Abstract
Neurada procumbens Linn. possesses a wide range of phytochemical and bioactive entities such as flavonoids and polyphenols that decrease the oxidative stress and enhance synaptic plasticity, resulting in accelerated healing processes. Almost all parts of the plant have been used in the traditional system of medicine for various disorders including its use as an anticonvulsant, sexual tonic, and anti-rheumatic agent. This study is designed to evaluate the anxiolytic and antidepressant activities of the methanolic crude extract of N. procumbens (Np.Cr) based on its use in traditional medicine and presence of the phytochemical constituents. Aqueous methanolic extract of Np.Cr was prepared under reduced pressure using rotary evaporator, and different chemical constituents were identified by phytochemical screening. Light/dark exploration, elevated plus maze (EPM), and hole board tests were used to assess the anxiolytic activity, while forced swim and tail suspension tests were used to assess the antidepressant potential of the crude extract. Treatment groups treated individually with 3 different doses; i.e., 50, 100, and 200 mg/kg of Np.Cr, showed a dose-dependent increase in time spent in light compartment and in open arms as well as increased number of head poking by the experimental animals. Np.Cr showed significant antidepressant potential (P < .05) as evident from marked decrease in behavioral despair. Findings of the present study assure that N. procumbens possess significant anxiolytic and antidepressant potential with the demands of further investigations to identify the active compound(s) responsible for these pharmacological effects.
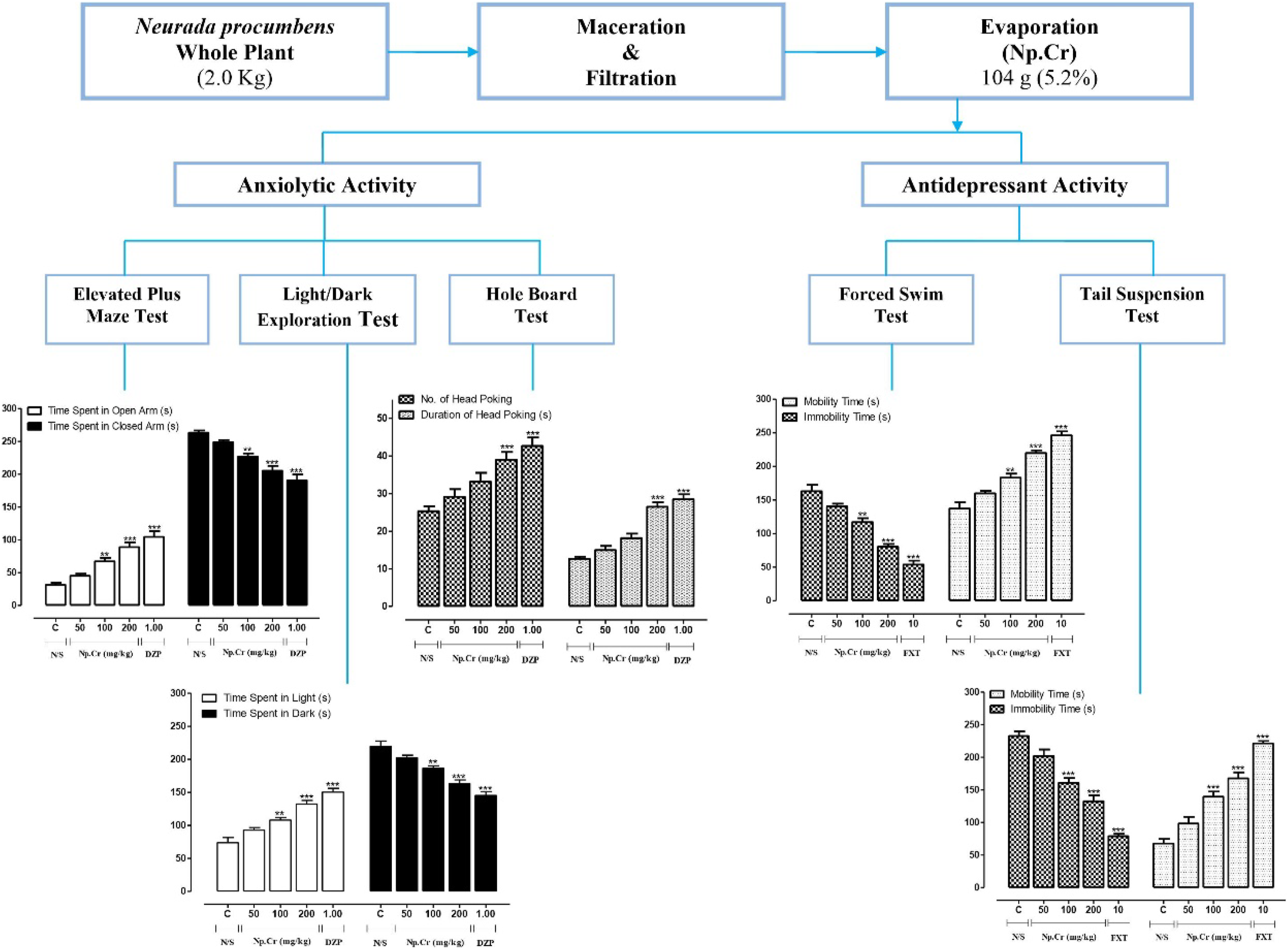
Introduction
Anxiety, the most common psychiatric disorder, may be defined as an uncomfortable feeling of vague fear or apprehension, affecting almost one fourth of the population at least once in lifetime. 1 Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) identifies generalized anxiety disorder (GAD) as the most common anxiety disorder among the 6 main types of anxiety disorders.2,3 Depression though considered as a single clinical identity, it is a group of several different disorders, each with its own definition, typical signs and symptoms, diagnostic criteria, and treatment. Depression is often defined in terms of major depressive Disorder and is characterized by depressed mood or loss of interest or pleasure in daily activities, with at least 4 additional symptoms of depression from amongst the depressed mood, loss of interest, fatigue, insomnia, changes in weight or appetite, difficulty in concentrating, and suicidal thoughts.2,3 Both anxiety and depression, being undertaken in present research, do not have any cure though treatments exist. An additional problem with the treatment of depression is its relapse. These disorders have long been present and people tried to treat them with different herbs. Almost three fourth of the world population still uses herbs for the treatment of various disorders and illnesses because plants offer a cheap source of drugs with high therapeutic value. 