Abstract
Radiotherapy is a very important tool in the treatment of cancer; nevertheless, its side effects are a hindrance to its use. The present study is designed to evaluate glucosamine effects against radiation-induced brain oxidative stress and depression-like effect in rats. Four groups of female Wister rats were used as control, irradiated (4 × 2 Gy), glucosamine (1 g/kg P.O), and glucosamine + irradiated group. The behavioral responses are estimated. The brain hippocampi of the rats are separated to evaluate oxidative stress biochemical parameters and glycogen synthase kinase pathway in addition to the biogenic amines. Irradiation exposure led to disturbances in the behavioral assessments (forced swimming test, light–dark box, and open field test) and a significant decrease in brain GSH, neurotransmitters (serotonin, norepinephrine, and dopamine), phosphatidylinositol 3 kinase (PI3K), and phosphorylated protein kinase-B (p-AKT) levels. Additionally, MDA and ROS levels increased significantly post-irradiation along with the phosphorylated glycogen synthase kinase (p-GSK3). Glucosamine administration before irradiation caused improvement in the behavioral valuations and the biochemical parameters in the brain as well. Glucosamine might be used as a radioprotector to improve brain function and as an antidepressant drug. It could be promising as a future therapy in managing depression occurring during radiotherapy.
Introduction
Radiotherapy is considered a primary treatment modality, a neoadjuvant or adjuvant therapy with chemotherapy for brain cancer. Although recent radiation techniques aim at allowing the delivery of higher radiation doses to the target site and reducing the damage to normal tissues, but unfortunately, the neurocognitive decline is still a detrimental factor considerably affecting the quality of life. 1 In addition, the diagnosis and subsequent radiotherapy for head and neck cancer patients may have effects on the psychosocial state, consequently influencing their treatments and may lead to suicide. Depression and anxiety are the most important psychopathological complications of brain radiotherapy.2,3 Exposure to whole-body gamma radiation (8 Gy) significantly caused neural disorders. 4 Recently, it has been found that brain irradiation (8 Gy, 2 fractions) induced microvascular cognitive impairment in juvenile murine unilateral hippocampal synaptic plasticity. 5
The brain is affected by radiation exposure according to age, brain region, and sex. Of the different brain regions, the cortex and the hippocampus are mostly sensitive to irradiation. The hippocampus is an active site of neurogenesis in the mammalian brain and is considered the most affected brain region regarding depression-like effects.6–9 It is well known that, oxidative stress produced after radiotherapy destroys neurons mainly by apoptosis, inflammatory signaling, and reactive oxygen species (ROS), which oxidize the cellular components, such as proteins, lipids, and DNA. This subsequently decreases the volume of the hippocampus, potentially leading to a major depression. 10 The brain and neurons are more vulnerable to oxidative stress because of their high lipid content and oxygen consumption. 11 Hence, ROS are one of the main causes of neurodegeneration, and their involvement in the pathogenesis of major depression is unequivocally established. 12 Likewise, the potency of oxidative stress is correlated to the severity of the depression. 13 Thus, suitable antioxidants, which scavenge ROS, could be an effective approach for preventing and treating depression, especially in patients undergoing radiotherapy.
