Abstract
The incidence of emergence delirium (ED) is higher in preschool children undergoing tonsillectomy and/or adenoidectomy. The purpose of this study was to determine the median effective dose (ED50) of dexmedetomidine (DEX) for the inhibition of ED in preschool children by using probit regression analysis. A total of 140 anesthesia records were retrieved and divided into seven groups based on the infusion rate of DEX: .2, .25, .3, .35, .4, .45, and .5 μg·kg−1·h−1. The Pediatric Anesthesia Emergence Delirium Scale (PAEDS) was used to assess ED in preschool children, and ED was defined as a PAEDS score ≥ 10. Probit regression analysis revealed that the ED50 and ED95 of DEX were .31 μg·kg−1·h−1 (95% CI: .29–.35) and .48 μg·kg−1·h−1 (95% CI: .44–.56), respectively. Probit(p) = −2.84 + 9.28 × ln (Dose), (χ2 = 1.925, P = .859). The PAEDS score was significantly increased in the ED group, and the rate of bradycardia was significantly decreased in the ED group compared with the without ED group (27.3% vs 54.1%, P = .02). DEX can effectively inhibit the ED in preschool children undergoing tonsillectomy and/or adenoidectomy, however, bradycardia was the main complication.
Introduction
General anesthesia (GA) is frequently used in preschool children undergoing tonsillectomy and/or adenoidectomy, and some children experience ED due to pain, residual sedation, and throat suffocation, including the destructive behavior observed during the recovery period from GA.1,2 In addition, preoperative fear, anxiety, venipuncture, mask application, and loneliness due to separation from parents are all risk factors for ED in preschool patients. 3 Prophylactic medication is the most common method to minimize anxiety in children, which facilitates the process of anesthesia induction, and various sedatives are continuously intravenously infused intraoperatively to reduce the incidence of ED before children are transferred to the postanesthesia care unit (PACU). 4
Midazolam is a traditional sedative anxiolytic agent that is widely used for premedication to reduce the incidence of ED. 3 A previous report confirmed that oral administration of .5 mg/kg midazolam can effectively reduce separation and anxiety in preschool children. 5 However, some adverse effects associated with midazolam were increased, such as respiratory depression, changes in postoperative behavior, paradoxical reactions, and cognitive dysfunction. 6 More importantly, some studies demonstrated that midazolam was not effective in preventing ED when compared with other sedative agents, such as α-2 receptor agonists and propofol. 7
Dexmedetomidine (DEX) has been proven to be an “arousable” sedative; it is a novel highly selective α-2 adrenoreceptor agonist and is considered an alternative premedication for ED in children. 8 DEX is used for sedation and analgesia as well as an anxiolytic effect without respiratory depression, thus producing a similar natural sleep state for its stable recovery of the central nervous system (CNS). 9 Recently, DEX has been explored as a premedication in the inhibition of ED in children, 10 and numerous studies have confirmed that DEX can effectively reduce the incidence of ED in children.11–13 However, some adverse effects, such as hypotension and bradycardia, limit its clinical application.14,15 Although different studies have provided different doses of DEX,16,17 there is no consensus on the optimal dose of DEX to prevent ED in pediatric patients.
Emergence Delirium (ED) in children is mainly related to behaviors such as crying sadly, kicking, hallucinations, disorientation, cognitive, and memory impairment during emergence from GA. 18 Timely management of the ED in the PACU is necessary and requires more sedative and analgesics, which prolong discharge from the hospital. To date, studies on effective doses of DEX are very limited despite the perception that DEX may prevent ED in children.
The aim of this dose-response trial was to determine the ED50 of DEX for the inhibition of ED in preschool children between the ages of 3 and 7 years who underwent tonsillectomy and/or adenoidectomy with GA.
Patients and Methods
Patients
The anesthesia record data of all the preschool children who underwent tonsillectomy and/or adenoidectomy between January 2020 and December 2022 were selected for this study from the information center of Anqing Medical Center, Anhui Medical University.
