Abstract
Objective
In this study, we investigated the cause of the AngII dose elevation in aqueous humor of primary open-angle glaucoma (POAG) patients.
Methods
Enzyme-linked immunosorbent assay (ELISA), western blotting were used to detect concentration of Angiotensin Converting Enzyme 2 (ACE2) and Prolylcarboxypeptidase (PRCP). AngII and AngII + Recombinant PRCP were injected into anterior chamber of mouse eye. Mouse Intraocular pressure (IOP) was measured every week, mouse eye sections were conducted Hematoxylin-and-Eosin (H&E) staining, Masson’ staining and Immunofluorescence staining. Western blotting and Immunofluorescence staining assays to detected fibrosis of trabecular meshwork cells. Mass spectrometry was used to identify proteins of aqueous humor.
Results
PRCP dose are decreased in aqueous humor of POAG patients. There is a negative correlation between PRCP and AngII levels in aqueous humor and between PRCP levels and the IOP. PRCP treatment reverses fibrosis of trabecular meshwork (TM) and prevents IOP elevation induced by AngII. Exogenous PRCP rescues fibrosis induced by AngII in HTMCs. Proteome profiling detected 502 differentially expressed proteins.
Conclusion
Our study found PRCP dose was decreased in POAG patients’ aqueous humor, and it might cause high level of AngII. Restoration of PRCP rescued fibrosis of TM cells and ameliorated IOP in AngII treatment mouse.

Introduction
Glaucoma is the leading cause of irreversible blindness. It is characterized by progressive loss of retinal ganglion cells and irreversible visual deficiency and POAG is the most common type.1,2 Currently, nearly 60.5 million people worldwide are affected by this indolent disorder, and it is estimated that this will climb to 111.8 million by the year 2040. 3 Elevated intraocular pressure is a major risk factor for glaucoma. 4 The flow of aqueous humor generates the IOP and maintains the normal eye shape. Aqueous humor is produced by the ciliary body and flows out mainly through the trabecular meshwork. Hence, any functional defect of TM cells promotes IOP elevation.5,6
The renin-angiotensin-aldosterone system (RAS) plays essential roles in terms of controlling sodium balance and the volume of body fluid, and thus regulates blood pressure. 7 The RAS of local tissues influences long-term changes in certain organs, including the eye.8-11 RAS components have been detected in the eye and have been linked to the development of eye disease.12-15 Notably, in animal models, ACE (Angiotensin Converting Enzyme) inhibitors lower the IOP, suggesting that such inhibitors may be valuable in humans.16,17 We previously reported that the dose of angiotensin II (AngII) was elevated in the aqueous humor of POAG patients, and that this was positively correlated with IOP. In terms of downstream actions, we found that AngII promotes NOX4 expression in TM cells and tissues, increasing the levels of reactive oxygen species (ROS) and activating the TGFβ/Smad3 pathway to induce fibrosis-related changes in gene expression. 18 These results suggest that an elevated AngII dose is a risk factor for POAG, although the cause of such elevation has remained unclear.
ACE2 is the key enzyme responsible for converting AngII to Ang1-7 in blood; another such enzyme is prolylcarboxypeptidase. 19 The enzyme that is preferentially expressed in the aqueous humor and might thus be responsible for the observed elevation in the AngII level requires further investigation. In this study, we used ELISA to measure the ACE2 and PRCP levels in aqueous humor. We found that ACE2 levels did not differ between POAG and cataract patients but PRCP levels were lower in the former. PRCP levels are negatively correlated with AngII concentrations and the IOP. Extension of these findings to in vivo and in vitro models of POAG have shown that exogenous PRCP treatment rescues the AngII-induced fibrotic changes in both murine TM and cultured human TM cells (HTMCs). This suggests that restoration of normal dose of PRCP in the aqueous humor of POAG patients would potentially treat glaucoma.
To confirm the decreases in PRCP detected by Western blotting and ELISA assays, we used unbiased mass spectrography (MS) to assess the proteome of the aqueous humor. We screened 10 aqueous humor samples (five from POAG patients and five from cataract surgery controls) and identified 1871 proteins, of which 502 were differentially expressed proteins (DEPs). PRCP levels were lower in POAG patients. This improves our understanding of the eye environment in POAG patients, opening new avenues for research.
Materials and Methods
Aqueous Humor Collection
Demographic Features of the Study Population.