4 Herbal treatments could be viewed as supplemental or alternative treatments. Research on medicinal plants has advanced globally, proving the pharmacological potency of many plant species in a range of animal models in the pursuit of novel compounds useful for the treatment of neurological illnesses. 5 The vast majority of herbal medications whose psychotherapy potential has been evaluated in a variety of animal models reflect this. These investigations have given valuable information for the creation of new isolated active phytoconstituents and new pharmacotherapies derived from medicinal plants.6,7 The aim of the present study was to screen the indigenous medicinal plants, especially of Cholistan origin, for their anxiolytic and antidepressant potential, as Cholistan Desert is enriched with diverse flora of plants. Neurada procumbens Linn. (Neuradaceae) commonly known as “Chapri” was selected on the basis of its conventional use by the inhabitants of Cholistan Desert. The plant has scientifically been explored for various pharmacological activities like antioxidant, acetylcholinesterase inhibitory, tyrosinase inhibitory, α-amylase inhibitory, and α-glucosidase inhibitory activity. 8 Scientific exploration of this plant may open up cheap and effective treatment options for anxiety and depression.
Materials and Methods
Plant Material
Whole plant of N. procumbens Linn. was collected from the desert area of Bahawalpur region of Pakistan, and identified by botanist. Plant specimen was preserved in the herbarium of the Department of Pharmacology, Faculty of Pharmacy, the Islamia University of Bahawalpur, Pakistan, and the voucher number (NP-WP-05-15-94) was kept for future reference. The plant was dried under shade after washing, and any extraneous matter was removed carefully.
Preparation of the Crude Extract
Coarse grinding of plant material was carried out with a grinder. Ground material was then macerated with 70% methanolic water for 3 days at room temperature. Macerated plant material was first filtered through muslin cloth and then through filter paper. Residue was subjected to soaking and filtration for two more times. Filtrate was then subjected to evaporation, using a rotary evaporator (Heidolph Laborota 4000 efficient, Germany), at low temperature and under reduced pressure. Thick viscous paste of crude extract of plant was further dried in hot air oven at 40°C. The obtained semisolid extract was then weighed, labeled, and stored at a temperature below 0°C, in an air tight container. Percent yield of the extract was calculated. 9
Chemicals
Pure chemicals of research-grade were used in the study. Chemicals including diazepam and fluoxetine were purchased from Sigma-Aldrich, USA.
