Abstract
Hibiscus mutabilis L (Malvaceae) was a traditional Chinese medicine with significant anti-inflammatory activity. We isolated 3 compounds from the flowers of H mutabilis L , including a new flavonoid, (1″R)-8-(1-(3,4-dihydroxyphenyl)ethyl)-3,3',5,7-tetrahydroxy-4′-methoxyl flavone (
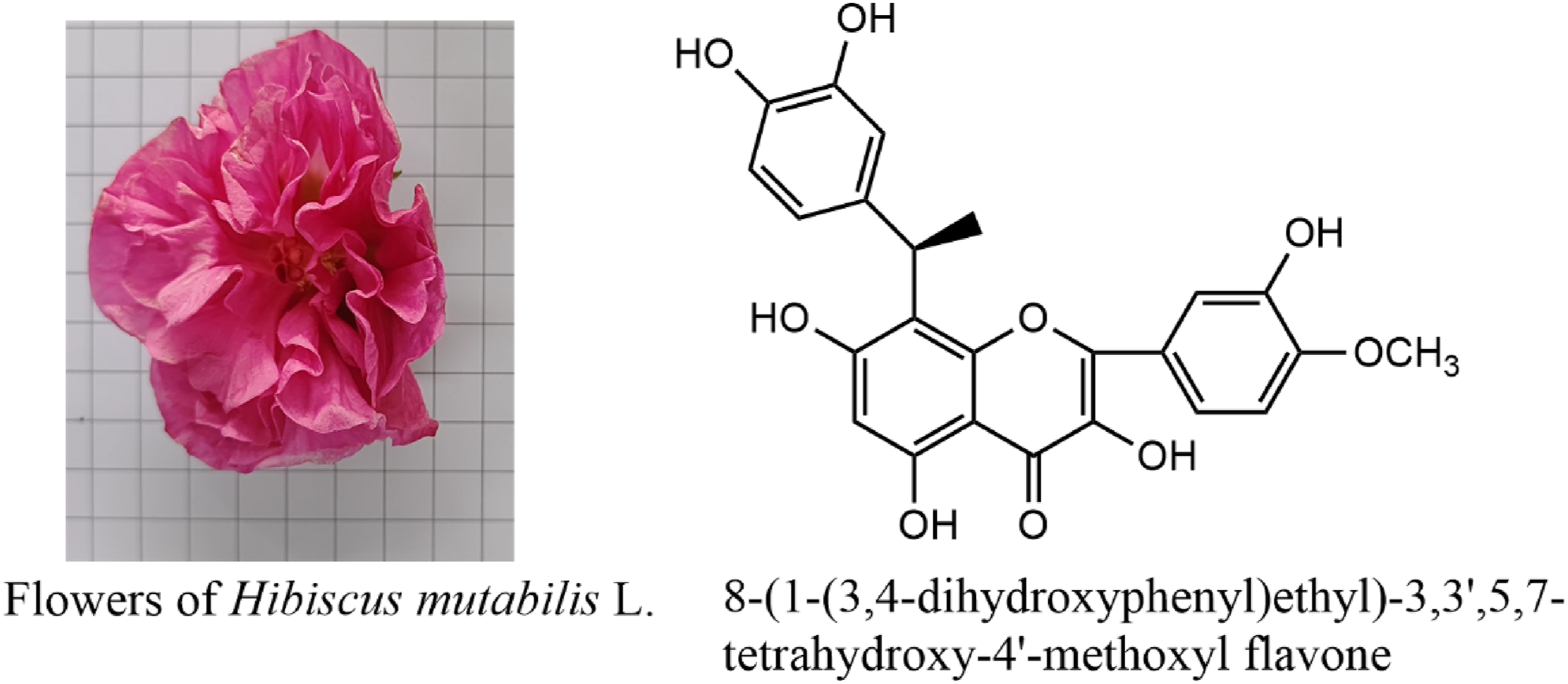
Introduction
Medicinal plants plays a significant role in health care because of their special active ingredients. It was reported that around 80 % of the global population still relies on botanical drugs. 1 Lots of medicinal plants extracts show therapeutic effects to some specific diseases. For example, Astragalus membranaceus (Fisch). Bunge extract is beneficial to prevent asthma, 2 broad-spectrum cannabis oil can ameliorate fibromyalgia, 3 and so on. Medicinal plants contribute significantly to human livelihood and development.
