Abstract
Recently, there has been a radical change in understanding of the nature of drugs based on highly diluted solutions. It has been established that their activity does not depend on the content of the original substance in dilutions, but is a consequence of the technological processing (TP) of dilutions with vibration, which accompanies each dilution during the preparation of solutions and, among others, leads to the formation of nanoparticles with certain properties. Repeated vibration treatment leads to the appearance of modifying activity that is absent in the original substance, and these effects of TP solutions can be exerted without direct contact with their targets, which clearly indicates the physical nature of the TP solution’s activity. In the framework of this article, a statistically significant effect of TP antibodies to the insulin receptor on glucose consumption by CHO cells both with and without contact exposure, as compared with control (P < 0.05) was shown in the vast majority of the experiments. The obtained results shed light on a possible source of activity of drugs based on TP antibodies, which should be associated with the applied vibration effect and can manifest itself both with contact exposure and without it.

Keywords
Introduction
Relatively recently, studies have started to appear on the search for pathways underlying the effects of highly diluted substances. Among the various hypotheses, it can be highlighted that for such dilutions of substances, experiments have detected long-lived nano- and microparticles of unknown nature,1–3 which emerge in aqueous solutions during serial dilution combined with intensive vibration treatment. In the process of preparing multiple serial dilutions, the molecules of the original substance can be engaged in the emergence and stabilization of nanobubble structures formed during an intensive mechanical process.4–9 A number of authors claim that molecules of the original substance can be retained even in high dilutions due to the flotation effect.1,10 However, the activity of high dilutions is associated not so much with the presence of an insignificant number of the original substance molecules, as with the presence of nanoassociates that self-organize during multiple dilutions with shaking.11,12 At the same time, the biological activity of high dilutions is related to changes in their physical-chemical properties.3,12–14 Apparently, the ability to emit in the millimeter range shown for nanoparticles can make an important contribution to the realization of the effects of high dilutions.15,16
During the research on high dilutions of biologically active substances, it has been found that they exert a modifying effect (ME), which is specific (selective), aimed only at the target of these biologically active substances. 17 ME manifests itself in a conformational transformation of the target sufficient to activate a biological pathway in the body with the participation of this target, followed by a therapeutic effect. Furthermore, it has been found that the activity of dilutions appears in cases when they are subjected to vibration-mechanical treatment combined with exposure to the electromagnetic field during preparation,3,18 and as a result nanoassociates are formed in solution. 3 Revealing of the importance of the preparation technology contributes to the idea of highly diluted (HD) solutions, which, in this case, are accumulators of external vibration effects rather than “small doses”. 19
To date, we have studied ME mainly using HD antibodies, which has resulted in the development of a new class of drugs. Technological treatment of solutions consisting in a repeated procedure of serial dilution of the original substance accompanied by external rhythmic effect received the official name “gradual technology” after the General Pharmacopoeia of Russian Federation Monograph “Biological medicines prepared using gradual technology” was developed and put into effect for drugs containing high dilutions of antibodies (GPM.1.7.0001, SP of the RF XV). Therefore, drugs that have been subjected to a similar procedure will be called “gradualized drugs”. The main repeatedly confirmed advantage of technologically processed (TP) drugs is their high safety.20,21
Recently, the ability of TP drugs to affect the target without direct contact with it has been discovered: it has been shown that the previously known ME of TP antibodies to interferon-gamma17,22 can be exerted even when the effector (TP antibodies) and the target (interferon-gamma) are in closely spaced test tubes.23–26 It has been found that molecules with a large molecular weight 27 using as a target (or nanoparticles with a complex surface shape 16 forming during effector’s preparation) possess a more pronounced ability to interact without direct contact between the effector and the target.
Following the research on the mechanisms underlying the biological activity of TP drugs, this paper shows that the biological effects of TP antibodies (TPA) can be realized without direct contact between them and their targets. To demonstrate this effect, an oral hypoglycemic drug has been used, which contains antibodies to the beta subunit of the insulin receptor as the main active ingredient sprayed on an excipient, isomalt. The antihyperglycemic effect of the drug is based on its influence on the phosphorylation of the insulin receptor beta subunit. 28 The drug stimulates insulin-dependent glucose uptake 29 and adiponectin secretion in vitro 30 reduces plasma glucose levels, and improves glucose tolerance in vivo31,32 In clinical trials, the drug has shown its efficacy in the treatment of type 1 and type 2 diabetes mellitus, as well as impaired glucose tolerance.33–35 To carry out this work, a standard approach was chosen allowing the effect of samples on glucose consumption by CHO cells to be analyzed in vitro. All experiments were conducted in the same way and in parallel, with the only difference that 1 half of the samples was added directly to the cells and the other half was placed in containers without direct contact with the cells, which allowed us to show a single mechanism of the drug activity for interactions with the target molecule.
