Abstract

Keywords
Introduction
Preparations based on technologically processed antibodies (TPA) are obtained using a standardized technology of multiple vibrational processing of the initial substance in combination with sequential dilution at each stage of preparation. Their mechanism of action involves changing the conformation of the target 1 due to the spatial transformation of the hydration shells of proteins. 2 At the same time, it has recently been shown that the dilution process is an optional step and preparations that have a modifying effect on their target can be obtained without the dilution step. 3 The presence of dilution may be irrelevant, even if technically retained. 4
TPA-based drugs are used in the treatment of a wide range of diseases, including neuropsychiatric. For example, TPA to S100B in experiments in vivo exhibited anxiolytic, antidepressant,5,6 nootropic, and neuroprotective activity.7-10
Prospekta is prepared on the basis of a solution of antibodies that have been subjected to technological treatment (sequential dilution) as well as additional exposure to a physical factor (electromagnetic signal) (see Methods). Both types of treatment enable the drug to exert a modifying effect on the “target”. In the first case, the effect is exerted on the S100B protein, in the second, probably via biological feedback mechanisms, on the electrophysiological activity of the brain. 11
Prospekta has been shown to improve recovery in rats after hemorrhagic stroke, 12 exhibit nootropic activity in a focal cerebral ischemia model, 13 as well as demonstrate procognitive effects 14 and modulate EEG characteristics 15 . In randomized, double-blind, placebo-controlled clinical trials, Prospekta was found to be an effective and safe treatment for cognitive, behavioral, and mental disorders in patients with vascular dementia, 16 cognitive impairment in patients recovering from an ischemic stroke,17,18 ADHD in children, 19 and post-infectious asthenia in patients recovering from COVID-19. 20
Cognitive disorders, especially in the post-stroke period, are often aggravated by an anxiety-depressive component,21,22 which makes the study of the anxiolytic effect of Prospekta particularly relevant.
This article is devoted to the experimental study that uses the open field test 23 to assess the effect of different daily administration frequencies of the drug, as its specific composition makes it impossible to conduct conventional pharmacokinetic studies.
Materials and Methods
Samples
The test sample was an active component of the drug “Prospekta” (TPA to S100B protein, modified, 10 000 units of modifying activity). Polyclonal rabbit antibodies (Abs) to S100B protein (initial concentration 2.5 mg/mL) were produced by AB Biotechnology (UK) by affinity purification in accordance with the standards of good manufacturing practice. Technological processing of Abs included a series of successive centesimal dilutions and controlled hydrodynamic treatment at each dilution step. In addition, the solution of Abs to S100B protein was treated with an electrical signal, which introduced an additional modification into the initial substance. For this purpose, electroencephalograms (EEG) of healthy volunteers without any signs of abnormalities were recorded in accordance with the recommendations of the International Federation of Clinical Neurophysiology. A software was used to convert the recorded brain biopotentials into a combined (averaged) EEG signal. The resulting signal, pre-modulated by amplitude-frequency characteristics, was then applied as a potential difference to a water-alcohol solution containing Abs specific to S100B protein. This intermediate product was subsequently subjected to further gradual processing. For dilutions at each stage, a water-alcohol solution was used (1:100 at each stage), except for the last dilution, for which purified water was used. The theoretical reduction in the initial concentration of antibodies in the final dilution exceeds 1024 times. However, ultra-high dilutions of substances were tested using a number of methods of molecular analysis such as NMR, 24 inductively coupled plasma atomic emission spectroscopy (ICP-AES), inductively coupled plasma mass spectroscopy (ICP-MS), 25 dynamic light scattering, and phase microscopy. 26 The results obtained confirm the presence of molecules in such solutions, where the presence of any active molecules is theoretically unlikely. Most likely, the “carrier” of activity, determining the special properties of high dilutions of substances, are spontaneously formed nanoassociates. 27
We have previously established that high dilutions of the substance have a modifying effect on the initial substance, which allowed us to develop quality control methods for gradualised preparations.28-31
For sequential dilution of the initial substance in combination with vibration processing an automated microfluidics unit was used. It consisted of an Atlas syringe pump (Syrris Ltd., UK), a flow microreactor (microfluidic chip) and a computer with a software for automatic solution preparation. 32
Purified water treated only by the serial dilution method was used as a placebo. The samples were provided by OOO “NPF “MATERIA MEDICA HOLDING” in encrypted and ready-to-use form. To conduct a blinded experiment with placebo and three different doses of the same drug, vials with placebo and drug were marked for administration to rats in the morning, afternoon and evening, while of the three vials intended for a single administration of the drug, two contained placebo, for a double administration - one contained placebo, for a triple administration - all vials contained the drug under study. Decoding of the samples was carried out after receiving all the experimental data and the results of statistical processing.
