Abstract
This study aims to elucidate the anti-hypoxia mechanism of sesamoside, an active component of
Introduction
With the advancement of modern transportation and tourism, an increasing number of individuals have the opportunity to visit high-altitude regions and experience their majestic scenery. However, high altitudes’ unique low-pressure and hypoxic environments challenge human health. Among these, high-altitude pulmonary edema (HAPE) stands as a severe altitude-related disease that has garnered significant attention from the medical community. HAPE occurs when individuals rapidly ascend to high altitudes, exposing themselves to low-pressure, hypoxic conditions, which lead to abnormal accumulation of fluid in the lungs, manifesting as symptoms such as dyspnea, cough, and the production of frothy sputum.1,2 In severe cases, it can even be life-threatening, with a mortality rate as high as 50%.3,4 The treatment and prevention strategies for HAPE are continuously being explored and improved. Existing treatment methods primarily include symptomatic treatment, causal treatment, and pharmacological therapy, aiming to alleviate patient symptoms, reduce the extent of pulmonary edema, and improve patients’ quality of life.5-7 However, these treatment methods still have limitations and shortcomings in practical applications, such as inconsistent treatment and significant side effects.
Environmental factors such as high-altitude hypoxia and low pressure lead to pathological and physiological changes such as excessive elevation of pulmonary arterial pressure, increased pulmonary capillary permeability, and dysregulation of fluid transport, which trigger HAPE. Firstly, hypoxia activates transcriptional factors such as Hypoxia-inducible factor (HIF), thereby regulating the transcription and translation of numerous genes. 8 These changes in gene expression can affect energy metabolism, homeostasis, and neuroendocrine systems and disrupt fluid balance and oxidative stress responses.9-11 These molecular alterations collectively contribute to the pathogenesis of HAPE in the lung tissue. Secondly, the increased permeability of pulmonary capillaries plays a pivotal role in the development of HAPE. Bronchoalveolar lavage fluid from HAPE patients shows high protein levels, inflammatory cells, increased red blood cell count, and C-reactive protein, indicating that HAPE is a form of exudative edema. 12 Animal studies suggest that vascular endothelial growth factor levels rise significantly under hypoxic conditions, increasing blood flow and damaging the vascular endothelium. NO is a vasodilator. In hypoxic conditions, individuals susceptible to HAPE experience a significant decrease in NO levels regulated by NO synthase (NOS). 13 Schemer and colleagues discovered that long-term residents of high-altitude areas have significantly higher NO levels in their lung tissues than newcomers. 