Abstract
Dexmedetomidine can effectively decrease the incidences of emergence agitation (EA) in adult patients, but there are major side effects related to increased dose of dexmedetomidine. The purpose of this study was to determine the median effective dose of dexmedetomidine in the prevention of EA among geriatric patients undergoing major open surgery with general anesthesia. A total of 50 geriatric patients were enrolled in this study. Dexmedetomidine 0.5 μg·kg−1·h−1 continuous intravenous infusion was administered to the first patient. The next dose was increased or decreased by .05 depending on the response of the previous patient, according to the Dixon up-and-down method. An “effective” or “ineffective” response was determined based on the Riker sedation-agitation score (RSAS), we defined “effective” as RSAS<5, and “ineffective” as RSAS≥5. The ED50 of dexmedetomidine in prevention of EA was .30 μg·kg−1·h−1 (95% CI, .27–.33) and the predicted ED95 was .42 μg·kg−1·h−1 (95% CI, .38–.51). The incidence of bradycardia was significantly increased in the group without EA compared to the group with EA (57.1% vs 13.6%, P = .002). The ED50 of dexmedetomidine in prevention of EA was .30 μg·kg−1·h−1 (95% CI, .27–.33) and the predicted ED95 was .42 μg·kg−1·h−1 (95% CI, .38–.51). Bradycardia was the main complication.
Introduction
Post-anesthesia EA was first described by Eckenhoff in 1961. EA was first characterized as confusion, agitation, disorientation, and hallucinations during emergence from general anesthesia. 1 EA usually occurs during the initial 30 and 60 min in recovery from general anesthesia, and it may last from 45 min to 48 h in some patients. 2 The incidences of EA in adults have been reported to differ according to the variability of surgery, patient population, and the methods used to assess EA. The incidence of EA ranges from 3.7% to 30%, and its incidence is also associated with the use of a urinary catheter or an endotracheal tube and with postoperative pain.3-6
The risk factors for EA and measures to prevent EA in adult patients are unclear and still being studied. Factors that are significantly related to EA include having an endotracheal tube, urinary catheter, postoperative pain (numerical rating scale, NRS ≥ 5), anesthesia based on inhalation, smoking history, and younger age.7,8 Although the ethology of EA is not clear because of the different recovery rate in different brain areas, inhalational agents with higher incidence of EA may explain why, less soluble agents have faster recovery from general anesthesia. 9
Dexmedetomidine is an elective α2-receptor agonist that has sympatholytic, analgesia, sedation, anti-anxiety, and stress-relief effects, and dexmedetomidine exhibits those properties by inhibiting the release of norepinephrine mediated through α2 adrenergic receptors in the locus ceruleus and spinal cord. 10 In addition, dexmedetomidine exerts hypnotic properties by activating the endogenous sleep-promoting pathway. 11 Some studies have indicated that dexmedetomidine significantly improves the subjective sleep quality after surgery. 12 Dexmedetomidine produced a natural sleep state that stabilized the recovery time of the central nervous system, thereby reducing the occurrence of agitation, which is more optimal approach compared to the use of sedative-hypnotics (such as benzodiazepines) that may increase the incidence of EA. 13
Dexmedetomidine is a recently developed drug, it has been widely used to prevent postoperative EA in children, and it effectively reduces EA incidence without side effects.14-16 Many recent studies have demonstrated that intraoperative dexmedetomidine infusion also decreases the incidence of EA in adult patients but has side effects including intraoperative hypotension and bradycardia.17,18 However, different doses have been examined in different studies,19,20 so there is no consensus on the appropriate dose of dexmedetomidine in geriatric patients to prevent EA. Therefore, it is necessary to identify the safe dose range for dexmedetomidine to effectively prevent EA and avoid drug side effects.
Materials and Methods
Design
We conducted a prospective, double-blinded, dose-response trial. The primary study outcome was to optimize the median effective dose (ED50) of dexmedetomidine for the prevention of EA in geriatric patients undergoing major open surgery with general anesthesia. The secondary outcomes included changes of MAP, HR, NRS of pain after extubation, and incidence of bradycardia.
