Abstract
Objective
Ciprofol is a new sedative anesthetic drug that can be used for gastrointestinal endoscopy and induction of general anesthesia, but the appropriate dosage for use in elderly patients has not been determined. Sufentanil is a commonly used opioid in clinical practice, and this study was designed to induce anesthesia in elderly patients using sufentanil in combination with ciprofol. However, the optimal dosage of ciprofol when it is co-administered with sufentanil has not yet been established. This study was designed to find the median effective dose (ED50) and 95% confidence interval (95% CI) of ciprofol for intravenous anesthesia when combined with sufentanil.
Methods
We studied 57 patients who were scheduled to undergo a diagnostic upper gastrointestinal endoscopy. According to age, it was divided into two groups: 65∼74 years old (group A) and over 75 years old (group B). Using the modified Dixon sequence test method, intravenous bolus of 0.1 μg/kg sufentanil was given 3 min before ciprofol is administered, the initial dose of ciprofol was 0.4 mg/kg, the upper gastrointestinal endoscopy was placed after reaching the depth of sedation, and vital signs and adverse events were recorded at each perioperative time point (T0-T7).
Results
In the group A, when combined with 0.1 μg/kg sufentanil, the ED50 of ciprofol to inhibiting responses to insertion of upper gastrointestinal endoscopy was 0.23 mg/kg, and the 95% CI was 0.09∼0.30 mg/kg; in the group B, the ED50 was 0.18 mg/kg, and the 95% CI was 0.13∼0.22 mg/kg.
Conclusion
The ED50 of ciprofol in combination with sufentanil (0.1 μg/kg) for upper gastrointestinal endoscopy in elderly patients: 0.23 mg/kg in group A and 0.18 mg/kg in group B.
Introduction
Gastrointestinal endoscopy is the common and reliable diagnostic and treatment method for gastrointestinal diseases. 1 However, as an invasive treatment method, routine gastrointestinal endoscopy without sedation/anesthesia often causes varying degrees of pain, discomfort, stress, and anxiety to patients, leading them to fear and even refuse gastrointestinal examinations. Pain-free gastrointestinal endoscopy reduces the patient’s fear of the procedure on the one hand, and greatly reduces the patient’s stress response on the other, making it less difficult for the endoscopist to perform the procedure and increasing the positive detection rate of gastric cancer. Due to these advantages, painless gastrointestinal endoscopy is now widely used in clinical practice. 2
Ciprofol, the latest 2,6-disubstituted phenol derivative, was independently developed in China and is currently used for painless gastrointestinal endoscopy and anesthesia induction. 3 Ciprofol is a new intravenous anesthetic drug with rapid onset of action, smooth and rapid awakening, complete recovery of consciousness, and low incidence of injection pain.3,4 Although the sedative effect of ciprofol is similar to that of propofol, its absorption, distribution, metabolism, and excretion processes are faster, the incidence of hypotension and respiratory depression is low, and it is safe. 5 Its indications include sedation during gastrointestinal endoscopy, induction of general anesthesia, etc. After phase II and III clinical trials, the instructions recommend a dose of up to 0.4 mg/kg of ciprofol for GI endoscopy,4,6-8 but the dose may vary between ages and when different drugs are used in combination.
Sufentanil, as the opioid with the strong analgesic effect at present, is applied in painless gastroscopy diagnosis and treatment, which can reduce the pain of patients during the examination process, and not only reduce the dosage of the ciprofol, but also reduce the incidence of respiratory depression, nausea and vomiting, and other adverse events, and show the advantage of a more stable hemodynamics, while achieving a sufficient depth of sedation. 9 In this study, the ED50, ED95, and 95% CI of ciprofol were calculated using a modified sequential test for the combined application of sufentanil to suppress the response to upper gastrointestinal endoscopic placement in older patients.
Materials and methods
The trial was approved by the Ethics Committee of the Affiliated Hospital of Guizhou Medical University (number: 2023079K) and registered by the China Clinical Trials Registry (registration number: ChiCTR2300074160).
Figure 1 depicts a high-level overview of the study, 57 patients who voluntarily underwent upper gastrointestinal endoscopy in the Affiliated Hospital of Guizhou Medical University from August 2023 to September 2023 were included as study subjects. All patients signed the informed consent form. The flowchart of the participants in the study.
Participants
The inclusion criteria for the trial were as follows: elderly patients over 65 years of age undergoing upper gastrointestinal endoscopy under intravenous anesthesia; American Society of Anesthesiologists (ASA) physical classification status of I and II; any gender; BMI ≥18 and ≤30 kg/m2, and those who can understand the trial protocol and sign the informed consent form.