2 Endothelin-1 (ET-1) is a potent vasoconstrictor synthesized in pulmonary endothelial cells, where it regulates the contraction of vascular smooth muscle cells and maintains vascular tension. Studies found that individuals susceptible to HAPE in high-altitude hypoxic environments have increased levels of ET-1 in their lungs. 14 The hypoxic high-altitude environment induces pulmonary vascular endothelial cells to secrete ET-1 and other vasoactive substances and adhesion molecules. These endothelial cells mediate the adhesion and aggregation of inflammatory cells through ET-1, contributing to the remodeling of pulmonary vessels and the onset of pulmonary hypertension. 15
Also, alveolar fluid clearance is impaired, and systemic inflammatory responses may contribute to the pathogenesis of HAPE. In high-altitude environments, the volume of alveolar fluid depends partly on the overflow of fluid from pulmonary vessels, primarily associated with hypoxic pulmonary arterial hypertension, and partly on the reabsorption rate by alveolar epithelium, determined by sodium transport in these cells. Hypoxia disrupts sodium transport in pulmonary epithelial cells, obstructing alveolar fluid clearance. Aldose reductase (AKR1B1, AR) is a member of the aldo-keto reductase superfamily of proteins in various human tissues and organs. 16
AR converts glucose to sorbitol and plays a role in the polyol metabolic pathway.17,18 Sorbitol’s high polarity prevents it from freely crossing the cell membrane, accumulating inside cells, and creating a hyperosmotic state. Hyperglycemia can change the intracellular ratio of reduced nicotinamide adenine dinucleotide phosphate to nicotinamide adenine dinucleotide via AR, resulting in increased production of reactive oxygen species within cells, intensifying severe oxidative stress and leading to apoptosis. 19 Nuclear factor kappa B (NF-κB) binds to promoters in the nucleus that encode inflammatory factors, initiating transcription and playing a role in the development of inflammation. 20 Inhibiting AR significantly reduced the transcriptional activity of NF-kappa-B and eased inflammation. 21 Inflammatory responses play a role in the development of HAPE by increasing pulmonary vascular permeability and pulmonary arterial pressure. 22 In high-altitude hypoxic conditions, NF-κB is activated, initiating various inflammatory factors, such as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6), initiates systemic inflammatory responses and binds to receptors in lung tissues after a series of inflammatory reactions, causes lysosomal damage and leakage, ultimately leading to lung injury. 23
In terms of treatment, different treatment measures should be taken based on the severity and progression of HAPE. For mild HAPE patients, general treatment measures such as rest and oxygen inhalation can be taken to alleviate symptoms. For severe HAPE patients, more aggressive treatment measures such as mechanical ventilation, diuretics, and pulmonary artery pressure-lowering drugs are needed to reduce the degree of pulmonary edema and improve the patient’s vital signs.