Subjects and Setting
A total of 50 geriatric patients (age ≥ 70 years) with an American Society of Anesthesiologists (ASA) physical status I or II and undergoing major open surgery were enrolled in this study. The medical ethical committees of AnQing Municipal Hospital of Anhui Medical University approved this study (Approval date: 20 December 2018), and the study was registered in the Chinese Clinical Trial Registry (Registration number: ChiCTR1900023769). All patients provided written informed consent.
The inclusion criteria are as follows: patients aged more than 70 years, scheduled for open chest or abdominal surgery and without bradycardia, and patients agree with this study protocol and participate in this trial.
The exclusion criteria are as follows: hypotension, hypertension, bradycardia, tachycardia, arrhythmia, pre-existing cardiopulmonary disease or mental health problems, renal or hepatic functional abnormalities.
Major open surgery includes resection of esophageal or lung cancer, radical resection of gastric carcinoma, radical resection of colon or rectal carcinoma, and so on.
Study Protocol
All patients were transported to the operating theater, where they were subjected to standard monitoring electrocardiography, heart rate (HR), pulse oximetry, and bispectral index (BIS). A catheter was inserted into the radial artery under local anesthesia with 1% lidocaine to continuously monitor mean arterial pressure (MAP).
Intravenous etomidate .25 mg·kg−1, sufentanil 0.5 μg·kg−1, and cisatracurium . 2 mg·kg−1, were administered for anesthesia induction; then propofol 3 ∼ 6 mg·kg−1·h−1 and remifentanil .1 ∼ 0.2 μg·kg−1·min−1 were administered for anesthesia maintenance with a BIS between 40 and 60 during surgery. Patients were sent back to post-anesthesia care unit (PACU) at the end of the surgery.
Dexmedetomidine 0.5 μg·kg−1·h−1 continuous intravenous infusion was administered to the first patient after intubation, and the infusion was stopped at the end of the surgery. The next dose was increased or decreased by .05 depending on the response of the previous patient, according to the Dixon up-and-down method. An “effective” or “ineffective” response was determined based on the RSAS, we defined “effective” as the RSAS <5 and “ineffective” as the RSAS ≥5.
Measurements
MAP and HR were monitored every 5 min during surgery and in the PACU until the end of the study.
Emergence is defined as the interval within 2 minutes after extubation. The RSAS was assessed by an independent anesthesia assistant who was blinded to the dose of dexmedetomidine in this study, and another anesthesia nurse blinded to the dose assessed pain intensity by the NRS (patients described their pain intensity on a number from 0 to 10, the higher scores represent the worse pain) at the time point of extubation, and 5 min, 10 min, 15 min, and 30 min after extubation in the PACU. Rescue analgesics, such as flurbiprofen axetil, tramadol, and opioids were intravenously administered when the NRS ≥5.
A 20% decrease in MAP compared with the baseline triggered administration of 5 to 10 mg intravenous ephedrine, and a decrease in HR of less than 45 beats·min−1 .5 mg triggered administration of intravenous atropine.
Statistics
At least six “effective–ineffective” pairs were required for statistical analysis according to Dixon’s up-and-down method; therefore, patient enrollment continued until six crossover pairs was observed. 22 There were nine “effective–ineffective” pairs in our study, so the sample size is sufficient.
The average of all “effective–ineffective” pairs was used to calculate ED50 using Dixon’s up-and-down method. The number of responders at each dose was used to plot a sigmoid dose-response curve. Probit regression was performed and the interpolation was used to obtain ED50 and ED95.
The various parameters were statistically analyzed using the SPSS 17.0 (SPSS Inc., Chicago, IL, USA). Parametric data were evaluated with independent samples t test, and frequency data with the chi-square test. P < .05 was considered statistically significant.
Results
Fifty-eight patients were enrolled in this study. Three patients did not consent to participate and five patients were excluded from this study. The participants’ flow during the study is shown in Figure 1. Subject cohort flow diagram.
