Abstract
Chronic kidney disease (CKD) is an important factor that contributes to the increase of all-cause morbidity and mortality in the group of non-communicable diseases, and it is also recognized as a strong and independent risk factor that contributes to cardiovascular disease (CVD). CVDs are a consequence of the action of a large number of risk factors among which are traditional and non-traditional. These risk factors have been the subject of a large number of studies which partially explained the unfavorable cardiovascular (CV) outcome of CKD patients. Therefore, valid studies about clinical and biohumoral predictors are of particular importance, especially in the early stages of renal disease, that is, in patients with creatinine clearance below 60 ml/min/1.73 m2 when preventive measures are most effective. Among potential predictors of adverse CV outcome are biomarkers of inflammation (Interleukin-18—IL-18), oxidative stress (ischemia-modified albumin—IMA; superoxide dismutase—SOD), acute kidney injury (kidney injury molecule-1—KIM-1; neutrophil gelatinase–associated lipocalin—NGAL), and microribonucleic acids (specific microRNA-133a). In this review, we tried to confirm the relationship between risk factors of CKD and CVD and newer, less frequently examined biomarkers with the occurrence of incidental CV events in renal patients.
Keywords
Epidemiology of Cardiovascular Diseases in Chronic Kidney Disease
Chronic kidney disease (CKD) is an increasingly important factor contributing to the all-cause morbidity and mortality in the group of non-communicable diseases and is recognized as a strong and independent risk factor contributing to cardiovascular diseases (CVD).1,2 Heart failure (HF), coronary artery disease (CAD), cardiac arrhythmias, and sudden cardiac death are among the most common causes of increased morbidity and mortality in CKD, and severe cardiovascular (CV) events account for almost 50% of all deaths in the renal disease population.3,4 Patients on a chronic dialysis treatment have a mortality rate that is ten to twenty times higher than the population of patients without a diagnosis of CKD.5-7
Timely diagnosis and therapy would not only improve outcomes of CKD and the quality of life of patients but also give rise to a significant reduction in the number of hospitalizations and consumption of medical resources and thus the overall reduction of direct and indirect costs of treatment.
Risk Factors for Cardiovascular Disease
The reason for the marked increase in the number of CV events with CKD progression has not yet been sufficiently investigated, but it is considered to be largely due to the complex mechanism of action of traditional and non-traditional CV risk factors.
Risk factors for cardiovascular diseases.
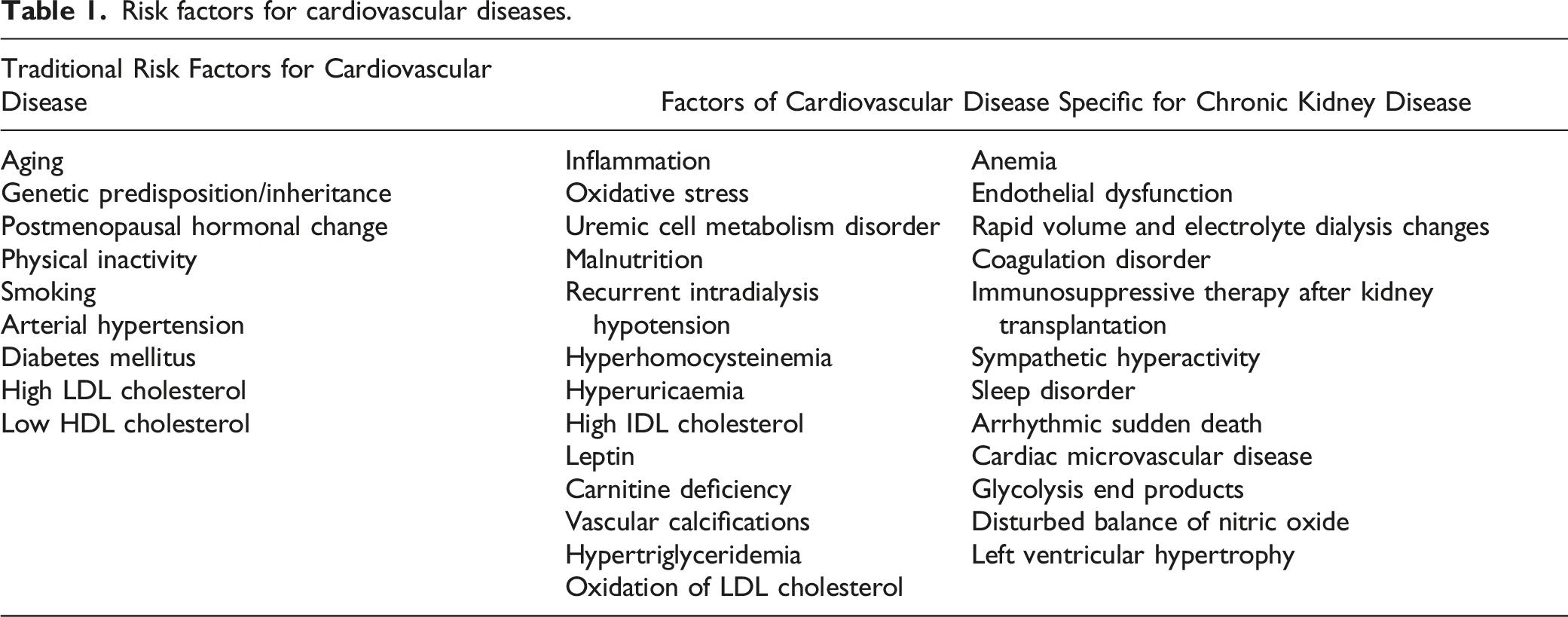
Non-traditional risk factors are related to CKD progression and represent another, broad group of CV risk factors that are quite characteristic of CKD and their prevalence increases with declining glomerular filtration rate (GFR). They are often referred to as uremia-related CV risk factors, the most important of which are anemia, microinflammation, oxidative stress, malnutrition, and secondary hyperparathyroidism (Table 1). 10