24
In recent years, with the deepening of research on natural drugs, more and more natural compounds have been discovered to have potential anti-hypoxic and anti-altitude disease effects. Among them,
Materials and Methods
Reagents
Sesamoside and dexamethasone were purchased from Shanghai Yuanye Bio-Technology Co., Ltd. Cocl2 was purchased from Tianjin Kemiou Chemical Reagent Co., Ltd. BCA protein assay kit, reverse transcription kit, and DEPC-treated water were purchased from Yisheng Biotech Co., Ltd. Cell lysis buffer, PMSF, and SDS-PAGE protein loading buffer were from Beyotime Biotechnology Co., Ltd. SYBR Green qPCR Master Mix was purchased from Sevier Biotechnology Co., Ltd. TNF-α and IL-6 ELISA kit were purchased from Nanjing Jiancheng Bioengineering Institute.
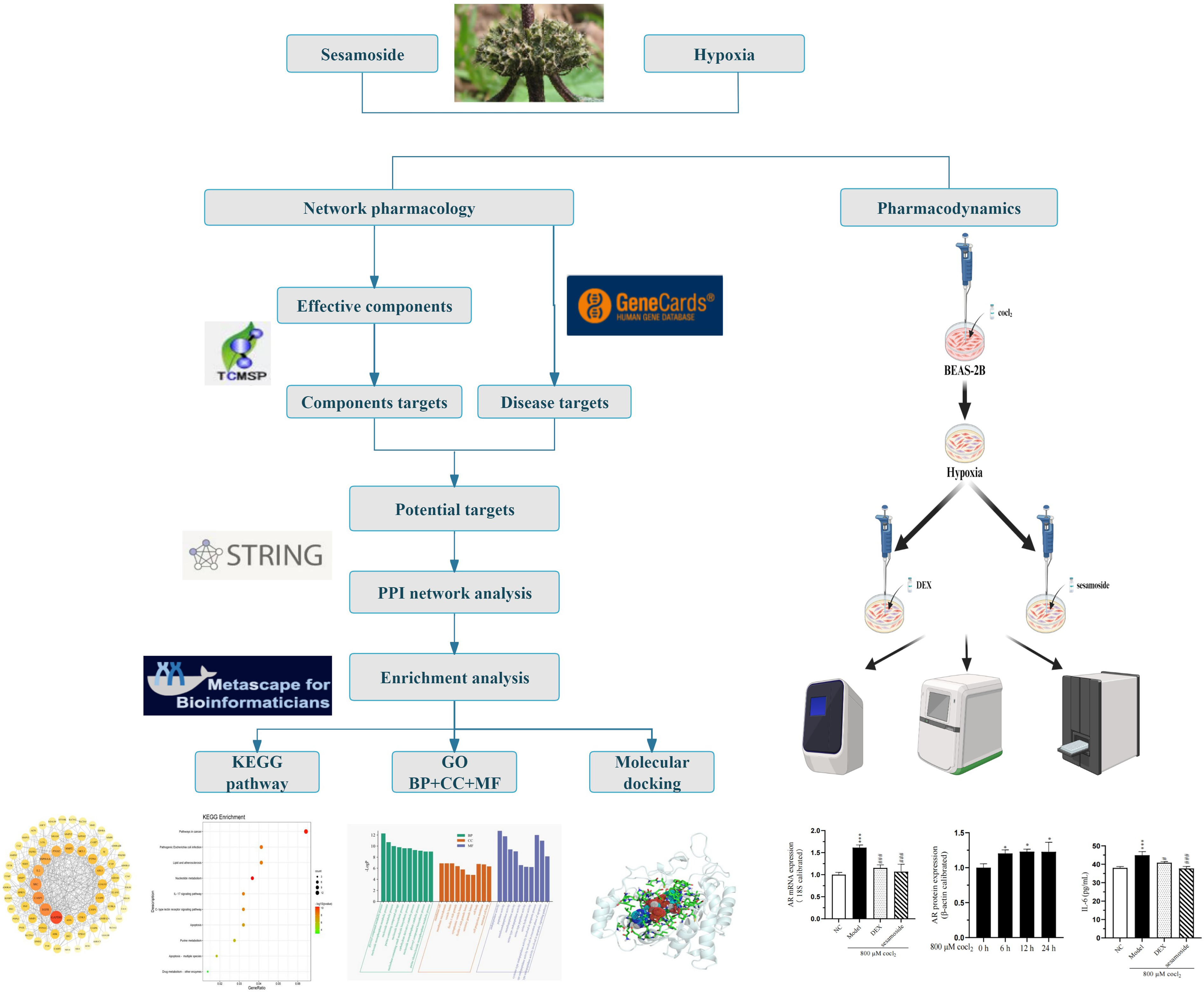
Network Pharmacology Analysis
Collection of Targets for Sesamoside Extracted From Phlomis younghusbandii Mukerjee
First, the PubChem database was used to identify the chemical structure (https://pubchem.ncbi.nlm.nih.gov/). The compound’s SMILES (Simplified Molecular Input Line Entry System) code was obtained, and the compound’s three-dimensional structure was downloaded. The compounds were then screened using the Swiss Target Prediction and TargetNet (https://targetnet.scbdd.com) (Probability >0) online databases to obtain predicted targets. Finally, gene names of the retrieved targets were converted and standardized using the UniProt database (https://www.uniprot.org/). 28
Drug-Disease Target Prediction
Using the words ‘hypoxia’ and ‘sesamoside’ we searched for disease-related targets in the GeneCards (https://www.genecards.org/), OMIM (https://www.omim.org/), and TTD (https://db.idrblab.net/ttd/). Then, we input the compound targets obtained in the aforementioned method and the disease targets into Venny 2.1.0 to identify the intersecting set. We summarized the intersecting genes to create a Venn diagram for sesamoside-hypoxia.
Construction of Component-Target Networks
The selected sesamoside and hypoxia targets were used to construct an interaction network diagram of targets and compounds through the Cytoscape platform (V3.9.1) (https://www.cytoscape.org/). Core targets were screened, and their degree values were calculated. Degree Centrality: In a network, the greater the degree of a node, the higher its degree of centrality, suggesting that the node is more important within the network.
GO Functional Enrichment Analysis and KEGG Pathway Enrichment Analysis
The selected core targets were analyzed using Metascape (https://metascape.org/), setting the species to ‘
Construction of PPI Network and Screening of Core Targets
The drug-disease targets identified in section ‘2.1.2’ were analyzed using the STRING database (https://string-db.org).