Drugs
All the doses of plant extract (Np.Cr), diazepam, and fluoxetine were prepared in normal saline and administered as 10 mL/kg via intraperitoneal route.
Phytochemical Screening
Preliminary Phytochemical Examination
Secondary metabolites of plants are responsible for the biological activities exhibited by the plants. The crude extract of N. procumbens was screened for different primary and secondary phytochemical constituents like alkaloids, glycosides, saponins, tannins, flavonoids, terpenes, carbohydrates, and proteins.10,11
HPLC Analysis
Stock solutions of reference standards were made and then diluted with ethanol to reach a final concentration of 50 μg/mL. Ethanol was used as a solvent for polyphenols at a concentration of 10 mg/mL. Samples were made fresh before analysis or kept at 4°C if not to be tested for more than 1 hour. Detector (UV-Vis), column C18 (25 cm × 4.6 mm, 5.0 μM), and SIL-20A auto sampler (Shimadzu Scientific Instruments, Kyoto, Japan) were used. The analytical method employed a linear gradient system which consisted of 1% (v/v) acetic acid (solvent A) in water and ethanol (solvent B). The gradient elution started with 15% for 0–15 minutes, 45% for 15–30 minutes, and 100% for 30–45 minutes (end analysis). The flow rate was 1 mL/min, and absorbance was detected at 280 nm. Identification and quantitative analysis were conducted by comparison with standards.12,13
Total Phenolic Content
The Folin–Ciocalteu method was selected to measure TPC of the extract. A standard curve was plotted using gallic acid for calibration. In 50% ethanol solution, different concentrations of gallic acid were prepared. 1 mL of extract (1 mg/mL) was mixed with 1 mL of Folin–Ciocalteu’s phenol reagent. After 5 minutes, 10 mL of 7% Na2CO3 solution was added to the mixture with the addition of 13 mL of distilled water and mixed thoroughly. Mixture was kept in dark for 60 minutes after which the absorbance was recorded at 750 nm. Results are expressed in mg of gallic acid equivalent (mg GAE/g) per gram of dried sample.
Total Flavonoid Content
Total flavonoid content was determined by using rutin (quercetin) as a standard. Quercetin was used to make the calibration curve. Different concentrations of quercetin were prepared in 50% of ethanol solution. Then to 1 mL of extract (1 mg/mL), 0.5 M NaNO2 and 0.3 M AlCl3.6H2O were mixed. 1 M NaOH was added after 5 minutes. Solution was mixed thoroughly, and absorbance was measured at 506 nm. The total flavonoid content was expressed as mg quercetin equivalent per gram (mg QE/g) through the calibration curve.
Free Radical Scavenging Activity by DPPH Assay
Free radical scavenging activity of crude extract of N. procumbens was measured by using DPPH. In brief, 50 mL of DPPH (0.2 mg/mL) in ethanol was prepared. This solution (2 mL) was added to 1 mL of extract (0.5 mg/mL) in ethanol at different concentrations. The mixture was shaken vigorously and allowed to stand at room temperature for 30 minutes. Absorbance was measured by using spectrophotometer at the wavelength of 517 nm. Reference standard compound being used was ascorbic acid, and experiment was carried out in triplicate.14,15
Experimental Animals
Male Swiss albino mice were housed in the animal house of pharmacology research lab, Faculty of Pharmacy, the Islamia University of Bahawalpur. Standard laboratory conditions (12 hours light/dark cycle at 22 ± 2°C) were maintained for animals, and they were fed with standard rodent pellets and water ad libitum. Guidelines approved by Pharmacy Research Ethics Committee of the Islamia University Bahawalpur were followed during studies.
