Abstract
Isoproterenol (ISO), a chemically synthesized catecholamine, belongs to β-adrenoceptor agonist used to treat bradycardia. The β-adrenergic agonist is an essential regulator of myocardial metabolism and contractility; however, excessive exposure to ISO can initiate oxidative stress and inflammation. This study aims to investigate the molecular mechanisms underlying ISO-induced cardiac remodeling, the protective efficacy of resveratrol (RSVR), and its liposomal formulation (L-RSVR) against such cardiac change. Wistar albino rats were evenly divided into 4 groups. Control group, ISO group received ISO (50 mg/kg, s.c.) twice a week for 2 weeks, and RSVR- and L-RSVR-treated groups in which rats received either RSVR or L-RSVR (20 mg/kg/day, p.o.) along with ISO for 2 weeks. ISO caused a significant elevation of the expression levels of BAX and MEF2 mRNA, S100A1 and cytochrome C proteins, as well as DNA fragmentation in cardiac tissue compared to the control group. Treatment with either RSVR or L-RSVR for 14 days significantly ameliorated the damage induced by ISO, as evidenced by the improvement of all measured parameters. The present study shows that L-RSVR provides better cardio-protection against ISO-induced cardiac injury in rats, most likely through modulation of cardiac S100A1 protein expression and inhibition of inflammation and apoptosis.
Introduction
Isoproterenol (ISO) is a catecholamine chemically synthesized, 1 and it belongs to the β-adrenergic receptor agonist used to treat bradycardia conditions and cardiac arrest. 2 The β-adrenergic agonist is an essential regulator of myocardial metabolism and contractility 3 ; likewise, catecholamine has many therapeutic uses in maintaining blood pressure and normal cardiac function. However, when catecholamines are produced in excess, their oxidative metabolism creates quinines, which react with oxygen to form reactive oxygen species (ROS) that lead to changes in the structure, function, and metabolic characteristics of the myocardium. 4 Excessive exposure to ISO can initiate the release of inflammatory cytokines such as interleukin-6 and 1β (IL-6 and IL-1β) and tumor necrosis factor- α (TNF-α) 5 ; additionally, the levels of apoptotic and necrotic markers were increased in rats exposing to a single subcutaneous dose of ISO causing myocardial cell damage. 6 Therefore, we hypothesized that using an antioxidant would be a promising approach to prevent ROS generation and protect against cardiac injury induced by ISO.
Resveratrol (RSVR) is a natural polyphenol found in many plant species, especially grapes. 7 RSVR has various properties, including antioxidant, anti-inflammatory, antiapoptotic, and anti-tumor effects.8-10 Besides that, RSVR has been shown in preclinical research to have protective benefits against numerous diseases, such as diabetes, cancer, neurological disorders, and cardiovascular disease.11,12 The antioxidant activity of RSVR exerts through decreasing ROS production and maintaining mitochondria function.13,14 Additionally, RSVR upregulates γ-glutamylcysteine synthetase by activating Nrf2 and the rate-limiting enzyme for glutathione (GSH) synthesis. 15 Consistently, RSVR increases endothelial GSH content. 16
The cardioprotective effects of RSVR are demonstrated by inhibition of caspase-3 activity and expression, downregulation of TGF‐β1 expression, and reduction of collagen fiber deposition. 17 Previous studies show significant oxidative stress reduction and upregulation of iNOS mRNA expression result in decreased cardiomyocyte apoptosis. 18 Kaga et al showed that RSVR has cardioprotective effects in vivo and in vitro through increased heme oxygenase (HO-1), vascular endothelial growth factor (VEGF), and increased induction of thioredoxin (Trx-1). In a myocardial infarction (MI) rat model, phosphorylation of Akt mediates the translocation of catenin, a crucial mediator during angiogenesis and development, into the nucleus, where it increases the expression of VEGF, Bcl2, and survival while decreasing the number of cardiomyocytes apoptosis. 19
Although the RSVR is beneficial in cardioprotective effects, its use is limited due to less favorable pharmacokinetic properties. In addition, RSVR was demonstrated to have low water solubility and extensively metabolized in the liver and intestine leading to poor oral bioavailability. 20 Liposome formulation-based drug delivery systems can overcome these constraints and improve RSVR bioavailability. This study aims to evaluate the molecular mechanisms of L-RSVR compared to RSVR in ISO-induced cardiac remodeling by measuring markers of heart injury and inflammation in serum and assessing the expression of apoptosis-associated genes, B-cell lymphoma 2 (Bcl2), Bcl2 associated X-protein (BAX), and myocyte enhancer factor 2 (MEF2); besides the expression of the cardiac performance proteins S100 calcium-binding protein (S100) and cytochrome C.