This study was a retrospective trial, and the data of all the children were screened. During the data collection period, the data of the study subjects were kept confidential, and all methods were carried out in accordance with relevant guidelines and regulations.
Study Protocol
We reviewed the anesthesia record data of children with continuous intraoperative intravenous infusion of DEX. The inclusion criteria of this study were as follows: preschool children between 3 and 7 years with American Society of Anesthesiologists (ASA) I. Intravenous propofol 1.5 mg·kg−1, sufentanil .5 μg·kg−1, and cisatracurium .2 mg·kg−1 were administered for anesthesia induction, and then intravenous pumped propofol 3–6 mg·kg−1·h−1, remifentanil .1–.2 μg·kg−1·h−1, and cisatracurium 2–5 μg·kg−1·min−1 for intraoperative maintenance. We intravenous pumped DEX 0.2-0.5 μg·kg−1·h−1 for intraoperative maintenance after endotracheal intubation. All of the children were treated with the same anesthesia regimen. The exclusion criteria included abnormal critical organ function, congenital diseases, asthma, mental, and neurological defects, administration of anxiolytic or anti-psychotics preoperative period, and incomplete anesthesia data. Children with a postoperative CHEOPS score >6 and intolerable were considered analgesic insufficiency, and received intravenous parecoxib sodium .5–1.0 mg·kg−1, and these patients were also excluded.
We divided the patients into seven groups based on the rates of DEX intraoperative continuous infusion: .2, .25, .3, .35, .4, .45, and .5 μg·kg−1·h−1. A total of 168 anesthesia record data were retrieved, and 25 incomplete data were excluded: one child was excluded in the .25 dose group due to congenital disease, one child was excluded in the .45 dose group due to mental retardation, and one child was excluded in the .50 dose group due to left upper limb neurological dysfunction. After the patients’ data were screened, 140 children were ultimately included.
Measurement
The Ramsay Sedation Scale (RSS) for children is scored as follows: 1 = anxious, restless; 2 = quiet and cooperative; 3 = responsive to the commands; 4 = quick responsive to a light tap; 5 = slow in response to a light tap; 6 = no response at all. In this trial, an unsatisfactory level of sedation was defined as an RSS of 1, and an RSS ≥ 2 was defined as satisfactory sedation. 19
The Parental Separation Anxiety Scale (PSAS) for children is as follows: 1 = easily separated from parents; 2 = whimpers, but easy to reassure; 3 = crying and not easy to reassure, but not attached to parents; 4 = crying loudly and attached to parents. Successful parental separation was defined as a PSAS score ≤ 2 in this trial. 20
The Mask Acceptance Scale (MAS) is a behavioral assessment for children receiving a mask by a 4-point scale during the mask application, and the MAS score is as follows: 1 = excellent (very easy to accept mask); 2 = good (mild fear of mask, easy to comfort); 3 = fair (moderate fear of mask, but not easy to comfort); and 4 = poor (extreme fear of mask, too difficult to comfort). “Satisfactory” mask acceptance was defined as an MAS score ≤ 2 in this trial. 20
The Children’s Hospital Eastern Ontario Pain Scale (CHEOPS) for children is scored as follows: 1 = no cry, 2 = moaning or crying, 3 = screaming; 0 = smiling, 1 = composed, 2 = grimace; 0 = positive, 1 = none or complaints other than pain, 2 = pain complaints; 1 = torso neutral, 2 = torso shifting or tense, shivering, restrained; 1 = not touching, 2 = reach or touch, grab; 1 = legs neutral, 2 = legs squirming kicking or drawn up tensed, standing, restrained. The total CHEOPS score was the sum of scores on 6 items, with a minimum score of 4 and a maximum score of 13. Postoperative incomplete analgesia was defined as a CHEOPS score ≥6. 21
The Pediatric Anesthesia Emergence Delirium Scale (PAEDS) for children is scored as follows: 1, the children have eye contact with the caregivers; 2, the behavior of the children is purposeful; 3, the children are alert to the surrounding environment; 4, the children are restless; and 5, the children are extremely sad and difficult to comfort. The first 3 items are scored as follows: 4 = not at all; 3 = a little bit; 2 = quite a bit; 1 = very much, 0 = super much. The final two items are scored as follows: 0 = not at all; 1 = a little bit; 2 = quite a bit; 3 = very much; 4 = super much. The total PAEDS score was the sum of the scores on 5 items, with a minimum score of 0 and a maximum score of 20. ED was defined as a PAEDS score ≥ 10 in this trial. 22
Parental Separation Anxiety Scale (PSAS) scores were assessed when the children were separated from their parents before anesthesia, and MAS and PAEDS scores were assessed immediately and at 5, 15, 30, and 45 minutes after extubation in the PACU.