Acute Toxicity Assay
Guidelines of Organization for Economic Co-operation and Development (OECD) were followed to perform acute toxicity studies. Male Swiss albino mice (18–30 g) were divided into 5 groups, consisting of 5 mice each. Normal baseline behavioral parameters of each mouse were observed and recorded. Experimental animals were fasted overnight but received water ad libitum. One group of mice received normal saline (10 mL/kg) and other 3 groups received the plant extract (Np.Cr) at the doses of 1, 3, and 5 g/kg i.p., respectively. Behavioral changes and response parameters, that is, alertness, grooming, hyperactivity, convulsion, sweating, lacrimation, urination, rightening reflex, corneal reflex, pain response, touch response, gripping strength, and mortality, were observed and recorded at .5, 1, 2, 4, 6, 12, 24, and 48 hours. 16
Anxiolytic Activity of Crude Extract of Neurada procumbens
For evaluation of anxiolytic activity in all the tests, experimental animals were divided into 5 groups consisting of 6 mice each. Control group received normal saline (10 mL/kg, i.p.) and standard group received diazepam (1 mg/kg i.p.). Three treatment groups were treated with Np.Cr at the doses of 50, 100, and 200 mg/kg (i.p.), respectively.
Light/Dark Exploration Test
The apparatus consisted of two adjoining boxes (25 × 25 × 25 cm3) with a wall between them. One box was covered at the top and was painted black (dark room), and the other box was open from the top and was painted white (light room). The light box was illuminated with 40-watt lamp placed at 25 cm height. The center wall had a hole through which mouse could freely pass into both the boxes. Apparatus was placed in a room having dim illumination. Animals were treated with respective group treatment, and after 30 minutes, they were placed on apparatus at the hole, facing the dark box. The time (in seconds) spent by each mouse in light and dark box was recorded for 5 minutes. 17
Elevated Plus Maze Test
EPM test is the most widely used and validated test to measure anxiety in rodents. 18 Apparatus consisted of 4 arms of which two were open and two were closed. Open arms (35 × 5 cm2) were crossed with closed arms (35 × 5 × 20 cm3) at a center point (5 × 5 cm2). EPM was elevated to a height of 50 cm from the ground and placed in a room having dim illumination. Animals were treated with respective group treatment, and after 30 minutes, they were individually placed on EPM apparatus at the center, facing one of the closed arms. The time (in seconds) spent by each mouse on open and closed arms was recorded for 5 minutes. 18
Hole Board Test
Apparatus consisted of a wooden chamber (40 × 40 × 25 cm3) having 16 evenly spaced holes on it (each of 2.5 cm in diameter) through which mice could peep. Apparatus was elevated to a height of 25 cm from ground. Apparatus was placed in a room having dim illumination. Animals were treated with respective group treatment, and after 30 minutes, they were placed on hole board apparatus. The number of head pokes and duration of head pokes by each mouse were recorded for 5 minutes. 19
Antidepressant Activity of Crude Extract of Neurada procumbens
For evaluation of antidepressant activity in all tests, experimental animals were divided into 5 groups consisting of 6 mice each. The control group received normal saline (10 mL/kg, i.p.), and the standard group received fluoxetine (10 mg/kg i.p.). Three treatment groups were treated with Np.Cr at the doses of 50, 100, and 200 mg/kg (i.p.), respectively.
Each group of mice was treated i.p. as described above.
Forced Swim Test
Among the in vivo tests available for measuring antidepressant activity, FST is the most widely used and validated test. 20 Apparatus consisted of transparent plexiglass cylinder (20 cm × 12 cm). It was filled with water (24 ± 1°C) to 15 cm depth. 20 Animals were given a pretest swimming session of 15 minutes each, 24 hours before final test session. Animals were treated with respective group treatments immediately after pretest session, 6 hours before final test session, and 30 minutes before final test session. After 30 minutes of final dose or 24 hours after pretest session, each animal was individually placed in the plexiglass cylinder for final swimming session of 5 minutes each. Duration of immobility was noted for each animal for 5 minutes. Mouse was considered immobile when no attempts were made to escape, other than movements necessary to keep its head out of water. 21
Tail Suspension Test
TST apparatus consisted of wooden chamber (70 cm high). A rod was fitted between side walls of the chamber, at a height of 60 cm from ground or 10 cm from the top of the apparatus. Animals were hung with the rod by placing adhesive tape one inch from the tip of tail. Animals were given a pretest session of 15 minutes each, 24 hours before final test session. Animals were treated with respective group treatments immediately after pretest session, 6 hours before final test session, and 30 minutes before final test session. After 30 minutes of final dose or 24 hours after pretest session, each animal was individually hung with rod for final test session of 5 minutes each. Duration of immobility was noted for each animal for 5 minutes. Mouse was considered immobile when it passively hung with rod with no efforts to escape. 22
Results
Phytochemical Screening
Preliminary Phytochemical Examination
Phytochemical analysis revealed the presence of variety of metabolic constituents like saponins, flavonoids, phenols, coumarins, terpenes, tannins, and phlobatannins in the crude extract of N procumbens (Np.Cr).
