Abstract
The natural radiation background contributes to the dose of ionizing radiation received by the whole population. However, the telluric component of the natural background radiation is not homogenous on Earth: while the average effective dose has been estimated to be 2.4 mSv/year worldwide, certain regions are considered as high natural background radiation areas (HBRA). To investigate the specificities of a continuous exposure to low-dose-rate irradiation, we reviewed data of the major HBRA from 98 studies published between 1973 and 2023. Three conclusions were drawn: 1) the dose received by the HBRA inhabitants is much lower than values assessed on hot spots : at Ramsar (Iran), 260 mSv/year were assessed at the highest hotspots but the maximal estimated dose-rate received by inhabitants is 80 mSv/year; 2) when DNA or chromosome breaks, cancer or accelerated aging are used as endpoints, no significant difference was observed between cells from HBRA and non-HBRA inhabitants; 3) conversely, adaptive response effect was systematically observed on ex vivo lymphocytes from HBRA inhabitants when they are exposed to a high dose ranging for 0.25 to 4 Gy. A mechanistic model based on the radiation-induced nucleoshuttling of the ATM protein provides an explanation to these last two conclusions.
Keywords
Introduction
The natural background radiation is composed of cosmic rays, (neutrons and other particles) telluric radioactivity (uranium and thorium daughters including radon gas) and radioactivity from living things (notably potassium-40 and carbon-14). Thus defined, the natural background radiation significantly contributes to the dose of ionizing radiation (IR) received daily by the whole population. 1 In addition to the cosmic, telluric and the food and water components, a current increase of medical use of IR represents to date, mostly in developed countries, 1 about 20% of dose in excess to be considered in the total population exposure. However, an essential radiobiological difference between medical exposure and natural background radiation is the dose-rate : while the doses received in the medical context are generally delivered at high dose-rate (higher than 2 Gy/min in radiotherapy; higher than 20 mGy/min in CT scan; higher than 2 mGy/min in mammography exams during a short time 2 ), the natural background radiation consists in a low and continuous dose-rate of the order of nGy/min (here, the term “continuous” has been preferred to “chronic” since “chronic” may suggest some regular time intervals during which the exposure is nil).
While the cosmic component, that represents up to 20% of the contribution of the final dose, can be considered as constant, the telluric component of the natural background radiation is not homogenous on Earth: the calculated average effective dose is 2.4 mSv/year worldwide but certain regions are considered as high natural background radiation areas (HBRA). 3 This is notably the case of Ramsar (Iran), Kerala (India), or Changjiang (China).4-6 In these HBRA, the dose-rate can reach up to 260 mSv/year at certain physical spots.7,8 Let us remind that these values can exceed the annual dose limits authorized for workers (6 and 20 mSv/year for category B and A exposed workers, respectively). 9 We are however aware that it is difficult to compare natural background radiation doses with those that are received by workers since they are not delivered with the same scheme. However, in the Sievert system, both are based on the same principle of cumulation of the dose.
The exposure delivered at a very low and continuous dose-rate raises the question of the concomitance of the formation of radiation-induced (RI) DNA damage and the repair process. It also raises the question of a potential adaptive response through one or several generations and the potential risks of cancer or accelerated aging on exposed subpopulations. In order to investigate the molecular, cellular and clinical features that would be specific to these regions and to draw conclusions about the biological effects produced by low dose-rates, we have therefore surveyed the radiobiological data concerning the major HBRA from 98 studies published between 1973 and 2023.
