Abstract
Keywords
Introduction
Myocardial infarction (MI) is the most severe manifestation of coronary artery disease, affecting over 7 million people globally each year.1-3 MI includes acute myocardial infarction (AMI), which is further classified into ST-segment elevation myocardial infarction (STEMI) and non-ST-segment elevation myocardial infarction (NSTEMI).4,5 AMI is associated with high mortality and poor prognosis, with heart failure being a leading cause of death. 6 Multimorbidity, defined as the presence of two or more chronic health conditions, is common among MI patients, significantly increasing the risk of mortality both during hospitalization and long-term, posing challenges to healthcare systems.7,8 Despite advancements in treatment, patients with congestive heart failure (CHF) continue to face a poor prognosis, with a 5-year mortality rate of 40%–50%.9-12 MI and heart failure often co-occur, exacerbating adverse outcomes, reducing quality of life, and lowering survival rates.13,14 Therefore, investigating MI in patients with heart failure is critical for improving outcomes and identifying potential risk factors.
Researchers are increasingly focusing on simpler, more accessible vital signs. Respiratory rate, measured as breaths per minute, is regulated by neurons in the medulla oblongata and is one of the four key vital signs used to evaluate a patient’s clinical condition.15-17 Studies have linked respiratory rate to long-term mortality in specific populations. For instance, one study showed that mean nocturnal respiratory rate, assessed via polysomnography, was independently associated with all-cause mortality in older adults over 6- and 9-year follow-up periods. 18 Another study on MI inpatients found an independent association between respiratory rate and all-cause mortality over a 5-year follow-up. 19 Additionally, research on acute coronary syndrome identified nighttime respiratory rate as a predictor of mortality. 20 However, the relationship between respiratory rate and mortality in MI patients with concurrent heart failure remains underexplored.
This study aims to investigate the relationship between low and high respiratory rates and in-hospital mortality in a large cohort of American adults admitted to intensive care for AMI and CHF.
Method
Data Source
This quantitative cohort study was conducted in Turkey between November 2023 and February 2024, using data from the publicly accessible Medical Information Mart for Intensive Care IV (MIMIC-IV) database, a collaboration between Beth Israel Deaconess Medical Center and the Laboratory for Computational Physiology at MIT.21-23 The data, spanning 2008-2019, were downloaded from https://mimic.physionet.org/. MIMIC-IV contains comprehensive clinical information, including patient demographics, laboratory results, clinical diagnoses, and medical records.
24
To access MIMIC-IV version 2.0, we completed the “Protecting Human Research Participants” exam after a National Institutes of Health (NIH) training course, with certification issued to the first author, Kai Zhang (Certification Number: 11639604). The database use was approved by the Institutional Review Boards of Beth Israel Deaconess Medical Center (2001-P-001699/14) and MIT (No. 0403000206), and patient consent was waived due to the retrospective and de-identified nature of the data.25-28 This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. A total of 16,012 patients were initially enrolled, but exclusions were made for those under 18 (n = 12), lacking outcome data post-ICU admission (n = 17), or without a diagnosis of AMI (n = 10,926). Figure 1 details the screening process. Flowchart of patient selection.
Study Population and Data Extraction
Data extraction was performed using PgAdmin PostgreSQL (v10.18, Bedford, USA) and Navicat Premium (v15.0.12). Diagnosis information was retrieved from the ‘diagnoses_icd’ and ‘d_icd_diagnoses’ tables. Acute myocardial infarction (AMI) and congestive heart failure (CHF) were identified based on the International Classification of Diseases (ICD). CHF was defined according to the ACC/AHA guidelines as clinical heart failure (stages C or D), 29 while AMI was defined based on the Third Universal definition of Myocardial Infarction. 30 ICD-9-CM codes (before 2015) and ICD-10-CM codes (after 2015) were used to identify CHF and MI from primary inpatient diagnoses. Specific codes are provided in the supplementary table (Table S1). Patients without ICU records were excluded, as vital signs were available only for ICU admissions. To avoid data duplication, only the first ICU admission was included.