The consecutive patients receiving dexmedetomidine using Dixon’s up-and-down method are shown in Figure 2. Twenty-two patients manifested with EA, and twenty-eight patients did not have EA. Effective(●) and inffective(♦) sequences of dexmedetomidine dose required for prevention of emergence agitation, and arrow represents “effective–ineffective” pairs.
Demographic characteristics, ASA status, BIS, types of surgery, time to extubation, duration of surgery, and time of stay in PACU.

Data are presented as the mean ± SD or the number. △P
The ED50 of dexmedetomidine in the prevention of EA among geriatric patients was .32 μg·kg−1·h−1 (95% CI, .26–.38) according to Dixon’s up-and-down method. A dose-response curve was plotted by probit analysis and shown in Figure 3. Logistic regression analysis of the dose-response curve and confirmed that the ED50 of dexmedetomidine for the prevention of EA in geriatric patients was .30 μg·kg−1·h−1 (95% CI, .27–.33) and the ED95 was .42 μg·kg−1·h−1(95% CI, .38–.51). Dose-response curve of dexmedetomidine for prevention of emergency agitation. Probability unit vs dose. ED50 is .3 μg·kg−1·h−1 (95% CI, .27–.33) and the ED95 was .42 μg·kg−1·h−1(95% CI, .38–.51).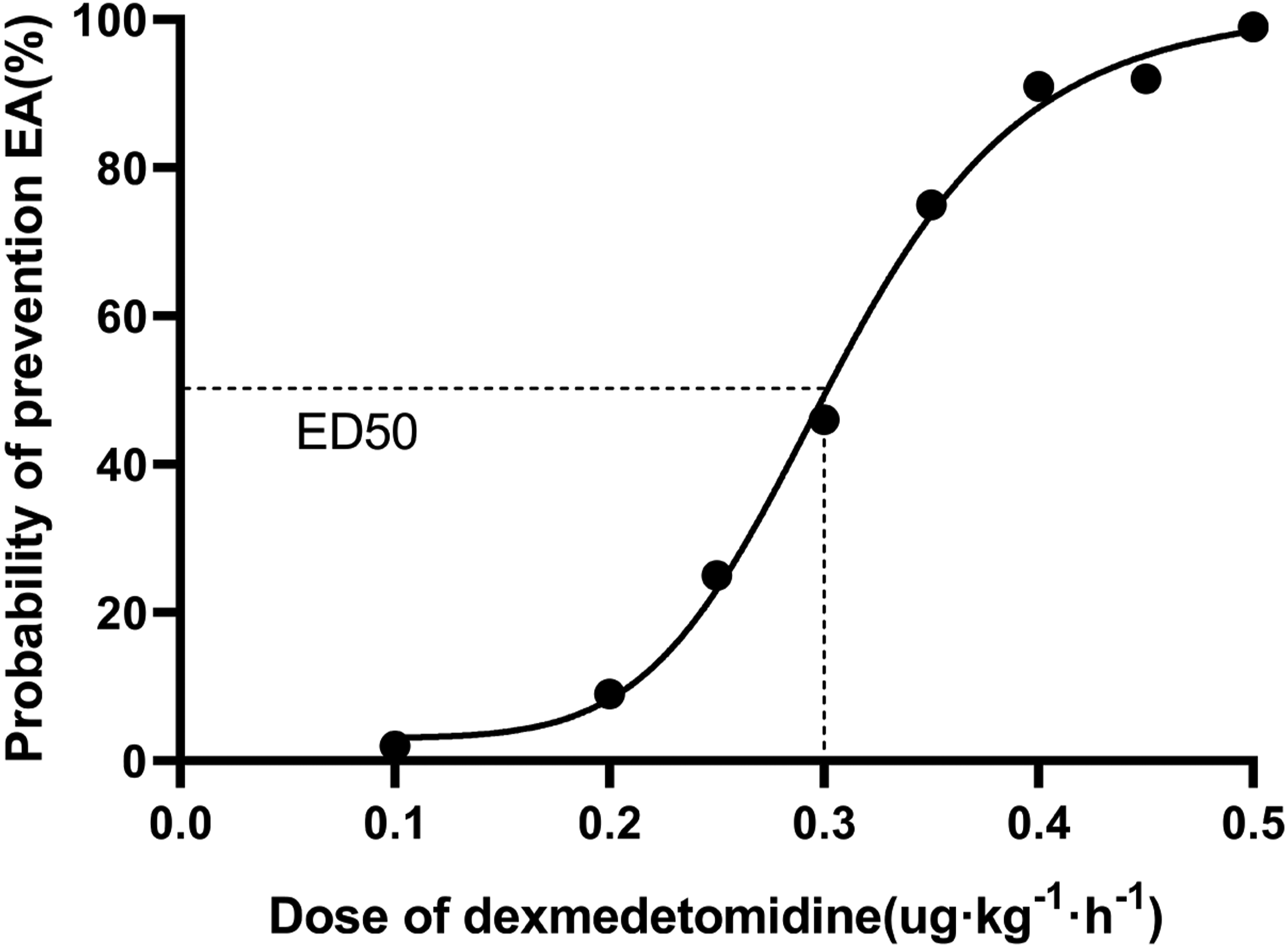
The changes of MAP, HR, and NRS of pain are shown in Figure 4 A, B, and C. (A) MAP was increased in group with EA at the time point of extubation and 5 min, 10 min, and 15 min after extubation than in group without EA, P < .001. (B) The HR was incresead in group with EA at the time of extubation and 5 min, 10 min, and 15 min after extubation than in group EA, P < .001. (C) The NRS of pain was increased in group with EA at the time point of extubation and 5 min, 10 min, 15 min, and 20 min after extubation than in the group without EA, P < .001. (D).
There are sixteen patients manifested bradycardia in the group without EA and only three in the group with EA; the incidence of bradycardia was significantly increased in the group without EA compared to the group with EA (57.1% vs 13.6%, P = .002), Figure 4(D).
Discussion