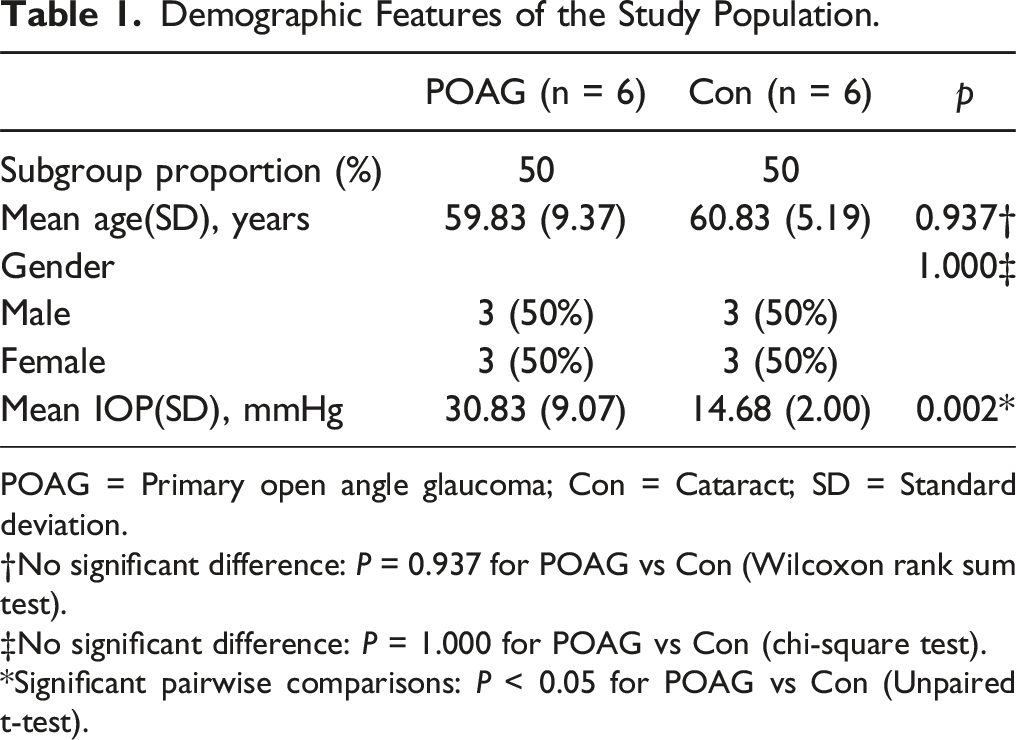
POAG = Primary open angle glaucoma; Con = Cataract; SD = Standard deviation.
†No significant difference: P = 0.937 for POAG vs Con (Wilcoxon rank sum test).
‡No significant difference: P = 1.000 for POAG vs Con (chi-square test).
*Significant pairwise comparisons: P < 0.05 for POAG vs Con (Unpaired t-test).
Demographic Features of the Study Population.
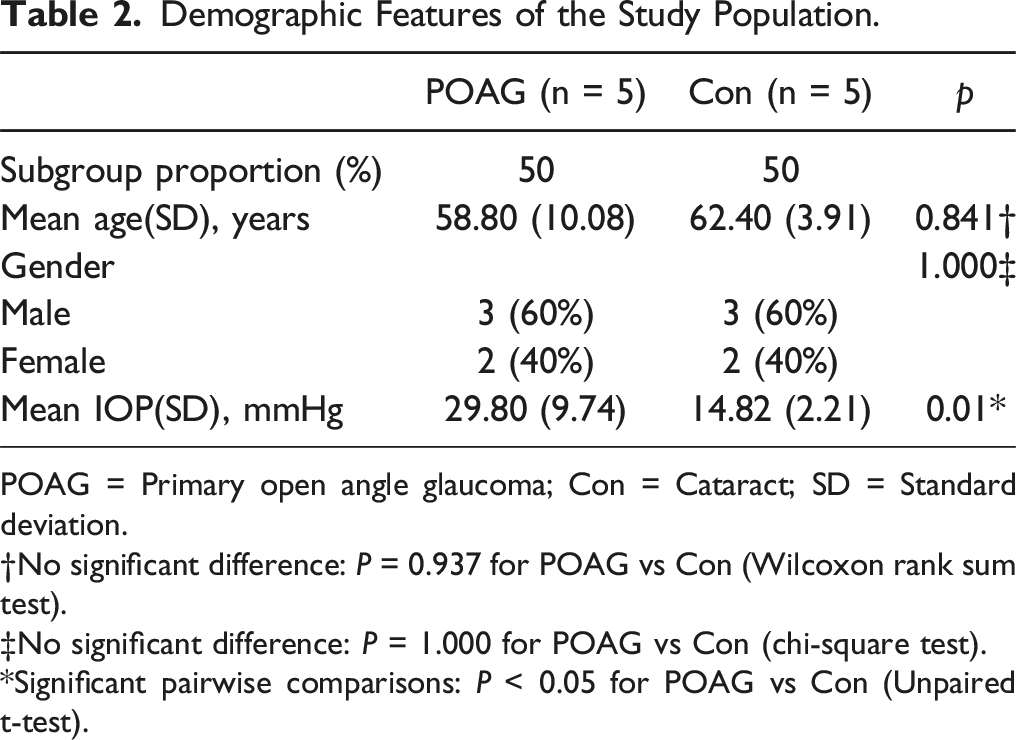
POAG = Primary open angle glaucoma; Con = Cataract; SD = Standard deviation.
†No significant difference: P = 0.937 for POAG vs Con (Wilcoxon rank sum test).
‡No significant difference: P = 1.000 for POAG vs Con (chi-square test).
*Significant pairwise comparisons: P < 0.05 for POAG vs Con (Unpaired t-test).
