no discernible change in color, morphology or shape. Outcomes were very similar to those of control group. There was no sign of necrosis or apoptosis as represented in Figure 2. It is possible to argue that the plant extract does not harm the liver or kidney tissues. The glomerulus, interstitial tissues, and renal tubules were all within normal limits. There was no evidence of hepatic or renal tissue inflammation. Histopathological Analysis of Acute Toxicity Studies in Tissues of Mice *CV = Central Vein, DC = Distal Convoluted Tubule, PC = Proximal Convoluted Tubule
Findings of Sub-acute Toxicological Research
Impact of Plant Extract on Histopathological Findings and Hematological Parameters
Impact of Plant Extract on Hematological Findings

The values are represented as mean ± S.E.M. To interpret data by comparing with the Control group, Dunnett’s test had been applied.
#:data is not significant as compare to control group.
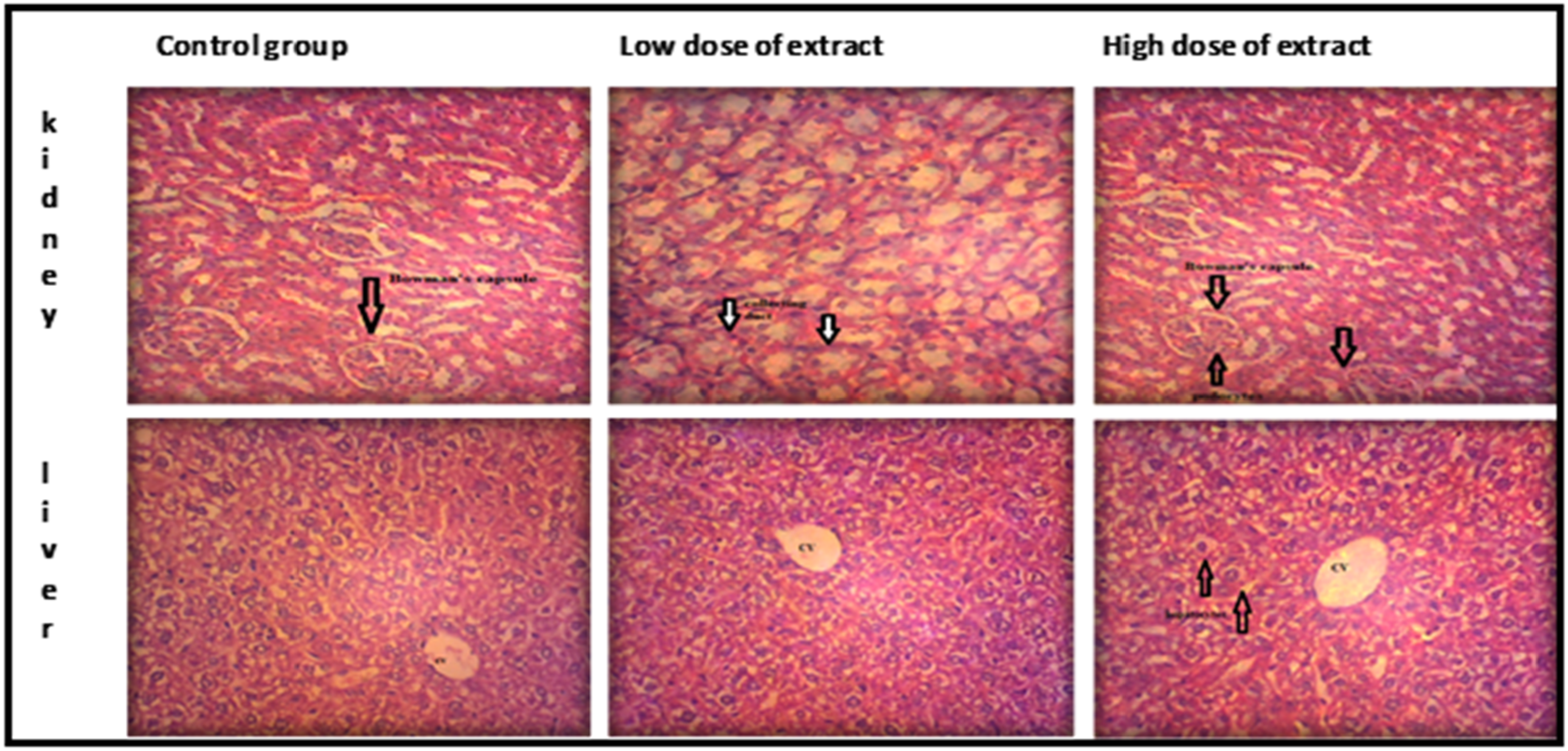
Histopathological Findings of Tissues of Mice
Findings of Nephroprotective Activity
Impact of Plant Extract on Weight of the Body
Effects of Plant Extract on Weight of the Body
Mean ± SEM (n = 4), where Cis = cisplatin (5 mg/kg), Cis + Pr1 = cisplatin + plant (400 mg/kg/14 days), Cis + Pr2 = cisplatin + plant extract (600 mg/kg/14 days
Impact of P. roxburghii Extract on Renal Weight of Rats
Impact of Plant Extract on Kidney Weight, Plasma Creatinine, Plasma Na+ and K+ of Animals