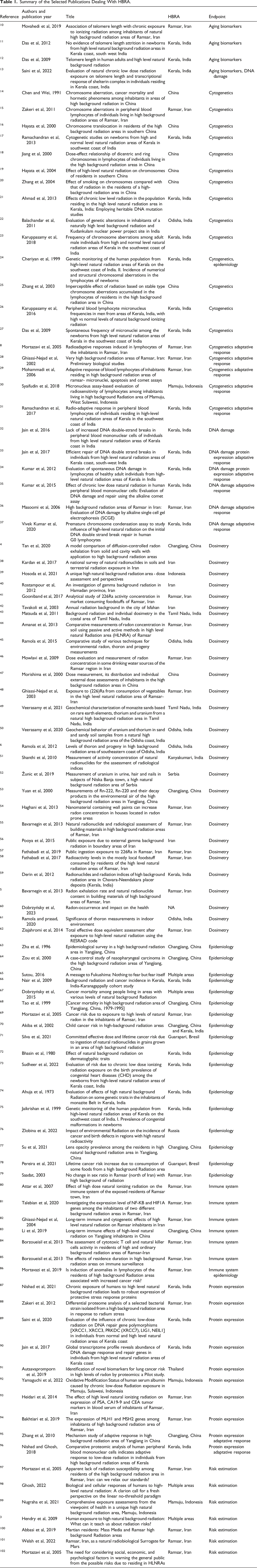
Summary of the Selected Publications Dealing With HBRA.
HBRA: Definitions and Average Dose Rates
We have analysed 98 peer-reviewed articles published in the 1973-2023 period. These papers revealed 6 major HBRA including Kerala (28% of reports), Ramsar (34%), and Changjiang (16%) (Table 1).
Ramsar (Iran)
Summary of the Radiobiological Features of the Major HBRA.

aNHBRA: non-HBRA.
bFrom dosimetry calculations from internal exposure from food and water intakes.
Kerala (India)
The HBRA of coastal Kerala, situated in the southwest India, presents wide variation in dose and dose-rates (ranging from less than 1 to 45 mSv/year; average 4 mSv/year) and a large size of its population. The Kerala HBRA is a narrow coastal line stretching from Neendakara panchayat (Kollam district) in the south to Purakkad panchayat (Allapuzzha district) in the north. The reason for this high natural radiation background level is the presence of uneven distribution of monazite and other heavy minerals such as ilmenite, rutile, zircon, garnet, etc. Monazite contains approximately 9% Th232 and 0.3% U238 in Kerala.106,113 The great variability of doses received by the population is also found in biological studies. Considering that the “control” group gathers individuals receiving less than 1.5 mSv/year, like in Ramsar, 3 groups of exposed individuals are generally defined in Kerala: the HBRA1 group of individuals receiving from 1.5 to 5 mSv/year; the HBRA2 group of individuals receiving from 5 to 15 mSv/year and the HBRA3 group of individuals receiving more than 15 mSv/year.31,35,73,90,96 For example, in the study of Kumar et al, the average dose-rate received by the “HBRA1 group” was 2.69 mSv/year and varied from 1.07 to 5.55 mSv/year. In most of the studies concerning Kerala, the groups of individuals labeled “HBRA” do not receive more than 2.4 mSv/year, the world average value1,114 (Tables 1 and 2).
Changjiang (China)
The Changjiang district is an administrative subdivision of the Jiangxi region situated at the east of China. There, the main sources of high-intensity natural background radiation are the nuclides of the decay products of Th232 and U238 present in the environment. 16 The average exposure dose-rate is 5.88 mSv/year (maximum 6.86 mSv/year). 110 Once again, a large variability in annual doses exists inside this district depending on the natural concentrations of radionuclides in construction materials, the age of the buildings and the layout of houses in the hamlets. There was a fairly wide and heterogeneous distribution of indoor and outdoor environmental radiation. In one study, for example, indoor radiation doses due to exposure to natural radionuclides present in building materials may be approximately twice higher than outdoor 47 and some studies consider the external exposure as well as the internal exposure, attributable to food and water intake. Thus, the annual dose varies drastically, depending on the age of the individuals and the season of the year. Indeed, since children and the elderly are more likely to stay indoors, the dose-rates are higher for these age groups. 47 The annual dose-rates for the HBRA groups vary from 1.0 to 45 mSv/year and have been found much lower than the maximal physical measures of the hottest geological spots. 23 Besides, the average dose received by the HBRA group individuals is very close to the average annual exposure worldwide (Tables 1 and 2).