HPLC Analysis
The HPLC fingerprinting confirmed the presence of quercetin (Rt. 2.77 minutes), gallic acid (Rt. 3.76), chlorogenic acid (Rt. 8.48), vanillic acid (Rt. 13.12), p-coumaric acid (Rt. 17.64), and m-coumaric acid (Rt. 21.22) in the crude extract of N procumbens (Figure 1). HPLC peak graph of the crude extract of Neurada procumbens (Np.Cr).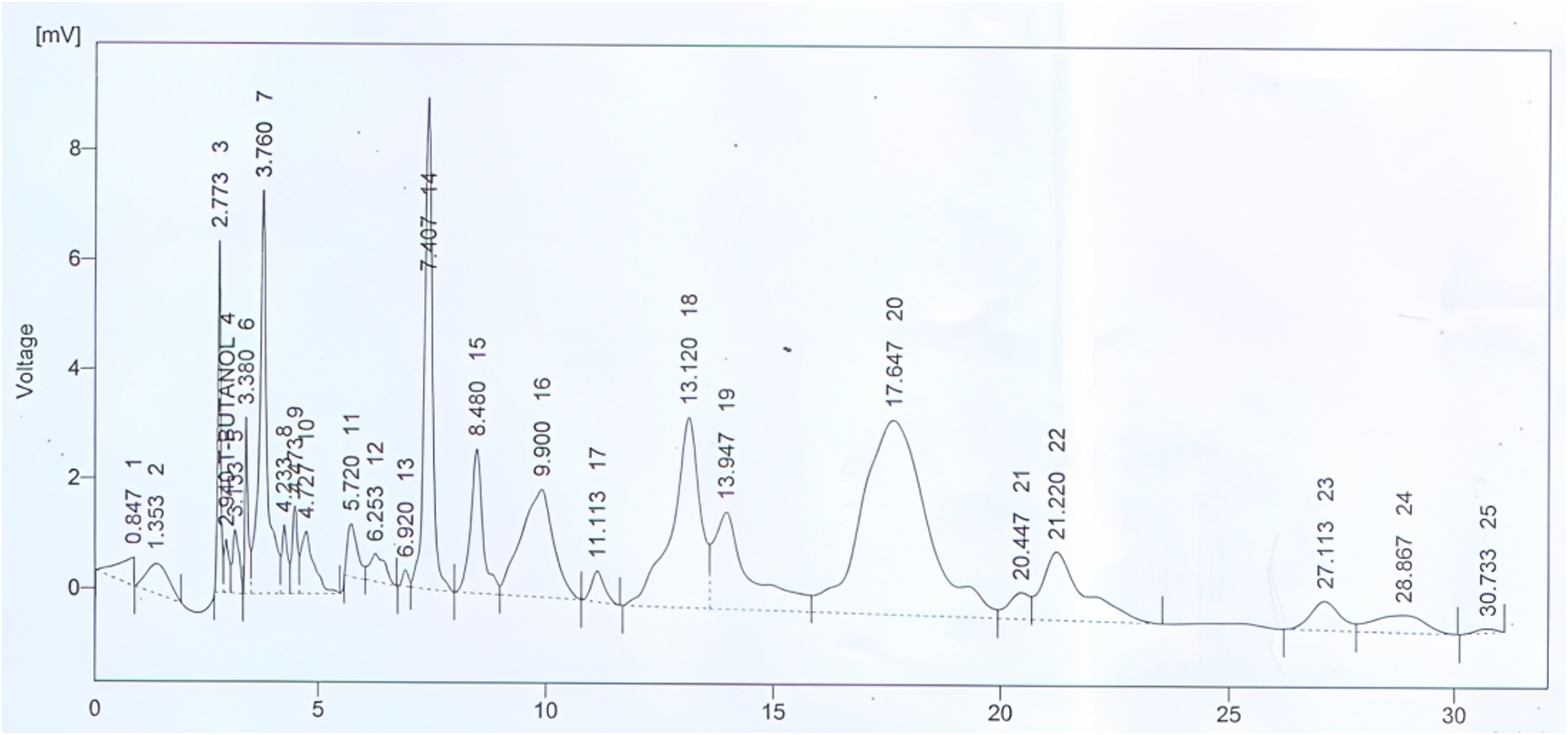
Total Phenolic Content
The crude extract of N. procumbens (Np.Cr) was found to possess phenolic contents 78.8 ± 2 mg GAE/g of the extract.