Animals
Sexually mature male Wistar rats (n = 65) with a body weight of 256 ± 3 g were obtained from the STEZAR breeding facility (Vladimir region, Russia).
The rats were housed in polycarbonate and polysulfone cages, 3 animals per cage (dimensions 475*350*200 mm, floor area 1662.5 cm2, AWTech, Moscow, Russia) and in polycarbonate and polysulfone cages, 5 animals per cage (cell dimensions 545*395*200 mm, floor area 2152.75 cm2, AWTech, Moscow, Russia). The corn substrate Rehofix MK 2000 (JRS, Germany) was used as a litter in accordance with the housing standards. The air temperature in the vivarium was 21.6°C–24°C, relative humidity was 45-65%, and 12:12 h light-dark cycle was used. Rats were provided with ad libitum access to food and tap water. The animals were kept in accordance with the rules adopted by Directive 2010/63/EU of the European Parliament and of the Council of the European Union of September 22, 2010 on the protection of animals used for scientific purposes. All procedures with animals in the study were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of the Institute of General Pathology and Pathophysiology (protocol No. 5 dated 09 November 2022). The entire experiment was executed in compliance with ARRIVE guidelines.
Experimental Design
Upon arrival animals were randomly assigned to 4 experimental groups using a random number generator, with an initially planned number of 17-18 rats per group. The sample sizes were determined based on a power analysis, which was performed by repeatedly generating simulated datasets using distribution parameters (mean, standard deviation, and distribution type) derived from previous studies for each of the evaluated open field test parameters. A total of 1000 simulated comparisons were conducted for each parameter, and sample size estimates were iteratively adjusted until the statistical power reached at least 80%. Rats were housed in 3-5 animals per cage, with animals from different groups distributed across the same cage to minimize potential cage effects.
The experimental phase of the study lasted 18 days, from January 9 to January 27, 2023, and included the arrival and grouping of animals, a quarantine period (9 days after arrival, during which daily health monitoring was conducted), weighing, administration of test samples, and behavioral testing.
Samples were administered intragastrically via a curved stainless steel gavage needle (16G) suitable for rats (Instech Laboratories, Inc) in the volume of 5 mL/kg 3 times per day in the morning (9:00), in the afternoon (14:00), and in the evening (19:00) during 5 consecutive days starting at the end of quarantine period. A 5-day administration schedule was selected based on previous data showing that the component of Prospekta TPA to S100B was ineffective after a single dose in the Vogel test and elevated plus-maze test in rats but produced a significant effect following repeated administration over five days. 5
To facilitate behavioral testing, the initiation of sample administration was organized in batches: treatment of animals from one group of cages began on one day, followed by the next group on the following day, and so on. This approach allowed for consecutive sample administration and behavioral testing over several days. To ensure blinding, all samples were provided in encrypted vials. For the single administration group (Prospekta_1x), only one vial per day contained the active drug, while the other two contained placebo; for the double administration group (Prospekta_2x), two vials contained the drug and one placebo; for the triple administration group (Prospekta_3x), all vials contained the drug. Decoding of the sample allocation was performed only after data collection and statistical analysis.
On the sixth day of the experimental phase, the open field (OF) behavioral test was conducted. Testing was conducted sequentially from the first to the last cage, ensuring that animals from different groups were tested each day. A total of 11 to 15 animals were tested per day. The OF test was performed daily at 9:00 a.m. in a sound-attenuated room under controlled temperature and illumination conditions; behavioral parameters were analyzed by an experimenter blinded to the group allocation.