Materials and methods
Cell Line and Culture Maintenance
Chinese hamster ovary cell line (CHO-S) was obtained from Thermo Fisher Scientific (USA, R80007) and used at 11-24 passages. The culture was maintained in Hybris-1 serum-free culture medium (PanEco, Russia, C740p) with
All procedures with cells were performed in a class II type A2 microbiological safety cabinet (BMB-II-“Laminar-C”-1.2, ZAO “Laminar Systems”, Russia). To avoid cross-contamination, the samples were placed in different plates, which were incubated on different shelves inside the incubator.
Preparation of Samples
The samples were used as tablet dosage forms and stored at room temperature in dark place. All preparations were made under clean conditions (cleanliness class D) in a laminar flow hood, using sterile automatic pipettes and sterile pipette tips. Glass vials for preparing samples were sterilized in a steam autoclave (Tuttnauer 2540 ML, Germany). The vial caps were treated with 70% ethanol and UV for 30 minutes. Purified water was obtained using a Milli-Q purification system (Millipore, Darmstadt, Germany).
TPAs to the β-subunit of the insulin receptor (InsR-β) and to endothelial NO-synthase (eNOS) are active pharmaceutical ingredients of commercial drug Subetta (OOO “NPF “MATERIA MEDICA HOLDING”, Russia). Affinity purified rabbit polyclonal antibodies to InsR-β (2.5 mg/mL) and to eNOS (1 mg/mL) were manufactured (Ab Biotechnology Limited, UK) in accordance with the GMP requirements for starting materials. TPA to InsR-β and TPA to eNOS were produced by the GMP-compliant manufacturing facility of OOO “NPF “Materia Medica Holding” in accordance with the technology described in the United States Patent 8,535,664. 36 Briefly, technological processing of antibodies to eNOS and to InsR-β consisted in sequential multiple dilutions followed by controlled intensive hydrodynamic (mechanical) treatment at each dilution step. The procedure of hydrodynamic treatment was as follows: the samples were shaken vigorously with impact by hand with a controlled frequency of about 4 Hz (21 strokes in about 4.8 s). For the preparation of all dilutions (1:100 at each stage), a water-alcohol solution was used, except for the final 1, which was made with purified water. The theoretical level of reduction in the concentration of the original antibodies is at least 10−24 times. Therefore, if physical aspects of heterogeneity of solutions described in the scientific literature 10 are not taken into account, the theoretical concentration of initial antibodies might be 2.5 × 10−24 mg/mL (TPA to InsR-β) or 1.0 × 10−24 mg/mL (TPA to eNOS). Molecular analyses performed using NMR, 37 plasma-atomic emission spectroscopy (ICP-AES) or inductively coupled plasma-mass spectroscopy (ICP-MS), 10 dynamic light scattering and laser phase microscopy 1 support the presence of molecules even at dilutions beyond the point at which any active molecules are theoretically expected to be present. The resulting solution was sprayed in a fluidized bed unit on isomalt powder (using a solution: isomalt weight ratio of 1:5) and dried with warm air. The resulting saturated isomalt powder was blended in a mixer with excipients to obtain a mass for tableting; compacting it in a tablet press produced experimental tablets for the study.
TPAs to eNOS and S100 B protein are active pharmaceutical ingredients of the commercial drug Divaza produced by OOO “NPF “MATERIA MEDICA HOLDING” (Russia). Affinity purified rabbit polyclonal antibodies to S100 B (2.5 mg/mL) and to eNOS (1 mg/mL) were manufactured (Ab Biotechnology Limited, UK) in accordance with the GMP requirements for starting materials. TPAs to eNOS and to S100 B (and the resulting tablets) were produced using a protocol similar the 1 described above.
A similar dosage form (tablets) was used as a placebo control, but instead of antibodies, glycine buffer, solvent of polyclonal antibodies, was used as the original substance for saturation of the isomalt powder.