Total Flavonoid Content
The crude extract of N. procumbens (Np.Cr) was found to possess flavonoid contents 63.5 ± 1.6 mg quercetin equivalent/g of the extract.
Free Radical Scavenging Activity by DPPH Assay
Free Radical Scavenging Activity of Ascorbic Acid and the Crude Extracts of Neurada procumbens (Np.Cr) in DPPH Assay.

The results are expressed as mean ± SEM of 3 observations for each sample, and values are expressed as percent of inhibition.
Acute Toxicity Assay
Np.Cr was found to be safe up to the dose of 5 g/kg in acute toxicity studies conducted in mice. No signs of toxicity and mortality were observed up to 5 g/kg dose.
Anxiolytic Activity of Crude Extract of Neurada procumbens
Light/Dark Exploration Test
Np.Cr increased the average time spent by mice in the light box and decreased the time spent in the dark box. For treatment groups of Np.Cr at 50, 100, and 200 mg/kg, the average time spent in light was recorded to be 108.7 ± 3.56 seconds, 132.2 ± 5.56 seconds, and 139.5 ± 3.75 seconds, respectively, while the average time spent in dark was 186.2 ± 3.21 seconds, 163.0 ± 5.65 seconds, and 156.3 ± 3.62 seconds, respectively (Figure 2). Effects of the crude extract of Neurada procumbens (Np.Cr) and diazepam (DZP) on the time spent in light and dark in light/dark exploration test. All values are given in mean ± SEM. N = 6. P < .001 (***) and P < .05 (*) as compared to control (ANOVA followed by Bonferroni test).
Elevated Plus Maze Test
Np.Cr showed the dose of 100 mg/kg and 200 mg/kg showed significant anxiolytic potential in the treatment groups that received the doses of 100 mg/kg and 200 mg/kg, by increasing the time spent in open arms (67.17 ± 5.25 seconds and 88.83 ± 7.14 seconds) and decreasing the time spent in closed arms of EPM (226.7 ± 5.03 seconds and 205.3 ± 7.16 seconds). Results of Np.Cr 200 mg/kg were highly significant (Figure 3). Effects of the crude extract of Neurada procumbens (Np.Cr) and diazepam (DZP) on the time spent in open and closed arms in elevated plus maze test. All values are given in mean ± SEM. N = 6. P < .001 (***) and P < .05 (*) as compared to control (ANOVA followed by Bonferroni test).
Hole Board Test
Effects of Np.Cr on Number and Duration of Head Poking in Hole Board Test.

All values are given in mean ± SEM. N = 6. P < .001 (***) and P < .05 (*) as compared to control (ANOVA followed by Bonferroni test).
Antidepressant Activity of Crude Extract of Neurada procumbens
Duration of mobility and immobility exhibited by mice of standard and treatment groups in 5 minutes (300 seconds) swimming session were compared with that of the control group. Fluoxetine markedly decreased the duration of immobility and increased the duration of mobility in FST and TST.
Forced Swim Test
Duration of mobility was increased and duration of immobility was decreased by Np.Cr. The duration of mobility by mice treated with Np.Cr at 100 mg/kg and 200 mg/kg was found to be 198.2 ± 5.91 seconds (P < .01) and 229.7 ± 4.12 seconds (P < .001), respectively, while the duration of immobility was found to be 101.8 ± 5.91 seconds (P < .01) and 70.33 ± 4.12 seconds (P < .001), respectively (Figure 4). Effects of the crude extract of Neurada procumbens (Np.Cr) and fluoxetine (FXT) on duration of mobility and immobility in forced swim test. All values are given in mean ± SEM. N = 6. P < .001 (***) and P < .05 (*) as compared to control (ANOVA followed by Bonferroni test).
Tail Suspension Test
Effects of Np.Cr on Duration of Mobility and Immobility in Tail Suspension Test.

All values are given in mean ± SEM.N = 6. P < .001 (***) and P < .05 (*) as compared to control (ANOVA followed by Bonferroni test).