H mutabilis L (Malvaceae) is a medicinal deciduous shrub, originally came from China and widely planted throughout the world.
4
The leaves, flowers and roots of the plant can be used as medicine. H mutabilis L reportedly contains a range of active constituents, including flavonoids, organic acids, amino acids, and other compounds.
5
And flavonoids are considered as the most important active components of H mutabilis L.
6
The separated flavonoids from H mutabilis L display identified bioactivities such as anti-influenza
7
and anti-oxidative activity,
8
among others. This paper shows the isolation and structure elucidation of a new flavonoid, named (1″R)-8-(1-(3,4-dihydroxyphenyl)ethyl)-3,3',5,7-tetrahydroxy-4′-methoxyl flavone ( Structures of compounds 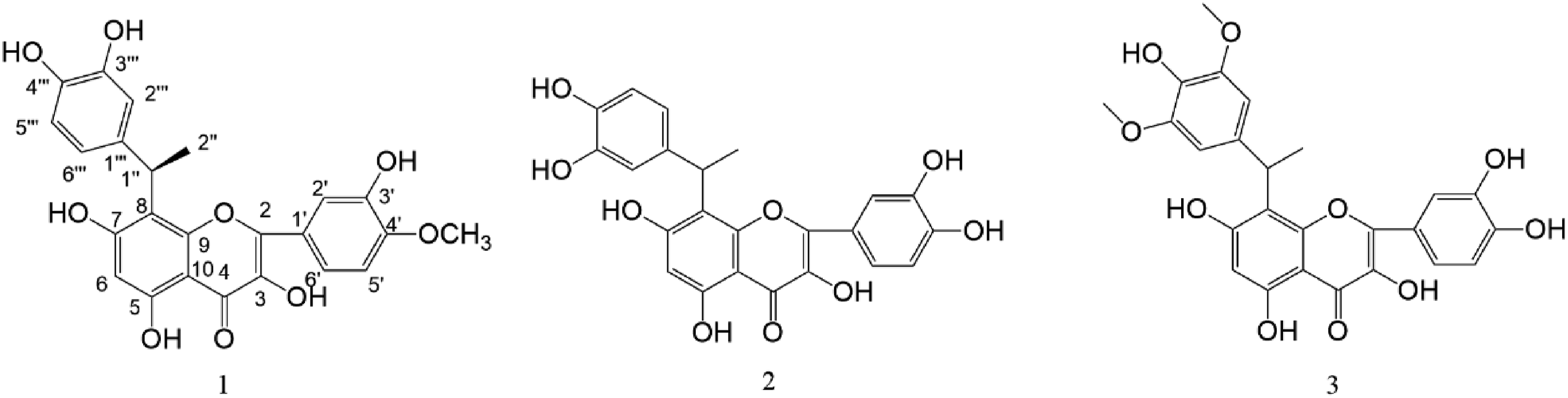
Materials and Methods
General
Column chromatography: silica gel (200-300 mesh, Qingdao hailang silica gel desiccant Inc., China), and MCI gel (8A801, Mitsubishi chemical Inc., Japan). Semi-prep HPLC: 7725i-049, Chengdu laipu technology Inc., DAD detector at 210 and 280 nm, AQ-C18 column (10 × 210 mm, Welch). 1H NMR and 13C NMR spectra: Bruker AVIII HD-600 spectrometer (600 and 151 MHz, resp.). HR-ESI-MS: UPLC-Q-Orbitrap-HRMS spectrometer (Thermo Scientific, USA). Multi label Plate Reader (Spectra MAX M5, Molecular Devices).
Plant Material
We collected the flowers of Hibiscus mutabilis from the H mutabilis L. planting base, Sanhe Town, Chengdu, Sichuan. The flowers were identified by Dr Fei Long (Chengdu University of TCM, Chengdu, China).