Guarapari (Brazil)
The City of Guarapari, on the coast of Espırito Santo State, Brazil is also considered as an HBRA because of its soils naturally rich in monazite containing U238 and Th232. According to the measurements performed at different points of this city in 2002, the average external exposure is 2.5 mSv/year but higher dose-rates, particularly assessed on the beach, are up to 19 mSv/year 115 (Tables 1 and 2).
Mamuju, Sulawesi (Indonesia)
In the Mamuju HBRA in Indonesia, recently discovered by the Indonesian National Nuclear Energy Agency (BATAN), the annual effective dose-rate ranges from 17 to 115 mSv/year, with an average of 32 mSv/year. This exposure is due to high concentrations of K40, U238, Th232 and Ra226 and Rn222 their main radioactive decay products.92,99 Calculation of cumulative lifetime dose suggests that Mamuju residents could receive up to 2.2 mSv/year on average (Tables 1 and 2).
Odisha (India)
The coastal area of Odisha, in eastern India is a well-known HBRA area rich in monazites and rutiles. High levels of radiation are caused by the presence of large amounts of naturally occurring radioactive material in rocks, soils, sands and water. Three radionuclides, 40K, 238U, 232Th and their radioactive decay products, 226Ra and 222Rn, contribute significantly to these levels. 50 The annual inhalation dose-rate in homes in Odisha due to radon and its progeny, as well as thoron and its progeny, ranges from 0.11 to 2.67 mSv/year, with an average from 0.93 mSv/year, and from 0.42 to 11.33 mSv/year, with an average of 2.39 mSv/year, respectively. 61 In another study, the gamma dose rate was also measured in the indoor and outdoor environment of the area by using a portable gamma dosimeter. The outdoor dose rate averaged 2.35 mSv/year with a maximum of 5.43 mSv/year and the indoor dose rate averaged 3.14 mSv/year with a maximum of 7.9 mSv/year 18 (Tables 1 and 2).
HBRA: Analysis of the Biological Studies