Biomarkers of Cardiovascular Disease in Chronic Kidney Disease
Biomarkers of cardiovascular disease in chronic kidney disease.

Biomarkers of Mineral Metabolism Disorders
Among the disorders of mineral metabolism, there are disorders of serum phosphorus, calcium, and magnesium, increased values of vascular calcification promoters (alkaline phosphatase, osteocalcin, osteonectin, bone morphogenic protein, and fibroblast growth factor-23), and decreased vascular calcification inhibitors (fetuin-A, osteopontin, GLA-protein matrix, and Klotho coreceptor). Therefore, the KDIGO Guideline (Kidney Disease Improving Global Outcome) recommends these values specifically for the CKD patient population. 14 Thus, phosphorus values in the fifth stage of CKD are allowed up to 1.7 mmol/L with a tendency to normal values, if possible, and the target values of parathyroid hormone (PTH) change with the progression of CKD to reach the recommended values up to nine times higher compared to the referent values. 14
The three factors hyperphosphatemia, hypocalcemia, and deficiency of the active form of vitamin D lead to secondary hyperparathyroidism, non-traditional risk factor that contributes to the occurrence of vascular and valvular calcifications in the presence of the vascular calcification promoters and lack of vascular calcification inhibitors.
Low levels of magnesium, as well as high levels of calcium and phosphorus, are associated with a higher risk of HF, the development of vascular calcifications, and consequent numerous complications.12,15 With the progression of CKD, there is an increase in serum phosphorus, and hyperphosphatemia is highly prevalent in the final stage of CKD. Elevated phosphorus values represent a strong, independent risk factor for all-cause and CV morbidity, while studies show that phosphorus values higher than 2.1 mmol/L are associated with far higher mortality compared to patients with lower values in chronic hemodialysis. 16 Increased values of serum phosphorus contribute to the occurrence of increased CV morbidity and mortality based on the occurrence of vascular calcifications (calcification of the medial blood vessel wall—Mönckeberg’s arteriosclerosis) and even the occurrence of endothelial dysfunction. 17 Certain studies support the fact that the daily intake of large amounts of phosphorus through food leads to the occurrence of reduced production of nitric oxide. 18 Hyperphosphatemia even directly leads to the occurrence of myocardial fibrosis. 19 Significance of serum phosphorus as cardiovascular biomarker has also been described by CARDIA study; 3015 young men without kidney disease were followed and baseline serum phosphorus even in the range of the normal values predicted coronary atherosclerosis content after 15 years. 20
Promoters and inhibitors of vascular calcifications in chronic kidney disease.