Western Blotting
The detailed procedures used for Western blotting of aqueous humor samples and cell lysates are described in previous studies.18,20 Membranes were blocked with blocking buffer (Kermey, MF0501, China) and antibodies were diluted in antibody dilution buffer (Kermey, MX0402, China). The primary antibodies used detected PRCP (Proteintech, 15995-1-AP, China, 1:2000), GAPDH (Abways, AB0037, China, 1:5000), Fibronectin (HUABIO, ET1702-25, China, 1:3000), Collagen I (Proteintech, 14695-1-AP, China, 1:2000), and αSMA (Abways, CY5295, China, 1:2000). Immunocomplexes were revealed using a chemiluminescent substrate (Sparkjade ECL plus, ED0016-C, China).
siRNA Transfection
siRNAs were synthesized by GENERAL BIOL (Chuzhou, Anhui, China). Negative control (si-NC) siRNAs sequences were as follow 5′- UUCUCCGAACGUGUCACGUTT and 5′-ACGUGACACGUUCGGAGAATT. The human PRCP siRNAs sequences were as follow siRNA1: 5′-GAUCGUAACUACAGAUUUUTT and 5′-AAAAUCUGUAGUUACGAUCTT; siRNA2: 5′- GCAACUAGCAGUCUGGGAATT and 5′- UUCCCAGACUGCUAGUUGCTT; siRNA3: 5′- GUGGAGUAACUAAGGAUAUTT and 5′- AUAUCCUUAGUUACUCCACTT. Transfections were conducted using the Lipofectamine™ RNAiMAX reagent (13778150, Thermo fisher SCIENTIFIC) according to the manufacturer’s instructions.
Enzyme-Linked Immunosorbent Assays (ELISAs)
The levels of PRCP (YJ161508 ELISA kit, Shanghai Enzyme-linked Biotechnology Co., Ltd., China) and ACE2 (YJ023376 ELISA kit, Shanghai Enzyme-linked Biotechnology Co., Ltd., China) in aqueous humor samples were detected using ELISA assays, according to the manufacturer’s recommendations.
Animal Experiments
Female C57BL/6J mice (8 weeks of age) were purchased from GemPharmatech (Nanjing, China). Drugs were injected into the anterior chamber of the right eye using 35 G needles, as previously reported, 21 after the mice were anesthetized using a small-animal gas anesthesia machine (Shenzhen RWD Life Technology Co., Ltd, China) delivering isoflurane (2.5% v/v) in oxygen at a flow rate of 0.8 L/min for ∼2 min. Injections were performed once weekly. Mice received either AngII alone (1 μL 0.5 μM AngII diluted in physiological saline solution [PSS]) or AngII with recombinant PRCP protein (AngII with 10 μg/mL PRCP). IOPs (three repeats) were measured each week at 10 a.m. using the TonoLab instrument (Icare Finland Type TV02); the mean value was employed for analysis. To prevent infection, ofloxacin ointment was applied. Four weeks later, the mice were humanely sacrificed and the excised eyes were fixed for later immunostaining. All animal experiments complied with the Association for Research in Vision and Ophthalmology Statement. All animal experiments were approved by the Animal Ethics Committee of Zhengzhou University (approval no. ZZU-LA20220729).
Hematoxylin-and-Eosin (H&E) staining, Masson’s trichrome staining, and immunofluorescence staining of tissue sections
The methods used for H&E staining and immunofluorescence staining of tissue sections are described in previous publications.18,20 Masson’s trichrome staining was performed using a Masson’s trichrome stain kit (Solarbio, G1346, China) according to the manufacturer’s recommendations.
Pharmacological Agents
Where indicated, cells were treated with AngII (100 nM, HY-13948, MedChemExpress, China) for 48 h alone or in combination with recombinant PRCP protein (10 ng/mL, M19977, AbMole, China).
Human Trabecular Meshwork Cell Culture
Immortalized (SV40-transformed) HTMCs were purchased from iCell Bioscience Inc. (Shanghai, China); these had been derived from primary HTMCs (ScienCell Research Laboratories, Catalog Number 6590; Lot Number 16930; CA Number 0002981). The cells were maintained in DMEM/F12 medium (CD0001, SparkJade, China) supplemented with 15% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 mg/mL streptomycin. Experiments were performed on HTMCs passaged five to seven times after receipt. Alternatively, primary TM cells were isolated and validated as described in previous studies18,20 using DEX treatment followed by analyses of myocilin expression and release, consistent with the published recommendations. 22 We employed cultured primary TM cells from two separate donors; all experiments used cells that had been passaged between five and eight times. The collection of human eye material was approved by the Medical Ethical Committee of Henan Eye Hospital (approval no. HNEECKY-2022[18]).
Immunofluorescence Staining of Cells
The methods used for immunofluorescence staining of cells are described in previous publications.18,20 All antibodies were diluted in blocking solution. Donkey anti-rabbit IgG (H + L) 488 (Jackson ImmunoResearch Inc., 711-545–152) or donkey anti-mouse IgG (H + L) Cy3 (Jackson ImmunoResearch Inc., 715-165–150, USA) secondary antibodies were used to detect rabbit and mouse IgGs, respectively. After nuclear counterstaining with DAPI solution (MedChemExpress, HY-D1396, China) for 5 min, cells on coverslips were mounted onto glass slides and images were captured via a Leica DMi8 confocal microscope. The primary antibodies used included those against fibronectin (1:500, HUABIO, ET1702-25, China), collagen I (1:500, Proteintech,14695-1-AP, China), and αSMA (1:500, Abways, CY5295, China).