Hemodynamic parameters such as the mean arterial pressure (MAP), heart rate (HR), and pulse oxygen saturation (SpO2) were recorded.
Statistical Analyses
Sample size was calculated using PASS 11 (NSCC, LCC, Kaysville, UT), (the Cochran - Armitage test for trend in proportions), 23 based on our calculation, the incidence of ED was 84, 70, 52, 34, 19, 9, and 4% for the seven DEX infusion doses of .2, .25, .3, .35, .4, .45, and .5 μg·kg−1 h−1, respectively. Our calculations showed that a sample size of 17 patients in each group could provide 81% power with a linear trend, using a two-tailed Z test and maintaining an α level of .05, our total sample size of 140 patients is sufficient.
SPSS 21.0 was used to analyze the data. Normally distributed data are expressed as the mean ± standard deviation (
Results
Anesthesia records from 168 children were retrieved. Records from 25 children were excluded due to being incomplete, and three children were excluded due to congenital disease, mental retardation, and left upper limb neurological dysfunction. Ultimately, 140 children were included in the current study. The children’s flow of this study is shown in Figure 1. Subjects cohort flow diagram.
Demographic Characteristics of all the Children, Duration of Surgery, and Duration of Extubation.

Data are expressed as
△P value: Chi-square test. ※P value: One-way ANOVA. Duration of extubation was defined as the time between the end of anesthesia and the removal of the tracheal tube.
Demographic Characteristics of all Children, Duration of Surgery, and Duration of Extubation.

Data are expressed as
Probit regression analysis revealed that the ED50 of DEX for the inhibition of ED in preschool children undergoing tonsillectomy and/or adenoidectomy was .31 μg·kg−1·h−1 (95% CI: .29–.35), and the ED95 was .48 μg·kg−1·h−1 (95% CI: .44–.56). Probit(p) = −2.84 + 9.28 × ln (Dose), (χ2 = 1.925, P = .859). The probability of ED inhibition in children with DEX was determined by probit regression analysis, and the dose-response curve was plotted and shown in Figure 2. Dose-response curve of DEX for the inhibition of the ED in preschool children undergoing tonsillectomy and/or adenoidectomy. Probability unit vs Dose of DEX.