Materials and Methods
Chemicals
RSVR raw powders were obtained from Sigma Chemical Co (St. Louis, MO, USA), while ISO ampoules were obtained from a local pharmacy. L-RSVR was purchased from Lipolife, Drakes Lane Industrial Estate, UK. The manufacturer has characterized this formulation as liposome bubbles, in which RSVR is encapsulated. These liposomes have a particle size of less than 200 nm with neutral Zeta potential. RSVR and L-RSVR were dissolved in 1 % carboxymethylcellulose (CMC).
Animals and Treatments
A total of thirty-two adult male Wister rats weighing between 150 and 180 grams were used in this study. The rats were obtained from the Animals Care Centre at the College of Pharmacy, King Saud University, Riyadh, Saudi Arabia. A week before the procedure work began, animals were allowed to adapt to the laboratory conditions. They were kept at a temperature (25 ± 2) with 12 hours of light and dark cycle and provided with food and water. Research protocol approved by King Saud University’s Scientific Ethics Committee (KSU-SE-22-111) on 29/01/2023.
After 1 week of acclimation to laboratory conditions, rats were randomly assigned into 4 groups (8 rats/group). Normal control group (1): rats were given 1% CMC (p.o.); ISO group (2): rats received a subcutaneous dose of ISO (50 mg/kg) twice a week for 14 days; RSVR group (3): rats were treated daily with RSVR (20 mg/kg, p.o.). 22 L-RSVR group (4): rats were given an oral daily dose of L-RSVR (20 mg/kg). 21 Rats in groups 3 and 4 were treated daily with RSVR and L-RSVR, respectively, for 14 days along with ISO (50 mg/kg, s.c, twice a week).
Rats were subjected to carbon dioxide in gradually increasing concentration and sacrificed by decapitation. Blood samples were collected; serum was separated by centrifugation at 3000 rpm for 15 min. Some parts of the heart tissues were removed and collected in 10 % formalin for histopathological studies, other parts were immediately frozen in liquid nitrogen and stored at −80°C for western blot analysis. The rest of the heart tissue was homogenized (20 % w/v) in phosphate buffer and centrifuged at 3000 rpm for 15 min., and then the supernatant was collected and stored at −80°C.
Biochemical Serum Analysis
Serum creatine kinase-MB (CK-MB) was evaluated using kits acquired from Randox Laboratories (UK) according to the manufacturer’s instructions. TNF-α and caspase-3 levels were estimated using ELISA kits obtained from R&D Co (USA) following the producer’s manual.
Western Blot Analysis
Protein expressions of cytochrome c and S100 in the heart tissues were evaluated using Western blot analysis. Using RIPA Lysis Buffer (Thermo Fisher Scientific, USA) that contains protease inhibitors, the cardiac cells were lysed. Protein concentrations were determined by Direct Detect® infrared spectrometer (Millipore, USA). Then, the lysates were subjected to SDS-PAGE electrophoresis, and the proteins were resolved and transferred to polyvinylidene difluoride (PVDF) membranes (Sigma-Aldrich, USA). After that, the membranes were blocked with 5% non-fat milk for 2 hours. To follow, the membranes were incubated overnight at 4°C with primary antibodies against cytochrome c and S100, all obtained from Santa Cruz (USA). Following a wash with buffer, the membrane was incubated at room temperature with the secondary antibody conjugated with horseradish peroxidase for 1 hour. The BioRad Molecular Imager (USA) was used to detect protein bands with ECL reagents (Immobilon, USA). To test the accuracy of our results, β-actin, the housekeeping protein, was used as an internal control.