There were 2 approaches to analyzing samples: 2.1 To study the direct effect on cells, 3 tablets of each test samples (Subetta, placebo and Divaza) from the same batch, respectively, were placed in sterile vials; 10 mL of the culture medium Hybris-1 without insulin (PanEco, Russia, C740p-1) was added. Next, the vials were incubated at 37°C for 30 minutes (TS-1/80 SPY (JSC Smolenskoye SKTB SPU)), then at room temperature for another 5 minutes. After that, the contents were mixed using Vortex V-1 plus (Biosan, Latvia) until the tablets were completely dissolved and filtered using PVDF-0.45/25 μm syringe filters (Macherey-Nagel, Germany, 729219.400). The resulting solutions (hereinafter referred to as ‘filtrates’) were added to plate wells in accordance with the described scheme (Figure 1). 2.2 To study the non-contact effect of the drug, tablets of each test samples (Subetta, placebo and Divaza) from the same batch, respectively, were thoroughly comminuted. 35 mL of purified water (Milli-Q) was added to 84 g of the resulting powder. The resulting paste was thoroughly mixed and transferred to sterile containers. Then plates with the cells were placed above them in such a way that the bottom and partially the side walls of the plate were covered with the paste (Figure 2). Scheme of the experiment to study the direct effect of the test samples on CHO-S cells. Scheme of the experiment to study the non-contact effect of the test samples on CHO-S cells.
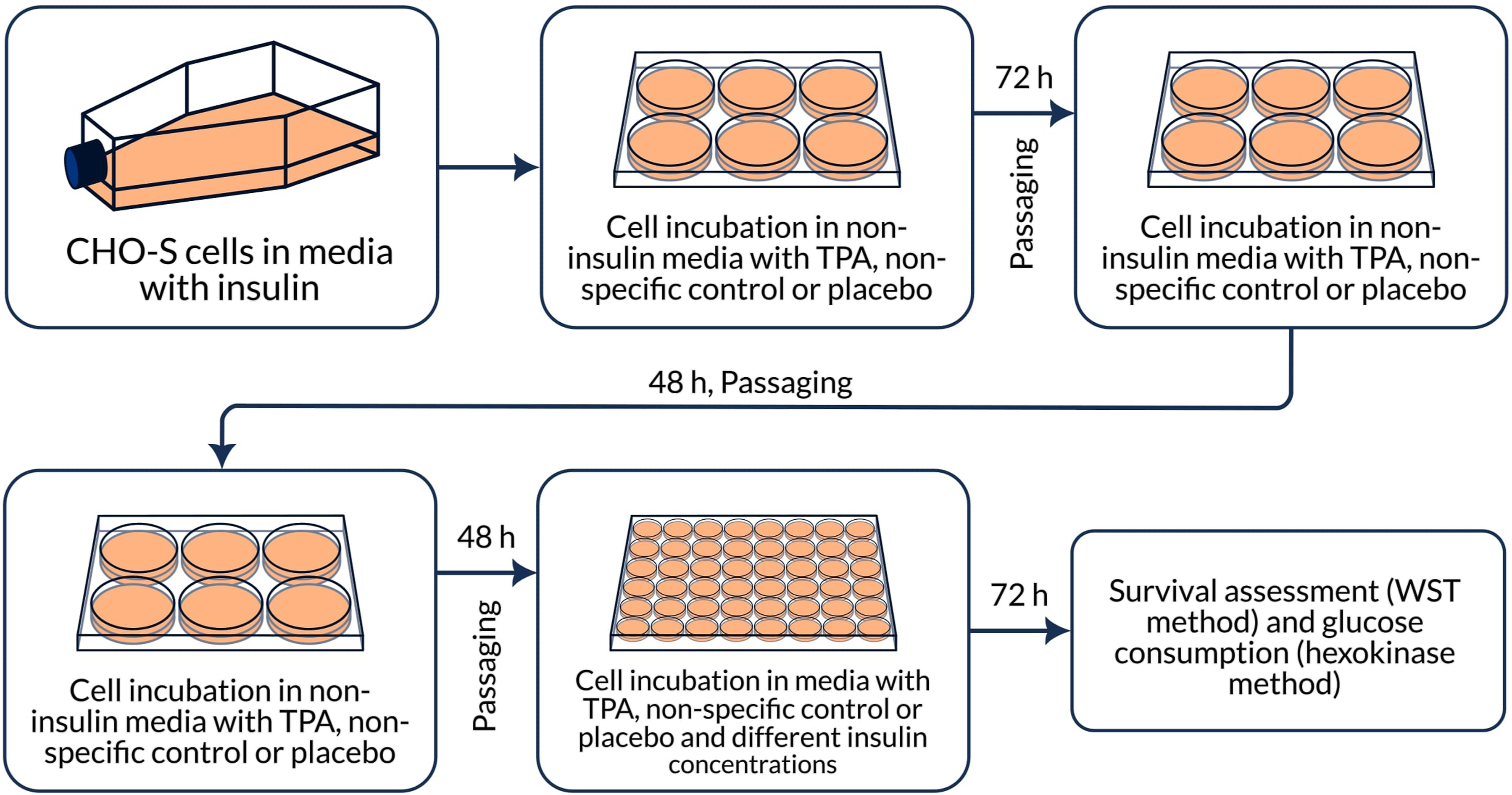
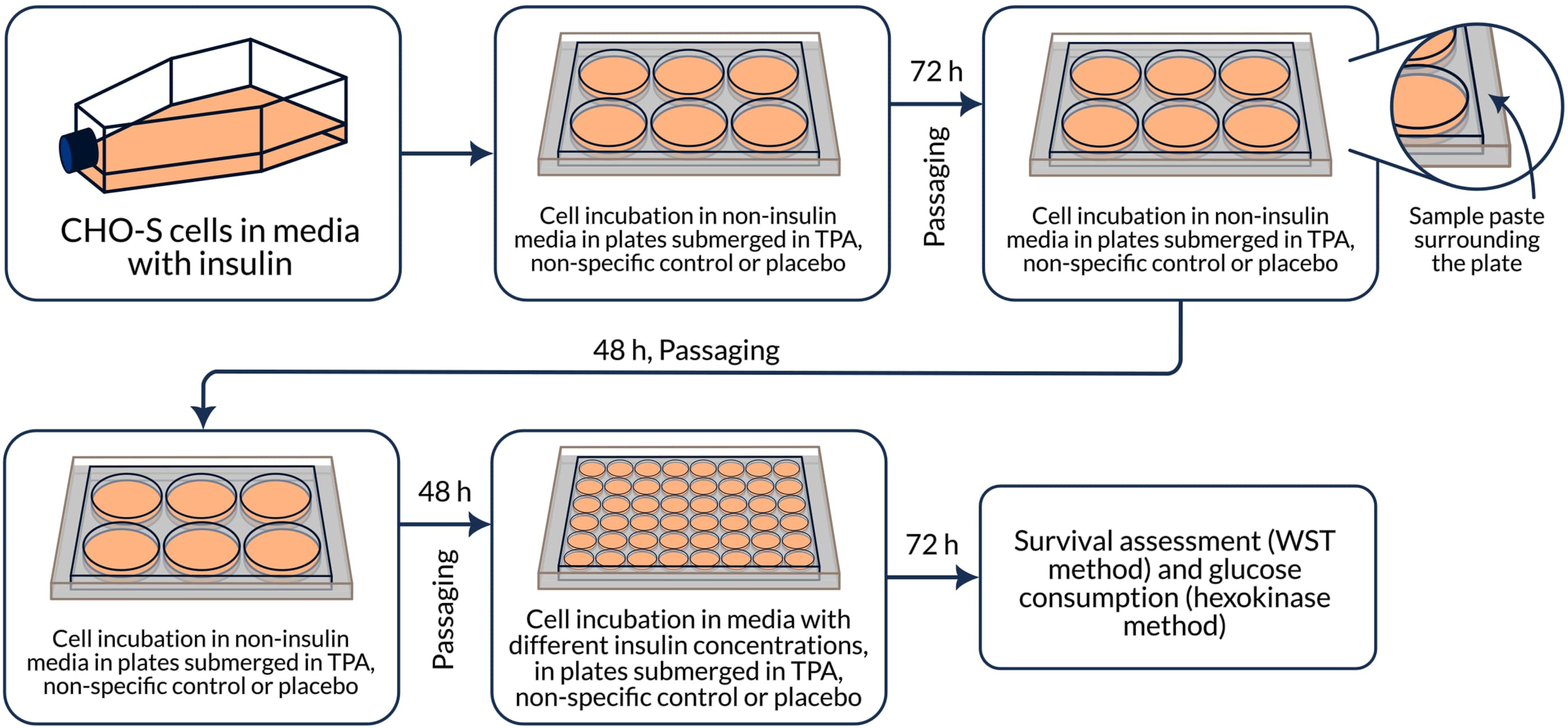
All samples were prepared by a single operator on the same day and under the same conditions. All tablet samples for testing were provided by OOO “NPF “MATERIA MEDICA HOLDING”. The samples were tested blindly and decoded after the experiment results had been obtained and statistically calculated.