During the quarantine period and behavioral testing, six animals were excluded from the study for health-related or procedural reasons. Three animals (two from the Prospekta_3x group and one from the placebo group) were excluded due to signs of lethargy observed during daily monitoring. Additionally, three animals (one from the Prospekta_3x group, one from the Prospekta_2x group, and one from the placebo group) were excluded because they escaped from the open field arena during testing, making behavioral data collection impossible. The final group sizes were as follows: placebo (n = 16), Prospekta_1x (n = 18), Prospekta_2x (n = 16), Prospekta_3x (n = 15).
At the end of the experimental procedures, all animals were humanely euthanized by decapitation using a guillotine, in accordance with the institutional guidelines and approved protocols. Euthanasia was performed without causing unnecessary suffering, in a designated area away from other animals. The carcasses were removed only after confirmation of death by the responsible personnel.
Open Field Test
The open field (OF) apparatus for rats consisted of a gray polyvinyl chloride chamber (uncoated), 97 cm in diameter and 42 cm in wall height. The floor was divided into three rows of equally sized sectors. In the center of the arena, the illumination was 500 lx, in the zone 2/3 it ranged from 430 to 450 lx, and near the wall it was from 330 to 370 lx. The animals were habituated in darkness for at least 60 min prior to testing.
Animal behavior was assessed visually and recorded using a video camera mounted above the arena on a tripod. Each animal was placed near the wall of the arena and observed for 5 min. Upon completion of testing for each animal, the floor of the arena was cleaned with a reusable cloth soaked in a 6% hydrogen peroxide and dried with paper towels. After testing, each animal was temporarily placed in an auxiliary cage and later returned to its home cage.
During testing, RealTimer software (version 20, OpenScience, Russia) recorded the latency to enter the center, the time spent in the center and in the zones 1/3 and 2/3 of the arena, the number of unsupported and wall-supported rearings, head dips, latency to the first long grooming episode, number of short- and long-duration grooming episodes, and the frequency of defecation and urination. Data were collected over a 4-minute period. In addition, the proportion of time spent in the center and zones 1/3 and 2/3 of the arena, the mobility coefficient and the total vertical activity were calculated. In parallel, video footage of each animal was recorded during testing. The videos were processed using EthoVision XT software (version 14.0.1322, Noldus, the Netherlands) to create motion tracks. Based on the tracks, the distance travelled per minute and within specific arena zones (1/3, 2/3 of the arena zone and the center), average movement speed, and total immobilization time were calculated.
Statistical Analysis
The analysis and visualization of the data were carried out using the environment for statistical computing R version 4.2.2. (R Foundation for Statistical Computing, Vienna, Austria). The normality of data distribution was assessed using the Shapiro-Wilk test, and the homogeneity of the variances was assessed using the Bartlett’s test. Group comparisons were performed using one-way ANOVA with post hoc Student’s t-test when the data met the assumptions of normality and homogeneity of variance. If at least one group violated these assumptions, the Kruskal–Wallis test with post hoc Dunn’s test was applied. For categorical variables with two levels, Fisher’s exact test followed by pairwise comparisons was used. Poisson regression was applied where appropriate, and quasi-Poisson regression was used in cases of overdispersion. The latency period was analyzed using Kaplan–Meier survival analysis with post hoc comparisons. The Holm correction was applied to adjust for multiple comparisons. The differences were considered statistically significant at P < .05.