30
The species was set to ‘
Molecular Docking of Sesamoside With AR
The SMILES code of the compound was obtained from PubChem (https://pubchem.ncbi.nlm.nih.gov/), and the three-dimensional structure of the compound was downloaded and saved in SDF format. The three-dimensional structure and protein structure of AR were downloaded from PDB (https://www.rcsb.org/). ‘
CCK-8 Colorimetric Assay
CCK-8 analyzed the cell viability of cocl2 on BEAS-2B cells; cells were treated with 0-, 10-, 300-, 500-, 700-, or 900 μM cocl2 for 24 h, respectively. After cell treatment, CCK-8 was added and incubated at 37°C for 1-4 h, the absorbance was measured at 450 nm.
Cell Culture
Human bronchial epithelial cells (BEAS-2B) were cultured in a MEM complete medium with 10% fetal bovine serum and 1% antibiotics (100 UI/ml penicillin and 100 μg/mL streptomycin). Cells were divided into 4 groups: (1) NC group (blank control), (2) hypoxia group (cells treated with 800 μM cocl2 for 6 h), (3) DEX + hypoxia group (cells pre-treated with 1 mM dexamethasone for 24 h, followed by 800 μM cocl2 for 6 h), (4) sesamoside + hypoxia group (cells pre-treated with 200 μM sesamoside for 6 h, followed by 800 μM cocl2 for 6 h).
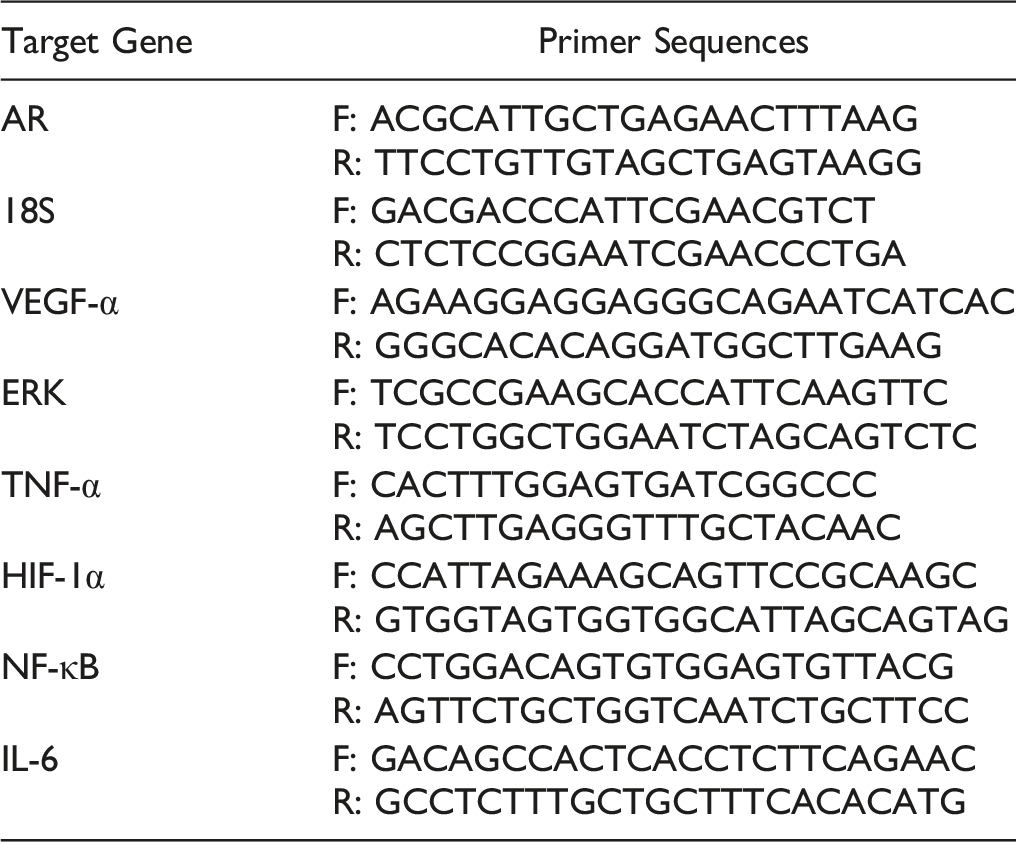
Quantitative Analyses of mRNA Expression by qPCR
Western Blots Analysis
Protein concentrations for cell lysates were determined using a BCA protein assay kit. Proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, transferred to polyvinylidene difluoride membranes, and blocked with 5% BSA for 1 h at room temperature. Proteins were then detected using primary antibodies incubated overnight at 4°C, followed by secondary antibodies for 1 h at room temperature.