Real-Time PCR Analysis of BAX, Bcl2 and Mef2
Firstly, total RNA was extracted from heart samples using the SV total RNA isolation system (Promega, Madison, USA). Then, the extracted RNA was reversely transcribed into cDNA using a cDNA synthesis kit (MedChemExpress, USA). The amplification was performed in a CFX96 cycler (Bio-Rad, USA) using an RT-PCR kit (Stratagene, USA). Gene expression of Bcl2, BAX, MEF2, and β-actin (Integrated DNA Technologies, Belgium) was detected using SYBR green master mix (MedChemExpress, USA).
DNA Fragmentation
To detect DNA fragmentation, agarose gel electrophoresis was employed. The DNA bands were visualized using the Gel Documentation System (UVP, USA). DNA fragmentation was assessed by applying the diphenylamine reagent method. 23
Histopathological Analysis
The formalin-fixed heart samples were dehydrated, then they had been fixed on paraffin wax. After that, heart specimens were excised into 5-μm thick sections and stored in 10% formalin. Following deparaffinization, sections were used for histopathological examination after hematoxylin and eosin (H&E) staining. The sections were dehydrated and examined using a light microscope.
Statistical Analysis
All values were calculated using the GraphPad Prism program (GraphPad Software, San Diego, CA, USA), and all statistical comparisons were made using the one-way analysis of variance test followed by Tukey’s post hoc test. Data are expressed as mean ± standard error of the mean and a P-value ≤.05 was considered significant.
Results
RSVR and L-RSVR Reduce Circulating Serum CK-MB
Data represented in Figure 1 show the effects of RSVR or L-RSVR on CK-MB activity in the serum of control and all treated groups. Rats exposed to ISO exhibited a significant (P ≤ .001) increase in serum CK-MB activity compared to control rats. Treatment with RSVR or L-RSVR for 2 weeks produced a marked (P ≤ .001) decrease in serum CK-MB activity. The L-RSVR-treated group showed a superior amelioration effect for the cardiomyopathy compared to the RSVR-treated groups. RSVR and L-RSVR prevent ISO-induced cardiac injury in rats. Treatments with RSVR or L-RSVR amended serum CK-MB levels. Statistical analysis was conducted using a one-way ANOVA test, followed by Tukey’s post hoc test, data are expressed as mean ± SEM, (n = 8). ***P < .001.
RSVR and L-RSVR Decrease Serum TNF-α and Caspase-3 Levels
Administration of ISO-induced serum inflammatory and apoptotic responses which were confirmed by the significant increase in serum levels of TNF-α (P ≤ .001) and caspase-3 (P ≤ .001) compared with the control group as shown in Figure 2. Treatment with RSVR or L-RSVR significantly reduced the levels of these markers in the serum of ISO-administered rats. RSVR and L-RSVR prevent ISO-induced cardiac injury in rats. Treatments with RSVR or L-RSVR decrease serum TNF-α and caspase-3 levels. Statistical analysis was conducted using a one-way ANOVA test, followed by Tukey’s post hoc test, data are expressed as mean ± SEM, (n = 8). *P < .05 and ***P < .001.