Extraction and Isolation
We extracted the air-dried flowers of H mutabilis L. (20 kg) with 90% EtOH (2 × 160 L, 2 h) under reflux. A brown residue (3.34 kg) was obtained after concentrating the EtOH extract under reduced pressure, which was suspended in H2O (3.5 L) and then partitioned sequentially with EtOAc (3 × 3.5 L). Seven fractions (Fr.1∼Fr.7) were obtained from the EtOAc extract (80.0 g) with a gradient elution of petroleum ether-EtOAc (100:1-0:1, v/v). Ten subfractions (Fr.4-1-Fr.4-10) were obtained from Fr.4 (12.64 g). Compound
Brown powder; [α]
Cell Viability
Cell viability was evaluated by CCK-8 assay
9
in RAW264.7 macrophages. The macrophages were plated at a density of 3 × 104 per well into 96-well plates and incubated for 24 h in a humidified atmospheres with 5% CO2 at 37°C. After the addition of
Griess Assay
The effect of
Elisa Assay
Compound
Western Blot Analysis
RAW264.7 macrophages (5 × 105 per well) were seeded in 6-well plates and incubated for 24 h, and then pre-treated with
Results
Structural Elucidation of Compound 1
Compound 1H-1H COSY and HMBC correlations of compound 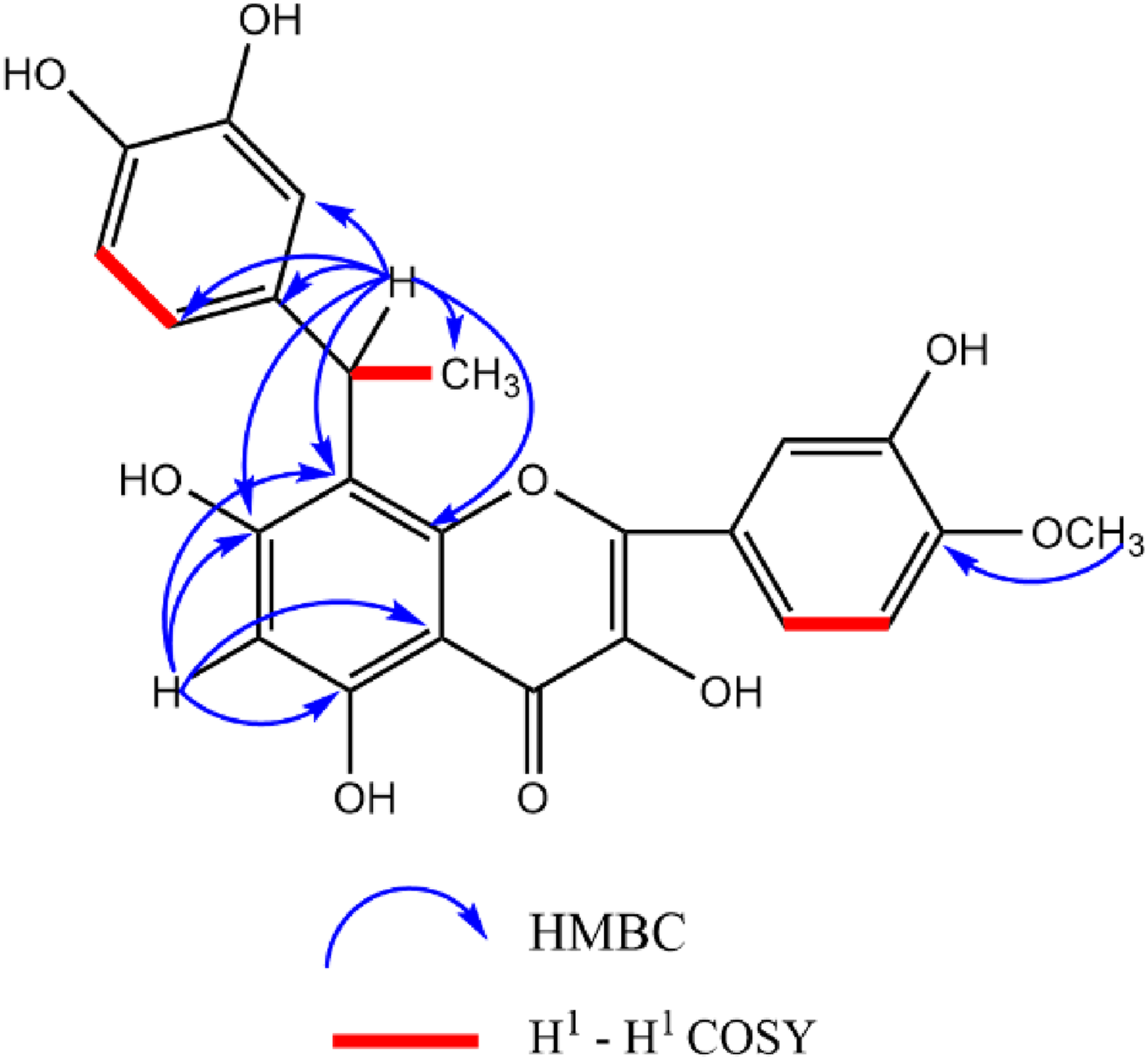
Two known compounds were identified by comparison of spectra data with those reported in the literatures as (1″R)-8-(1-(3,4-dihydroxyphenyl)ethyl)quercetin (
Effect of Compound 1 on Cell Viability of RAW264.7 Macrophages
The potential cytotoxicity of Cell viability of compound 
Effect of Compound 1 on NO Production in RAW264.7 Macrophages
The levels of NO production in macrophages were measured using the Griess reagent Kits. Treatment with LPS markedly increased NO production in macrophages. Administration of The inhibitory effect of compound 
Effect of Compound 1 on IL-6 and TNF-α Production in RAW264.7 Macrophages
The levels of IL-6 and TNF-α in RAW264.7 cells were investigated using the Elisa Kits. In comparison with the control group, LPS treatment increased IL-6 and TNF-α concentrations (Figure 5). Treatment with The inhibitory effect of compound List of Abbreviations.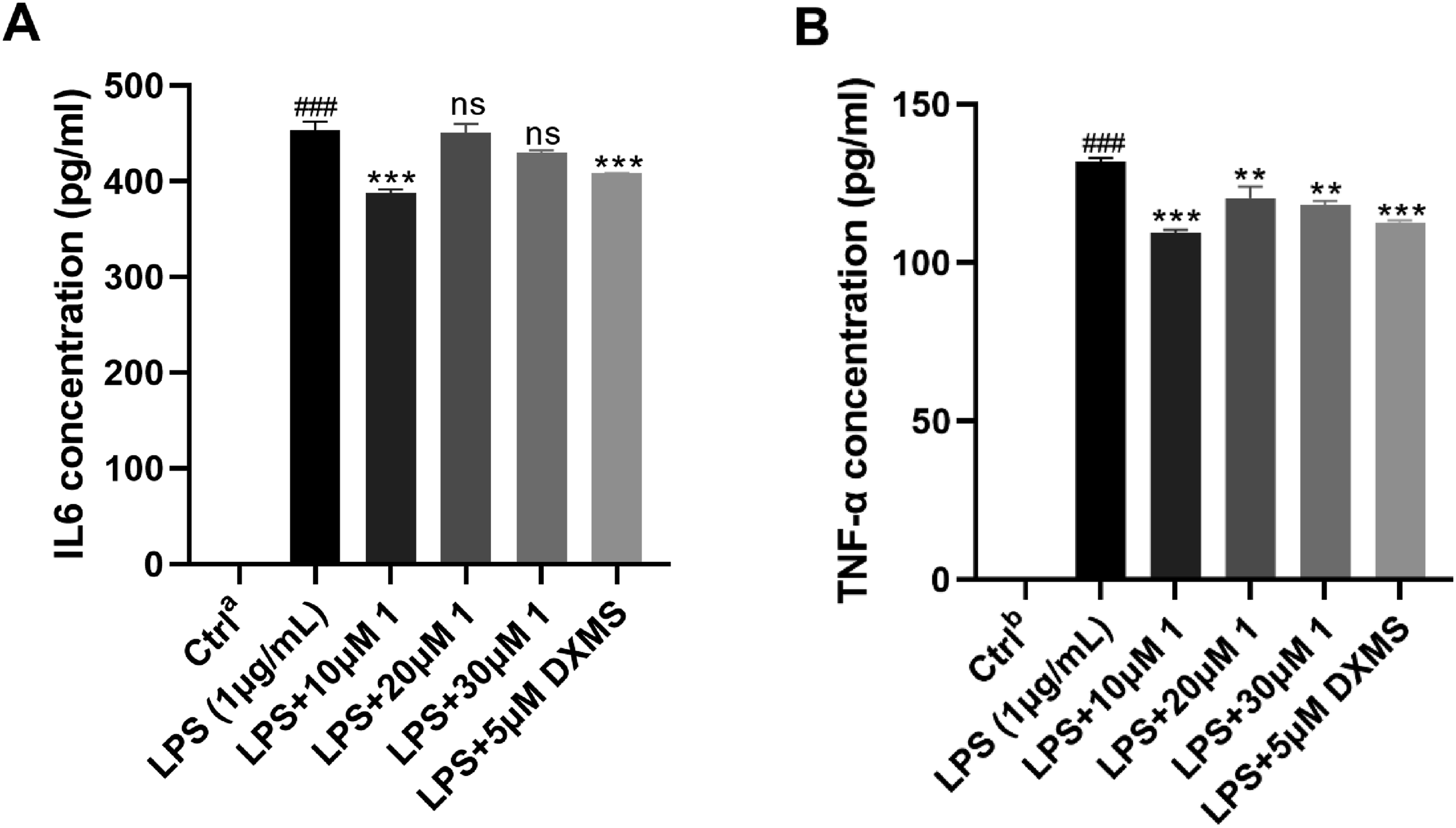