The results of our dose-response trial show that the ED50 of dexmedetomidine for the prevention of EA in geriatric patients was .30 μg·kg−1·h−1 and ED95 was .42 μg·kg−1·h−1, respectively. There were no patients who suffered any major complications, such as residual sedation, respiratory depression, or delay in extubation. There were 22 patients with EA and 28 patients without EA, and there was no significant difference in demographic characteristics (Table 2) between the two groups.
Dexmedetomidine is a highly selective alpha 2 adrenoreceptor agonist with a short half-life. It is used to prevent EA in adult patients in recent years. A bolus dose (range from .25 μg·kg−1 to 1.0ug·kg−1) or continuous infusion (0.4 μg·kg−1·h−1) of dexmedetomidine can reduce EA in adults after general anesthesia but was associated with increased hypotension, delayed extubation, residual sedation, and prolonged PACU stay.20,23 It is important to optimize the dose of dexmedetomidine to prevent EA and decrease complications in geriatric patients. We report the ED50 and ED95 determined using Dixon’s up-and-down method, as well as the effective dose of dexmedetomidine for the prevention of EA in order to decrease complications related to an excessive dose of dexmedetomidine.
Using Dixon’s up-and-down method to estimate the ED50, it requires a small sample size and relatively accurate results. Tracheal intubation increases the risk of EA by approximately 5-fold, and postoperative pain of NRS ≥5 doubled the risk of EA. 7 Various studies have reported on different doses of dexmedetomidine for prevention of EA. A retrospective cohort study showed that dexmedetomidine continuous infusion at a rate of .2–.7 µg kg−1 h−1 and suggested that dexmedetomidine decreased the incidence of EA, but bradycardia was a main side effect. 24 Considering the use of tracheal intubation and the high level of postoperative pain after major open surgery in geriatric patients, we selected .5 μg·kg−1·h−1 of dexmedetomidine as the initial dose. Kim HS et al. selected a dosing increment of dexmedetomidine of .1 in children. 25 We used this increment in our preliminary study, but it was inappropriate for geriatric patients because of their great differences in hemodynamics. Thus, we selected .05 as the dosing increment of dexmedetomidine.