Cell Culture Experiments
Direct Effect of Subetta on CHO-S Cells
In this experiment, the direct effect of Subetta was studied when it was added directly to the cells in the culture medium. The technique was initially based on 38 and adapted to the experiment requirement. The temperature (19 °C–25 °C) and humidity (15%–75%) were monitored. The experiments were performed over a 1-year period.
At the first stage, CHO-S cells were adapted to the conditions of insulin deficiency in the culture medium in the presence of Subetta or placebo (control), or Divaza (as non-specific control). For this, after routine cultivation, the cells were rinsed with the Versene solution (PanEco, Russia, P080p) and placed in Hybris-1 culture medium without insulin. Three 6-well culture plates (Corning, USA, 3516) were used: 1 for Subetta, 1 for placebo, and 1 for Divaza (Figure 1). The cells were placed into the plates at a concentration of 3 × 105 cells/ml, 2 mL per well. Then 400 μl of filtrates of the samples was added. The final volume in the wells was 2400 μl. Then the cells were incubated for 72 hours at a temperature of 37°C, 5% CO2 in a humidified atmosphere (CO2 incubator (Binder, Germany)). At the end of the incubation, 1200 μl of cell suspension was taken from each well after careful resuspending, and 800 μl of medium and 400 μl of filtrates of Subetta or placebo, or Divaza were added. After 48 hours of incubation at 37°C and 5% CO2, this procedure was repeated.
Previously, it has been shown that the effect of Subetta on InsR-β is enhanced in the presence of insulin. 28 Therefore, at the second stage of the experiment, exogenous insulin was added to the cells. First, the cells were transferred to 48-well culture plates (Corning, USA, 3548), 1 for the drug and 1 for placebo. Hybris-1 culture medium without insulin (500 μl per well) was added into the outer wells. Cell suspension (250 μl per well) with a concentration of 3 × 105 cells/ml was added into the central wells. Then stock solutions (250 μl per well), which consisted of filtrates of the tested samples (1/6 of the final volume in the well) and the culture medium with different concentrations of recombinant human insulin (0 μg/ml, 0.5 μg/ml, 5 μg/ml, and 50 μg/ml) (PanEco, Russia, F062), was added to the central wells (Figure S1). The final volume in all wells was 500 μl.
After 72 hours of incubation, the cell suspension from all wells was taken for the analysis of cell survival (WST method with cell proliferation reagent WST-1 (Roche Diagnostics, Germany, 11644807001)) and glucose consumption (hexokinase method with hexokinase (420 U/ml) + G6P-DH (210 U/ml) (Megazyme, USA, E-HKGDH)); it was the third day of incubation in the presence of insulin and the tested samples (Figure 1).
Non-contact Effect of Subetta on CHO-S Cells
In this experimental series, the possibility of a non-contact effect exerted by Subetta on CHO-S cells was assessed. The non-contact effect meant the potential effect of the drug on cell proliferation and glucose consumption when the plate was surrounded by a tablet paste, without direct contact between the cells and the tablet paste.
To study the non-contact effect, we conducted experiments using the same technique as with the direct effect. The difference was that instead of filtrates of drugs, serum-free cell culture medium Hybris-1 without insulin (400 μl) was added to CHO-S cells (at a concentration of 3 × 105 cells/ml, 2 mL per well). The plates were immersed in a paste prepared from tablets of Subetta or placebo, or Divaza during the whole experimental period. In each experiment, 3 plates were also used: 1 for each of the test samples.