Mean ± SEM, (n = 4), and Cis: cisplatin (5 mg/kg), Cis + Pr1: cis + plant extract (400 mg/kg/14 days).
Cis + Pr2: cis + plant extract (600 mg/kg/14 days. Results are considered significant (*) if
*
#
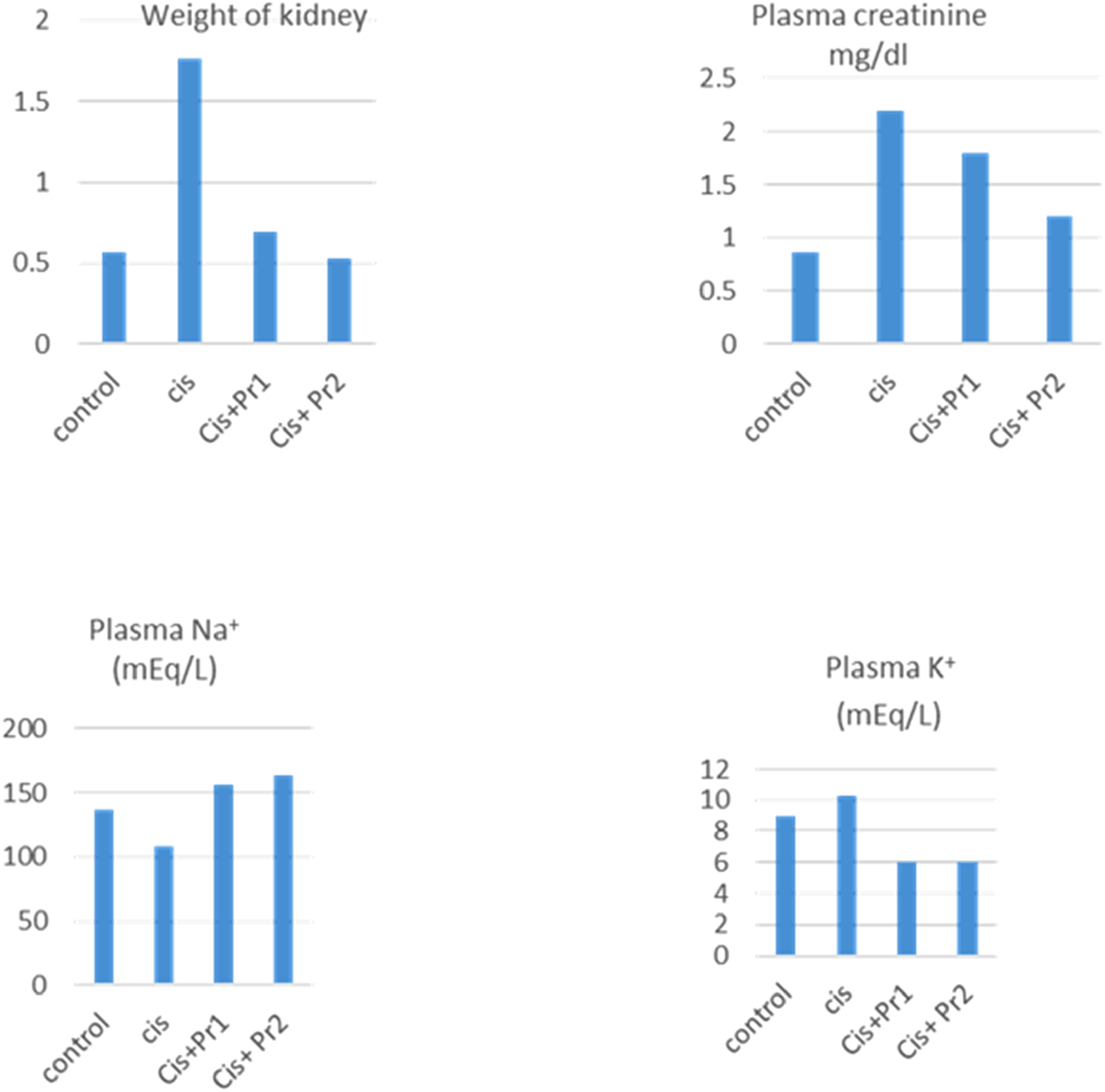
Graphical Representation of Impact of Herbal Extract on Kidney Weight, Plasma Creatinine, Plasma Na+ and K+ of Animals
Impact of Plant Extract on Plasma Creatinine
The effect of plant extract on the plasma creatinine of animals given cis is shown in the table. After the plant extract was administered for 14 days, results were obtained. In conclusion, cisplatin treatment raised the plasma creatinine level. However, at doses of 400 and 600 mg/kg, co-administration of Cis + Pr1 and Cis + Pr2 significantly decreased the level of creatinine in plasma. (Table 4, Figure 4B).
Impact of Plant Extract on Plasma Na+ of Cis-Treated Animals
The effect of plant extract on sodium levels in the plasma of the experimental groups is shown in the table no 4. Overall, cisplatin treatment decreased the amount of sodium in plasma, but when Cis + Pr1 and Cis + Pr2 were given together at rates of 400 mg/kg and 600 mg/kg, the amount of Na+ in plasma increased dramatically. (Table 4, Figure 4C).
Influence of Plant Extract on Plasma K+ of Cis-Treated Rats
The effect of extract on potassium in the plasma of experimental animals receiving Cis is shown in the table. Overall, cisplatin therapy disrupted the potassium content in plasma; however, co- administration of Cis + Pr1 and Cis + Pr2 at 400 mg/kg and 600 mg/kg,markedly improved the concentration of K+ in plasma of treated animals.(Table 4, Figure 4D)