Glucosamine hydrochloride is a natural constituent of glycoproteins mainly found in connective tissues and gastrointestinal mucosal membranes. Therefore, it is considered an effective therapeutic agent for treating osteoarthritis, 14 gastritis, and inflammatory bowel diseases, 15 as well as lung injury. 16 Glucosamine is found to be a major constituent of brain glycogen, in addition, it has a protective effect against brain damage,4,17 but its role in managing depression-like effect has not been established yet. Glucosamine is considered an effective compound in the health care field as it could alleviate oxidative stress through its multiple antioxidant activities as glucosamine showed a significant reducing power and superoxide/hydroxyl-radical scavenging ability. 18
Glycogen synthase kinase-3 (GSK3) is a serine/threonine kinase, first found as a phosphorylation-dependent inhibitor of the glycogen synthase metabolic process.19,20 It is widely expressed in many tissues with the highest level in the brain.21,22 GSK3 hyperactivation could contribute to the development of mood disorders, such as major depression, bipolar disorder, and hyperactivity-related illnesses. The inhibition of induced p-GSK3 activity could be a common therapy target for all brain disorders. One of the key mechanisms that suppress GSK3 activation is phosphorylated protein kinase-B (p-AKT)-mediated phosphorylation of GSK3 on serine-9.21,23 This regulating mechanism of AKT on GSK3 needs the activation of phosphatidylinositol 3 kinase (PI3K) for AKT to be phosphorylated and activated, allowing it to phosphorylate GSK3 (p-GSK3) and inhibit its activity.23,24
Consequently, the present work is designed to evaluate the effectiveness of glucosamine as an antidepressant and a radio-protective agent, through measuring the oxidative stress markers and singling pathway of PI3K/p-AKT/p-GSK3 in the brain. Thus, glucosamine may be included as a novel supportive drug in the treatment protocols of cancer patients suffering from depression through their exposure to radiotherapy.
Material and Methods
Animals
Forty Wister premature female albino rats weighing 90–110 g were obtained from the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt. Animals were housed for at least 1 week in the laboratory room of the Animal Care Facility. They were kept in a plastic cage under controlled environmental conditions; room temperature (24°C–27°C), humidity (60% ± 10%), and alternating 12-h light–dark cycle. Food (standard pellet diet) and water were allowed ad libitum. All animals were held and administered the drug by trained staff, and all efforts were made to minimize the number of animals utilized and to decrease their suffering. Adequate care was done, and parameters of health general well-being and indicators of pain and distress were monitored during the experiment period according to documented guidelines. 25 The Research Ethics Committee of the National Center for Radiation Research and Technology (REC-NCRRT), Cairo, Egypt, approved the experimental protocol set from 28.01.2021 (approval code: 10A/21). The reporting of this study conforms to ARRIVE 2.0 guidelines. 26
Irradiation Process
The rats were exposed to fractionated doses of whole-body gamma irradiation (2 Gy for four consecutive days) to induce brain dysfunction and a potential depression-like effect.4,5 The irradiation process was performed at the NCRRT, Egyptian Atomic Energy Authority (EAEA), Cairo, Egypt, using a Canadian Gamma Cell-40 biological radiator (137Cesium), manufactured by the Atomic Energy of Canada Limited, Ontario, Canada, and it is characterized by a uniform distribution of rays. The irradiation chamber’s dimensions are 10 cm (height) X 40 cm (diameter), which accommodated a complete animal group at a time. The radiation dose rate was .655 rad/sec at the time of exposure. The irradiation process was performed in the morning; it takes 6 min for each fraction dose. The Dosimetry Department members at the NCRRT have carried out the dosimetry dose validation on a scheduled basis to ensure the dose rate of the gamma-ray source, the absorbed dose received by the animals, and the uniformity of dose via dose mapping measurements.
Treatment
Glucosamine sulfate (Joflex capsules, 500 mg) purchased from Pharaonia Pharmaceuticals Co., Cairo, Egypt, was used in the investigation. The dose was 1 g/kg b. wt orally. Distilled water was added to the finely powdered glucosamine sulfate, mixed on a Vortex mixer for 1 min, and sonicated for 5 min or until all solids dissolved. Each 100 g rat body weight was administered with .5 mL.28
Experimental Design
Forty rats were randomly classified into four groups (n = 10/group) as follows: Group 1 (control): rats were administered distilled water. Group 2 (irradiation): rats were administered vehicle alone (distilled water) and exposed to fractionated doses of whole-body gamma radiation at a dose level of 8 Gy 4 (2 Gy for 4 consecutive days). Group 3 (glucosamine): rats treated with 1 g/kg glucosamine, P.O. 27 Group 4 (glucosamine + irradiation): rats treated with 1 g/kg glucosamine, P.O., 1 h before each irradiation dose. Behavioral tests were performed 24 hrs after the last dose of the irradiation process. Then the animals were sacrificed under deep anesthesia using urethane (1.2 g/kg) 28 and human-killed by cervical dislocation and brain tissues were dissected out, rinsed with saline, dried on a filter paper, and hippocampi were rapidly excised according to the technique of Sierakowiak et al. 29 The hippocampal tissues were homogenized in ice-cold phosphate buffer saline (PBS) to prepare 10% (w/v) homogenates and kept at −80°C for biochemical analyses.