ELISA
Cell supernatant was collected. According to the manufacturer’s instructions, the concentrations of TNF-α and IL-6 were measured with ELISA kits.
Statistical Analysis
The data were expressed as mean ± standard deviation. Significant differences were expressed as
Results
Intersection of Disease Targets and Drug Targets
Disease-related targets were retrieved using the GeneCards, OMIM, and TTD databases, and standard targets were predicted using the Swiss Target Prediction and TargetNet online databases. The common targets were obtained by intersecting the component targets with disease targets using Venny 2.1.0 (https://bioinfogp.cnb.csic.es/tools/venny/), resulting in 78 targets (Figure 1). These 78 intersecting genes were inputted into the STRING database for PPI analysis. Using degree as the criterion for evaluation, 24 genes with a degree more significant than the average were identified (Table 1). Venn diagram of sesamoside anti-hypoxia targets. Key Targets of Sesamoside in Anti-Hypoxia. Table 1 24 genes with a degree more significant than the average were identified by PPI.

Network Pharmacology Analysis of the Target Pathway of Sesamoside for Anti-Hypoxia
The 78 intersecting genes obtained from the sesamoside anti-hypoxia analysis were imported into the STRING database for PPI analysis. As mentioned in section 2.1.3, the higher the degree value of a node, the more critical it is within the network. Therefore, genes with a degree more significant than the average were identified as genes using degree as the criterion. The interaction network diagram for sesamoside’s anti-hypoxia effect was obtained. Nodes located centrally with more edges, larger sizes, and darker colors will likely be core targets (Figure 2A). The number of adjacent nodes is related to the probability of being a core gene; higher numbers indicate a greater likelihood of being a core target. The results identified 24 genes closely associated with sesamoside’s anti-hypoxia effect, among which AR was found. Subsequently, AR will be used as the gene for molecular docking. (A) Critical targets of sesamoside in the treatment of anti-hypoxia. (B) The GO enrichment analysis of critical targets. (C) The KEGG pathway enrichment analysis of critical targets. (D) Sesamoside-AR molecular docking pattern.
The selected core targets were analyzed using the Metascape online database for GO functional enrichment analysis and KEGG pathway enrichment analysis. GO functional enrichment analysis revealed that sesamoside is mainly involved in sugar and lipid metabolism, membrane vesicles, and peptidase activity in hypoxia-induced HAPE (Figure 2B). KEGG pathway analysis found that it primarily involves lipid metabolism, nucleotide metabolism, and the IL-17 inflammation signaling pathway (Figure 2C).
Binding Energy of Sesamoside With AR.