The exclusion criteria were those with contraindications to deep sedation/anesthesia or previous sedation/anesthesia accidents; those with known allergy to eggs, soya products, opioids and their antidotes, propofol, etc; chizophrenia and severe depressive states; obstructive sleep apnea; acute upper respiratory tract infection within 1 week, or asthma attack; uncontrolled severe hypertension; gastrointestinal obstruction with retention of gastric contents; long-term sedative and analgesic drug use, and allergy.
Grouping
The trial was divided according to age into group A: 65∼74 years, and group B: over 75 years old. This study did not set up a control group and aimed to explore the use of ciprofol in combination with sufentanil in the ED50 of elderly patients. Ciprofol is a white liquid, and sufentanil is a clear liquid, so there is no blinding. The dose used in the latter patient was determined based on the negative/positive of the previous case.
The study used a modified sequential approach, with sample sizes based on crossover inflection points obtained during the trial. The dose of the drug used in the first patient is recommended in the manual and the dose of the drug used in the next patient is determined by the previous patient. If there is a positive response to endoscopic placement, the dose of the drug is increased in the next patient, and if not, the dose of the drug is decreased until there are 8 crossovers and the trial is stopped (a crossover inflection point is defined as a positive case turning into a negative case). At least 20 patients were included in each group.
Intervention
Patients included in this trial had no preoperative medication, intravenous access was opened upon admission to the room, Ringer sodium lactate injection 5 kg/ml was infused, recorded baseline vital signs, patients were placed in the left lateral recumbent position, oxygen was administered using a face mask at medium flow (6∼10 L/min), and sedation/anesthesia levels were assessed using the Modified Observer’s Alertness/Sedation scale (MOAA/S). Assessment of the MOAA/S score was started as soon as the push of ciprofol was started, and scoring was performed every 10 sec until induction was successful (MOAA/S<3). No further scoring was performed during the gastroscopy until the end of the gastroscopy immediately, after which MOAA/S scoring was performed at 2 min intervals until the patient was fully awake (MOAA/S≥4, 3 times).
The MOAA/S score was assessed by the anesthesiologist using the following criteria: 5 score, responds readily to name spoken in normal tone; 4 score, lethargic response to name spoken in normal tone; 3 score, responds only after name is called loudly and/or repeatedly; 2 score, responds only after mild prodding or shaking; 1 score, responds only after painful trapezius squeeze; and 0 score, no response after painful trapezius squeeze.
Patients were transferred to the recovery room after the procedure, where their HR, SpO2, NIBP, and MOAA/S were monitored. The modified Aldrete score was used to assess recovery. Patients are only allowed to leave the recovery room if they have achieved an Aldrete score of 9 or more on three consecutive occasions.
If the mean arterial pressure (MAP) is less than 30% of the basal value during anesthesia, ephedrine 3∼9 mg is given intravenously (exact dose to be decided by anesthetist); if the heart rate (HR) is less than 50 beats/min, atropine 0.2∼0.5 mg is given intravenously (exact dose to be decided by anesthetist); when the pulse oximetry (SpO2) is less than 90% or/and the respiratory rate (RR) is less than 8 beats/min, mask-assisted ventilation is given.
All patients were treated by one attending anesthesiologist, and all the procedures were performed by physicians with at least 5 years of endoscopic work. The incidence of adverse reactions (e.g., choking, swallowing, and body movements) was recorded, and the time of induction, duration, fully alert, and recovery were recorded.
We defined the induction time as initiation of intravenous ciprofol to MOAA/S<3; duration of procedure is the time from the gastroscopy insertion to exit; fully alert time is the time from the last dose to full alertness (Aldrete≥9, 3 times); and recovery time is the time from the last dose to patient discharge.
Identification of ED50 and ED95
The Dose distribution determined using the Dixon’s modified up-and-down method.10,11 The administration regimen was as follows: the initial dose of ciprofol was set at 0.4 mg/kg in two groups, with a dose difference of 0.05 mg in each case. The initial dose of ciprofol was administered intravenously with .1 μg/kg of sufentanil 3 minutes prior to the administration of ciprofol, and the upper gastrointestinal endoscope was placed after a depth of sedation was achieved (MOAA/S<3).
“Successful sedation” was defined as no endoscopic placement reaction within 5 minutes after administration of the test drug. Then, the next patient received a low-level dose. Conversely, a reaction to endoscopic placement within 5 minutes was defined as a “failed sedation,” which the next patient received a high-level dose. And the criteria for upper gastrointestinal endoscopy insertion reaction: choking, swallowing, and/or body movement reactions were occurred during pharyngeal cavity implantation or within 5 minutes after upper gastrointestinal endoscopy implantation. If an upper GI endoscopic reaction was evident and interfered with the endoscopist’s examination, an additional dose of ciprofol was given at 1/2 the initial dose until the endoscopic reaction resolved until eight positive-negative crossover points were obtained.