Results
Assessment of Time-Related Behavioral Indicators
In the OF test, animals typically experience stress and anxiety and prefer to remain in the dimly lit periphery near the arena wall, avoiding the brightly lit center. Preparations with an anxiolytic effect reduce the time spent in the periphery and promote animal’s exploratory activity – an increase in its movement in a less closed and more illuminated area of the arena. According to time-related indicators, Prospekta showed the most pronounced anxiolytic effect when it was administered twice. When placed in the zone 1/3 of the arena, the animals with double administration of the drug spent significantly less time in this zone (10% less than the placebo group, P < .05, Figure 1A). These animals also spent significantly more time in the 2/3 OF zone, with a 4.5-fold increase compared to placebo (P < .05, Figure 1B). The triple and single administration groups also showed increased time spent in zone 2/3, by 1.5 and 3.2 times compared to placebo, but to a lesser extent than the double administration group (P > .05). The time spent by rats in the center of the arena increased across all Prospekta dosage regimens: 5.6, 7.5 and 1.6 times, for single, double and triple administration, respectively (P > .05 vs placebo, Figure 1C). Animals receiving the drug twice entered the center more quickly than other groups (“Latency of the first entrance to the center”), however, the difference compared to placebo was not statistically significant (Figure 1E). Results of the Study of the Effect of Prospekta Administration Frequency on the Time Indicators in the Open Field Test: (a) Time Spent in 1/3 Zone of the Arena, (b) Time Spent in 2/3 Zone of the Arena, (c) Time Spent in the Center of the Arena, (d) Time of Immobility, (E) Latent Period of the First Entry to the Center. The Data are Shown as Boxplots, Statistically Significant Differences From Placebo (P < 0.05) are Marked With *; Statistically Significant Differences From the Prospekta_3x Group are Marked With #. (A–D) Kruskal-Wallis Test Followed by the Dunn Test With the Holm Correction for the Multiplicity of Comparisons; (E) - Kaplan-Meyer Survival Analysis With Post-Hoc Test
Immobilization time in a novel environment is commonly considered as the main indicator of stress. In such conditions, the animal typically experiences intense fear and freezes. The group receiving Prospekta twice daily demonstrated a statistically significant reduction in immobilization time compared to both the placebo and triple administration groups – by 26.7% and 29.9%, respectively (P < .05 in both cases, Figure 1D).
Assessment of Locomotor Activity Indicators