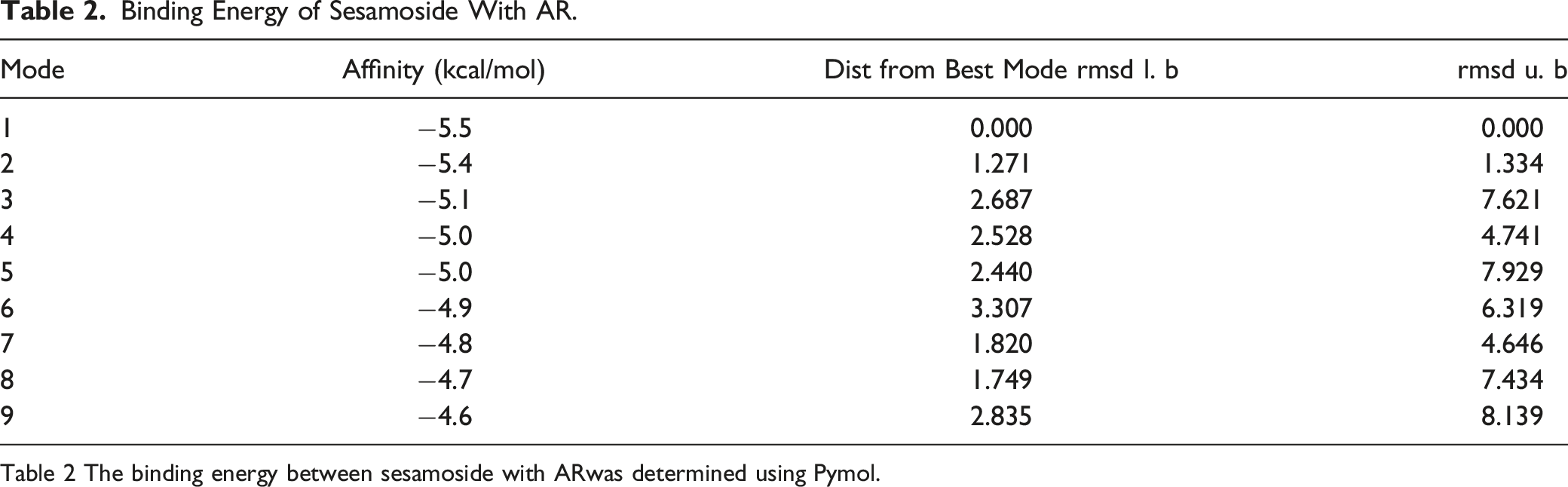
Table 2 The binding energy between sesamoside with ARwas determined using Pymol.
Sesamoside Reduced AR-Related Gene Expression Induced by Hypoxia
To validate the results of the network pharmacology analysis that sesamoside may exert its anti-hypoxia effect by regulating AR, we used cocl2 to treat BEAS-2B cells and established an in vitro hypoxia model. BEAS-2B cells were treated with a concentration gradient of 0-900 μM cocl2 for 24 h, cell viability was assessed using the CCK-8 assay. As shown in Figure 3A, cocl2 inhibited BEAS-2B cell viability with a 50% inhibitor concentration of 800 μΜ. Then we used 800 μΜ cocl2 stimulated BEAS-2B cell for 0-, 6-, 12-, or 24 h to detect the expression of AR, and found that the up-regulation trend of AR was the most significant at 6 h, therefore, we chose 800 μΜ treatment for 6 h to conduct subsequent experiments (Figure 3B and C). (A) The cell viability of cocl2 on BEAS-2B cells. (B) The mRNA expression of AR after cocl2 treatment. (C) The protein expression of AR after cocl2 treatment. (D) The mRNA expression of AR with sesamoside pre-treatment. (E) The mRNA expression of ERK with sesamoside pre-treatment. (F) The mRNA expression of VEGF-α with sesamoside pre-treatment. (G) The mRNA expression of HIF-1α with sesamoside pre-treatment.

To determine the effects of sesamosaide on anti-hypoxia, cells were divided into 4 groups: (1) NC group (blank control), (2) hypoxia group, (3) DEX + hypoxia group, (4) sesamoside + hypoxia group. Compared to NC group, the mRNA expression of AR, ERK, and VEGF-α was significantly increased; however, when cells were pre-treated with DEX or sesamoside, the expression of AR, ERK, and VEGF-α was significantly decreased (Figure 3D-F). HIF-1 is a nuclear protein with transcriptional activity, associated with hypoxia adaptation, inflammation development, and tumor growth. In our research, the mRNA expression of HIF-1α was down-regulated compared with the NC group after hypoxia treatment, and sesamoside pre-treatment had no significant impact on its expression (Figure 3G).
Sesamosed Alleviates the Inflammatory Response Caused by Hypoxia
To determine the effect of sesamoside on hypoxia-induced inflammation response, we examined the transcription and secretion of cytokines TNF-α, IL-6 and IL-10. The qPCR and ELISA results revealed that compared with NC group, the expression of TNF-α and IL-6 was up-regulated and IL-10 was down-regulated in hypoxia group. Meahwhile, sesamoside pre-treatment improved this situation somewhat (Figure 4A-E). These findings suggest that sesamoside alleviates the inflammatory response caused by hypoxia. (A) The transcription of TNF-α. (B) The transcription of IL-6. (C) The transcription of IL-10. (D) The secretion of TNF-α. (E) The secretion of IL-6.