Expose and Outcome
The main exposure variable of this study was the respiratory rate. Respiratory rate, as the core variable, is mainly detected automatically by machines and recorded electronically to ensure the objectivity of data collection. 31 Respiratory rate measurement at baseline was taken following a 15 min passive supine position rest. Participants were asked to remain silent and still during the measurements. The database only records patients’ data after admission, so we used the first respiratory rate level on the hospitalized days as the baseline respiratory rate. The recorded initial respiratory rate was treated as a continuous variable, and patients were categorized into three groups based on their respiratory rate tertiles on the first day of ICU admission: Q1 (≤17 breaths per minute), Q2 (17-20 breaths per minute), and Q3 (>20 breaths per minute). Our primary outcome of interest was in-hospital mortality, identified as a binary indicator variable in the discharge records. ICU mortality was exclusively determined during the first ICU admission.
Covariates
Informed by prior literature and clinical insights, we identified the following covariates: (1) Demographic variables encompassing sex, age, and race; (2) Comorbidities, specifically chronic obstructive pulmonary disease (COPD), diabetes, hepatic failure (Hep F), Melanosis coli (MC), and diabetes; (3) Medical procedures, including ventilation and intubation; (4) Medication usage, consisting of norepinephrine, dopamine, epinephrine, phenylephrine, and vasopressin; (5) Basic vital signs such as temperature, heart rate, and systolic blood pressure (SBP); (6) Blood biochemical indicators, encompassing anion gap (AG), blood urea nitrogen (BUN), chloride, creatinine, hemoglobin (Hb), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), platelet count, potassium, sodium, red blood cell distribution width (RDW), red blood cell (RBC) count, and white blood cell (WBC) count; and (7) Acute Physiology Score III (APSIII) and Sequential Organ Failure Assessment Score (SOFA). Due to the fact that Severity scores can reflect the state of a disease and are closely related to respiratory rate and mortality, this article includes two common Severity scores in the MIMIC database, including Acute Physiology Score III (APSIII) and Sequential Organ Failure Assessment Score (SOFA), as variables for consideration.
Statistical Analyses
First, continuous variables were summarized as mean ± standard deviation (SD) or median. Normality and homogeneity of variance determined the choice between t-tests and Mann-Whitney U tests for analysis. Categorical variables were expressed as case numbers (percentage) and analyzed using the chi-square test (or Fisher’s exact method).
Second, to mitigate confounding effects, multivariate regression analysis was employed to elucidate the relationship between respiratory rate and outcome variables. Model 1 included no adjustments. Model 2 adjusted for age, gender, and race, while Model 3 further adjusted for comorbidities (COPD, diabetes, MC, HepF). Model 4 extended adjustments to medical procedures (Vent, Intubated), medication usage (Norepinephrine, Dopamine, Epinephrine, Phenylephrine, Vasopressin), basic vital signs (Temperature, Heart Rate, SBP), blood biochemical indicators (AG, BUN, Chloride, Creatinine, Hb, MCH, MCHC, MCV, Platelet, Potassium, Sodium, RBC, RDW, WBC). Model 5 incorporated additional adjustments based on Model 4, including APSIII and SOFA. Variance inflation factor (VIF) assessed multicollinearity (threshold: 5), with highly collinear variables eliminated to prevent overfitting.
Furthermore, we evaluated the non-linear relationship between respiratory rate and in-hospital mortality using smooth curve fitting (penalized spline method) and restricted cubic spline regression. The inflection point was identified via two-piecewise logistic regression and a recursive algorithm, with statistical significance set at
Finally, to enhance the analysis’s reliability, we conducted subgroup analyses to test for sensitivity, utilizing interaction tests to detect effect differences across subgroups.
All statistical analyses were performed using R software (version 4.1.1) and Free Statistics software (version 1.7). We used multiple imputation with chained equations to impute values for missing data. Free statistics software is a statistical software based on R language for data analysis, which has been widely used in multiple previous studies.32-34 Two-sided probability values ≤0.05 were considered statistically significant. This cross-sectional study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines.
Results
Baseline Characteristics of Selected Participants
Characteristics of the Study Population (N = 5056).