The RSS was significantly decreased in the ED group compared with the without ED group (Figure 3A), however, the PSAS, MAS, CHEOPS, and PAEDS scores were significantly increased in the ED group compared with the without ED group (Figure 3B-E), respectively. The rate of bradycardia was significantly decreased in the ED group compared with the without ED group (27.3% vs 54.1%, P = .02, Figure 3F). Comparison of RSS, PSAS, MAS, CHEOPS, PAEDS scores and the rate of bradycardia in two groups. (A) Ramsay Sedation Scale. (B) Parental Separation Anxiety Scale. (C) Mask Acceptance Scale. (D) Children’s Hospital Eastern Ontario Pain Scale. (E) Pediatric Anesthesia Emergence Delirium Scale. (F) the rate of bradycardia was significantly decreased in the ED group. **P < 0.01 and ***P < 0.001.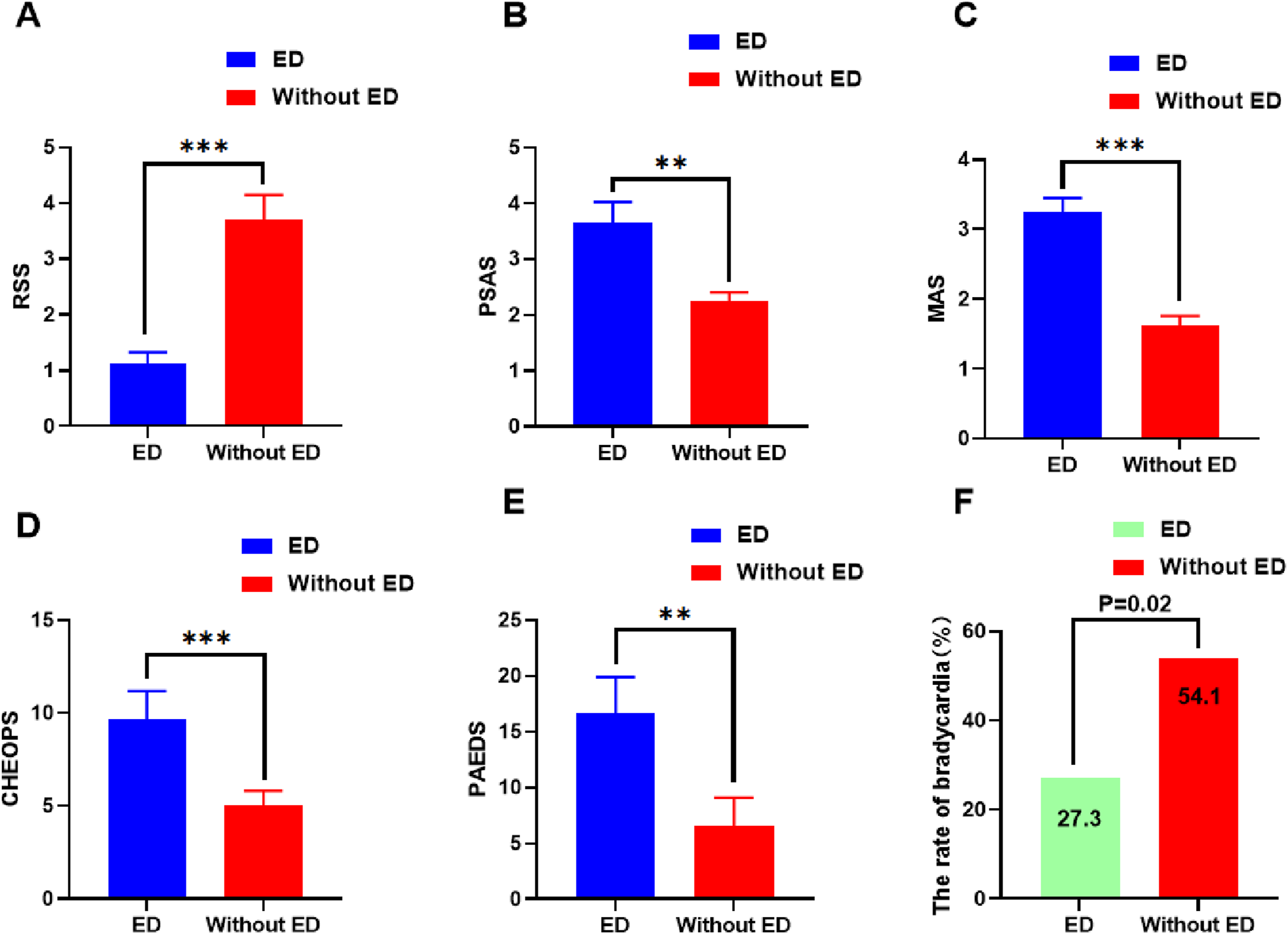
The changes in MAP and HR are shown in Figure 4 A and B in the two groups. The MAP and HR in the ED group were significantly increased at the time of extubation and 5, 10, and 15 min after extubation compared with the without ED group. Hemodynamic changes in two groups. (A) The MAP was significantly decreased in the withou ED group. (B) The HR was significantly decreased in the withou ED group. ***P < 0.001.