Mass Spectrometry, Data Processing, and Analysis
Sample Preparation
Lysis of aqueous humor cells, reduction, alkylation, and digestion were performed using sample preparation by easy extraction and digestion methods reported previously. 23
Liquid Chromatography-tandem MS
Nanoflow reverse-phase chromatography was performed using a NanoElute ultra-high performance liquid chromatography system (Bruker). Mobile phases A and B were water with 0.1 vol% formic acid and acetonitrile with 0.1 vol% formic acid, respectively. Peptides were separated over 45 min at a constant flow rate of 400 nL/min on a 25 cm × 75 µm column of 1.9 µm ReproSil-Pur C18-AQ particles (Dr. Maisch) using the gradient set: 0-35 min, 4-23% B; 35-40 min, 23-32% B; 40-42 min, 32-95% B; and 42-45 min, 95% B. The NanoElute platform was coupled online to a timsTOF Pro mass spectrometer (Bruker) via a capillary ion source (ion source voltage, 1.5 kV). MS data were acquired using the diaPASEF method as described previously, with slight modifications. 24
Data Processing for DIA Data Analysis
All mass spectrometric data were analyzed using the directDIA approach of SpectronautTM (Biognosys, v18) with the standard settings and the human Swiss-Prot database containing 20,404 sequences (downloaded in February, 2023). After SpectronautTM searching, the results were exported and normalized using the median centering method to correct for sample loading differences. We employed the “Wu Kong” platform (https://www.omicsolution.com/wkomics/main/) to input missing values using the K-nearest neighbor (k-NN) method. 25 Only proteins for which fewer than 50% of values were missing for at least one case in each of the POAG and cataract groups were input. For proteins for which quantitative information was available for one case but not another, we filled the missing values using half of the minimum values. After missing value input and log2 conversion, differential expression analysis was performed using the Student’s t-test of the R platform. We employed various packages (examples: “ggplot2,” “pheatmap”) to analyze and present the results. Gene ontology analysis was performed using the DAVID platform (https://david.ncifcrf.gov/).
Statistical Analysis
All analyses employed GraphPad Prism version 7.0 software. The age difference between POAG and cataract patients was analyzed using the chi-square test. All results are presented as means ± SDs of the results of at least three independent experiments. Data were compared employing the unpaired Student’s t-test (between two groups) or one-way analysis of variance (ANOVA; three or more groups). P < 0.05 was considered to indicate statistical significance.
Results
PRCP Expression is Negatively Correlated with AngII Levels and the IOP
As ACE2 is the key enzyme responsible for converting AngII to Ang1-7 in blood,
26
we first assayed ACE2 levels in a cohort of 48 aqueous humor samples from 28 cataract patients and 20 POAG patients, as described in a previous study.
18
Notably, there were no significant differences in the ACE2 level between aqueous humor samples collected from POAG patients and cataract surgery controls (Figure 1(A)). However, as PRCP is an isoenzyme of ACE2, we also measured the PRCP levels in the same samples; the PRCP levels were significantly lower in POAG patients (Figure 1(B)). Next, we subjected samples from each of six independent POAG and cataract patients to Western blotting, the clinical information was described in Table 1. This showed that the immunoreactivity of a single band migrating at the known Mr of PRCP was clearly reduced in most POAG samples (Figure 1(C)). Consistently, quantitative assessments confirmed that the PRCP level was significantly decreased in POAG patients (Figure 1(D)). PRCP dose are decreased in the aqueous humor of POAG eyes and are negatively correlated with the IOPs. (A) ELISA-based quantification of ACE2 concentrations in aqueous humor samples from control (cataract) patients (n = 28) and POAG patients (n = 20). (B) ELISA-based quantification of PRCP concentrations in aqueous humor samples from control (cataract) patients (n = 28) and POAG patients (n = 20). (C, D) Western blotting detection of PRCP in 12 human aqueous humor samples (top) from control (cataract) patients (n = 6) and POAG patients (n = 6). The loading levels were verified employing Ponceau staining (bottom). (C) Quantification of PRCP intensities normalized to the Ponceau staining levels of the protein bands in C (D). (E) A negative correlation between the PRCP and AngII concentrations of the 48 patients of (A) (R = –0.373 and P < 0.01). (F) A negative correlation between the PRCP concentrations and IOPs of the 48 patients of (A) (R = –0.50 and P < 0.001). Data are means ± SDs, *P < 0.05; **P < 0.01; ***P < 0.001, n.s.: not significant, unpaired Student’s t-test.
Next, we considered the relationship between the levels of PRCP and AngII, and their relationships with IOP. The Pearson correlations between the PRCP levels and the concentrations of AngII in the 48 samples indicated that the PRCP level was negatively correlated with the AngII concentration in aqueous humor (Figure 1(E)). Similarly, the PRCP level was negatively correlated with the IOP (Figure 1(F)). Together, the data suggest that PRCP is responsible for regulation of AngII levels in aqueous humor, in turn influencing the IOP.
To validate the specificity of PRCP antibody, we knock down PRCP in primary HTMCs using siRNA and detected PRCP expression with western blot assay, the results were showed in sFigure1. Our data revealed PRCP band signals were decreased when knock down RPCP with three different siRNAs, which indicating this antibody precisely recognized PRCP.