Measurements
Subjects were observed for endoscopic reactions during the endoscopic consultation. Criteria for upper gastrointestinal endoscopic reactions: choking, nausea and vomiting, and/or somatic movement reactions when the endoscope is inserted into the pharyngeal cavity or within 5 min after insertion.
Respiratory and hemodynamic indices, induction time, awake time, time out of the room, and incidence of adverse effects during endoscopy were recorded for each group.
Statistical Analyses
Statistical analysis and processing were performed using the SPSS 25.0 statistical package. Normally distributed measures were expressed as mean ± standard deviation (
Results
A total of 57 patients were enrolled and finished the experiment. The flowchart of the study is shown in Figure 1. In this experiment, 27 patients were included in group A, there were 15 negative and 12 positive reactions; 30 patients were included in group B, there were 17 negative and 13 positive reactions.
Procedure and Sedation-related Outcomes
Characteristics of Participants.

Note: Data are presented as mean ± standard deviation (SD), or number and number of subjects. Abbreviations: BMI, body mass index. *, we define P < 0.05 as a significant difference between groups.
The majority of the 57 patients enrolled had comorbidities of other systems, hypertension was observed in 33.3% vs 66.7% (P = 0.012*), diabetes was observed in 22.2% vs 0% (P = 0.006*), and history of surgical interventions was observed in 22.2% vs 3.33% (P = 0.030*). There were significant differences in hypertension, diabetes, and surgical history between the two groups, which may be related to the small sample size included. The age-specific prevalence of hypertension increased with older age in both men and women, 12 so the number of hypertension cases in group B is significantly higher than in group A.
The induction time, duration of procedure, fully alert time, and recovery time were similar between the two groups (P > 0.05).
Primary and Secondary Outcomes
Group A included 27 patients, of whom 15 were negative reactions with the ED50 of .23 mg/kg (95% CI 0.09∼.30 mg/kg) and the ED95 of 0.40 mg/kg (95% CI 0.32∼1.26 mg/kg); Group B included 30 patients, of whom 17 were negative reactions with the ED50 of 0.18 mg/kg (95% CI 0.13∼.22 mg/kg) and the ED95 of 0.27 mg/kg (95% CI 0.23∼.50 mg/kg).