RSVR and L-RSVR Diminish the ISO cardiac Remodeling via Modulating protein Expression of cytochrome C and S100
The effects of RSVR or L-RSVR on the protein expression of cytochrome C and S100 in the hearts of normal and ISO-challenged rats were determined by western blotting (Figure 3). The exposure to ISO caused a significant upregulation in cytochrome C (P ≤ .001) and S100 (P ≤ .001) protein expression relative to the control group. RSVR or L-RSVR significantly ameliorated ISO effects on these proteins (P ≤ .001) by restoring their regular expression levels. The use of RSVR-based liposomes showed more pronounced effects on these proteins compared to RSVR treatment (P ≤ .01, P ≤ .001). RSVR and L-RSVR downregulate cardiac cytochrome C and S100 expression in ISO-injected rats. Representative blots show changes in the expression of cytochrome C and S100 in all treated groups. Statistical analysis was conducted using a one-way ANOVA test, followed by Tukey’s post hoc test, data are expressed as mean ± SEM, (n = 8). **P < .01 and ***P < .001.
RSVR and L-RSVR Inhibit cardiac Cell Damage in Response to ISO
Genomic DNA integrity was used to assess the protective effects of RSVR or L-RSVR. In agarose gel, ISO-injected rats showed a smeared band that reflected DNA fragmentation; however, RSVR or L-RSVR were effective (P ≤ .001) against ISO-induced cell damage and kept the DNA intact (Figure 4). Representative agarose image shows variations in the DNA fragmentation in all treated groups. Statistical analysis was conducted using a one-way ANOVA test, followed by Tukey’s post hoc test, data are expressed as mean ± SEM, (n = 8). ***P < .001.
RSVR and L-RSVR Diminish the ISO cardiac Remodeling via Modulating gene Expression of BAX, Bcl2, and MEF2
The effects of RSVR or L-RSVR on the gene expression of BAX, Bcl2 and MEF2 in the heart of normal and ISO-injected rats were determined by RT-PCR (Figure 5). The exposure to ISO caused a significant upregulation in BAX and MEF2 (P ≤ .001) and reverse downregulation in Bcl2 (P ≤ .001) gene expression relative to the control group. RSVR or L-RSVR significantly ameliorated ISO effects on these genes (P ≤ .001) by restoring their regular expression levels. Concomitant use of L-RSVR with ISO showed further additive effects on these proteins compared to RSVR treatment (P ≤ .01). RSVR and L-RSVR downregulate cardiac BAX and MEF2/β-actin gene expression and upregulation of cardiac Bcl2/β-actin gene expression in ISO-injected rats. Representative agarose images show changes in the expression of the above genes in all treated groups. Statistical analysis was conducted using a one-way ANOVA test, followed by Tukey’s post hoc test, data are expressed as mean ± SEM, (n = 8). **P < .01 and ***P < .001.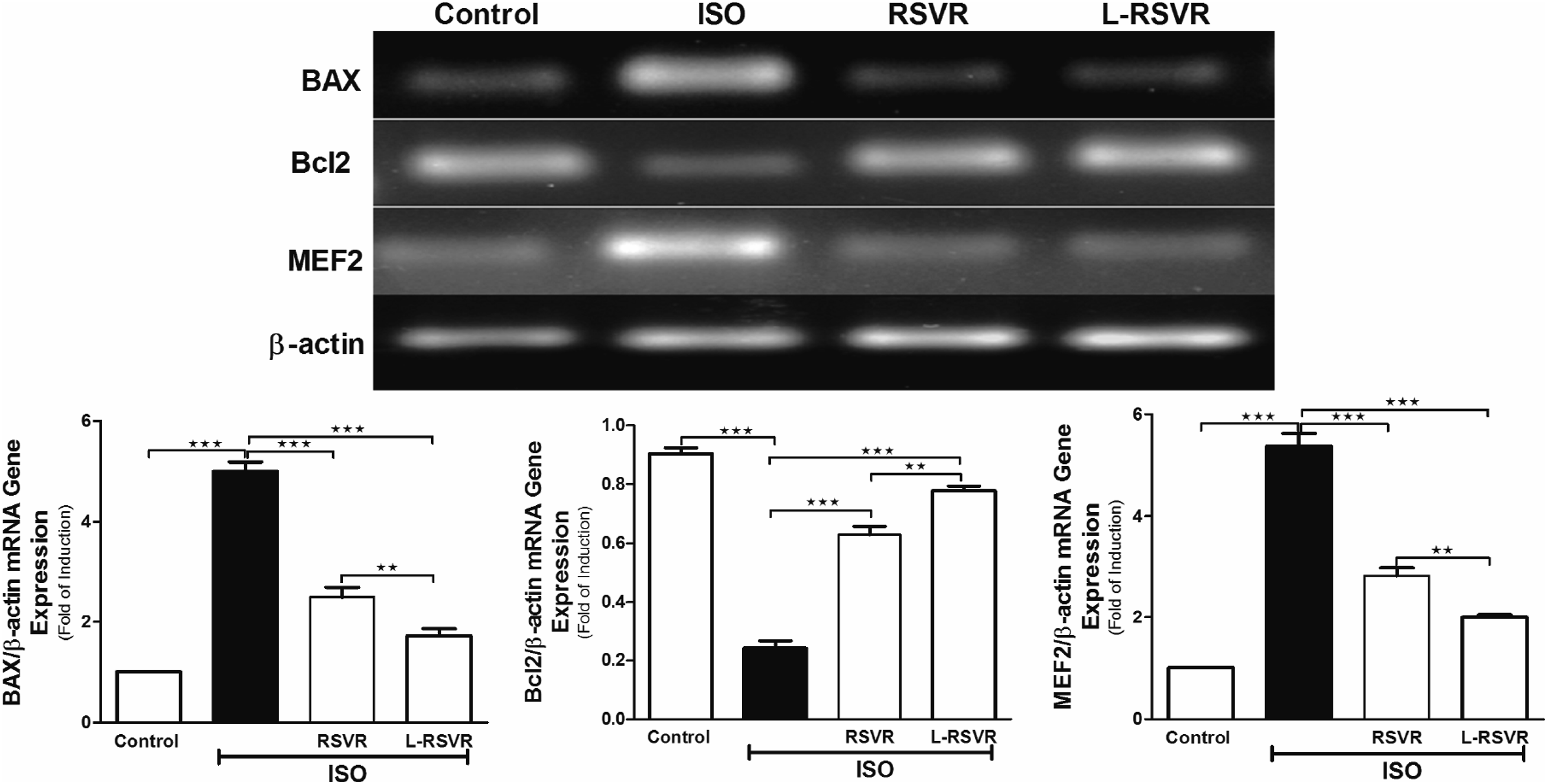