Discussion
This retrospective dose-response trial was designed to determine the ED50 of DEX for the inhibition of ED in preschool children undergoing tonsillectomy and/or adenoidectomy. The probit regression analysis showed that the ED50 was .31 μg·kg−1·h−1 (95% CI: .29–.35), and the ED95 was .48 μg·kg−1·h−1 (95% CI: .44–.56). Fifty-five children developed ED, and 85 children did not develop ED. The demographic characteristics were not significantly different between the two groups.
Our previous studies often used the Dixon sequential method to determine the ED50 of drugs. The advantage of this method is that a small sample is sufficient, but the disadvantage is that the results are not accurate enough because the sample size at the extreme dose is only 1–3, which leads to serious bias.24,25 In this study, a large sample of 140 children were included in a wide range of dose groups (7 doses), and thus, the results were less biased and highly reliable.
Eckenhoff first described the “neuroexcitation” observed during emergence under GA in the 1960s, but the patients were in a state of free consciousness. 26 Patients present with an “unpleasant state of extreme excitement” often associated with pain, anxiety, fear, and more complex neurobehavior. 27 Different surgical categories lead to different rates of ED, ranging from 10 to 80%. 28 Patient characteristics, anesthetic techniques, type of surgery, and different methods used to assess ED all contribute to different rates of ED. Once ED occurs in the PACU, the symptoms first need to be adequately assessed, including the level of analgesia, sedation, consciousness, and delusion. Children with severe ED may harm themselves or others, so caregivers in the PACU must quickly consult about the children’s medical history, the type of surgery, and style of anesthesia to identify the most appropriate management. Factors of severe ED may include acute pain, changes in the surrounding environment, or restrictive factors, and for children undergoing tonsillectomy and/or adenoidectomy, attention is given to oral secretions, blood clots, or postoperative sensory changes.
Dexmedetomidine (DEX) is a highly selective α-2 receptor agonist and is widely used in clinical sedation due to its short half-life. In recent years, numerous studies have demonstrated that DEX effectively prevents ED in adult patients at doses ranging from .25 to .4 μg·kg−1·h−1 but is accompanied by an increased risk for dose-related adverse effects such as hypotension, residual sedation, prolonged duration of extubation, and retention in the PACU.17,29 Few studies have examined the effects of DEX on ED in pediatric patients, and optimization of the DEX dose is crucial to prevent ED and reduce complications in pediatric patients. The ED50 we calculated herein using probit regression analysis effectively inhibited ED in children and did not increase the dose-related adverse effects, such as not prolonging extubation time (Table 2). There is no consensus on whether DEX causes bradycardia. The dose of DEX, the type of surgery, and the evaluation method of ED are all important factors responsible for the different conclusions.30–32 The similarity of this study and previous results lies in that DEX effectively inhibited ED in children, but bradycardia seemed inevitable. 33 There were 46 patients with bradycardia in the non-ED group and 15 patients in the ED group (54.1% vs 27.3%, P = .02, Figure 3F).