Histopathological Analysis of Kidneys
The normal group did not exhibit any abnormalities, according to a histological investigation of kidney tissues from slides belonging to several experimental animal groups. The glomeruli and Bowman capsule were both intact and in a normal state. Both the proximal and distal tubules’ textures were typical. There was no evidence of bleeding, interfacial damage, or obstruction in the control group. While cisplatin-treated group displayed many degenerative alterations. The collecting tubules were necrotic and the glomerulus was atrophied. Additionally, glomerular hypertrophy was noted. However, tubules with intact glomerulus revealed modest histological damage after 400 mg/kg of Pr and Cis.Conversely, the treatment of Cis + Pr at a dose of 600 mg/kg reduced the harm caused by cisplatin. There was little damage to the distal and proximal tubules. These facts are shown in Figure 5. (A) Photomicrography of Renal Parts of Rats Control. (B) Cis. (C) Cis + Pr1(400 mg/Kg). (D) Cis + Pr 2 (800 mg/Kg) BC: Bowman’s Capsule, RT: Renal Tubules, AG: Atrophied Glomerulus
Findings of Diuretic Effect
Impact of Plant Extract on Urine Output and Diuretic Action in Animals
Impact of Plant Extract

The values are expressed as mean ± SEM.To interpret data by comparing with Control group, Dunnett’s test had been applied.