RSVR and L-RSVR Improve Histological Architecture and Decrease Collagen Deposition in ISO-injected rats
Histopathological examination of the H&E-stained heart sections of control rats showed normal heart tissue with normal features of myocardium cells, blood vessels, and endomysium (Figure 6(A)). The Administration of ISO caused focal areas of degeneration and inflammatory cellular infiltration in myocardial cells with many congested blood vessels (Figure 6(B)). Heart sections from RSVR or L-RSVR-treated rats showed an improvement in the alterations induced by ISO injection (Figure 6(C) and (D). Effect of RSVR and L-RSVR on cardiac histological changes induced by ISO injection in rats.: Photomicrographs of H&E-stained heart sections of (A) control rats displays a normal heart tissue with normal features of myocardium cells, blood vessels, and endomysium; (B) Sections obtained from ISO-injected rats show inflammatory cellular infiltration and focal areas of degeneration in myocardial cells with many congested blood vessels (black arrow); the representative sections from (C) RSVR-treated rats and (D) L-RSVR-treated rats exhibit an improvements of the alterations induced by ISO injection (H&E ×200).
Discussion
ISO is a synthetic catecholamine and non-selective β-adrenergic receptor agonist; it is widely used in experimental studies to induce cardiac remodeling. 24 Myocardial hypertrophy, fibrosis, and apoptosis are the common features of cardiac remodeling. These structural changes can ultimately disrupt the normal functioning of the heart and may lead to heart failure. ISO-induced myocardial hypertrophy is a well-characterized model that resembles chronic sympathetic stimulation, the main pathological hallmark of a dysfunctional heart.25,26 High doses of ISO cause oxidative stress in the heart, which leads to a sequence of biochemical, molecular, and morphological changes in cardiac muscle cells. 27 Despite substantial advances in understanding the mechanisms involved in cardiac remodeling, therapeutic interventions are still needed in order to mitigate pathological cardiac remodeling after maladaptive sympathetic stimuli.