One of the first conclusions of the above literature analysis of dose-rates values was that the dose effectively received by the HBRA inhabitants is much lower than the values assessed on hot spots (Tables 1 and 2). This is notably true for Ramsar HBRA with more than 260 mSv/year assessed at the highest hotspots while the maximal estimated dose-rate received by Ramsar inhabitants is 80 mSv/year (for a small subset). In the 63 peer-reviewed articles dealing with the biological consequences of an exposure to IR in HBRA, several biomarkers were applied, notably cytogenetics (28%), DNA damage (10%), protein expression (9%), immune system biomarkers (11%), aging biomarker (6%) and epidemiology and cancer incidence (36%). Some of the studies deal with two or more biomarkers. Among these studies, 28% are about adaptive response and 72% involved a single dose (Figure 1). Relative Proportions of the Different Biomarkers Established in the 98 Peer-Reviews Analysed. (A) In Direct ex Vivo Cells Studies. (B) Adaptive Response Studies vs Direct ex Vivo Cells.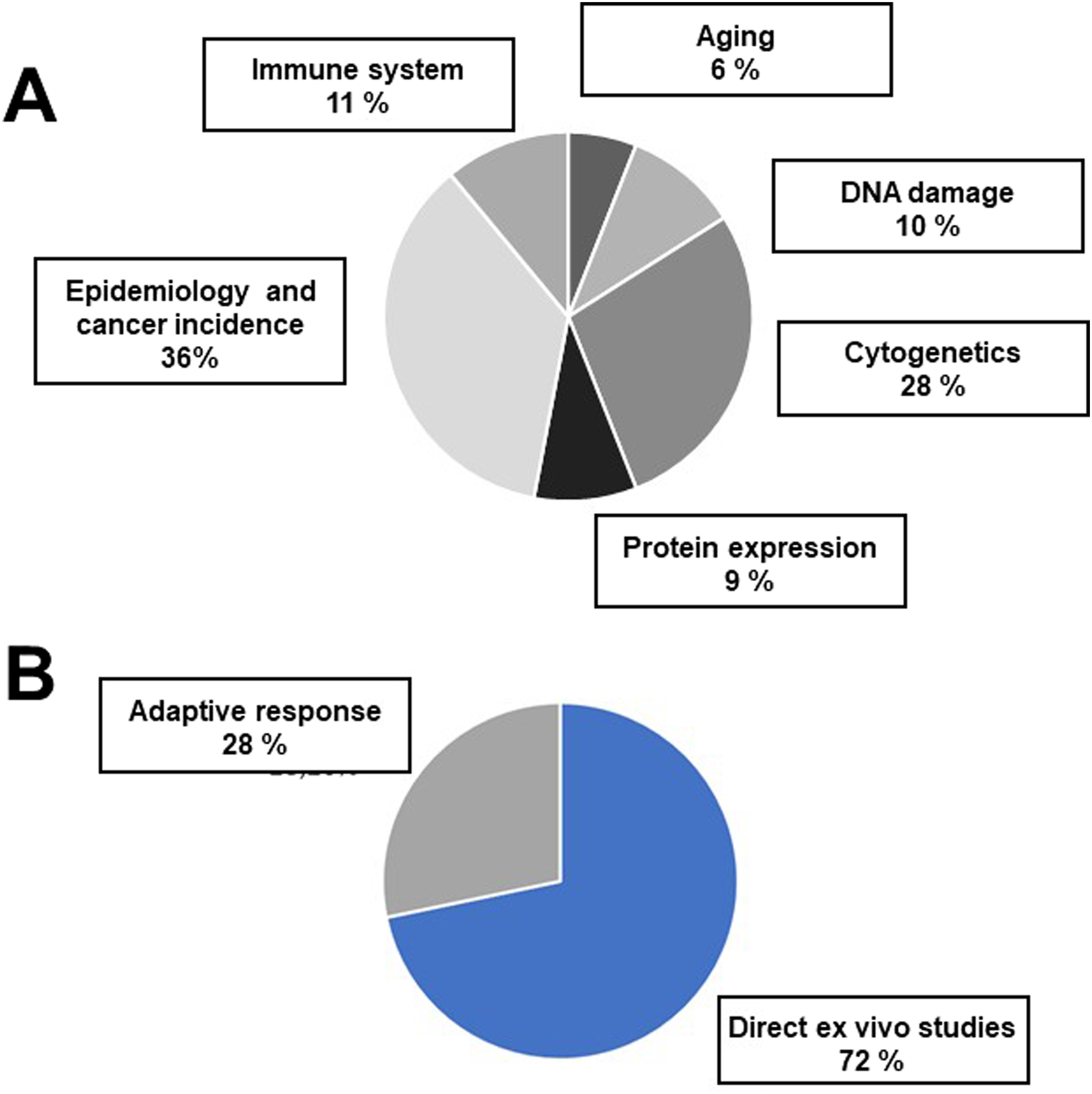
Cytogenetics
When chromosome aberrations or micronuclei are taken as endpoints, almost all studies (90%) did not reveal significant differences between the yield of cytogenetic events for HBRA and NHBRA individuals. Besides, the small subset of studies showing differences elicited discrepancies: all the conclusions drawn by the authors point to very small, rarely significant differences between the HBRA and NHBRA regions. For example, Ramachandran et al 17 showed no differences between the 27 295 studied newborns in Kerala HBRA and NHBRA areas, either through karyotype abnormalities or through the frequency of stable and unstable chromosomal aberrations. Considering micronuclei as endpoints, similarly, Das et al, 27 and Karupassamy et al, 26 did not observe any significant difference in basal micronuclei levels between newborns and the general population.