The group of endogenous inhibitors of vascular calcifications includes individual molecules that were also tested as markers of CV resistance in CKD. 25 Fetuin-A, Klotho coreceptor, osteopontin, osteoprotegerin, and matrix Gla-protein are just some of the tested inhibitors. Fetuin-A is considered an inhibitor of ectopic calcifications, including arterial calcifications, and its lower values have been noted in the hemodialysis population, and some studies have linked this marker to increased mortality in dialysis patients.26,27 Lower Klotho coreceptor values are associated with more severe forms of CAD. 28 There is also the association of serum osteoprotegerin with CV risk factors in CKD. 29
Cardiospecific Biomarkers
Certain diagnostic markers, such as troponin (a marker for acute myocardial infarction) and natriuretic peptides (markers for heart failure), have also been shown to be useful biomarkers for risk assessment and prediction of future CV events in CKD population. 13
Troponin is the most controversial biomarker of CV disease. In contrast to the general population, troponin turned out to be a better prognostic marker of CV outcome than a marker of acute CV events in the CKD group of patients. 30 A meta-analysis involving a number of studies and a large number of asymptomatic patients concluded that elevated troponin levels were associated with severe, diffuse CAD in dialysis and nondialysis patients and with unfavorable echocardiographic findings and poorer survival in transplant patients. 31 Other differences in relation to the general population concern cutoff values of troponin that would be of diagnostic importance. A large number of institutions use ten times higher values compared to values in the general population, but also the dynamics of increase in a function of time.
BNP (B-type natriuretic peptide) and NT-proBNP (amino-terminal fragment of BNP) as indicators of decompensated HF can also be used to diagnose and predict CV disease in CKD population. However, their usual, standard reference values used in the general population should be higher in CKD, as is the case with troponin. This is due to the reduced clearance of these markers and the corrected value should be adjusted to creatinine clearance if it is less than 60 ml/min/1.73 m2. An increase of BNP level reaching about 200 pg/mL has been previously reported in patients with reduced GFR in the absence of cardiac dysfunction, and NT-proBNP reference values of 1200 pg/mL have been recommended in patients with CKD.32,33 Studies involving dialysis patients have confirmed the predictive power of NT-proBNP for poorer survival and increased risk of CV mortality.34-36
Biomarkers of Inflammation
Given the importance of inflammation as a factor influencing the development of atherosclerosis, a large number of studies have examined individual inflammatory biomarkers to predict CV events in the population of patients with CKD. The most commonly tested biomarkers were C-reactive protein, interleukin-1, interleukin-6, and tumor necrosis factor.37,38
C-reactive protein (CRP) is the best known and most commonly used marker of an acute phase of systemic inflammation in the general population and the CKD population. 39 Elevated CRP values are a predictor of coronary events in the general population, and several studies support the association of this marker with all-cause mortality in patients with end-stage kidney disease.40,41 Although the upper normal value of CRP is usually 0.5 mg/dL in the general population, CRP concentration greater than 10 mg/L indicates microinflammation present in a large number of CKD patients and is a significant non-traditional risk factor for CV events. 42