The sequential doses of ciprofol co-administered with sufentanil for intravenous anesthesia in the upper gastrointestinal endoscopic using the up-and-down method are shown in Figures 2–3. The ED50, ED95, and 95% CI for each group are shown in Table 2. Figure 4 shows the changes in the vital signs documented against elapsed sedation time. Stepwise dose adjustment of ciprofol with low-dose sufentanil (0.1 μg/kg) using the up-and-down method. Does-effect curve fitting. The ED50, ED95, and 95% CI of Ciprofol to Inhibiting Responses to Insertion of Upper Gastrointestinal Endoscopy in Groups A and B (mg/kg). Changes in vital signs against elapsed sedation time. Note: HR, Heart rate; MAP, Mean arterial pressure; SpO2, peripheral oxygen saturation; RR, respiratory rate. T0, before anesthesia induction; T1, sufentanil was injected 3 minutes after injection; T2, ciprofol injection was complete; T3, endoscope through the mouth; T4, 5 min after endoscope through the mouth; T5, endoscope withdrawal from the mouth; T6, 5 min after the termination of anesthesia; T7, time of leaving the resuscitation room.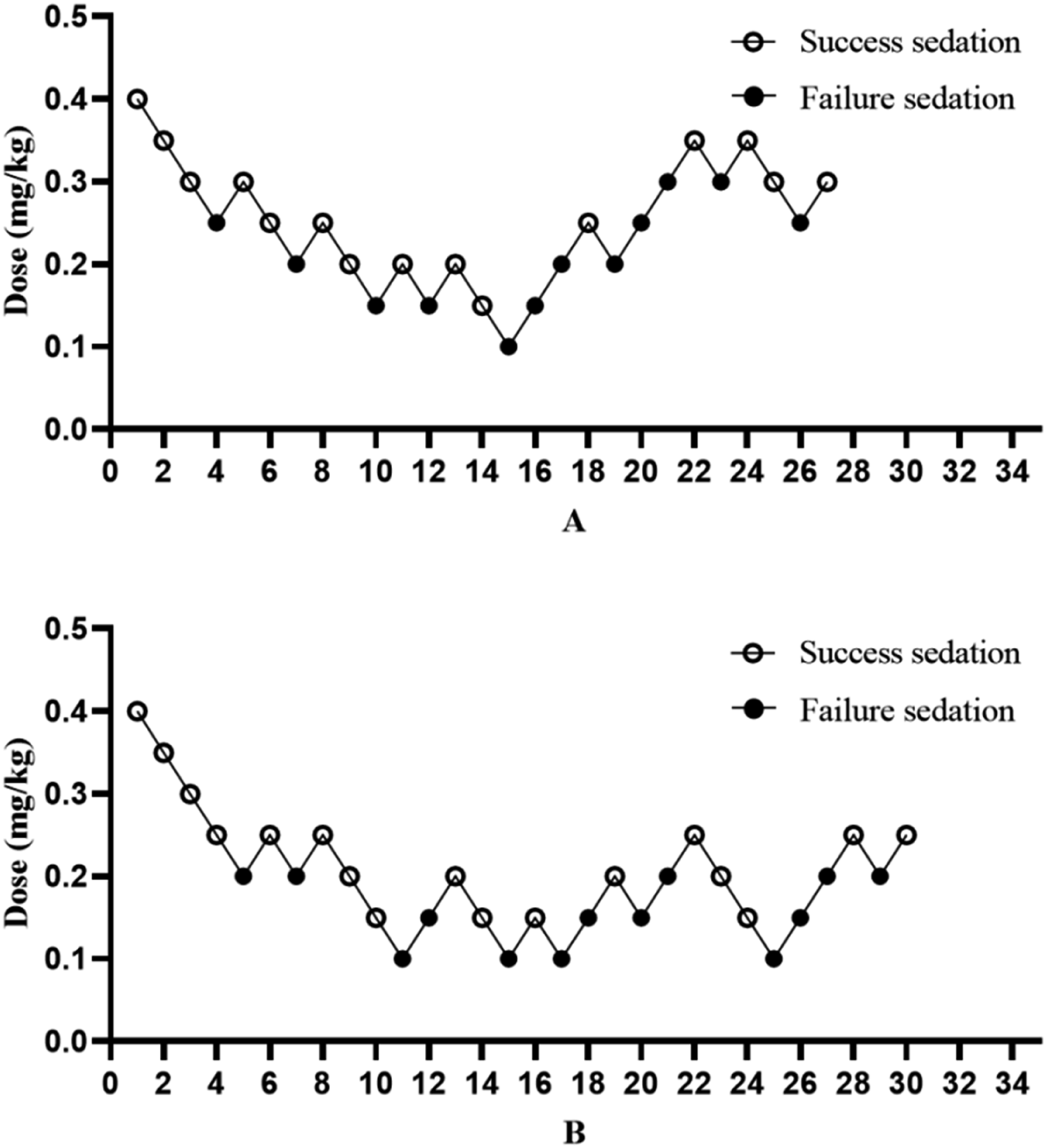



The adverse Events
Incidence of Adverse Events.

Note: apnea: thoracic motion disappeared for >30 sec; respiratory depression: defined as RR <8 bpm and duration >30 sec; hypotension: defined as MAP decrease 20% from baseline and duration >2 min from the time of first treatment to discharge; *, we define P value <0.05 as a significant difference between the groups.
Among the fifty-seven patients included in the experiment, no adverse drug reactions such as intravenous pain, muscle rigidity and significant vertigo were observed. No hypotension or bradycardia requiring intervention with ephedrine or atropine occurred during this study.
Discussion
Upper gastrointestinal endoscopy is a common method of diagnosing and treating gastrointestinal diseases. Compared with normal gastrointestinal diagnosis and treatment, painless gastrointestinal diagnosis and treatment not only effectively reduces the incidence of adverse reactions during endoscopy but also provides a more comfortable diagnosis and treatment, but the unregulated use of sedative/anaesthesia drugs may increase the risk of respiratory and cardiovascular complications. 13
Gastric cancer is the fifth commonest cancer worldwide, and it is the second most frequently diagnosed cancer in China. 14 As the population ages, an increasing number of older people are becoming concerned about their health problem. For elderly patients, painless gastrointestinal treatment is the better choice.