DNA Damage
When DNA damage were taken as endpoints, all studies showed generally the same amount of DNA strand breaks or less in cells derived from HBRA individuals than in NHBRA individuals. For example, by considering that immunofluorescence γH2AX foci reflects DNA double-strand breaks (DSB), Jain et al 32 showed a frequency of γH2AX foci equal to 0.078 ± 0.004 for HBRA inhabitants exposed to an average dose-rate of 11.04 ± 3.57 mSv/year, in comparison to the NHBRA population showing a frequency of 0.095 ± 0.009 γH2AX foci. 32
Immune System Biomarkers
When biomarkers of immune system were taken as endpoints, the conclusions were not consensual, with some change in the proportions of all the blood cell types studied except for a decrease in the CD107a-positive cell population among the HBRA inhabitants. Other studies, such as Li et al, 83 focusing on immune cell populations and biomarkers of inflammation, showed no difference among cell subtypes, to the exception of 5 markers of inflammation showing a slight increase (IFN-c, MCP-1, sIL-6R, EGFR, and CRP). 83
Aging Biomarkers
All studies concerning the length of telomeres concluded that no aging effect was observed after exposure to IR in HBRA. Regarding aging biomarkers, molecular investigations were performed only the Ramsar region in Iran and the Kerala region in India. It is noteworthy that the dosimetry data of these regions in these studies is not systematically provided in all the reports. 94 However, even though the methods of calculation or assessment of the dose were unequal among the selected studies, one can reasonably conclude that the exposure to IR in HBRA does not significantly accelerate aging effect.
Epidemiology and Cancer Incidence
Lastly, the great majority of reviewed studies did not show significant differences of cancer incidence between HBRA and NHBRA cohorts. In some epidemiological studies, researchers concluded to a decreased risk of leukemia-induced-death 70 and a negative correlation between lung cancer and radon exposure. 69 In addition to these negative correlations, which run against the fundamental assumption of an increased incidence of RI cancers or congenital malformations, several studies, such as Nair et al, 66 or Jaikrishan et al, 75 showed no correlation between cancer incidence or congenital malformations and exposure to IR in HBRA.
The Particular Case of Adaptive Response Experiments
The above analysis of the biological data suggests that there is no significant “HBRA effect” for a given cumulated dose received by ex vivo blood cells. However, 28% of the publications dealt with the adaptive response scenario that consisting in the succession of two IR doses, d and D separated by a period of time Δt, namely d Δt + D; with d < D).116,117 In the present cases, d illustrates the priming dose received during exposure to IR in HBRA by HBRA inhabitants i.e. the cumulated dose that directly depends on the time of exposure in HBRA (or in non-HBRA inhabitants for controls). Besides, historically, the first report related to the adaptive response originated from experiments with a dose (d = 0.1-0.01 µCi/ml for 2-3 days) delivered with tritium contamination, i.e. equivalent to a low and continuous dose rate exposure followed by a challenging dose (D = 1.5 Gy X rays) applied to ex vivo cells at high dose-rate.
118
Like in the study of Olivieri et al,
118
the adaptive response experiments analyzed in the HBRA reports were based on a dose d delivered at low and continuous (HBRA) dose-rate, a negligible Δt period of time and a challenging dose D delivered at higher dose-rate. For the great majority of cases, the authors sampled ex vivo lymphocytes from HBRA inhabitants exposed to a dose d and thereafter applied the challenging dose D on lymphocytes in vitro (Figure 2). Schematic Illustration of the Induction of the DSB and the ATM Monomers according to the Irradiation Scenario. After a Single Dose D, a Certain Number of DSB are Induced and Thereafter Repaired as a Function of Time. In Parallel, a Certain Number of ATM Monomers are Induced From ATM Dimers, Diffuse to the Nucleus and Trigger the DSB Repair. In the Case of an Adaptive Response Scenario (d + Δt + D), DSB and ATM Monomers Induced by the Priming Dose d Cumulate with Those Directly Induced by the Challenging Dose D. In the Case of an Exposure to a Single Dose Delivered at Low-Dose Rate, DSB and ATM Monomers Result From the Accumulation of Infinitesimal Number of DSB and ATM Monomers Induced Daily. In the Case of an Adaptive Response Scenario with Priming Dose Delivered at Low-Dose Rate, Resulting in Cumulated Doses Delivered at Low Dose-Rate, DSB and ATM Monomers Result From the Accumulation of Infinitesimal Number of DSB and ATM Monomers Induced Daily Cumulate with Those Induced by the Challenging Dose D.