Depression-like Behavioral Tests
For all the behavioral tests, rats have been trained for 1 day before the test. At the end of the experiment, they were allowed to perform the tests separately. Each animal was videoed for 5 min, and then a manual recording of the events was done in a blind way with codes to avoid any bias. The behavior tests were done for each group parallel together as 3 rats were taken for each test separately to avoid animals being exposed to overstress due to behavior tests.
Forced Swimming Test
FST is a specific test for assessing depression-like effect. Rats were individually forced to swim in transparent cylindrical glass containers (40 cm in height and 22 cm in diameter) filled with 20 cm of water (23°C–25°C). Animals were trained for the test 1 day before the test. Each animal was allowed to swim for 5 min to record the immobility time, latency to immobile, and climbing time according to the method of Yankelevitch-Yahav et al. 30 Then, each rat was removed from the water, dried with a towel, and returned to its cage. An increase in the duration of immobility is indicative of depressive-like behavior. 31
Light–Dark Box Test
LDBT is a way of measuring anxiety and depression in rodents. The apparatus for the LDBT consists of a box (42 × 21 × 25 cm) divided into a small (one-third) dark part and a large (two-thirds) lightened part. 32 A restricted opening (3 × 4 cm) connects the two chambers. Prior to introducing each rat to the area, it was cleaned with 10% alcohol to eliminate possible bias due to odors that may remain from the previously tested rats. At the start of the test, each animal was placed in the brightened chamber and videoed for 5 min. Latency time to enter the dark, number of transitions (enter and exit) between two compartments, time spent in the light compartment, and time spent in the dark compartment were assessed. Rats naturally select dark places while escaping bright places. 33 A longer time spent in the light part reflected the depressive-like effect.
Open Field Test
OFT assists the general locomotor activity and depression of rats and adjusts the habituation of other tests. 34 Each animal was placed in the central point of an opened square box (30 × 30 × 15 cm) divided into 16 squares of equal area. Rats were allowed to freely explore the apparatus for 5 min. Then, they were observed for the following parameters: latency time (i.e., time taken to move, a longer time spent in the center zone reflected the depressive-like effect of the rats), ambulation to express locomotor activities (i.e., number of squares crossed), number of grooming events (i.e., body cleaning with paws and picking at the body with mouth), and number of rearing events (i.e., rats stand on hind legs or with their forearm against the wall or in the free air). Prior to introducing each rat to the area, it was cleaned with 10% alcohol to eliminate possible bias due to odors that may remain from the previously tested rats. 35
Oxidative Stress Biomarkers in the Hippocampus
Lipid peroxidation (LPO) was estimated in the hippocampi homogenate by thiobarbituric acid assay, which is based on malonaldehyde (MDA) reaction with thiobarbituric acid creating thiobarbituric acid reactive substances (TBARS), a pink color complex was formed, and the absorbance was measured at 532 nm according to Yoshioka et al. 36 Reactive oxygen species (ROS) were measured using a kit purchased from LifeSpan BioSciences, Inc., USA (Cat. No: LS-F97590). Reduced glutathione (GSH) was determined by a colorimetric assay kit purchased from Bio Diagnostic, Egypt, according to Beutler et al. 37
Enzyme-Linked Immunosorbent Assay (ELISA) Detection of Biogenic Amines and Phosphorylated Glycogen Synthase Kinase (p-GSK3) Pathway Parameters
The homogenized hippocampi were used for estimating biogenic amines (i.e., serotonin [ST], norepinephrine [NE], and dopamine) and p-GSK3 pathway parameters using ELISA kits. The dopamine kit was purchased from Eagle Biosciences, Inc., USA (Cat. No: DOU39-K01). The ST (catalog no: MBS9362408), NE (catalog no: MBS269993), p-GSK3 (catalog no: MBS730623), phosphorylated protein kinase-B (p-AKT) (catalog no: MBS702819), and phosphatidylinositol 3 kinase (PI3K) (catalog no: MBS702819) kits were purchased from MyBioSource, Inc., USA. All parameters were estimated according to the catalog instruction guidelines.