Propofol has long been the most commonly used intravenous anesthetic drug, and it is widely recognized in clinical practice. Propofol has the characteristics of fast onset of action and quick recovery, however, the use of propofol is associated with more pronounced adverse effects, such as injection pain, respiratory depression, hypotension, nausea and vomiting, and so on. 15
Ciprofol, a new sedative/anesthetic drug independently developed in China, has been a highly discussed drug in recent years. Currently, several clinical studies have found that, compared with propofol, ciprofol has less injection pain, a lower incidence of adverse effects, and superior stability in respiration and circulation.4-8 In addition, the induction times of ciprofol (0.4 mg/kg) and propofol (2 mg/kg) were close to each other and both were successfully induced within 1 minute. The time from the discontinuation of anesthetic drug maintenance to full alertness and other recovery-related durations was the same for ciprofol and propofol. 16
The Dixon’s modified up-and-down method used in this study is a common test method for exploring the optimal dose of a drug to be used. It is able to give a reflection of the potency strength of the drug in a shorter period of time and smaller sample size. In this study, an equal difference increment/decrement method was used to determine the dose for the next patient based on the dose used in the previous patient. There are two methods of increasing or decreasing the dose gradient of the test drug that are more commonly used in sequential testing, one is the equal proportional increase or decrease method 17 and the other is the equal difference increase or decrease method.18,19 Equivariate increases and decreases make it easier to control the dose increases and decreases, facilitate the calculation and dosing of the next patient, save trial time and reduce the difficulty of dosing, and also shorten the study period.
The results of this study showed that the ED50 and ED95 for the use of 0.1 μg/kg sufentanil combined with intravenous injection of ciprofol for upper gastrointestinal endoscopy to suppress the somatic motor response in patients in different age groups were as follows: in group A, the ED50 was 0.23 mg/kg (0.09∼0.30 mg/kg), and the ED95 was 0.40 mg/kg (0.32∼1.26 mg/kg); in group B, the ED50 was 0.18 mg/kg (0.13∼0.22 mg/kg), and the ED95 was 0.27 mg/kg (0.23∼0.50 mg/kg).
In this study, we used sufentanil plus ciprofol for induction of anesthesia and recorded perioperative vital signs. We only recorded HR, MAP, SpO2 and RR for T0-T7 and did not perform statistical analysis of these data. The reason for this was the small sample size included in this study, and secondly, the dose used was different between each patient, and the number of patients who used the same dose was small. It was undeniable that after reaching the depth of anaesthesia (MOAA/S<3), the vital signs decreased to some extent compared to the basal values measured in the room, but the fluctuations were small.
In this trial, we did not record significant apnea, respiratory depression or bradycardia. Literature reports that some patients still experience injection pain when pushing ciprofol, 20 but the present study did not record patients with varying degrees of injection pain in the injected limb, which may be related to the analgesic effect of advance administration of sufentanil.
The ED50 and ED95 of ciprofol combined with 0.1 μg/kg sufentanil to suppress the upper gastrointestinal placement response were 0.23 mg/kg (65∼74 years old) and 0.18 mg/kg (over 75 years old), with stable respiratory circulation and definite sedation during the study. There was a significant difference in the dosage between different age groups, suggesting that in the course of clinical application, elderly patients should be considered to reduce the dosage of the drug. The recommended clinical dose of ciprofol was 0.4 mg/kg, 58% of the recommended dose for patients aged 65∼74 years, and 45% of the recommended dose for patients aged 75 years and above.
In this experiment, no interventions on heart rate and blood pressure were made with atropine or ephedrine, thus demonstrating the greater stability of the drug in terms of the circulatory system.
Before performing painless gastroscopy, patients were all required to visit the anesthesia clinic for risk assessment, which screened out patients with severe comorbidities. In this study, no significant drug injection pain or adverse events were found, which may be relevant.
In summary, the dose of ciprofol should be reduced with increasing age, and 0.23 mg/kg (65∼74 years old) and 0.18 mg/kg (over 75 years old) were recommended, not only to favor the maintenance of respiratory and hemodynamic stability, but also to provide a better comfortable medical experience for the patient.
Strengths and Limitations of This Study
This study may provide a medication reference for elderly patients when performing upper gastrointestinal endoscopy.
The limitation of this study is that the sample size depends on the number of crossover points, so it may not be possible to obtain the appropriate dose of the study drug accurately in the case of a small sample size.
In this study, no significant perioperative adverse events were recorded, which may also be related to the small sample size.
Research Ethics and patient Consent
The trial was approved by the Ethics Committee of the Affiliated Hospital of Guizhou Medical University (number: 2023079K) and registered by the China Clinical Trials Registry (registration number: ChiCTR2300074160). We certify that the study was performed in accordance with the 1964 declaration of HELSINKI and later amendments. Written informed consent was obtained from all the participants prior to the enrollment of this study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available due to the privacy policy but are available from the corresponding authors on reasonable requests