Stilbenoid polyphenols such as RSVR may play a role as cardioprotective agents against ISO-induced cardiac remodeling. Evidence has approved the antioxidant and anti-inflammatory activities of RSVR.28,29 Therefore, we evaluated the cardioprotective effect of RSVR or L-RSVR against ISO-induced cardiac remodeling in an experimental rat model. Investigating the possible roles of RSVR or L-RSVR in oxidative stress, inflammation, and apoptosis pathway, as well as protein expression of cytochrome C and S100.
In this study, cardiomyopathy in rats was manifested by elevated serum levels of CK-MB, the primary marker of cardiac damage. This change was accompanied by massive histopathological changes in the cardiac tissue. CK-MB is a cytosolic enzyme that leaks out from the damaged heart to the bloodstream, and its concentration reflects the severity of tissue damage. This study showed a significant elevation of CK-MB in ISO-injected rats, as shown in previous studies.30,31 However, treatment with RSVR or L-RSVR restored the normal CK-MB levels, as demonstrated in previous studies.32,33 Evidence showed that ISO could trigger the generation of oxidative free radicals that cause cardiac cell membrane rupture and cardiac enzyme leakage, primarily CK-MB. 34 In line with our study, previous animal studies had shown that ISO injection induced elevation in serum levels of cardiac enzymes, including CK-MB. 35 It is worth noting that the damage in cardiac myocytes during the ISO overdose is owing to insufficient oxygen and glucose supply, which subsequently increases the release of cardiac CK-MB. Our findings were supported by previous studies that showed a reduction of ISO-induced cardiac remodeling by RSVR administration to rats, as evidenced by the significant reduction of CK-MB, LDH, and troponin-I levels and decreased myocardium degeneration in the histopathological findings that were in line with.36,37
Inflammation is one of the critical features of ISO-induced cardiac remodeling. Activation of β-adrenergic receptor by sympathomimetic agent stimulates inflammatory response by increased expression of IL-1β, IL-6, and TNFα in the heart. 38 TNFα is an inflammatory cytokine involved in the pathogenesis of many cardiac diseases. It acts by triggering a series of inflammatory molecules, including other chemokines and cytokines. 39 In accordance with our findings, a study by Murray and his colleagues showed that ISO-induced higher expression of TNF-α, IL-1 β, and IL-6. 40 In addition, previous studies revealed improvement in left ventricular remodeling and downregulation of myocardial expression of the inflammatory cytokines such as TNF-α and IL-6, both known as proapoptotic stimuli after β receptor blockade.41,42 Sijo and his colleagues showed RSVR administration for hypertensive mice significantly decreased serum TNF-α and IL-6 after 8 weeks from administration, 43 which parallels our findings. In addition, evidence showed RSVR reduces TNF-α through regulating NF-κB activation. 44
Besides inducing inflammation, ISO-induced cardiac remodeling can trigger cell apoptosis, which is widely recognized as a key contributor to inflammation and oxidative stress. Both of these contribute to myocardial damage, as noted by Obeidat et al. 45 The caspases represent key enzymes in the apoptotic signaling pathway in cardiomyocytes. Caspase-3 remains inactive in normal cells; however, upon exposure to apoptotic stress, caspase activates other downstream caspases, such as caspase −6, −7, and −9, to break down the apoptotic cells before removal. 46 In line with our data, results reported by Yu Zhao showed an increase in caspase-3 and caspase-9 protein expression in the ISO group compared with the control group, which was ameliorated after difluoro-methylornithine treatment. 