Interestingly, 100% of HBRA studies involving adaptive response scenario were found positive, with the challenging dose D ranging from 0.25 to 4 Gy (Table 1). For example, according to Mortazavi et al, 8 lymphocytes from HBRA inhabitants showed less chromosomal aberrations (0.111 ± 0.003) than the cells from non-HBRA inhabitants (0.167 ± 0.004) after a dose D of 2 Gy. Hence, while single dose experiments performed in HBRA provide controversial data, the fact that adaptive response experiments performed in HBRA were found systematically positive raises an important question of radiobiology that is discussed below.
Discussion
Dose-Rate Received by HBRA Inhabitants is Lower than Those Assessed at High Spots
One of the first conclusions of the literature review concerning HBRA data is that the average dose-rate received by the HBRA inhabitants is much lower than those assessed at the hottest spots. One of the most representative examples of such statement is the case of Ramsar in which some spots may reach 260 mSv/year while the most exposed subgroups received 80 mSv/year on average (Tables 1 and 2). It is important to remind that the effective dose is intended for radiological protection applications and is not actually a real dose, as is also the case for equivalent dose which is needed for evaluating effective dose. Thus, to a given effective dose related to natural background radiation exposure, corresponds a different absorbed dose for each organ of the body. It is therefore very important to rigorously mention the effective dose received by HBRA inhabitants in the reports to avoid any confusion between maximal values assessed and average dose calculated or assessed.
How to Explain the Difference Between Single-Dose and Adaptive Response Experiments Data?
The data analysis performed in this report suggests that there is no significant “HBRA effect” for a given cumulated dose received by ex vivo blood cells (single-dose experiment) while the adaptive response experiments from ex vivo blood cells of HBRA inhabitants exposed to a challenging dose were found positive.
Crude Estimates of the Number of DNA Damage and ATM Monomers Induced by IR in NHBRA and HBRA.

With regard to the adaptive response scenarios, we have published in 2022 a paper that proposes a coherent biological interpretation of the adaptive response phenomenon from a model based on the RI nucleoshuttling of the ATM protein kinase (RIANS) model116,117 (Figures 2 and 3). Such interpretation is not based on the DNA damage but on the ATM monomers induced by IR. Briefly, after exposure to IR, the cytoplasmic ATM dimers dissociate into ATM monomers that diffuse in the nucleus. Once in the nucleus, ATM monomers phosphorylate the X variant of the H2A histone protein (γH2AX), which triggers the recognition of the RI DSB by the non-homologous end-joining (NHEJ) pathway, the most predominant DSB signaling and repair pathway in humans.119-121 However, the RIANS can be delayed by the overexpression of some cytoplasmic substrates of ATM (called X-proteins). These X-proteins hold SQ and TQ domains, specifically phosphorylated by ATM kinase after exposure to IR.
122
The X-proteins sequestrate the RI ATM monomers, decrease their flux in the nucleus and therefore affect the nuclear ATM kinase activity and the number of DSB recognized by NHEJ121,123,124 (Figure 3). An overexpression of cytoplasmic X-proteins and a delayed RIANS may cause radiosensitivity, RI cancer or RI aging.
125
For human radioresistant fibroblasts, about 40 DSB are induced per Gy per cell (the repair half-time of DSB is generally considered to be about 1 h). In the frame of the RIANS model, 106 to 108 ATM monomers are supposed to be emitted per Gy from cytoplasmic ATM dimers. The full ATM kinase activity is considered to be ensured for 24 h, at least.