Interleukin-6 (Il-6) is an inflammation protein biomarker produced by the liver, and it is a potential predictor of all-cause and CV mortality in the CKD population at different stages of kidney disease. It is shown that Il-6 is a better biomarker than acute phase proteins (albumin, CRP, and tumor necrosis factor) for mortality prediction. 43 Also, some other study showed that IL-6 (mean of two measurements) is a better inflammatory marker than CRP and TNF-α at predicting cardiovascular events in CKD nondialysis patients. 44
Recently, the first clinical studies were conducted in which the inhibition of interleukin was examined. Ridker et al. tested canakinumab, a human monoclonal antibody targeting IL-1β in reduction of cardiovascular event rates and improvement of renal function among post-myocardial infarction patients with CKD. The conclusion of this study was that IL-1β inhibition with canakinumab reduces major adverse cardiovascular event rates among high-risk atherosclerosis patients with CKD, particularly among those with a robust anti-inflammatory response to initial treatment. 45
Some experimental evidence suggests a role of IL-1 in kidney disease and hypertension and targeting IL-1 showed promising results in high cardiovascular risk patients and hemodialysis and renal transplantation patients. Recently published research by Schunk et al. demonstrated that IL-1α represents a central regulator of leukocyte-endothelial adhesion in myocardial infarction and in chronic kidney disease, so inhibition of IL-1α may serve as a novel anti-inflammatory treatment strategy for this population. 46
Interleukin-18 (IL-18) is an increasingly mentioned and tested biomarker of inflammation for the prediction of CV events in CKD population. IL-18 initiates a cascade of other proinflammatory cytokines which trigger a Th1 or Th2 lymphocyte response. This activation of lymphocytes triggers an immune response and accelerates the process of atherosclerosis. IL-18 has been shown to be present in high concentrations in atherosclerotic plaques and to contribute to their destabilization, which may result in acute ischemic events. 47 Based on previous studies, increased serum IL-18 levels are an important indicator of CV mortality in patients with CKD. 48 Also, IL-18 can be considered as a possible biomarker of future cerebrovascular, CV, and peripheral vascular diseases (PAD) in the final stage of CKD. 49 Today, we still do not know what are the limit values of this cytokine that would be important in the population of CKD patients.
Biomarkers of Oxidative Stress
Since oxidative stress has one of the dominant roles in the pathogenesis of atherosclerosis, it is important to investigate biomarkers of oxidative stress that would be predictors of CV events in CKD patients. 50 This association was observed in predialysis patients, patients on hemodialysis (HD), and peritoneal dialysis (PD). Among these markers are oxidized LDL cholesterol, myeloperoxidase, lipid, and protein peroxidation molecules. 51
The defense against increased oxidative stress goes through several lines of defense, and it is easiest to examine enzymes as potential biomarkers of oxidative stress. Superoxide dismutase (SOD) is the most effective intracellular enzyme in the first line of defense against oxygen-free radicals. 52 Some studies have linked SOD to certain CV diseases such as CAD.53,54 Ischemia-modified albumin (IMA) is one of the newer biomarkers of CV events related to ischemic conditions and increased oxidative stress such as ischemic heart disease. 55 This biomarker can most likely be considered as an indirect indicator of increased oxidative stress, ischemia, and hypoxemia. At the same time, IMA has been shown to be a predictor of CKD mortality in some studies. 56
In this group of important biomarkers, it remains to determine their dynamics and significant values in the CKD patients.