#: data is not significant as compare to control group.
adiuretic action is urine volume of Test group divided by urine volume of control group.
bdiuretic activity is urine volume of Test group divided by urine volume of Standard group.
Impact on Electrolyte Excretion
Effect on Excretion of Electrolyte
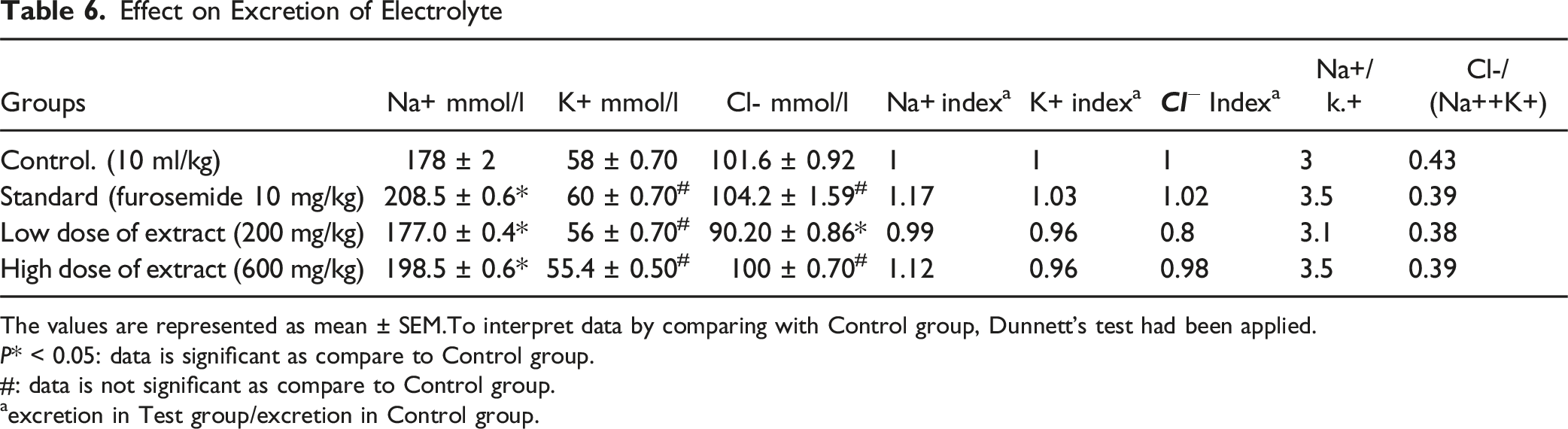
The values are represented as mean ± SEM.To interpret data by comparing with Control group, Dunnett’s test had been applied.
#: data is not significant as compare to Control group.
aexcretion in Test group/excretion in Control group.
Results of in Vitro Studies
Impact on Isolated Urinary Bladder of Rat
Rat bladder strips were subjected to an ethanolic extract of Tracings Representing. (A) Response of Plant Extract Regarding CCh Induced Spasm. (B) Response of Oxybutynin Regarding CCh Induced Spas. (C) Response of Plant Extract Regarding High K+ Induced Spasm. (D) Response of Verapamil Regarding High K+ Induced Spasm (A) Dose Response Curve Plotted for Spasms Caused by CCh and Relaxation Induced by Plant Extract. (B) Dose Response Curve Plotted for Spasms Produced by CCh and Rselaxation Caused by Oxybutynin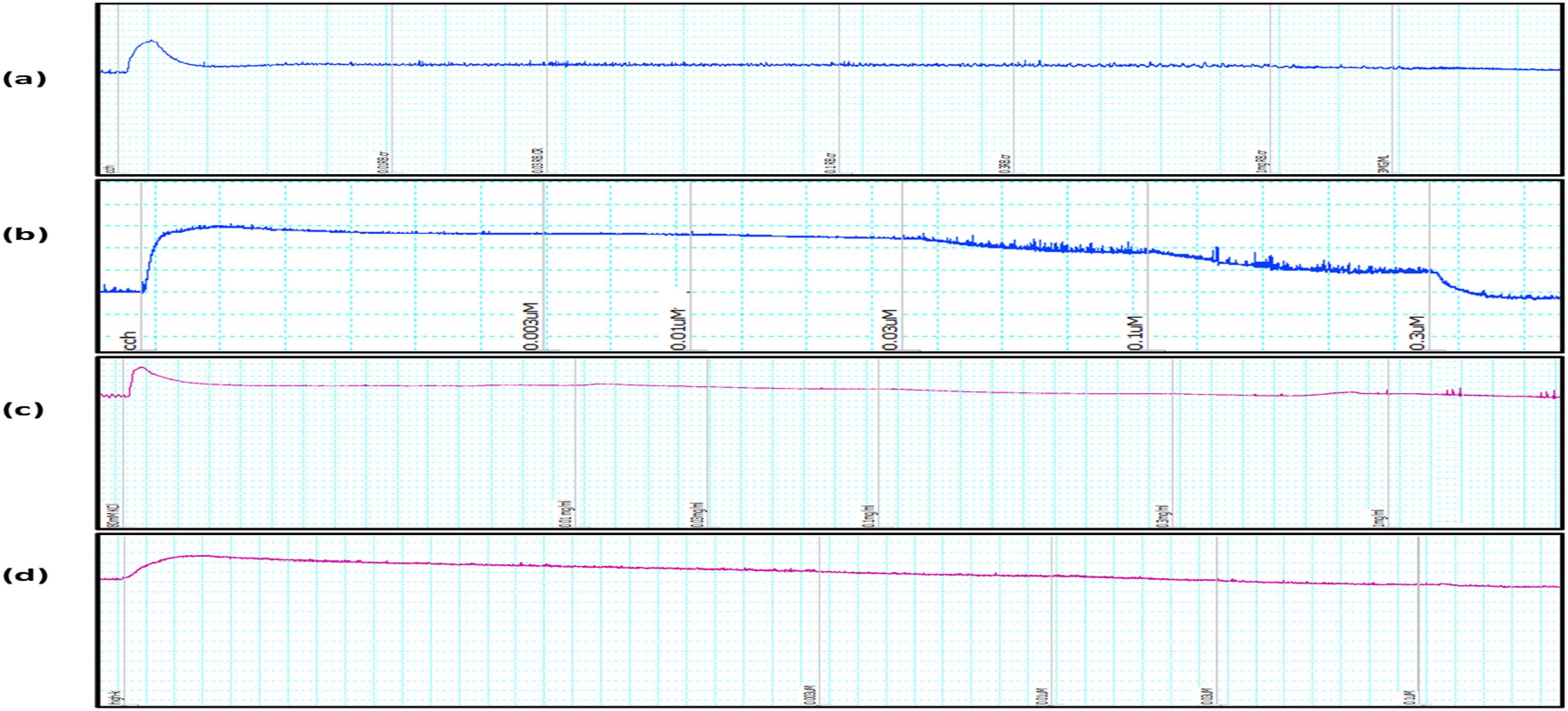

Network Pharmacology
Potential Bio-Active Compounds


Venn Diagram and Protein-Protein Interaction Network. Key: number of edges: 95; average node degree: 6.33; avg. Local clustering coefficient: 0.569; expected number of edges: 25 PPI enrichment