The plates were incubated for 72 hours at a temperature of 37°C, 5% CO2 in a humidified atmosphere. Then the medium was changed. For this, 1200 μl of cell suspension was taken from each well and 1200 μl of the medium (per well) was added. After 48 hours, the medium was changed again. The cells were then transferred to 48-well culture plates, 1 for the drug and 1 for placebo. This layout was chosen in order to avoid distant effects of the drug on the control group or vice versa during the experiment. In order to obtain reliable results and avoid a possible edge effect, all the edge wells of the plates were filled with cell culture medium. Hybris-1 culture medium (500 μl per well) without insulin was added into the outer wells. Cell suspension (250 μl per well) with concentration of 3 × 105 cells/ml was added into the central wells. Medium (250 μl per well) with different concentrations of insulin (0 μg/ml, 0.5 μg/ml, 5 μg/ml, and 50 μg/ml) was added to the central wells (Figure 2).
The final volume in all wells was 500 μl. After 72 hours of incubation, the cell suspension from all wells was taken for the analysis of cell survival and glucose consumption.
The Mann-Whitney test (P value <0.05) was used for statistical data processing. An increase in glucose consumption normalized to cell number calculated by analysis of cell survival (WST method with cell proliferation reagent WST-1 (Roche Diagnostics, Germany, 11644807001)) in the presence of the drug with statistically significant differences from the control group (placebo) at least 1 concentration of insulin was considered as a “Positive result”.
The figures present the data using 95% confidence intervals of the difference between the means for the placebo group and for the drug group. The inclusion of zero in the confidence interval indicates the absence of statistically significant differences between the placebo and drug groups at a significance level of P = 0.05.
Results
The effect underlying the “antihyperglycemic” activity of a drug containing TP antibodies to InsR-β was shown when the experiments were carried out in parallel under the same conditions, either with contact between samples and the target, or without direct contact between them. 10 pairs of experiments were conducted, including an experiment where a non-specific control was additionally tested – a drug containing TP antibodies that did not have hypoglycemic activity. Although there were experiments where statistically significant differences in glucose consumption by cells were found in the presence of the drug compared with the control group (placebo) only in the case of either contact exposure or non-contact exposure, in most of them, glucose consumption increased statistically significantly for at least 2 concentrations of insulin in the presence of the test drug, both with contact and non-contact effects on cell culture (Figure 3 and Summary Table 1). Comparison of 95% confidence intervals of the difference in the means between different concentrations of insulin and different types of exposure for each experiment individually. Dots indicate the differences in the average glucose consumption by cells in the presence of a placebo or drug, and lines indicate confidence intervals showing this difference with 95% probability. The y-axis shows the number of experiment (left) or the concentrations of added insulin, μg/ml (0 μg/ml (0.ins), 0.5 μg/ml (0.5.ins), 5 μg/ml (5.ins), and 50 μg/ml (50.ins)) (right). The occurrence of a statistically significant difference in glucose consumption in the presence of a drug or placebo depends on the type of exposure.

As mentioned earlier, in addition to the main group of experiments, a specificity test was performed using a non-specific control, Divaza, containing TP antibodies to other proteins (Experiment 10 in Figure 3). It has been shown that the effect observed is specific and seems to be related to TP antibodies to InsR-β.
After averaging the results obtained in 10 pairs of experiments, the following result was obtained (Figure 4): it can be clearly seen in the figure that statistically significant differences between the values in the Subetta and placebo groups for at least 2 insulin concentrations are observed not only when the samples were added directly to the cell medium, but also in the case of a non-contact exposure, ie, when a suspension of dissolved samples was placed around the plate. Based on Figure 4, it is possible that the observed effect is concentration-dependent. However, due to the fact that there were only 4 concentration points and the sample size was small, no statistically significant relationship between the effect and concentration was found in this set of experiments. Comparison of 95% confidence intervals of the difference in the means individually for different concentrations of insulin and different types of exposure for all 10 pairs of experiments. Dots indicate the differences between the average glucose consumption by cells in the presence of a placebo or drug, lines indicate confidence intervals showing this difference with 95% probability. The y-axis shows insulin concentration in the media (0 μg/ml (0.ins), 0.5 μg/ml (0.5.ins), 5 μg/ml (5.ins), and 50 μg/ml (50.ins)).
Discussion
In 15 out of 20 experiments, the drug containing TPA to InsR-β increased the glucose consumption by cells, regardless of the type of interaction between solutions in test tubes. At the same time, the differences within the same experiment, but with different types of contact, for different concentrations of insulin, are probably related to complex interactions of various intracellular signaling pathways. The main cellular mechanism which is involved here is the insulin signaling pathway, which is directly related to the regulation of cell proliferation,.39–41 When exposed to the drug, cells consume glucose more actively and, as a result, divide more actively. Therefore, when calculating glucose consumption, normalized by the number of cells, the effect of the drug is not always obvious. However, the general tendency to increase glucose consumption is pronounced.