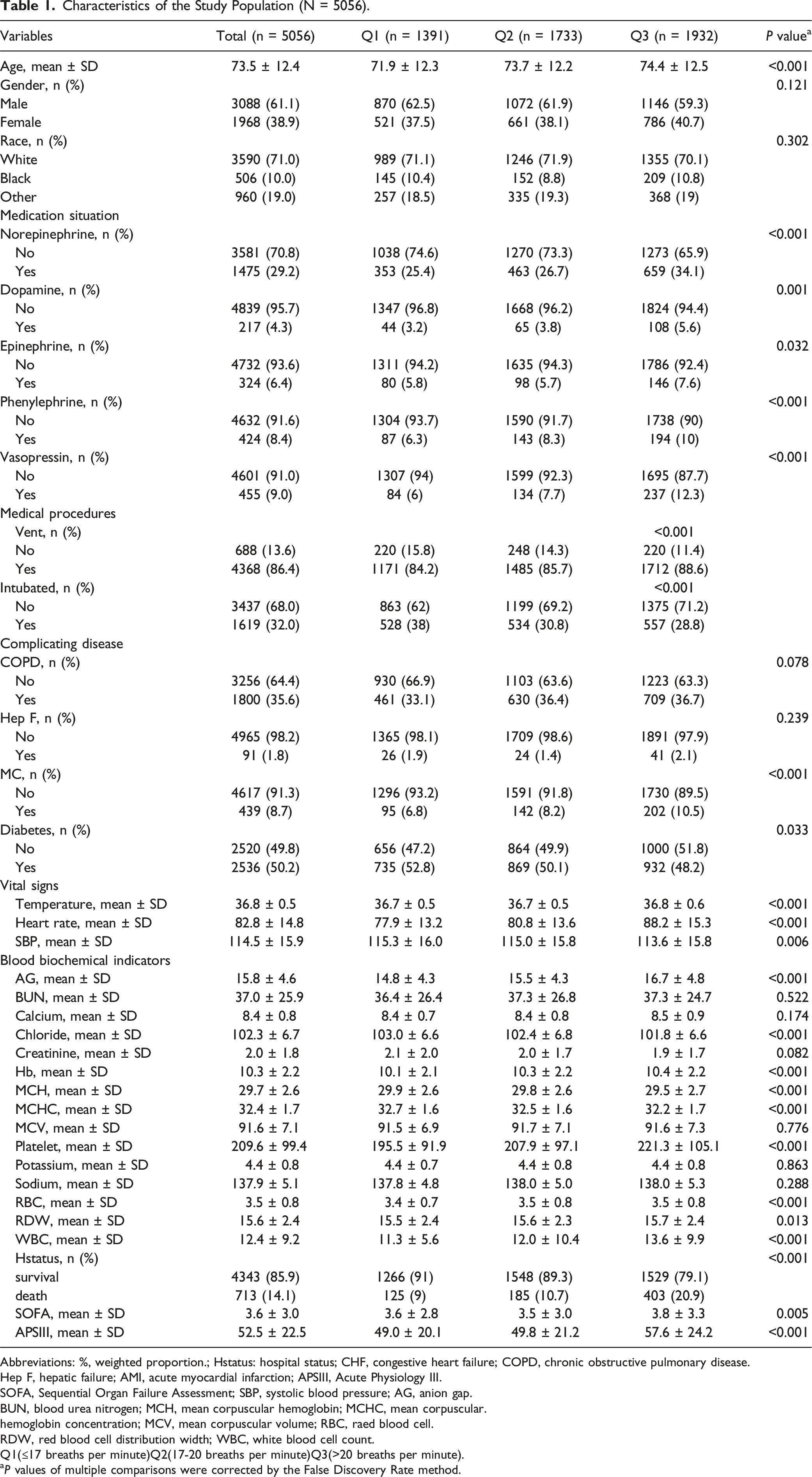
Abbreviations: %, weighted proportion.; Hstatus: hospital status; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease.
Hep F, hepatic failure; AMI, acute myocardial infarction; APSIII, Acute Physiology III.
SOFA, Sequential Organ Failure Assessment; SBP, systolic blood pressure; AG, anion gap.
BUN, blood urea nitrogen; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular.
hemoglobin concentration; MCV, mean corpuscular volume; RBC, raed blood cell.
RDW, red blood cell distribution width; WBC, white blood cell count.