The locomotor activity indicators obtained via video tracking showed that the animals with double administration of the drug exhibited the highest activity levels. For example, double administration of Prospekta significantly increased the distance travelled in the periphery of the arena (P < .05 vs Prospekta_3x group, Figure 2A) and in the 2/3 zone of the OF (P < .05 vs placebo, Figure 2B). In the center of the arena, the same rats travelled 1.9 times more distance than the placebo group (P > .05, Figure 2C). Based on the total locomotor activity, the most active rats were those receiving Prospekta twice daily. The total distance travelled by this group was 39% and 40.8% higher compared to the placebo and the Prospekta_3 x groups, respectively (P < .05, Figure 2 d). Indicators of Locomotor Activity: Distance Travelled in Various Sectors of the Open Field Arena. The Data are Shown as Boxplots, Statistically Significant Differences From the Placebo Group (P < .05) are Marked With *; Statistically Significant Differences From the Prospekta_3x Group (P < 0.05) are Marked With #. (A, D) Analysis of Variance Followed by a Pairwise Comparison of Groups Using the Student’s t-Test; (B, C) Kruskal-Wallis Test Followed by the Dunn Test. The Holm Correction for the Multiplicity of Comparisons was Applied in all the Comparisons.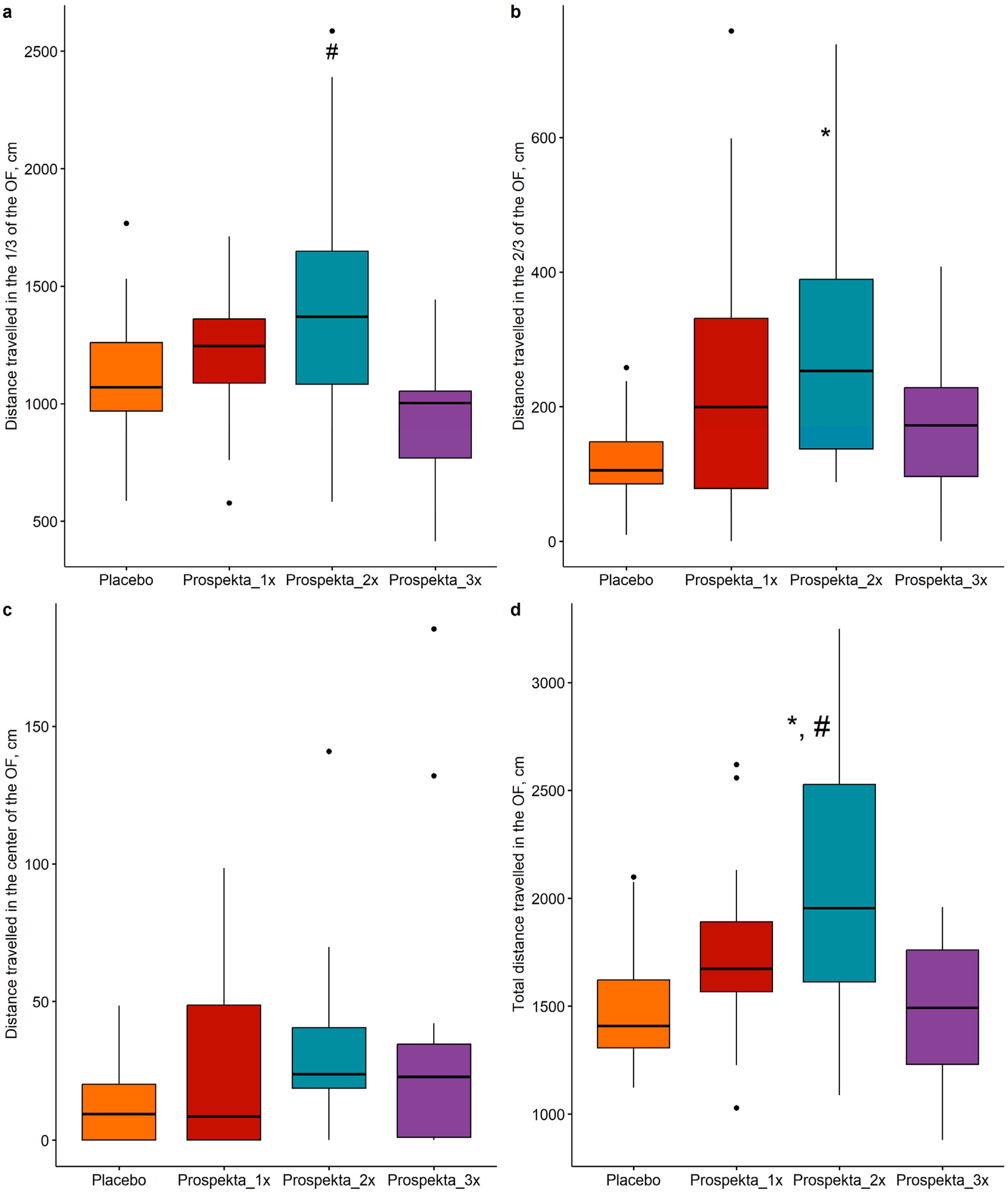
Additionally, the distance travelled per minute in the arena was calculated. At the beginning of test, all animals showed similar initial distance travelled. The group means differed by no more than 10% (P > .05, Figure 3). During the second and subsequent minutes of testing, the activity of all animals decreased. At the same time, a steeper decline in exploratory activity was observed for placebo and triple administration groups, whereas the decrease was more gradual in the double and single administration groups. The double administration group was more active than the others and consistently showed the highest distance travelled across all time intervals following the first minute (Figure 3). In addition, at the third minute, rats in the single administration group showed comparable mobility, which was significantly higher than in the triple administration and placebo groups. The Distance Travelled for Each Minute Spent in the “Open Field” Arena. The Data are Presented as Mean ± SD, Statistically Significant Differences From the Placebo Group (P < .05) are Marked With *; Statistically Significant Differences From the Prospekta_3x Group (P < .05) are Marked With #. (60-12 s, 120-180 s, 180-240 s) Analysis of Variance Followed by a Pairwise Comparison of Groups Using the Student’s t-test; (0-60 s) Kruskal-Wallis Test Followed by the Dunn test. The Holm Correction for the Multiplicity of Comparisons was Applied in all the Comparisons.