Statistical Analysis
The data were expressed as mean ± standard error (SE). All results were analyzed using one-way analysis of variance (ANOVA), followed by the Tukey–Kramer post-comparison test and graphs were plotted using Prism 5 (GraphPad Software, Inc., San Diego, CA, USA). Differences with P-values of less than .05 were considered statistically significant.
Results
It was noticed that no significant differences were detected in any of the tested markers (behavioral and biochemical) between control animals and those given glucosamine without being irradiated.
Effect of Glucosamine on Depression-like Behavioral Tests
The irradiated group revealed a significant increase in immobile time in the FST compared with the control group (160%). Meanwhile, the glucosamine + irradiation group recorded a significant decrease in the time of immobility by 75% in the FST compared with the irradiated group (Figure 1). However, the irradiated group showed a significant decrease in the number of entries/exits (Figure 2(A)) (77%), the number of crossed squares (Figure 2(B)) (60%), and the number of jumps (Figure 2(C)) (50%) compared with the control group in the LDBT. Pretreatment with glucosamine showed a significant increase in the number of entries/exits (50%), number of crossed squares (150%), and number of jumps (200%) compared with the irradiated group in the LDBT. The irradiated group showed a significant decrease in the time spent in the dark by 40% and a significant increase in the time spent in the light by 94%, compared to the control group. Administration of glucosamine before irradiation significantly increased the time spent in the dark by 67% and a significant decrease was observed in the time spent in the light by 63% compared to irradiated rats (Figure 2). As a result, glucosamine administration before irradiation increased the locomotive activity, which appears in the increased entry, crossed squares, and jumps. Effect of glucosamine on forced swimming test immobility time (A), latency to immobile (B), and climbing time (C). Each value represents mean ± SE. Statistical analysis was performed using one-way ANOVA, followed by the Turkey–Kramer multiple comparisons test. @ Significantly different from control group at P < 05. # Significantly different from the irradiated group at P < 05. Effect of glucosamine on dark/light box: Number of transitions between two compartments (A), time spent in the light compartment (B), time spent in the Dark compartment (C), and number of jumps (D). @ Significantly different from the control group at P < 05. # Significantly different from the irradiated group at P < 05.
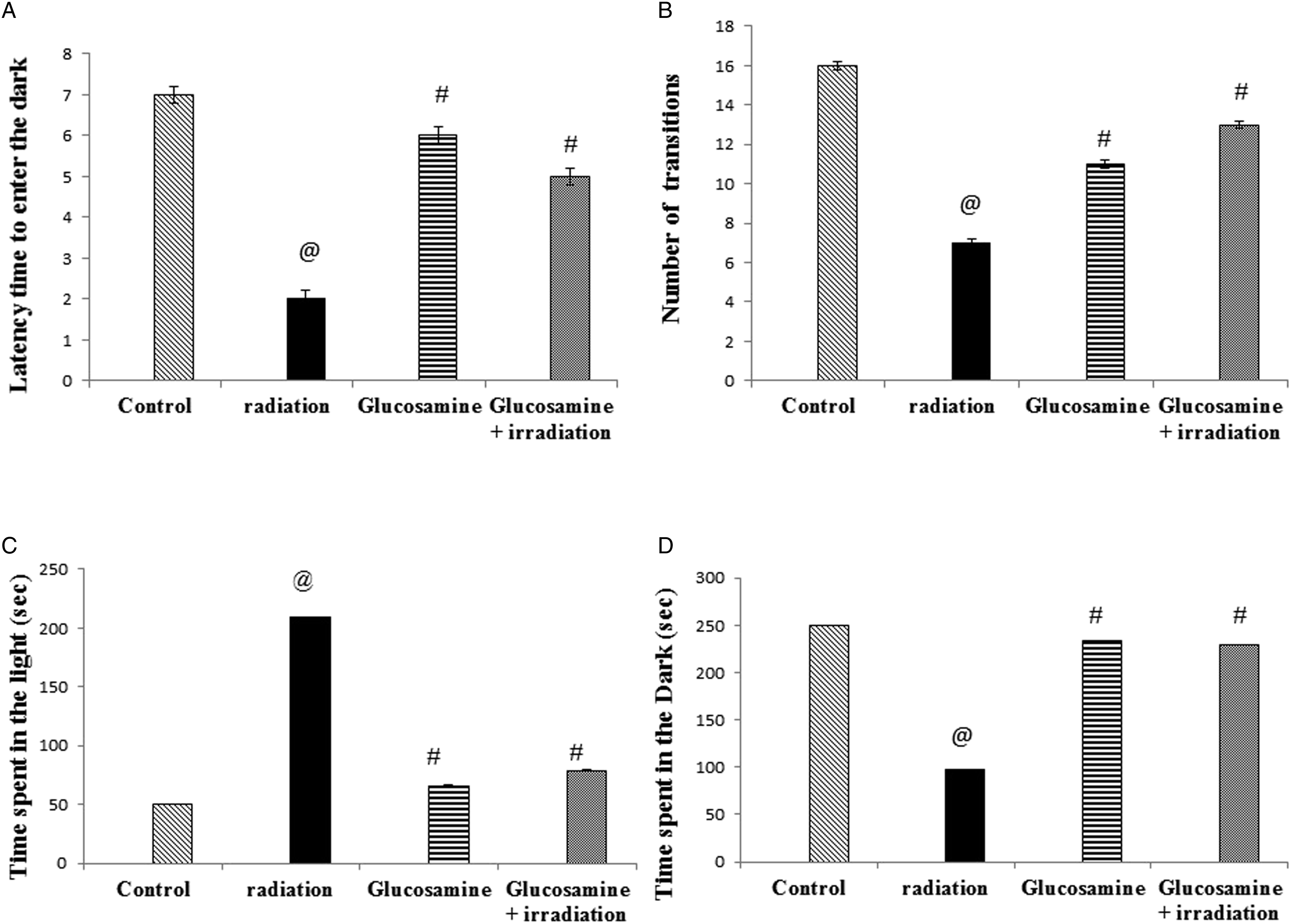
Rats exposed to gamma radiation showed a significant elevation in latency time (33%) (Figure 3(A)) and rearing (63%) (Figure 3(C)) compared with the control group in the OFT. Glucosamine administration before irradiation showed a significant decrease in the latency time (75%) (Figure 3(A)) and