Discussion
With the rapid development of modern pharmacology and bioinformatics, network pharmacology, as an emerging research approach, has shown significant potential in drug discovery and mechanism exploration. The anti-hypoxic mechanism of sesamoside, a natural compound with potential pharmacological value, is essential for developing novel anti-hypoxia drugs. Network pharmacology allows us to construct a complex network model that can capture the interactions between sesamoside and their potential targets and how these targets participate in hypoxia-related biological processes. The unique low-pressure and hypoxic environments at high altitudes pose significant health challenges, particularly in the form of HAPE, a severe altitude-related disease with high mortality rates. Given the limitations of existing treatments, there is a pressing need to explore novel therapeutic agents and understand their mechanisms of action. Through this network, we delved into the anti-hypoxia mechanism of sesamoside, we can identify critical nodes and pathways, which may be the key factors for sesamoside exerting anti-hypoxic effects.
Our network pharmacology analysis identified multiple targets associated with sesamoside’s anti-hypoxic effects, primarily involved in glucose and lipid metabolism, nucleotide metabolism, and inflammatory processes. We predicted the core targets of sesamoside against high-altitude hypoxia, including AR, GAPDH, EGFR, IL-2, and others. Notably, AR emerged as a crucial target, given its role in the polyol metabolic pathway and regulation of oxidative stress and inflammation. Therefore, it can be inferred that the role of AR is not limited to being a rate-limiting enzyme in the polyol pathway, but is likely a crucial factor affecting the entire body’s energy supply and sugar and lipid metabolism.
31
This hypothesis is consistent with our GO analysis results. Combined with the KEGG pathway enrichment analysis, the primary pathways involved in prevention and treatment include cancer, pathogenic
Our findings shed light on the anti-hypoxic mechanism of sesamoside, providing a theoretical basis and experimental foundation for its potential application in the prevention and treatment of hypoxic diseases, such as HAPE. The combination of network pharmacology and in vitro validation experiments has allowed us to identify critical targets and pathways through which sesamoside exerts its beneficial effects. While our study offers valuable insights, it is important to acknowledge the limitations of network pharmacology and the need for further experimental validation in animal models and clinical trials. Future research should focus on elucidating the precise molecular mechanisms underlying sesamoside’s anti-hypoxia effects and exploring its potential as a therapeutic agent for various hypoxic-related diseases.
Conclusion
In conclusion, by constructing a network model, predicting and validating target interactions, and integrating multiple omics data, the targets, signaling pathways, and biological functions of sesame glycosides in combating high-altitude hypoxia were explored and predicted. Our study underscores the potential of sesamoside as a novel anti-hypoxic agent with pleiotropic effects. The identification of AR as a key target of sesamoside’s actions sheds light on the underlying mechanisms of its anti-hypoxic effects. Experimental validation confirmed that sesamoside significantly modulates AR expression and reduces inflammatory cytokines, thereby mitigating hypoxia-induced cellular damage. This work provides a comprehensive theoretical basis and experimental foundation for the potential application of sesamoside in the prevention and treatment of hypoxic diseases.
Footnotes
Author Contributions
DS designed and organized the study, reviewing and editing, and provide fund support. MJ W performed the network pharmacology and experiments, prepared the original draft. YY Z performed the experiments, and provide fund support. XJ Z helped with data analysis and provide fund support. YR Z, HY Y and LZ helped with data acquisition and experimental materials prepar.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 32160165), the Natural Science Foundation of Tibet Autonomous Region (Grant No. XZ202201ZR0065G), Youth project of Xizang Minzu University (Grant No. 24MDQ06), Graduate Research Innovation and Practice Project of Xizang Minzu University (Grant No. Y2024012, Y2024004).