Restoration of PRCP Dose Rescues the IOP Elevation and the TM Fibrosis-Related Changes Induced by AngII in vivo
We used an in vivo mouse model of POAG to determine whether PRCP rescues fibrosis-related changes in the TM induced by AngII. Anterior chamber injections featured a vehicle control (physiological salt solution), and AngII either alone or in combination with recombinant PRCP protein (Figure 2(A)). AngII significantly increased the IOP compared to that of the control group, but this elevation was rescued by cotreatment with recombinant PRCP protein (Figure 2(B)). Cotreatment with PRCP rescues the IOP elevation and collagen deposition in the trabecular meshwork induced by AngII in vivo. (A, B) An in vivo mouse model was established by injecting AngII alone or with recombinant PRCP into the anterior chamber once weekly (A). The IOPs were measured every week (B) (n = 10 mice/group). (C) Representative eye sections from the treatment groups of (A) stained by H&E. The trabecular meshwork (TM) tissues are the boxed areas. (D, E) Representative images of eye sections from the treatment groups of (A) showing Masson’s trichrome staining of collagen fibrils (D). The staining intensities were quantified within the boxed TM areas. Data were derived from five eyes representative of all animals in each treatment group. Data are means ± SDs, ***P < 0.001, one-way ANOVA.
H&E staining showed that the TM morphology of eye was not affected by AngII treatment, either alone or with PRCP (Figure 2). However, parallel staining of sections with Masson’s trichrome showed that the collagen fibril intensities significantly increased after AngII treatment, but PRCP cotreatment prevented such changes. To explore further, we performed immunofluorescence staining and confocal microscopy. Consistently, TM staining for both Collagen type I alpha 1 (Col1) and Fibronectin (FN) was increased by AngII but not by the combination of AngII and PRCP (Figure 3). Quantitative analysis of the staining intensities verified that PRCP cotreatment completely rescued the increases in fibrotic-related protein levels induced by AngII. Thus, similar to the findings in vitro, PRCP rescued the fibrosis-related changes induced by AngII in vivo. Cotreatment with PRCP reverses the trabecular meshwork changes in fibrotic gene expression induced by AngII in vivo. (A, B) Representative confocal images of single eye sections from the treatment groups (PBS, AngII, or AngII + PRCP) immunostained for Col1 (A; green) and FN (B; green). The cell nuclei were stained with DAPI (blue). (C, D) The relative intensities of Col1 (C) and FN (D) staining were quantified in the trabecular meshwork (TM) tissues of the boxed areas. Data were derived from five individual eyes representative of all animals in each treatment group. Data are means ± SDs, ***P < 0.001, one-way ANOVA.
Restoration of PRCP Dose Rescues the Fibrotic Phenotype of HTMCs Induced by AngII in vitro
Exposure of HTMCs to AngII induces fibrosis-related changes involving upregulation of the extracellular matrix components FN and Col1 and the alpha smooth muscle actin (αSMA) level. To explore whether PRPC could reverse this phenotype, we treated transformed HTMCs with AngII in combination with recombinant PRCP protein. Indeed, Western blotting revealed that exogenous PRCP countered the elevations of the FN, Col1, and αSMA levels induced by AngII (Figures 4). Similar findings were apparent when primary HTMCs were cotreated with AngII and PRCP, indicating that the changes in the fibrosis-related protein levels were not artefacts of cell transformation. Exogenous PRCP rescues fibrosis-related changes in HTMCs induced by AngII. (A, B) Transformed HTMCs were treated with PBS, 100 nM AngII, or 100 nM AngII +10 ng/μL PRCP for 48 h and Col1, FN, and αSMA quantified via Western blotting by reference to a GAPDH loading control (A) Densitometric quantification of the levels of fibrosis-related proteins in the three independent experiments (B). (C, D) Primary HTMCs were treated with PBS, 100 nM AngII, or 100 nM AngII +10 ng/μL PRCP for 48 h and Col1, FN, and αSMA quantified via Western blotting by reference to a GAPDH loading control (C) Densitometric quantification of the levels of fibrosis-related proteins in the three independent experiments (D). Data are means ± SDs, *P < 0.05, **P < 0.01, a one-way ANOVA.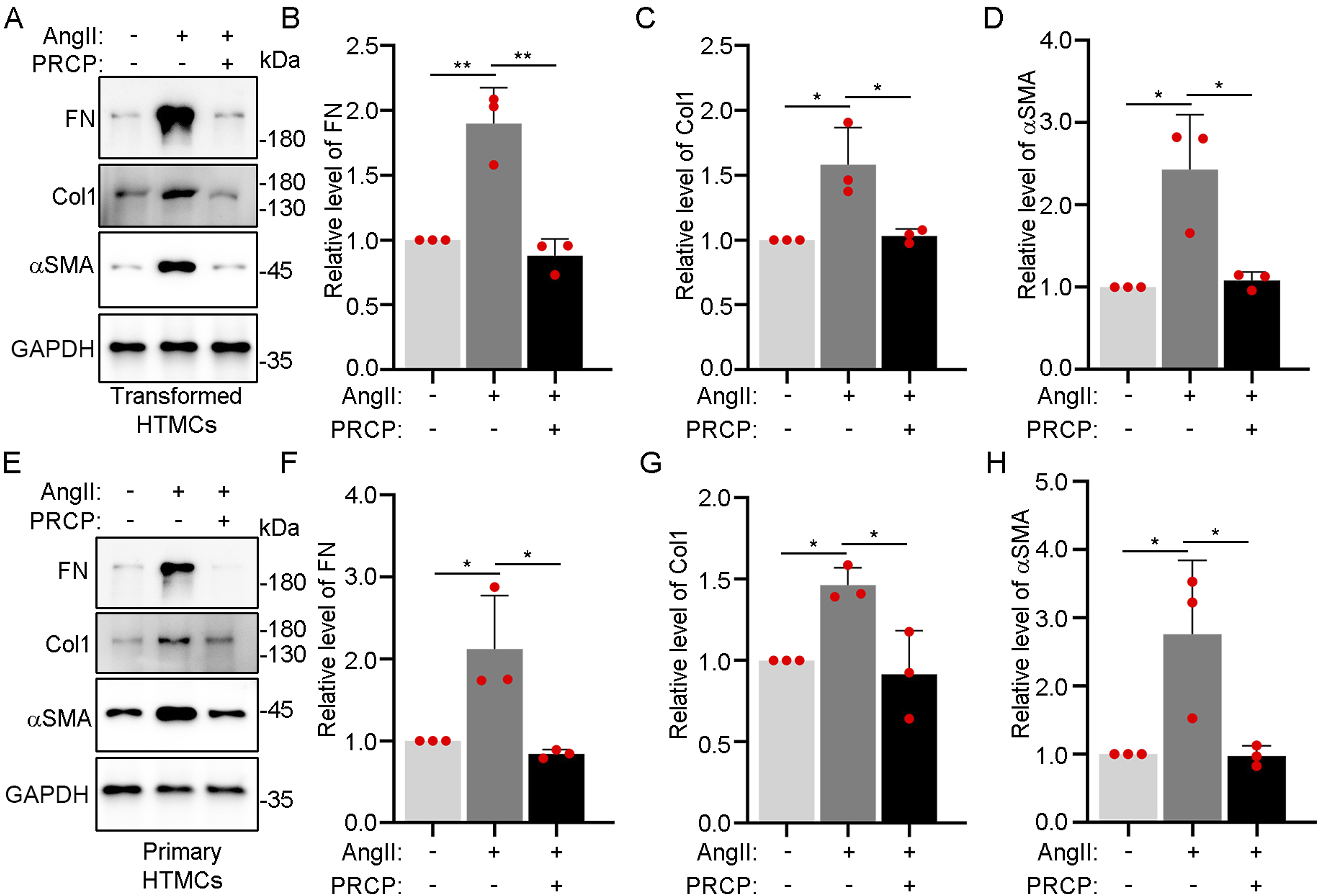