The dose of dexmedetomidine is a critical factor for maintaining MAP. Hals S et al. demonstrated that premedication with 1.0 μg·kg−1 oral dexmedetomidine decreases the MAP more than 25% compared with a .5 μg·kg−1 dose and saline placebo. 26 In our study, the dose does not reduce MAP and HR in the group without EA, on the contrary, the MAP and HR were significantly increased in the group with EA (Figure 4(A),(B)) because of the increased NRS (Figure 4(C)). The ED50 not only decreased EA but also maintained MAP and HR stability in geriatric patients.
While the efficacy of midazolam and dexmedetomidine intravenous infusion was comparable in the prevention of EA during short and simple surgery (mean duration of surgery 87 ± 44 min), 17 in another study with more complex surgery (mean duration of surgery more than 120 min), the incidences of EA were significantly lower in the group treated with dexmedetomidine, and complications—such as depth of sedation, hypotension, and respiratory depression—were higher in the group treated with midazolam. 27 In our major open surgery (mean duration of surgery 249 ± 40 min, Table 2), dexmedetomidine appeared to effectively prevent EA in geriatric patients without any complications.
There are many studies confirmed that dexmedetomidine causes bradycardia when used for the prevention of EA in geriatric patients. A meta-analysis including 842 participants come from 12 randomized controlled trials shown that dexmedetomidine can effectively prevent EA and has no significant reduction in the incidence of bradycardia. 28 A single study 19 and a retrospective cohort study (including 814 patients with intraoperative dexmedetomidine infusion) revealed a lower incidence of EA, and bradycardia was the main side effect. 29 Another meta-analysis including 10 trials and 1387 patients demonstrated that dexmedetomidine decreased the incidence of delirium in patients after cardiac surgery, but the incidence of bradycardia was increased. 30 Our study contained 3 patients with bradycardia in the group with EA and 16 patients with bradycardia in group without EA (57.1% vs 13.6%, P = .002, Figure 4(D)). We suggest that the optimal dose of dexmedetomidine should be reduced with the advancing of age, especially in geriatric patients. We should pay more attention to the incidence of bradycardia when dexmedetomidine infusion is administered to prevent EA among geriatric patients undergoing major open surgery.
Although dexmedetomidine can effectively decrease the incidence of EA, the bradycardia seems to be unavoidable; our dose-response trial has also shown that 16 patients with bradycardia in group without EA and 3 patients with bradycardia in the group with EA. It is a challenge for anesthesiologist to balance the dose of dexmedetomidine between prevention of EA and decrease the incidence of bradycardia; in future, a multicentre and large sample dose-response trial should conduct for appropriate dose of dexmedetomidine for the prevention of EA and greatly decrease the incidence of bradycardia.
There are three limitations to our study. The first is that the ED95 was predicted by probit regression. This method is not reliable for calculating the small percentage points of ED95 according to the Dixon up-and-down method; therefore, it is an inaccurate prediction, but it is well accepted. Although probit analysis may not be the best method, it is generally used to calculate the ED95 when the Dixon up-and-down method is used.31,32 The second limitation of the study is the absence of a control group. We compared the incidence of bradycardia only between the group with EA and the group without EA, and the sample size was too small. Because all of the patients in this study were inserted urinary catheter before anesthesia, so the third limitation is that we did not analyze the relativity between urinary catheter and EA.
Conclusions
This study demonstrates that the ED50 of intravenous dexmedetomidine infusion in the prevention of EA among geriatric patients undergoing major open surgery was .30 μg·kg−1·h−1 (95% CI, .27–.33) and the predicted ED95 was .42 μg·kg−1·h−1(95% CI, .38–.51). Bradycardia was the main complication.
Footnotes
Acknowledgments
The authors would like to thank all staff members in the Department of anesthesia and operating room of AnQing Municipal Hospitals of Anhui Medical University. The authors sincerely thank AiJiaoSun, M.D for his contributions to the dose-response trial design and his assistance with the study. The authors acknowledge Miss. Hui Zhou, who performed data extraction, and Dr. JingChong Dong and Dr. HuangXu, who helped review the study design and date analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Clinical Trial Registration
This study was registered in the Chinese Clinical Trial Registry (Registration number: ChiCTR1900023769) on June 11th, 2019.