Biomarkers of Acute Renal Injury
In addition to the role in the early detection of newly developed acute kidney injury, their role during CVD was examined. Neutrophil gelatinase–associated lipocalin (NGAL) and kidney injury molecule-1 (KIM-1) are the most commonly examined from the aforementioned group of markers of acute kidney injury and could potentially play a role in the pathogenesis of CKD and prediction of CV events. Thus, recent studies suggest a role for NGAL in the progression of CKD. 57 It has also been described that elevated plasma NGAL in CKD patients may be an independent predictor of future CV events. 58 It has been observed that increasing NGAL in urine increases the incidence of ischemic atherosclerotic events in CKD patients independent of GFR, albuminuria, and comorbidities. 59 Also, NGAL values are increased in atherosclerosis plaques, and its expression is upregulated in condition of ischemia, hypoxia, and myocardial infarction, and NGAL can be regulated by vascular inflammation response to ischemia. 60
In recent years, studies have examined the association between NGAL and coronary blood vessel diseases. The paper published by Katagiri et al. presented that serum NGAL level was positively and significantly associated with CAD severity, and the evaluation of both serum NGAL and BNP was useful for predicting CAD in patients without renal dysfunction and heart failure. This finding showed that serum NGAL might be a biomarker for CAD severity. 61
Review article published by Alves de Freitas et al. analyzed eight novel biomarkers in the prognosis of patients with atherosclerotic coronary artery disease. The main evidence suggested that elevated NGAL level is associated with better prognosis after cardiac arrest and with comorbid kidney injury. 62 Beside this, some recent data showed that NGAL may be a novel biomarker to aid in risk stratification in coronary heart disease patients. 63 It should be pointed out that monitoring of these markers could have significant importance for early diagnosis and clinical course, not only in patients with various forms of renal diseases but also in patients with cardiorenal syndrome, heart failure, cardiopulmonary bypass, and cardiothoracic surgery. 64
KIM-1 is released during the ischemic and toxic effects of agents on the proximal tubules of the kidney. Although initially tested and used as a urinary biomarker of acute kidney impairment, KIM-1 has been shown to be associated with CKD progression. 65 Recent studies suggest that elevated urinary KIM-1 may be associated with an increased risk of ischemic heart disease, HF, and all-cause mortality in patients with CKD, but further investigations of the said molecule as a potential biomarker of CV events are pending. 66
Wybraniec et al. showed in their study that excessive increase of urinary KIM-1 levels after coronary angiography may help identify CAD patients with poor 12-month prognosis. 67 This paper proved that urinary KIM-1 may be used as a combined biomarker of renal function worsening and long-term cardiovascular morbidity and mortality in patients referred for coronary angiography.
In another study, Paapstel et al. found independent associations between urinary liver–type fatty acid–binding protein and KIM-1 and aortic stiffness for the CAD patients with eGFR above 60 ml/min per 1.73 m2. Therefore, measurements of KIM-1 may help to further explore the interplay between early alterations in renal function and vascular damage. 68
Additionally, proteomic profiling of end-stage renal disease (ESRD) patients could lead to improved risk prediction and novel insights into cardiovascular disease mechanisms. Feldreich et al. revealed that plasma KIM-1 represents a risk marker for cardiovascular mortality and coronary artery calcification in three independent ESRD cohorts. 69
Microribonucleic Acids
Newer biomarkers of CV disease in the population of patients with CKD include micro-ribonucleic acids (microRNAs). MicroRNAs are a class of non-coding ribonucleic acid molecules that play an important role in regulating the expression of a large number of genes, especially those related to the normal functioning of the CV system. These molecules participate in the regulation of cell proliferation, differentiation, apoptosis, and tumorigenesis and are secreted into biological fluids such as blood and urine.70-72
A few number of previous studies concern the examination of microRNAs in the physiology and pathophysiology of the CV system. For example, microRNAs 1, 208a/b, 499, and 133a are most likely specific for the regulation of normal heart function and development.73,74Any pathological condition and disease that leads to dysregulation of these microRNAs can lead to HF progression, cardiac fibrosis, and dilatation complicated by ischemia and cardiac arrhythmias. 73
Atherosclerosis, as the basic pathophysiological process of CV disease, is regulated by a number of microRNA molecules. There are molecules involved in endothelial dysfunction (microRNA-31, 126), atherosclerotic plaque development (21, 155, 221), cholesterol homeostasis (122, 33a/b), neoangiogenesis (155, 210, 221, 222), and even instability and rupture of plaque (100, 127, 145). 73
Due to all the above, a large number of microRNAs have been tested as potential diagnostic and predictive biomarkers for various diseases within the CV system. 75 So far, over 2000 different microRNAs have been detected, including those potentially related to the pathogenesis of certain CV diseases. Altered expression was registered in patients with left ventricle hypertrophy (LVH), CAD, HF, peripheral artery disease, and stroke.76-80
A small number of studies have examined the association of one particular microRNA with the occurrence of CV events in the population of patients diagnosed with CKD, as well as its association with CV risk factors and other complications in CKD. In some studies, altered expression of a specific microRNA-133a molecule was demonstrated, as well as its association with certain CV diseases.75,80,81
Although promising, this group of biomarkers for the prediction of CVD is currently not sufficiently tested to be routinely used in patients with CKD, so their further examination is necessary, especially the definition of cutoff values in particular clinical conditions.