Top 10 Nephrotic Related Target Genes in Protein-Protein Interaction Network Ranked by Degree Method


Network Pharmacology of. (A) Potential Phytochemicals Network. (B) Association of Pythochemicals With Top 10 Hub Genes. (C) Top 10 Hub Genes and Kegg Pathway. (D) Top 10 Hub Genes, Phytochemicals and Kegg Pathway Association
GO Enrichment Analysis
Shiny GO 0/77: an online tool was used to perform GO enrichment analysis of 30 common genes. Figure 10 illustrates the top 10 Biological processes, Cellular processes and molecular pathways. For the identification of signal pathways associated with common genes Kegg pathway enrichment analysis of target genes was performed and sorted by Folding enrichment (FDR ≤ 0.05). Figure 11 represents the top 20 signal pathways. GO Enrichment Analysis of Target Genes Against Nephrotic Pathways Kegg Pathway Analysis of 30 Common Genes Depicts that These Genes are Associated With Various Metabolic and Signal Pathways

Discussion
The current study has been conducted to explore the potential of ethanolic extract of
Plant extract’s ability to inhibit carbonic anhydrase was measured using the Cl-/[Na+ + K+] ratio. The inhibitory activity of carbonic anhydrase is excluded by values between 0.8 and 1. But carbonic anhydrase activity can be considered with decreasing ratios.