The number of transitions between different zones of the arena was assessed visually; a transition was recorded only when all four paws entered a new zone. Placebo-treated animals were the least likely to enter the central zone of the arena. In contrast, animals receiving the drug once, twice or three times daily entered the center 2.7, 3.4 and 2 times more frequently, respectively. The double administration group entered the center statistically significantly more often than the placebo group (Figure 4A). The Number of Entries to the Center (a) and the Average Speed of Movement (B) for the “Open Field” Arena. The Data are Presented in the Form of Boxplots, Statistically Significant Differences From the Placebo Group (P < 0.05) are Marked With *; Statistically Significant Differences From the Prospekta_3x Group are Marked With #. (A) Quasi-Poisson Regression; (B) Analysis of Variance Followed by a Pairwise Comparison of Groups Using the Student’s t-Test. The Holm Correction for the Multiplicity of Comparisons was Applied in All the Comparisons.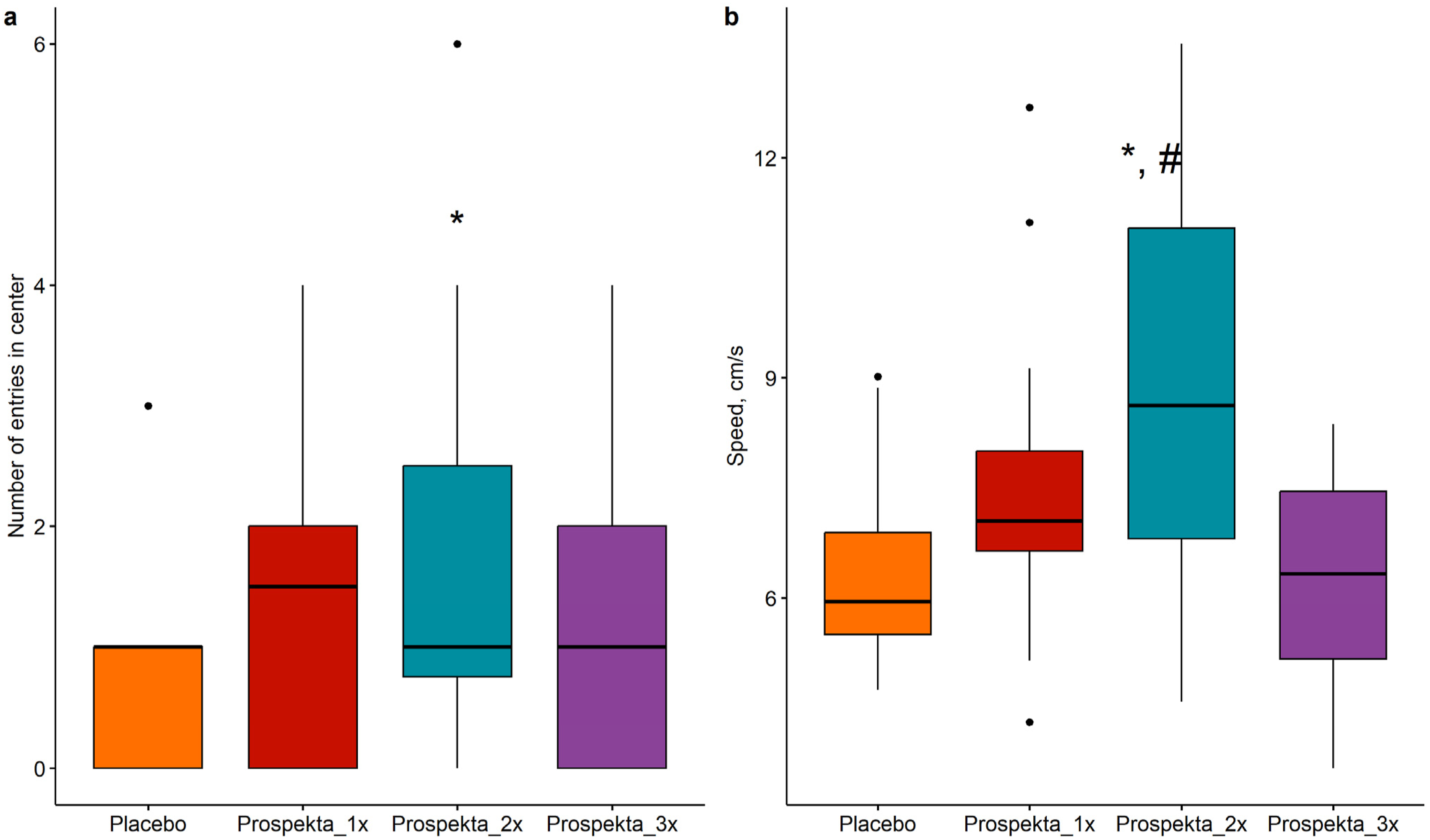
The average movement speed in the double administration group was 40.7% and 43% higher compared to the placebo and triple administration groups, respectively (P < .05, Figure 4B). These findings indicate a decrease in anxiety and an increase in exploratory activity in animals receiving double administration of Prospekta.