Effect of Compound 1 on iNOS, COX-2, P65 and p-P65 Protein Expression in RAW264.7 Macrophages
We carried out Western blot analysis on RAW264.7 macrophages to explore the potential anti-inflammatory mechanism of Effects of compound 
Discussion
Hibiscus mutabilis L was a traditional Chinese medicine with significant anti-inflammatory activity, normally applied to the clinical treatment of various inflammation-related diseases such as epidemic parotitis. 15 At present, there are few reports on the anti-inflammatory effect of H mutabilis L, and it’s basic research is weak. The present study aimed to investigate the potential anti-inflammatory effect of a new flavonoid extracted from H mutabilis L. The results revealed that the flavonoid inhibits LPS-induced IL-6 and TNF-α release and iNOS and COX-2 expressions in RAW264.7 cells, suggesting its potential as a therapeutic agent for inflammation treatment. Furthermore, our results realed that the critical mechanism by the flavonoid may exert it’s anti-inflammatory effect is through the down-regulation of NF-κB signaling pathway.
The study findings demonstrate that the lowest dose of the flavonoid exert a greater inhibitory effect to IL-6 and TNF-α vs the other 2 higher concentrations. This result indicate that the anti-inflammatory effect of the compound may be not dose-dependent. The sesult might be related to the complex mechanism by which flavonoids act on cells. First, flavonoids can affect cellular immune responses through multiple signaling pathways and contribute to the expression of downstream key immune proteins. Meanwhile, downstream key proteins may also act on cells through certain positive and negative regulatory mechanisms. Our results only demonstrate that the flavonoid can act on immunity through NF-κB signaling pathway. However, it is reported that flavonoids also can affect immunity via TLR, MAPK and other signaling pathway. 16 Second, the flavonoid may also act on other targets to affect related signaling pathway through crosstalk, thereby regulating the secretion of IL-6 and TNF-α. In addition, different types of cell lines also have different responses to the same drug. All of the above reasons may lead to a non dose-dependent response of the flavonoid to IL-6 and TNF-α in RAW264.7 cells.