47 Another study showed a reduction in caspase-3 expression after RSVS treatment of diabetic rats. 48 Besides that, there is a significant association between improved cardiac function and the prevention of cardiomyocyte apoptosis. In our study, we observed marked upregulation of BAX and downregulation of Bcl2 gene expression in the ISO-treated group compared to the control group. However, when either RSVR or L-RSVR was administered, the expression levels of BAX and Bcl2 mRNA were significantly restored to their normal state. In response to cardiac oxidative stress, BAX induces the release of mitochondrial cytochrome C, which activates caspase enzymes and leads to apoptosis, whereas Bcl2 limits the apoptosis progression by stopping BAX from releasing cytochrome C. Therefore, the ratio of Bcl2 to BAX is considered an essential indicator of cell susceptibility to apoptosis in various diseases. 49 Corroborating our findings, 1 study has shown that RSVR can block cardiomyocyte apoptosis, induced via the coronary microembolization model, by elevating the Bcl2/BAX ratio. 50 Furthermore, RSVR protects against mitochondrial oxidative stress and apoptosis in neonatal rat cardiomyocytes subjected to hypoxia/reoxygenation injury by restoring Bcl2/BAX ratio. 51
Our study showed upregulation of cytochrome C after ISO administration, which is a logical consequence of apoptosis and imbalance of BAX and Bcl2 levels. Cytochrome C is a main component in the electron transport chain, cell respiration, and energy production. The expression of cytochrome C and caspase-3 and 9 activity levels were significantly increased in ISO-induced myocardial infarction in rats. 52 The use of either RSVR or its liposomal form downregulated cytochrome C expression. Additionally, immunoblotting analysis revealed upregulation of S100A1 expression in ISO-induced cardiac remodeling; however, this effect was ameliorated after treating rats with either RSVR or L-RSVR. S100A1 is a calcium-binding protein predominantly found in the myocardium. Studies showed a correlation between S100A1 expression and cardiac contractile performance. 53 A higher expression level of S100A1 may be associated with ischemic heart diseases; thus, it can be used as a diagnostic marker. 54 A study by Most et al showed that extracellular S100A1 could protect cardiomyocytes via specific activation of the ERK1/2 signaling pathway from apoptosis. 55
Moreover, ISO-induced apoptosis in cardiac myocytes can be manifested as excessive DNA fragmentation. In more straightforward terms, mitochondrial dysfunction triggers a cascade of events leading to heart cell damage, DNA fragmentation, and, ultimately, heart remodeling. 56 Thus, DNA fragmentation is a critical sign of this remodeling, indicating that heart cells undergo apoptosis. Our investigation showed that RSVR partly protected cells from damage induced by ISO by protecting DNA integrity. RSVR works by activating sirtuin 1 (SIRT1), an enzyme that controls metabolic homeostasis and improves tissue pathophysiological changes in response to stress and aging. 57 The activation of SIRT1 not only helps to protect cells from oxidative stress but also inhibits the process of cardiac remodeling.58,59 In addition, the antioxidant properties of RSVR help to neutralize the free radicals that cause DNA fragmentation, provide a protective effect, and enhance the natural repair mechanisms. 60
Furthermore, cardiac remodeling is characterized by myocardial hypertrophy that collectively leads to a disturbance in the heart’s function. MEF2 plays an essential role in developing the hypertrophic response of cardiomyocytes to pressure overload, suggesting its possible contribution to adult heart pathology.61-63 MEF2 has 4 genes, MEF2-a, -b, -c, and -d, that are expressed in cardiac tissue, 64 and it is involved in β-adrenergic receptor signaling. 64 Several kinases are involved in the activation of MEF2 including Ca++/calmodulin-dependent protein kinase (CaMKII), mitogen-activated protein kinases (MAPKs) such as PI3K/Akt/p38/ERK5 along with several co-activators such as GATA4. 65 MEF2 is a transcription factor implicated in regulating genes that are involved in cardiac muscle morphology. MEF2 binds to specific DNA sequences called MEF2 response elements (MREs) present in the regulatory regions of various genes thus activating or repressing gene transcription involved in the regulation of hypertrophy, apoptosis, inflammation, actin dynamics, and stress signaling. 66 These genes encode for contractile proteins such as myosin heavy chain (MHC), atrial natriuretic factor (ANF), a putative cell-cycle inhibitor CHAMP, the insulin-regulated glucose transporter Glut4 and the transcription factor KLF5.65,67 Cardiac overexpression of a dominant negative MEF2C protein inhibits cardiomyocyte differentiation, which is partially mediated in part by decreased GATA4 and NKX2-5 expressions. 67 Transgenic mice with overexpression of MEF2A and MEF2C exhibited enlarged ventricles despite weaker ventricular contractility. Moreover, overexpressing MEF2D in mice can also result in pathological remodeling of the heart tissue.68,69