We also visualized changes in protein expression levels using immunofluorescence staining combined with confocal microscopy. Analyses of both transformed and primary HTMCs (Figure 5 and Supplemental Figure 2, respectively) showed that the increased levels of FN, Col1, and αSMA staining after AngII treatment were not apparent if recombinant PRCP protein was present. Quantitative measurements of the fluorescence intensities confirmed that PRCP treatment maintained the levels of fibrosis-related proteins at the normal (untreated) levels in AngII-treated cells. Together, these data indicate that exogenous PRCP rescues the fibrotic changes in HTMCs induced by AngII, supporting the suggestion that augmentation of low PRCP levels may counteract pathogenic changes in POAG patients. Imaging confirms the effects of exogenous PRCP on HTMCs treated with AngII. (A-C) Representative confocal microscopy images of transformed HTMCs stained by immunofluorescent antibodies detecting Col1 (green; A), FN (green; B) and αSMA (red; C) after 48 h of treatment with PBS, 100 nM AngII, or 100 nM AngII +10 ng/μL PRCP. Cell nuclei were stained with DAPI (blue). (D-F) The staining intensities of Col1 (D), FN (E), and αSMA (F) in transformed HTMCs determined in the three independent experiments of (A-C) (n = 10 cells/group/experiment). Data are means ± SDs, ***P < 0.001, one-way ANOVA.
Dramatic Changes in the Aqueous Humor Proteome of POAG Patients
To develop a better understanding of the protein composition of the aqueous humor of POAG patients, we subjected 10 samples (five from patients with POAG and five from normal controls) collected during cataract surgery to proteome analyses, the clinical information was described in Table 2. Using Nanoflow reverse-phase chromatography, proteomic profiling identified more than 1000 proteins in each sample with a nonsignificant trend toward greater protein abundance in POAG samples compared to controls (Figure 6). Around 800 proteins were common to all 10 samples; a further ∼2000 proteins were expressed in at least one sample. Principal component analysis (PCA) revealed that most POAG samples and all cataract control samples clustered apart, indicative of compositional differences. Notably, the high degree of separation between individual POAG samples likely reflects the marked complexity of POAG. Comparative proteomic analysis of POAG aqueous humor reveals extensive changes in protein composition and protein expression levels. (A) The total number of proteins identified via MS analysis in the aqueous humor of five control (cataract) and five POAG patients, respectively. (B) The uniqueness of the aqueous humor proteomes of all samples expressed as distribution counts. (C) Principal component analysis (PCA) of the aqueous humor protein components in the 10 samples of (A). (D) Volcano plot showing the differentially expressed proteins (DEPs) between the aqueous humors of POAG and cataract patients. Of the 1,871 proteins detected, 396 and 106 were upregulated and downregulated, respectively, as revealed by log2-fold changes > 1.5 or < –1.5, respectively; P < 0.05. (E, F) Biologic process enrichments as revealed by the upregulated DEPs (E) and downregulated DEPs (F) of (D), based on KEGG analyses. Data are means ± SDs, unpaired Student’s t-test.