How to prevent and handle ED in children is not yet clear. Some studies have proven that the preoperative use of nonpharmacological strategies to reduce preoperative anxiety in children can effectively reduce the incidence of ED.34,35 Although the effect of preoperative pharmacological intervention on the development of ED remains unclear, a recent randomized controlled trial compared oral benzodiazepine and tablet-based interactive distraction (TBID), and the results confirmed that anxiety baseline and mask-induced tolerance were improved in the TBID group, shortened hospital stay, and a lower incidence of ED. 36 In addition, nasal administration of DEX (1–2 μg/kg) 20 minutes before anesthesia induction significantly reduced the incidence of ED and led to less postoperative anxiety; more importantly, the patient’s recovery time was not delayed compared with placebo. 37 Recent studies have also demonstrated that preoperative oral DEX significantly reduced PAEDS scores (P < .05) and the incidence of ED compared with oral benzodiazepine (DEX 0% vs benzodiazepine 19%, P = .01). 38
Emergence delirium (ED) severely affected MAP and HR in the children. Abdel-ghaffar et al. confirmed that oral DEX at 1.0 μg/kg reduced MAP by 25% compared to the control group (equal volume of saline). 39 We divided all the children into two groups (ED group and without ED group) and found that MAP and HR in the ED group did not decrease compared with the basal value; however, the MAP and HR in the ED group were significantly increased at the time of extubation and 15 min after extubation. The levels of RSS were significantly decreased (Figure 3A), and the PSAS, MAS, CHEOPS, and PAEDS scores were significantly increased in the ED group, which resulted in an increase in MAP and HR (Figure 3B-E). Therefore, the ED50 measured in this study can effectively inhibit ED in children and maintain the stability of MAP and HR during the emergence period.
Although ED is transient, there are many potential risks, such as incision cracking, hemostatic gauze shedding, and bed falling, which should be considered by PACU caregivers and treated early or in a timely manner. In addition, ED can increase the incidence of adverse events, such as prolonged resuscitation time and susceptibility to asphyxia. There are some limitations in this study. The first limitation is that it is a retrospective study, and the researchers did not participate in the preoperative psychological counseling and anxiety relief of children, which is one of the important reasons for ED in children. Therefore, the results may have been biased. The second limitation is that each anesthesiologist treated ED with a different strategy, which affected the duration of ED and the degree of postoperative pain. Therefore, there may have been bias in the CHEOPS and PAEDS scores. Finally, although this is a large sample size study, it is also a single-center study and a prudent conclusion requires a large sample size and multicenter study.
In conclusion, DEX can effectively inhibit ED in preschool children undergoing tonsillectomy and/or adenoidectomy without complications such as respiratory depression, delayed extubation, and residual sedation, but bradycardia is the major perioperative complication, and the anesthesiologist should be vigilant. The primary caregivers of the PACU should accurately assess the awareness, agitation degree, and pain level when the child has an ED to assist the anesthesiologist in rapid treatment. Future studies require further all-cause analysis of ED and behavioral dysregulation in pediatric children to better develop strategies to prevent ED and improve the long-term consequences of ED.
Conclusions: This retrospective dose-response trial confirmed that DEX effectively inhibited ED in preschool children undergoing tonsillectomy and/or adenoidectomy. The ED50 of DEX was .31 μg·kg−1·h−1 (95% CI: .29–.35), and the ED95 was .48 μg·kg−1·h−1 (95% CI: .44–.56). Bradycardia was the main complication.
Footnotes
Acknowledgements
The author thanks the patients and doctors who participated in the study. The authors are very grateful to Professor Tao Pan, Anqing Medical Center of Anhui Medical University, for his guidance on this study.
Authors’ contributions
The study was conceived and designed by BYW and WBW; JBX collected the data; CYY, HX, and PZ analyzed the data; CYY and BYW drafted and corrected the manuscript, and all authors read and agreed to the submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Department of Anhui Education (NO: 2022AH052555) and Anhui Medical University (NO: 2022xkj247).
Ethical Statement
Clinical Trial Registration
The current dose-response trial was registered in the Chinese Clinical Trial Registry (NO.: ChiCTR2300069681) on 23/03/2023.