Q1(≤17 breaths per minute)Q2(17-20 breaths per minute)Q3(>20 breaths per minute).
a
Furthermore, compared to individuals with lower respiratory rates, those with higher respiratory rates exhibited several notable characteristics, including older age, increased levels of No epinephrine, Dopamine, Phenylephrine, Vasopressin, and Vented cases, as well as a decreased Intubated rate. Additionally, there was a higher prevalence of colon melanosis, smaller diameters, elevated body temperature, increased heart rate, reduced SBP, higher AG, lower blood chlorine, higher Hb, decreased MCH, lower MCHC, elevated platelet count, higher RBC count, increased RDW, and elevated WBC count. These individuals also had a higher mortality rate and APS score.
Association Between Respiratory Rate and In-Hospital Mortality in Acute Myocardial Infarction Patients with Congestive Heart Failure
Multivariable Logistic Regression to Assess the Association of Respiratory Rate With In-Hospital Mortality Rate.

Abbreviations: the unit of Respiratory Rate is breaths per minute, %, weighted proportion. CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; HepF, hepatic failure; AMI, acute myocardial infarction; APSIII, Acute Physiology III; SOFA, Sequential Organ Failure Assessment; SBP, systolic blood pressure; AG, anion gap; BUN, blood urea nitrogen; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular.
hemoglobin concentration; MCV, mean corpuscular volume; RBC, red blood cell; RDW, red blood cell distribution width; WBC, white blood cell count.
CI: confidence interval;OR: odds ratios, Ref: reference.
Model 1: No adjustment.
Model 2: Adjusted for demographic variables(sex, age, race).
Model 3: Adjusted for demographic variables, comorbidities (COPD, diabetes, MC, HepF).
Model 4: Adjusted for demographic variables, comorbidities, Medical Procedures(Vent, Intubated), Medication situation(Norepinephrine Dopamine Epinephrine Phenylephrine Vasopressin),Basic vital signs(Temperature Heart Rate SBP),Blood biochemical indicators(AG BUN Chloride Creatinine, Hb MCH MCHC MCV Platelet Potassium Sodium RBC RDW WBC).
Model 5: Adjusted for demographic variables, comorbidities, Medical Procedures, Medication situation, Basic vital signs, Blood biochemical indicators, APSIII, SOFA.
Threshold Effect Analysis of Relationship of Respiratory Rate With In-Hospital Mortality Rate.

Adjusted for demographic variables (sex, age, race), Concomitant disease(COPD, diabetes, MC, Hep F), Medical Procedures(Vent, Intubated), Medication situation(Norepinephrine Dopamine Epinephrine Phenylephrine Vasopressin),Basic vital signs(Temperature Heart Rate SBP),Blood biochemical indicators(AG BUN Chloride Creatinine Hb MCH MCHC MCV Platelet Potassium Sodium calcium RBC RDW WBC), APSIII, SOFA.
Abbreviations: %, weighted proportion. CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease.
Hep F, hepatic failure; AMI, acute myocardial infarction; APSIII, Acute Physiology III.
SOFA, Sequential Organ Failure Assessment; SBP, systolic blood pressure; AG, anion gap.
BUN, blood urea nitrogen; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular.
hemoglobin concentration; MCV, mean corpuscular volume; RBC, red blood cell; RDW, red blood cell distribution width; WBC, white blood cell count.
CI: confidence interval; OR: odds ratios, Ref: reference.
Dose–Response Relationships
In this investigation, we employed restricted cubic spline models (Figure 2) to explore nonlinear associations. Our results unveiled a J-shaped relationship between respiratory rate and in-hospital mortality in patients diagnosed with congestive heart failure, while accounting for potential confounding variables. Below a threshold of 19 breaths per minute, we observed a negative correlation between HR and the risk of in-hospital mortality (OR = 0.902 [95% CI 0.787∼1.035], Dose–Response Relationships between Respiratory Rate with In-hospital mortality rate odds ratio.
Subgroup Analysis
Subgroup analyses were conducted to assess potential effect modifications on the relationship between respiratory rate and in-hospital mortality in several subgroups. No significant interactions were detected in any of the subgroups when stratified by age, sex, race, dopamine, epinephrine, vasopressin, COPD, MC, and diabetes (see Supplemental Figure). It should be noted that, considering multiple testing, a
Discussion
This study represents the pioneering exploration of the association between respiratory rate and in-hospital mortality risk in patients diagnosed with Acute Myocardial Infarction (AMI) and Congestive Heart Failure (CHF). Specifically, our investigation focuses on discerning a J-shaped relationship between respiratory rate and in-hospital mortality among AMI and CHF patients admitted to the Intensive Care Unit (ICU), pinpointing an optimal respiratory rate of approximately 19 breaths per minute. This research contributes a novel, straightforward, and efficacious basic vital sign indicator for the early identification of mortality risk in AMI and CHF patients within the ICU.