Next, we used log2-fold changes >1.5 or < −1.5 with a P < 0.05 cutoff to identify differentially expressed proteins (DEPs) between the two groups. This revealed 502 DEPs, of which 396 were upregulated and 106 downregulated (Figure 6). Enrichment analyses of the DEPs in terms of the “biologic processes” of KEGG indicated that many fundamental processes were likely affected in the aqueous humor of POAG patients, including the immune response and metabolic and rhythmic processes, in line with a previous study. 27
Changes in DEPs Involved in Pathways and Cellular Compartments
Next, we subjected all DEPs to further KEGG pathway enrichment and cellular compartment analyses. The upregulated DEPs in POAG samples played roles in the metabolism, reactive oxygen species, cellular senescence, and ferroptosis systems (Figure 7). Changes in pathway and cellular compartment components in the aqueous humor of POAG patients. (A) KEGG database analysis identified significantly enriched pathways associated with upregulated DEPs in the aqueous humor of POAG patients. (B, C) KEGG database analysis of enriched cellular compartment components revealed by the upregulated (B) and downregulated (C) DEPs in POAG aqueous humor. (D, E) Volcano plots of DEPs related to exosomes (D) and the mitochondrion (E) Of the 898 exosome-related proteins detected, 107 and 57 were upregulated and downregulated, respectively. Of 224 mitochondrionrelated proteins detected, 70 and 4 were upregulated and downregulated, respectively, as revealed by log2- fold changes > 1.5 or < –1.5, respectively; P < 0.05. (F) Statistical analysis of RAS-associated proteins in the aqueous humors of POAG and cataract patients. Data are means ± SDs, *P < 0.05; **P < 0.01; ***P < 0.001, unpaired Student’s t-test.
Cellular compartment enrichment linked the upregulated DEPs in POAG patients to changes in the levels of cytosolic and nuclear proteins and also the extracellular exosome and mitochondria-associated signatures. The extracellular exosome signature was also enriched in compartment analysis of the downregulated DEPs. Of the 898 exosome-related proteins detected in the MS dataset, 107 and 57 were upregulated and downregulated, respectively; of the 224 mitochondrial proteins detected, 70 and 4 were upregulated and downregulated.
We explored whether proteins, the levels of which were affected by POAG, included those associated with the RAS, which is implicated in both IOP regulation and POAG pathogenesis. 12 Indeed, we detected significant changes in the levels of RAS-related components, including upregulation of AGT (the precursor of AngII). This supports a previous study that showed that AGT is expressed in the aqueous humor of POAG patients. 18 In addition, PRCP levels in aqueous humor markedly decreased, consistent with the results described above (Figure 1). Thus, we conclude that PRCP may regulate the AngII levels in aqueous humor and that changes in PRCP expression contribute to the pathogenesis of POAG.
Discussion
Using contemporary MS techniques, we believe that our study presents the most detailed proteomic landscape of the human aqueous humor derived to date.28,29 A total of 1871 proteins were detected, including 502 DEPs, the levels of which were affected by POAG (396 and 106 upregulated and downregulated DEPs, respectively). This provides a rich source of potentially new biomarkers that will aid further research on the pathogenesis of POAG.
Notably, the exosome-related proteins of the aqueous humor represented the largest group of altered proteins. A total of 164 extracellular exosome proteins were differentially expressed between the control and POAG samples, pointing to the presence of extracellular exosomes in the aqueous humor. Exosomes carry cargo molecules, including proteins, nucleic acids, and bioactive lipids, to target cells, and contribute to eye disease.30-32 They may thus play a role in POAG.
After proteomic profiling, we focused on the linkage between the local eye RAS and the pathogenesis of POAG. This logically followed from previous studies showing that elevated AngII levels in the aqueous humor of POAG patients are positively correlated with the IOP. Mechanistically, we showed that AngII promotes NOX4 expression, increasing ROS levels and thus activating the TGFβ/Smad3 pathway that triggers fibrotic changes in HTMCs. 18 However, the reason why AngII levels were elevated in POAG patients had remained unclear. In our study, Western blotting, ELISA, and proteomic analysis revealed significant downregulation of PRCP expression in the aqueous humor of POAG patients. Moreover, we found no evidence that ACE2, which is best known as the prime regulator of the blood levels of AngII, was involved in the local eye RAS. Importantly, in aqueous humor samples of both control and POAG patients, we observed a strong negative correlation between the PRCP and AngII levels and also between the PRCP level and the IOP. This indicates the existence of a general relationship between PRCP levesl and control of the eye AngII level. When PRCP levels decrease, the ensuing elevation in IOP contributes to the pathogenesis of POAG.