Discussion
The study was designed to explore the anxiolytic and antidepressant activity of N. procumbens based on its traditional use and phytochemicals reported. The potential of various extracts and naturally occurring chemicals in the prevention/treatment of chronic diseases has received a lot of attention.5,7 Extracts from plants may contain a variety of substances with advantageous biological characteristics, forming complex combinations with multi-target actions that are able to concurrently block or modulate a large number of important targets.23,24 Preliminary estimation of total phenols and flavonoids as well as HPLC was performed for phytochemical analysis of the Np.Cr. Phytochemical analysis of aqueous methanolic extract of N. procumbens revealed the presence of flavonoids, phenols, saponins, coumarin, and tannins. HPLC analysis of Np.Cr was used to explore the presence of plant constituents that may be responsible for therapeutic effects. Quantification of flavonoids and phenolic compounds was performed as both these plant constituents have been reported to exert a variety of pharmacological effects. Flavonoids, alkaloids, saponins, and phenolic acids of different plants have been shown to possess anxiolytic and antidepressant properties. 25 Flavonoids have inhibitory effects on monoamine oxidases, as alteration in monoaminergic system and enhancing GABAergic transmission are related to neurodegenerative diseases such as psychiatric disorders including depression and anxiety. 26 Quercetin has been shown to protect against stress-induced anxiety and depression in mice by increasing the serotonin concentration in the brain. 27 Phenols are known for their antioxidative and neuroprotective properties. For instance, different polyphenolic compounds have been shown to produce antidepressant-like effects in rodents and human by increasing synaptic plasticity and promoting adult hippocampal neurogenesis.28,29 Gallic acid shows a protective action against arsenic-induced anxiety and depression-like effects as well as memory impairment. 30 Vanillic acid produces antidepressant effects by the activation of Akt or ERK signaling pathway. 31 Drugs that increase serotonin level in the central nervous system such as selective serotonin reuptake inhibitors (SSRIs) have been used to delay ejaculation in the treatment of premature ejaculation.32,33 Short-acting SSRIs such as dapoxetine have been investigated for on-demand use in premature ejaculation. 34 Traditional use of N procumbens as sexual tonic points toward the possibility that the plant may also increase the serotonin level in the central nervous system as done by the selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs). Abnormalities in the functioning of serotonergic pathways are involved in the pathogenesis of depression and anxiety. 35 Elevated serotonin levels produce anxiolytic and antidepressant effects in human body. 36 As most of the antidepressant drugs aim at restoring the serotonin levels and functioning in the brain, so the underlying mechanisms for anxiolytic and antidepressant effects of the plant may be the increase in serotonin levels, that is, similar to SSRIs and SNRIs. Moreover, the phytoconstituents in the plant may be responsible for the biological effects of the crude extract of N. procumbens observed in this investigation. Our early phytochemical analysis identified flavonoids and polyphenols in the plant extract. Certain flavonoids have been found to bind to the GABA receptor’s benzodiazepine region with a high degree of affinity. Their overall bioavailability and, more specifically, their presence in the brain in vivo, appears to be key factors in how their effects on the CNS manifest. The effects on the CNS could possibly be explained by the presence of flavonoids in the crude extract of N. procumbens.
Flavonoids and phenols are most probably the phytochemicals responsible for the anxiolytic and antidepressant effects of N. procumbens, but further studies are required to isolate the flavonoids and phenols from Np.Cr in order to study the anxiolytic and antidepressant effects of individual chemical constituents and to establish the mechanism by which these constituents exert such effects.
Conclusion
The current study demonstrates the phytochemical profiling and anxiolytic and antidepressant activities of crude methanolic extract of N. procumbens. The phytochemical profiling shows the presence of alkaloids, flavonoids, phenols, saponins, and tannins that are associated with a wide variety of pharmacological activities. In vivo studies showed significant anxiolytic and antidepressant potential at the doses tested. Thus, present study validated the use of the plant in the management of anxiety and depressive disorders with the need of further exploration of phytochemicals of the plant.
Footnotes
Acknowledgments
The authors acknowledged the technical staff of their research laboratory for their assistance in carrying out the experiments.
Author Contributions
Ahmed Awais Khalid planned the project and performed the experiments under the supervision of Prof. Dr. Qaiser Jabeen. Faraza Javaid helped in the evaluation of acute toxicity of plant extract, performance of anxiolytic activity, and manuscript preparation. All authors read carefully and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