Additionally, the vertical activity of animals (wall-supported and unsupported rearing), autonomic responses (urination and defecation counts), orientation and exploratory activity (according to the parameter “Hole exploration”), and grooming indicators were assessed. No statistically significant differences were found between the groups for any of these additional parameters. However, the highest values of overall vertical activity and hole exploration, were observed for the double administration group (data not shown).
Discussion
We have shown that Prospekta, administered orally to Wistar rats twice a day for 5 days (daily dose of 10 mL/kg), exhibits the most pronounced anxiolytic effect. In rats receiving the drug twice daily, both exploratory and locomotor activity indicators showed a statistically significant increase. This included an increase in total distance travelled along with an enhanced exploratory motivation, reflected in a higher number of entries into the center and greater time spent in the more central zones of the open field.
In the case of triple administration of Prospekta, behavioral parameters were comparable to those observed in the placebo group. A single administration of Prospekta tended to increase the overall activity of the animals, however, the magnitude of the effect did not reach statistical significance in most parameters compared with placebo. Importantly, since animals were randomly assigned to groups at the beginning of the study, and animals exhibiting signs of lethargy or illness were excluded prior to analysis, the observed behavioral differences between groups cannot be attributed to pre-existing differences in animal condition or allocation bias. Repeated handling and the intragastric administration procedure may contribute to increased stress levels in all groups; however, all animals were subjected to identical handling protocols, and the experimental conditions were standardized across all groups. The difference in activity and anxiety-related parameters was observed only in the Prospekta_2x group, which suggests a specific effect of the treatment rather than a generalized response to stress.
The observed dose-response effect has an inverted U-shaped dose-effect curve, which is typical for dose-response curves of some anxiolytic drugs. Such curves were first described in rats evaluated using elevated plus maze test. 33 In this study, cannabidiol increased open arm entries (anxiolytic effect) at doses of 2.5-10 mg/kg, but not at 20 mg/kg. Similar inverted U-shaped dose-effect curves have been reported in other studies. For example, in spontaneously hypertensive rats, pre-stimulus inhibition was abolished by cannabidiol at 30 mg/kg, but not at 15 and 60 mg/kg. 34 In a rat model of reserpine-induced cognitive impairment, cannabidiol at 0.5 mg/kg attenuated the impairment, whereas a dose of 5 mg/kg was ineffective. 35
Due to the technological features of the preparation of TPA-based drugs, the conventional concept of “dose”, defined as the amount of a medicinal product administered over a certain period, cannot be fully applicable. It has been shown previously that the effect of a drug cannot be explained by the residual content of molecules of the initial substance in dilutions. The concept of “dosage regimen” includes not only the dose of the drug but also the frequency of its administration. In this study, the enhanced effect observed with double administration is probably related not to an increase in the conventional “dose”, but rather to a cumulative effect resulting from repeated administrations.