According to our results, ISO induced a remarkable elevation in the expression of the MEF2 gene, an effect that has been significantly abrogated by RSVR. The exact mechanisms through which ISO-induced MEF2 overexpression in the heart are not yet fully understood; however, excessive exposure to ISO may induce the release of Ca++ inside the heart cells, 70 activating calcineurin. Calcineurin triggers the dephosphorylation of MEF2, which subsequently becomes more active and increases the transcription of genes contributing to heart muscle hypertrophy. 71 To our understanding, there has been no in vivo report that links RSVR administration to the gene expression of MEF2. In general, several studies have revealed that RSVR can alleviate cardiac hypertrophy, 72 and according to our results, inhibition of MEF2 expression could represent a possible mechanism for this alleviation.
In support of the previous finding, histopathological examination of cardiac tissues revealed focal areas of congestion and degeneration in myocardial cells in ISO-injected rats. Previous reports confirmed the deleterious effect of ISO administration on cardiac tissue by showing degenerative changes, including focal myonecrosis, edema, and infiltration of inflammatory cells, 73 and increased cardiac fibrosis, as indicated by Masson staining. On the other hand, RSVR and L-RSVR restored normal heart tissue architecture with regular features of myocardium cells, blood vessels, and endomysium. Our results were parallel with previous studies which revealed the protective activity of RSVR. 73
This work provides new protective mechanisms of RSVR-based liposomes against ISO-induced cardiac remodeling despite some limitations including the animal sample size and design which were calculated and done in accordance with 3 Rs (replacement, reduction, and refinement).
Conclusion
In conclusion, RSVR has antioxidant, anti-inflammatory, and antiapoptotic properties. Both RSVR and L-RSVR ameliorated ISO-induced cardiac remodeling by reversing the changes in cardiac function markers, inflammation, cell death DNA damage, and cardiac cell degeneration, with more superior effects of L-RSVR than its native form. Given its beneficial influence in averting cardiac remodeling, RSVR emerges as a prospective prophylactic supplement in treating high-risk cardiac patients and potentially beneficial in treating other cardiovascular conditions. Nevertheless, further comprehensive investigations are warranted to fully understand RSVR mechanisms of action and establish the optimal dosage and timing for RSVR.
Footnotes
Acknowledgments
The authors would like to extend their gratitude to Researchers Supporting Project number (RSPD2024R822), King Saud University, Riyadh, Saudi Arabia for funding this work.
Author Contributions
Project administration: A.M.A.; Methodology: A.M.A., H.K.A., W.S.S., J.S.A., and S.M.A., Validation: Q.H.A., A.R.A., E.A.A. and E.N.A; Formal analysis: I.H.H., and W.S.S.; Writing the original draft: A.M.A., H.K.A., W.S.S., Q.H.A., and J.S.A.; All authors have read and agreed to the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Researchers Supporting Project number (RSPD2024R822), King Saud University, Riyadh, Saudi Arabia.
Institutional Review Board Statement
The animal experiment was conducted according to the Institutional Animal Care and Use Committee (IACUC) guidelines and was approved by the Research Ethics Committee at King Saud University (Ethical reference no. KSU-SE-22-111) on 29/01/2023.
Data Availability Statement
Data analyzed or generated during this study are available upon request from the corresponding author.