A substantial body of evidence indicates that aberrant respiratory responses serve as prognostic indicators in cardiac diseases.35,36 Quantification of these responses can be achieved through various stimuli. Patients displaying the most abnormal ventilatory responses during exercise stress testing experience an unfavorable prognosis, even among those with preserved exercise capacity. 36 Aberrant responses can also be elicited without physical exertion, triggered by chemoreflex stimulation that escalates ventilation in response to rising carbon dioxide levels or declining oxygen levels. Nevertheless, despite compelling evidence from reflex measurements, these tests are unlikely to integrate into routine clinical practice for post-Myocardial Infarction (MI) patients due to the requisites of specialized expertise, equipment, and patient cooperation. Consistent with us, a prospective cohort study suggests that respiratory rate provides strong prognostic information that is independent and can complement existing risk assessments. A simple and inexpensive assessment of respiratory rate should be considered as a supplementary indicator for risk assessment after acute myocardial infarction. 19 Another study focusing on acute coronary syndrome suggests that nighttime respiratory rate is a predictive indicator of mortality in patients with acute coronary syndrome. 20 However, we are the first study to investigate the association between respiratory rate and mortality in a population with acute myocardial infarction and heart failure. Our investigation extends prior research by exploring the association between respiratory rate levels, both high and low, and in-hospital mortality in hospitalized patients with AMI and CHF in the United States.
To investigate the non-linear relationship between respiratory rate and in-hospital mortality among individuals with heart failure, this study employed smooth curve fitting and generalized additive models. Subgroup analyses were conducted to assess primary findings’ consistency. The association between respiratory rate and in-hospital mortality exhibits a J-shaped pattern, signifying an inflection point at 19 breaths per minute. When the respiratory rate is less than the threshold of 19 breaths, the curve shows a decrease in mortality as the respiratory rate increases. This may be due to further improvement in ventilation, increased oxygen concentration, improved systemic metabolism, improved physical condition, and decreased mortality in clinical practice. In addition, low RR may be related to mental state, sedation, and mechanical ventilation, which requires further research in the future. These results suggest the potential benefit of maintaining a stable respiratory rate around 19 breaths per minute for congestive heart failure (CHF) and acute myocardial infarction (AMI) patients in the ICU. These findings underscore the importance of monitoring respiratory rate vigilantly in clinical practice, considering both low and high levels, and implementing timely interventions to enhance patient prognosis. Forced vital capacity (FVC) and FEV1, key indicators of pulmonary function, are established predictors of mortality, regardless of smoking habits, not only in cardiovascular disease but also in all-cause mortality.37,38 Furthermore, disease-specific studies focusing on interstitial pneumonia 39 and cystic fibrosis, 40 which are lung conditions associated with declining pulmonary function, have demonstrated a negative correlation between pulmonary function and respiratory rate. Counting respiratory rate is a simple, cost-effective method that does not require specialized equipment and can be applied across medical and care settings. Conversely, conducting pulmonary function tests can be challenging and potentially harmful, particularly for ICU patients, as they necessitate maximal effort from patients.41,42 Moreover, advanced wearable devices for automatic respiratory rate detection have recently emerged, 43 despite facing technical and cost-effectiveness challenges. As such devices become more prevalent, respiratory rate information will likely become more accessible in the near future. 43 Consequently, we can conclude that respiratory rate may serve as an alternative measure to pulmonary function in assessing mortality risk among ICU patients. Our study provides a new perspective on potential targets for maintain an appropriate respiratory rate at the management of ICU. Respiratory rate, as one of the most common vital indicators in ICU patients, maintaining an appropriate range is of great significance in improving prognosis. We believe that this strategy is feasible and promising and deserves the attention of clinicians.