126
From these numerical values, one can deduce that, in the frame of the RIANS model, doses lower than 25 mGy do not induce DSB but still produce tens of thousands ATM monomers per cell, at least. Let’s take a representative example. In the frame of the RIANS model, Ndiff, the number of ATM monomers that diffuse in the nucleus as a function of dose D and time-post-irradiation t obeys the following formula (1) for human radioresistant fibroblasts
63
: General Model for Adaptive Response Based on the RIANS Model. The Scenario d + Δt Produces a Significant Amount of Active ATM Monomers in the Nucleus before the Exposure to D. As a Result, More DSB are Recognized by Non-homologous End Joining (NHEJ) after d + Δt + D (Panel A) than after D (Panel B): Radiosensitivity and Radiosusceptibility Decrease. However, it is Noteworthy that Adaptive Response is Preferentially Observed in Radiosensitive (Group II) Cells. The Adaptive Response is Not Observed in Radioresistant (Group I) Cells since the Repair of DSB Induced by d + Δt is Complete.


The following conditions were taken: 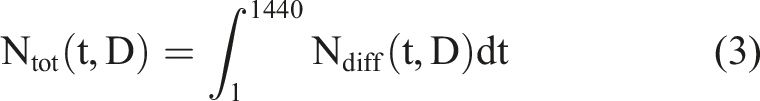
The numerical values obtained form this formula and shown in Table 3 confirmed that: - The numbers of the different biochemical type of DNA damage induced and remaining are negligible whether after an exposure to HBRA or to non-HBRA; - The quantity of ATM monomers accumulated in nucleus was found systematically much higher than the number of DNA damage; - When cells are exposed to HBRA, they provide a significant amount of ATM monomers (more than 106 per day) that can help in recognizing more DSB when exposed to a challenging dose. Such additional flux of ATM monomers may represent some additional percents of ATM monomers to those produce directly by the challenging dose. After exposure to non- HBRA, such flux is negligible.
Interestingly, a recent study published by Bugala and Fornalski reached similar conclusions about the radiation adaptive response for constant dose-rate irradiation in HBRA from another mathematical model. 127
Conclusions
The review of 98 studies published between 1973 and 2023 and dealing with HBRA suggest two conclusions: 1) the doses effectively received by the HBRA inhabitants are much lower than the values assessed on hot spots. This is notably true for Ramsar HBRA with more than 260 mSv/year assessed at the highest hotspots while the maximal estimated dose-rate received by Ramsar inhabitants is 80 mSv/year, which encourages authors to mention more precisely the dosimetry in the HBRA studies; 2) cells from HBRA inhabitants did not show consensual data when DNA damage, chromosome breaks, cancer or accelerated aging biomarkers were used as parameters; 3) a systematic adaptive response was observed in HBRA inhabitants when a second high dose was applied to ex vivo to lymphocytes. Indeed, the number of RI DNA damage appears to be too low to provide any significant “HBRA effect”. Conversely, HBRA exposures yield a significant and constant number of ATM monomers that may help cells in replying to additional doses. A mechanistic model based on the RI nucleoshuttling of the ATM proteins (the RIANS model) may provide an explanation for this last conclusion. Obviously, further experiments are needed to document the model proposed in this particular HBRA configuration.
Footnotes
Acknowledgements
J.R-V thanks the Centre National d’Etudes Spatiales (CNES) and Neolys Diagnostics for their financial support (PhD grant). All the authors have confirmed that the listed authors have authorized the submission of their manuscript via third party and approved any statements or declarations, notably the absence of conflict of interest and the list of funders.
Statements and declarations
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Commissariat General à l’Investissement (Programmes Investissement d’Avenir (INDIRA Project) and the Centre National d’Etudes Spatiales (ICARE Project).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