19
At low and high doses, the plant under study displayed a ratio of 0.38 and 0.39, respectively. It suggests that the plant may have the ability to inhibit carbonic anhydrase activity of renal tubules. The antispasmodic properties of a plant extract were studied using urine bladder strips that were separated. Both high K+ (80 mM) and carbachol (1 µM) caused contractions. At a dosage of 3 mg/mL, plant extract almost totally relaxed the contractions caused by carbachol. Plant extract reduced K+-induced contractions at a 1 mg/mL dosage. The plant extract’s EC50 value of 6.937 mg/mL for carbachol and 1.456 mg/mL for K+ indicated that it had higher calcium channel blocker activity. K+ causes smooth muscle contractions by depolarizing the membrane, which opens voltage-gated calcium channels. This causes the intracellular calcium level to rise and the contraction process to start.
33
Secondary metabolites found in plants are useful in the treatment of a number of illnesses. For instance, flavonoids have cytotoxic, antimicrobial, antiulcer, and spasmolytic effects.
34
It is considered that spasmolytic response of plant extract is because of the existence of flavonoids that has been verified by HPLC analysis. Network pharmacological analysis screened 5 potential phytoactive compounds. This analysis showed that 30 common genes were observed to have been associated with nephrotic syndrome after the Venn diagram. Protein-protein network showed the interactive effect of 30 common genes. Cytohubba plugin network analyzer, through the degree method, identified the top proteins (hub genes) interacting closely in the network. The current study endorsed and documented that degree metrics are an indicator of essentiality within the interaction network.
22
In the present study, ACE (angiotensin-converting enzyme) protein was observed to have the highest degree score. ACE plays a vital role in the renin-angiotensin-aldosterone system (RAAS).
35
RAAS plays an important role in sodium ion homeostasis and regulation of blood pressure. The kidney along with the cardiovascular system works together for the maintenance of these activities.
36
Kaempferol reduces the diabetic-induced nephrotoxicity.
37
In a recent study, HPLC analysis revealed the presence of Chlorogenic acid, kaempferol, para coumaric acid, salicylic acid, sinapic acid, and gallic acid in the extracts of
Limitation of Study
Further studies are needed to determine that exactly which component of the plant is responsible for nephroprotective and spasmolytic activity. Furthermore, additional analysis and research is needed to determine the precise mechanism of action of the plant regarding its diuretic activity, particularly its potential for natriuresis. It is vital to ascertain the precise component responsible for the natriuretic potential. Moreover, sample size calculation was done on the basis of reported literature and justification was not done because we don’t have the resources and necessary expertise to conduct a thorough sample size calculation. We don’t have enough information to estimate the variability of the outcome measure.
Conclusion
In conclusion, this study shows that the
Footnotes
Acknowledgements
We would like to express our sincere thanks and appreciation to Dr Imran, Chairman of the Department of Pharmacology, Bahauddin Zakariya University, Multan for the provision of animals and laboratory equipment required for the conduction of this research.
Ethical Approval
Ethical approval was attained from the Ethical Committee of Bahauddin Zakariya University, Multan (EC /07-PHL-2022-2024) dated 7th February 2023. Researchers agreed using the approved informed consent documented before their enrolment into the study.
Author Contributions
M.N., F.S., H.S., C.L.C., C.M and M.F.L designed the study and performed experiments.M.N., M.A.M and O.A. collected data and wrote the manuscript. All authors carefully read and approved the final manuscript.
Funding
The authors declared no funding to the research, authorship, and/or publication of this article.
Declaration of Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