It should be noted that in most experiments, an increase in glucose consumption by cells is observed even in the absence of insulin (0 μg/ml), which allows us to suggest the sensitization of the target molecules (insulin receptors) by the drug, and the effect, which was demonstrated earlier for another TP drugs 42 and for Subetta in particular. 28 In other cases, differences from the control group were found for concentrations of 0.5 μg/ml, 5 μg/ml, and 50 μg/ml, which seems to be associated with an increase in the concentration of a glucose transporter protein (GLUT4) in the plasma membrane when insulin is added 43 , an indirect effect on which was previously shown for Subetta. 29
Moreover, in most experiments, there is a similarity in the shape of the curves of glucose consumption by cells when analyzing contact and non-contact effects within 1 experiment. Since the experiments were carried out under absolutely identical conditions (except for the type of exposure to the drug), this suggests the presence of a single physical mechanism of action of the drug in any type of its interaction with cell culture. It has previously been shown that TP antibodies obtained using serial dilution technology combined with vibration-mechanical treatment can emit in the radio and IR ranges15,16,25,26,44. Furthermore, there is evidence of the effect of TP antibodies on their target molecules23–25,27,44. It is still not quite clear what processes and/or structures of solution underlie the emergence of emission. This issue requires further comprehensive research at the level of physics and chemistry. In particular, during this study, it has not been possible to control for the geomagnetic background, season, lunar cycle, and magnetic fields. These are the factors that can potentially influence the results. Cell-to-cell variation can also be attributed to the limitations of the study. However, it can already be assumed that the presence and properties of nanoassociates can play an important role, since their formation and rearrangement in highly diluted aqueous solutions are known to determine their physical-chemical and biological properties3,45.
Conclusion
A series of experiments was conducted to investigate the effect of TP antibodies to the insulin receptor on a glucose consumption by CHO cells, both with and without contact exposure. The results showed a statistically significant difference in a glucose consumption compared to the control group (P < 0.05) in most of the experiments. The difference didn’t correlate with the added insulin concentrations. These results obtained in the cell culture model demonstrate the ability of TP antibodies to exert an effect on target molecules, realized both with contact and non-contact exposure.
We believe that the results of this study not only bring us closer to understanding the nature of HDs, but also demonstrate the advantages of TP drugs, which include not only safety, but also high specificity of effect and good bioavailability of the drug.
Supplemental Material
Supplemental Material - Biological Medicines Prepared Using Vibration Processing Are Able to Influence Their Targets Without Direct Contact With Them
Supplemental Material for Biological Medicines Prepared Using Vibration Processing Are Able to Influence Their Targets Without Direct Contact With Them by Elena Don, Svetlana Zakharova, Sabina Yaroshenko, Anastasia Petrova, and Sergey Tarasov in Dose-Response
Footnotes
Author Contributions
Conceptualization, E.D., A.P. and S.T.; methodology, S.Y. and S.Z.; writing—original draft preparation, E.D.; writing—review and editing, A.P., S.T.; supervision, S.T.; project administration, A.P. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest in connec-tion with the authorship and/or publication of this article: S. Zakharova, S. Yaroshenko, A. Petrova, E. Don and S. Tarasov are employees of OOO “NPF “MATERIA MEDICA HOLDING”, fully or partly. OOO “NPF “MATERIA MEDICA HOLDING” sponsored the study, performed statistical analysis, made a decision to publish the work, and covered the current article processing charges, took part in the design of the experiments and the manuscript writing. The TPA mentioned in the manuscript are the substance (single, or 1 among other components) for commercial drugs produced by OOO “NPF “MATERIA MEDICA HOLDING”. Commercial drugs Subetta, Divaza are produced by OOO “NPF “MATERIA MEDICA HOLDING”. Patents on this substance and the drugs belong to OOO “NPF “MATERIA MEDICA HOLDING”.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was funded by OOO “NPF “Materia Medica Holding”, Moscow, Russia. The employees of OOO “NPF “Materia Medica Holding” took part in the design of the experiments and the manuscript writing, performed statistical analysis, and made a decision to publish the work. This research received no additional external funding.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