Biomarker Changes in Dialysis Patients
The dialysis procedure can lead to a change in the concentration of certain CV biomarkers, which may result in a decrease in their predictive power and inadequate assessment of CV risk. This is especially true of markers of inflammation and oxidative stress.
Inflammation in dialysis is due to bioincompatibility of dialysis membranes, inflammatory process within the vascular access, and insufficient sterility of dialysis fluid, while oxidative stress is the consequence of uremia, aging, and loss of residual renal function.82,83
Previous studies have confirmed that only one hemodialysis session increases some markers of inflammation such as pentraxin-3 and high-sensitivity CRP, but also increases markers of oxidative stress such as serum malondialdehyde. 84 Some markers can be removed during dialysis and purification from patient blood depending on the size of the molecule. This leads to the conclusion that one of the shortcomings of the examined biomarkers is the influence of dialysis on the concentration of biomarkers, which can lead to problems in the adequate interpretation of the obtained results and correlation with CV events.
Significance of Determining the Value of Individual Biomarkers as a Diagnostic, Prognostic, and Monitoring Parameter of Therapeutic Effects
And finally, a relatively small number of biomarkers tested so far are in official use in everyday clinical practice, given that the exact cutoff values have not been defined for the largest number of them. This applies not only to CKD but also to the general patient population. Each potentially new biomarker can be used to assess the therapeutic effect, to diagnose and predict the further course of the disease.
Of all the biomarkers listed in the text only for troponin and natriuretic peptides, we have clearly defined diagnostic values for the myocardial ischemic event and cardiac decompensation in the general population but debatable and somewhat defined values concerning the population of CKD patients.
For example, for meta-analysis, Kampmann et al. discuss cutoff values of troponin in patients with impaired renal function with acute myocardial infarction, and the following values are suggested: 42 ng/L for troponin I and 48 ng/L for troponin T, and for patients on dialysis, troponin T cutoff is much higher at 239 ng/L, but troponin I value for dialysis patients could not be established due to lack of data. 85 Also, there is a lack of data on troponin values after certain therapeutic procedures that would indicate their success.
For people who don’t have HF, normal BNP levels are less than 100 pg/mL. BNP levels over mentioned value may be a sign of HF. NT-proBNP normal levels are less than 125 pg/mL for people under 75 years old and less than 450 pg/mL for people over age 75. NT-proBNP levels over 900 pg/mL may be a sign of HF. As already mentioned, there are partially defined values in patients with impaired renal function: an increase in BNP level reaching about 200 pg/mL has been previously reported in patients with reduced GFR in the absence of cardiac dysfunction, and NT-proBNP reference values of 1200 pg/mL have been recommended in the same group of patients.32,33 We need more experiences that would compare the therapeutic effects of HF according to values.
Given the previous story, in the time to come, studies and research will determine an increasing number of potential biomarkers of adverse CV events, for the purpose of early detection of vulnerable patients within CKD population and timely application of preventive measures that will reduce CV morbidity and mortality. There is even less data regarding the monitoring of biomarkers as indicators of favorable or unfavorable effects of certain therapeutic measures.
Conclusion
Despite numerous achievements in the field, the mortality of patients with CKD is unsatisfactorily high. Although numerous attempts tried to find newer, more reliable biomarkers and predictors of CV morbidity and mortality in CKD patients and to define its cut-of values, it still remains unacceptably high. At the moment, “classic” biomarkers (anemia, urea, albumin, parameters of left ventricular hypertrophy) are the most reliable predictor of CV outcome in CKD patients. Therefore, there is still room for new research that would allow us to timely and efficiently prevent CV disease in CKD patients thus improving their survival. 86
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