This poses a question: might restoration of PRCP levels alleviate the degenerative changes in the eye that are induced by AngII? We addressed this using both in vitro and in vivo models in which AngII triggers fibrotic changes in TM cells and tissues. In both systems, exogenous PRCP readily rescued the upregulation of key fibrosis-related proteins induced by AngII and the elevation of IOP observed in vivo was similarly abrogated. Thus, our findings suggest that PRCP is a promising therapy for POAG. Notably, the preclinical treatments used here involved intravitreal injections; such invasive procedures are not ideal in terms of chronic maintenance therapy. Hence, further knowledge of why PRCP levels are reduced in POAG eyes may inform other treatment options. It must also be considered that PRCP has functions other than conversion of AngII to Ang1-7. For example, it regulates food intake by inactivating alpha-MSH. 33 Such activities would need to be considered if POAG therapy with PRCP were to be planned.
Limitations
Limitations of this study include several factors. Firstly, we used 10 mice per group in mouse model experiment, it needs big sample size to give a rigorous conclusion. Secondly, we collected 48 patients’ aqueous humor and IOPs to analyze levels of ACE2 and PRCP, and correlation between PRCP levels and IOPs, PRCP levels and AngII levels, these assays also need bigger sample size to test our conclusion. Additionally, we just used 5 POAG patients’ aqueous humor and 5 control patients’ aqueous humor to do MS, it also needed more samples to avoid bias caused by sample collection process.
Furthermore, it should be noted that PRCP enzyme converts AngII to Ang1-7, while Ang1-7 didn’t be involved in this study. AngII dose increased in POAG patients’ aqueous humor whether accompanied with Ang1-7 dose decreased, which need further investigation.
Conclusion
Our study found PRCP, not ACE2 might mainly regulated AngII dose in POAG patients’ aqueous humor. Our data indicated PRCP dose was decreased in POAG patients’ aqueous humor, and it might cause high level of AngII. Restoration of PRCP rescued fibrosis of TM cells in vitro and in vivo, and ameliorated IOP in AngII treatment mouse. Our proteomic findings identify many potential POAG biomarkers that deserve further investigation.
Supplemental Material
Supplemental Material - Decrease of Prolylcarboxypeptidase Dose of Aqueous Humor is Involved in the Pathogenesis of Primary Open-Angle Glaucoma via Finetuning of the Local Ocular Renin-Angiotensin System
Supplemental Material for Decrease of Prolylcarboxypeptidase Dose of Aqueous Humor is Involved in the Pathogenesis of Primary Open-Angle Glaucoma via Finetuning of the Local Ocular Renin-Angiotensin System by Jing Ren, Yuanyuan Xiao, Di Wang, Huiling Cui, Rumeng Zhao, Zilu Guo, Yuhao Wang, Shichao Zhu, Bo Tang, Jing Wang, Gang Wang, Huaying Wang, Xinyuan Hu, Rick F. Thorne, Shichao Duan, Haijun Li in Dose-Response
Supplemental Material
Supplemental Material - Decrease of Prolylcarboxypeptidase Dose of Aqueous Humor is Involved in the Pathogenesis of Primary Open-Angle Glaucoma via Finetuning of the Local Ocular Renin-Angiotensin System
Supplemental Material for Decrease of Prolylcarboxypeptidase Dose of Aqueous Humor is Involved in the Pathogenesis of Primary Open-Angle Glaucoma via Finetuning of the Local Ocular Renin-Angiotensin System by Jing Ren, Yuanyuan Xiao, Di Wang, Huiling Cui, Rumeng Zhao, Zilu Guo, Yuhao Wang, Shichao Zhu, Bo Tang, Jing Wang, Gang Wang, Huaying Wang, Xinyuan Hu, Rick F. Thorne, Shichao Duan, Haijun Li in Dose-Response
Supplemental Material
Supplemental Material - Decrease of Prolylcarboxypeptidase Dose of Aqueous Humor is Involved in the Pathogenesis of Primary Open-Angle Glaucoma via Finetuning of the Local Ocular Renin-Angiotensin System
Supplemental Material for Decrease of Prolylcarboxypeptidase Dose of Aqueous Humor is Involved in the Pathogenesis of Primary Open-Angle Glaucoma via Finetuning of the Local Ocular Renin-Angiotensin System by Jing Ren, Yuanyuan Xiao, Di Wang, Huiling Cui, Rumeng Zhao, Zilu Guo, Yuhao Wang, Shichao Zhu, Bo Tang, Jing Wang, Gang Wang, Huaying Wang, Xinyuan Hu, Rick F. Thorne, Shichao Duan, Haijun Li in Dose-Response
Footnotes
Acknowledgments
The authors would like to extend their gratitude to the patients for their participation in the study. And we also thanks Editor and Reviewers’ efforts to improve the quality of this study.
Author Contributions
Haijun Li and Shichao Duan conceived and designed the experiments. Jing Ren, Yuanyuan Xiao, Di Wang, Huiling Cui, Rumeng Zhao, Zilu Guo, Yuhao Wang, Shichao Zhu, Bo Tang, Jing Wang, Gang Wang, Huaying Wang, Xinyuan Hu performed the experiments. Haijun Li and Shichao Duan analyzed the data. Shichao Duan, Haijun Li, and Rick F. Thorne wrote the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China [U1904166]; Henan Medical Science and technology research plan [SBGJ2018072]; Henan Medical Science and technology research plan joint construction project [LHGJ20220044]; Henan province natural science foundation [242300421284].
Ehical Statement
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request. The proteomic data of aqueous humor analyzed during the current study are available in the supplementary materials. The datasets generated during this study are available at integrated proteome resources iProX with the dataset identifier IPX0006960001.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