Furthermore, except for IL-6, other inflammation related immune factors are significantly inhibited. And IL-6 can also be significantly inhibited at lowest dose of the flavonoid. The results suggests the flavonoid’s potential as a targeted therapeutic agent against inflammation. Due to various reasons, this article only detected key targets on limited signaling pathways, and further exploration is needed to fully elucidate the mechanism of the flavonoid’s anti-inflammatory effect.
In a word, our study revealed the potential of the flavonoid as an inflammatory therapeutic agent. Despite the interesting finding, our study has a few limitations. On the 1 hand, the study was conducted in vitro using RAW264.7 macrophages. Therefore, our study finding may not reflect anti-inflammatory effect of the flavonoid in vivo or in other cell lines. So it is necessary to further evaluate the anti-inflammatory effect using animal models or other cell lines. On the other hand, our study only analyze the effect of the flavonoid on downstream proteins in the NF-κB signaling pathway. Further mechanistic studies are needed to fully elucidate the specific targets and signaling pathways invoved.
Finally, we hope that our finding can provide reference for future studies.
Conclusion
In this study, 1 new flavonoid, named (1″R)-8-(1-(3,4-dihydroxyphenyl)ethyl)-3,3',5,7-tetrahy-droxy-4′-methoxyl flavone (
Footnotes
Author Contributions
P.-H. and L.-Y. have isolated and identified the compounds. Y.X.-G. and X.-Y. have analyzed the experimental data of plant chemistry. The bioactivity of compound 1 was evaluated by X.Q.-S and X.-W. The detail of the experiments were designed by P.-H. The manucript was written by L.-Y. under the supervision of P.-H.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