The J-shaped relationship between respiratory rate and in-hospital mortality in myocardial infarction (MI) patients with heart failure likely reflects multiple physiological and pathological mechanisms. For low respiratory rates, slow or deep breathing activates the parasympathetic nervous system, increasing vagal tone, which reduces heart rate and blood pressure, enhancing cardiac oxygen supply and decreasing the risk of arrhythmias and MI, thereby lowering mortality. Low respiratory rates also improve heart rate variability (HRV), a marker of cardiac adaptability, which is associated with reduced mortality. Furthermore, slower breathing enhances alveolar ventilation, improving oxygen exchange and arterial oxygen saturation—critical for cardiovascular function. Additionally, slow breathing reduces chronic inflammation and oxidative stress, key drivers of atherosclerosis and cardiovascular disease progression. In contrast, high respiratory rates, such as rapid shallow breathing, are linked to sympathetic overactivation, leading to elevated heart rate, blood pressure, and vasoconstriction, which increases cardiac workload and oxygen consumption. This heightened strain raises the risk of arrhythmias and acute myocardial ischemia, contributing to higher mortality. High respiratory rates also correlate with reduced HRV, indicating impaired autonomic regulation and increased susceptibility to stress-induced events like sudden cardiac death and heart failure. Moreover, rapid breathing can cause respiratory alkalosis, leading to coronary artery spasm, decreased oxygen supply, and arrhythmias, further elevating mortality risk. Lastly, high respiratory rates may promote inflammation and oxidative stress, accelerating atherosclerosis and worsening cardiovascular outcomes.
The employed methodology in this study offers notable advantages. Firstly, this study represents the inaugural effort to analyze the relationship between respiratory rate and mortality in patients with Acute Myocardial Infarction (AMI) and Congestive Heart Failure (CHF) utilizing the MIMIC-IV database. Secondly, given the retrospective nature of cohort studies, the presence of confounding factors necessitates consideration. To mitigate their influence inherent in observational studies, logistic regression analysis was employed with multiple models, and subgroup analyses were conducted with appropriate categorization. Thirdly, the non-linearity of the study was assessed through the use of a smooth curve fitting diagram, revealing consistent outcomes. Lastly, it is important to emphasize that no patients incurred harm or violation throughout the course of this investigation.
Several limitations should be acknowledged. First, as a retrospective study, it is subject to inherent limitations, including a lower level of evidence compared to prospective studies, underscoring the need for future validation through prospective case-control studies. Second, despite adjusting for as many variables as possible, unmeasured factors and residual confounding may still influence the results. Third, the inclusion of only American participants limits the generalizability of our findings to other populations. Lastly, potential sources of bias, particularly those related to differential errors in measuring disease or exposure, must be considered. Future research will utilize our own database.
Conclusions
Our population‐based cohort study provided the epidemiologic evidence that myocardial infarction patients complicated by congestive heart failure have a J‐shaped association between respiratory rate and in-hospital mortality. Clinicians should not ignore that respiratory rate is a main factor associating risk for mortality in patients. Additional studies are warranted to confirm our findings in prospective cohorts and to elucidate the potential mechanisms underlying the relationship.44,45
Supplemental Material
Supplemental Material - J-Shaped Association Between Respiratory Rate and In-Hospital Mortality in Acute Myocardial Infarction Patients Complicated by Congestive Heart Failure in Intensive Care Unit
Supplemental Material for J-Shaped Association Between Respiratory Rate and In-Hospital Mortality in Acute Myocardial Infarction Patients Complicated by Congestive Heart Failure in Intensive Care Unit by Kai Zhang, Yu Shi, Yu Han, Tian Yi Cai, Fang Ming Gu, Zhao Xuan Gu, Tianqi Zhang, and Mao Xun Huang in Dose-Response.
Footnotes
Acknowledgments
We appreciate Dr Jie Liu of the Department of Vascular and Endovascular Surgery, Chinese PLA General Hospital for statistics, study deign consultations and editing the manuscript.
Author Contributions
KZ contributed as First authors of this manuscript. YH, FMG, ZXG and RZ were responsible for the concept and design of the study. TQZ and TYC explain the analysis. KZ and MXH are responsible for data recovery. MXH is the primary corresponding author. All authors critically revised the important intellectual content of the paper and approved the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the Jilin Provincial Health Talent Special Fund (Project No. 2024WSZX-D18).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