For one of the components of the studied drug, TPA to S100B, different relationships between the administration volume and the magnitude of the effect have also been reported. For example, in the elevated plus maze test in rats a linear relationship was observed, with the highest administration volume used in the study (10 mL/kg) being the most effective. However, in the Vogel conflict test, TPA to S100B enhanced sensitivity to punishment for alcohol consumption in rats at a volume of 7.5 mL/kg (P < .05), but not at 2.5 and 10 mL/kg, indicating an inverted U-shaped relationship between effect and administration volume. 5
This bell-shaped effect can be explained by the activation of the negative feedback system, multiple binding sites for the substance under study, or specific features of ligand-receptor interaction. In this experiment, we studied the effect of Prospekta, the main target of which is S100B protein. This protein is widely distributed in the brain (not limited to this organ) and is involved in the regulation of numerous physiological processes. Apparently, the pleiotropic action of S100B, which is modulated by TPA, may partly account for the inverted U-shaped response observed in our study. In particular, in vitro TPA to S100B was shown to increase ligand binding to 5-HT1F, 5-HT2B, 5-HT2C, 5-HT3, and D3 receptors, while inhibiting binding to GABA-B receptor subtypes and sigma-1 receptors. 36 In functional tests, TPA to S100B acted as a 5-HT1A receptor agonist and as an antagonist at 5-HT1B, D3, and GABA-B receptors. 36 In addition, the electromagnetic component of the drug may also contribute to its effect by normalizing the electrophysiological activity of the central nervous system through biofeedback mechanisms. It is known that electromagnetic signals play an active role in synchronizing cell function and/or adaptive response, and may be characterized by specific electromagnetic patterns that can be used for therapeutic purposes. In particular, the electromagnetic information transfer through aqueous system (EMITTAS) procedure has demonstrated efficacy in the treatment of pain, depressive and anxiety disorders, etc. 37
Our study has several limitations. The duration of Prospekta administration was limited to five days based on our previous behavioral studies, 5 which may not be sufficient to capture long-term or cumulative effects of the drug. In our previous studies in models of ischemic and hemorrhagic stroke,12,13 a longer treatment period of at least two weeks was required to achieve therapeutic effects. It is worth noting, however, that these are severe and life-threatening conditions, which may explain the need for longer treatment periods to mitigate their delayed consequences, in contrast to the anxiolytic effect observed in healthy animals. In future studies, a longer administration period may be considered to evaluate whether the anxiolytic effect of the drug becomes more pronounced with extended treatment. Additionally, the anxiolytic effect was evaluated only using the open field test, which may not capture all aspects of anxiety-related behavior. The inclusion of additional behavioral paradigms, such as the elevated plus maze or the light-dark box test, in future studies would provide a more comprehensive evaluation of the drug’s anxiolytic potential. Finally, the study was conducted in healthy rats, and modeling anxiety or stress-related conditions in future experiments would enhance the clinical relevance of our findings.
In conclusion, the results of this study demonstrate a significant impact of the dosage regimen of Prospekta on its anxiolytic effect in Wistar rats. Twice daily administration of the drug was found to be the most effective.
Footnotes
Ethical Statement
Author Contributions
Experimental procedures and Data collection: Svitkin Kirill, Morozova Elizaveta and Krasheninnikova Anna; experimental design: Kardash Elena and Ganina Ksenia; analysis and interpretation of the data: Krasheninnikova Anna, Kardash Elena and Ganina Ksenia; writing manuscript: Emelyanova Alexandra; overall supervision: Tarasov Sergey and Epstein Oleg. After reviewing the final manuscript, all authors gave their approval.
Funding
The authors disclosed receipt of financial support for the research, authorship, and/or publication of this article: the study was funded by OOO “NPF “MATERIA MEDICA HOLDING”, Moscow, Russia. Employees of OOO “NPF “MATERIA MEDICA HOLDING” participated in planning the experiments and writing the manuscript, performed the statistical analysis, and made the decision to publish the work. OOO “NPF “MATERIA MEDICA HOLDING” covered the APC for the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Svitkin Kirill, Morozova Elizaveta, Krasheninnikova Anna, Kardash Elena, Ganina Ksenia, Emelyanova Alexandra, Tarasov Sergey are employees of OOO “NPF “MATERIA MEDICA HOLDING” (full or part-time). Epstein Oleg is the founder of OOO “NPF “MATERIA MEDICA HOLDING”. OOO “NPF “MATERIA MEDICA HOLDING” sponsored the study, performed the statistical analysis, made the decision to publish the work and covered the costs associated with the publication of the article, participated in planning the experiments and writing the manuscript. TPA to S100B are active pharmaceutical ingredients (separately or as one of the components) of the commercial drugs “Tenoten”, “Tenoten for children”, “Divaza”; TPA to S100 B modified is an active pharmaceutical ingredient of the commercial drug “Prospekta”, which are manufactured by OOO “NPF “MATERIA MEDICA HOLDING”. Patents and patent applications for this substance and drugs belong to Epstein Oleg/OOO “NPF “MATERIA MEDICA HOLDING”. The authors have fully disclosed these interests to Dose-Response journal.
Data Availability Statement
The data will be made available upon reasonable request to the corresponding author.