a significant increase in rearing (17%) compared with the irradiated group. Ambulation (Figure 3(B)) and grooming in tests (Figure 3(D)) were significantly reduced in the irradiated group (50% and 50%, respectively) compared with those in the control group. Animals that received glucosamine displayed a significant increase in ambulation and grooming compared with those in the irradiated group (100% and 25%, respectively) (Figure 3(B) and 3(D)). Effect of glucosamine on open field test: latency time (A), ambulation (B), rearing (C), and grooming (D). @ Significantly different from the control group at P < 05. # Significantly different from the irradiated group at P < 05.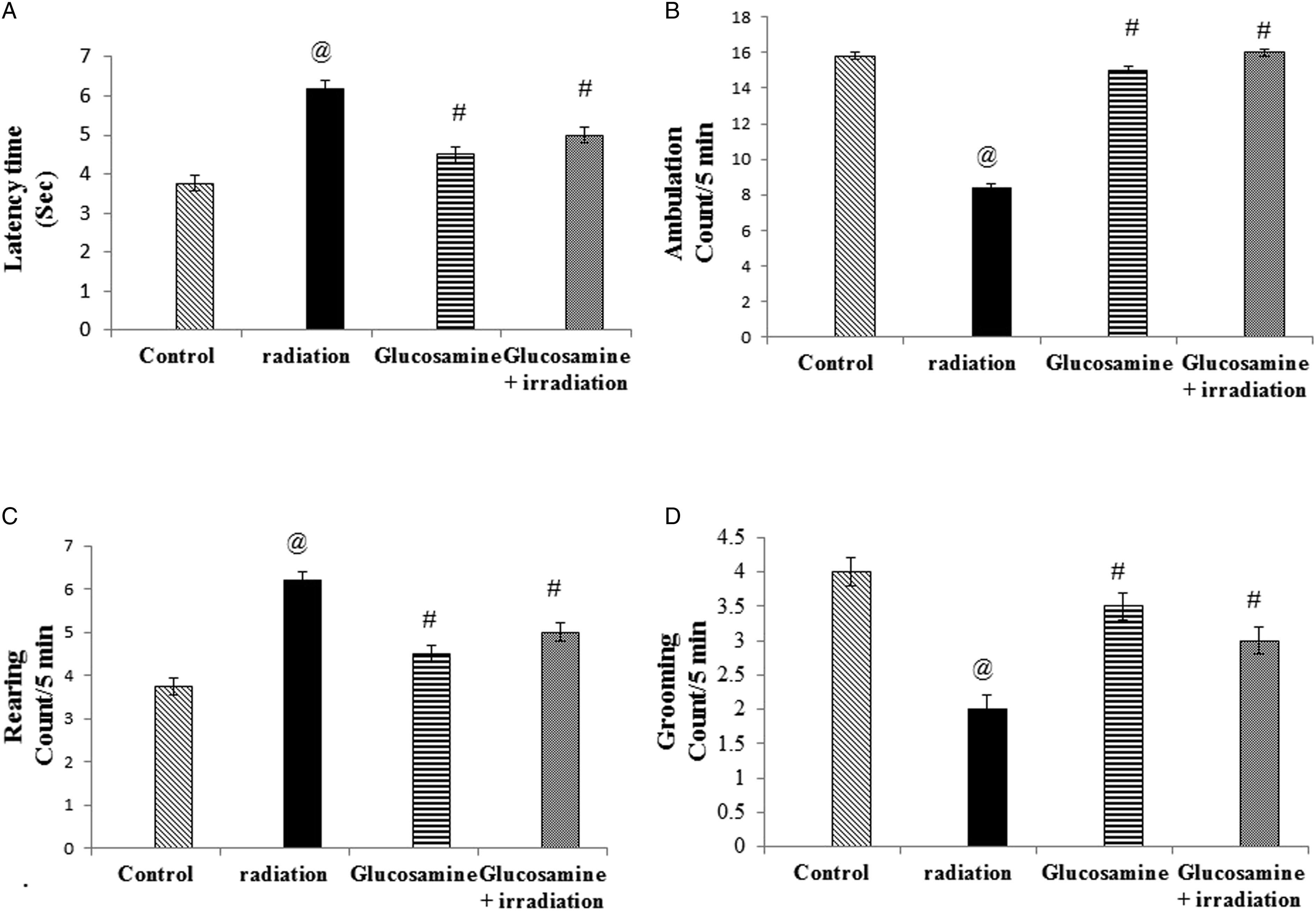
Effect of Glucosamine on Oxidative Stress Biomarkers (MDA, ROS, and GSH)
MDA and ROS contents in the hippocampal homogenate significantly increased after irradiation by 50% and 150%, respectively, compared to the control rats (Figure 4(A) and(c)). While GSH contents significantly reduced (58%) in the irradiated rats compared with those in the control group (Figure 4(B)). Pretreatment with glucosamine prevented the increase in MDA level (50%) and led to a significant increase in the GSH contents (250%) as compared to the irradiated group. Effect of glucosamine on MDA (A), GSH (B),the and ROS (C) concentrations in brain tissue of irradiated rats. @ Significantly different from the control group at P < 05. # Significantly different from the irradiated group at P < 05.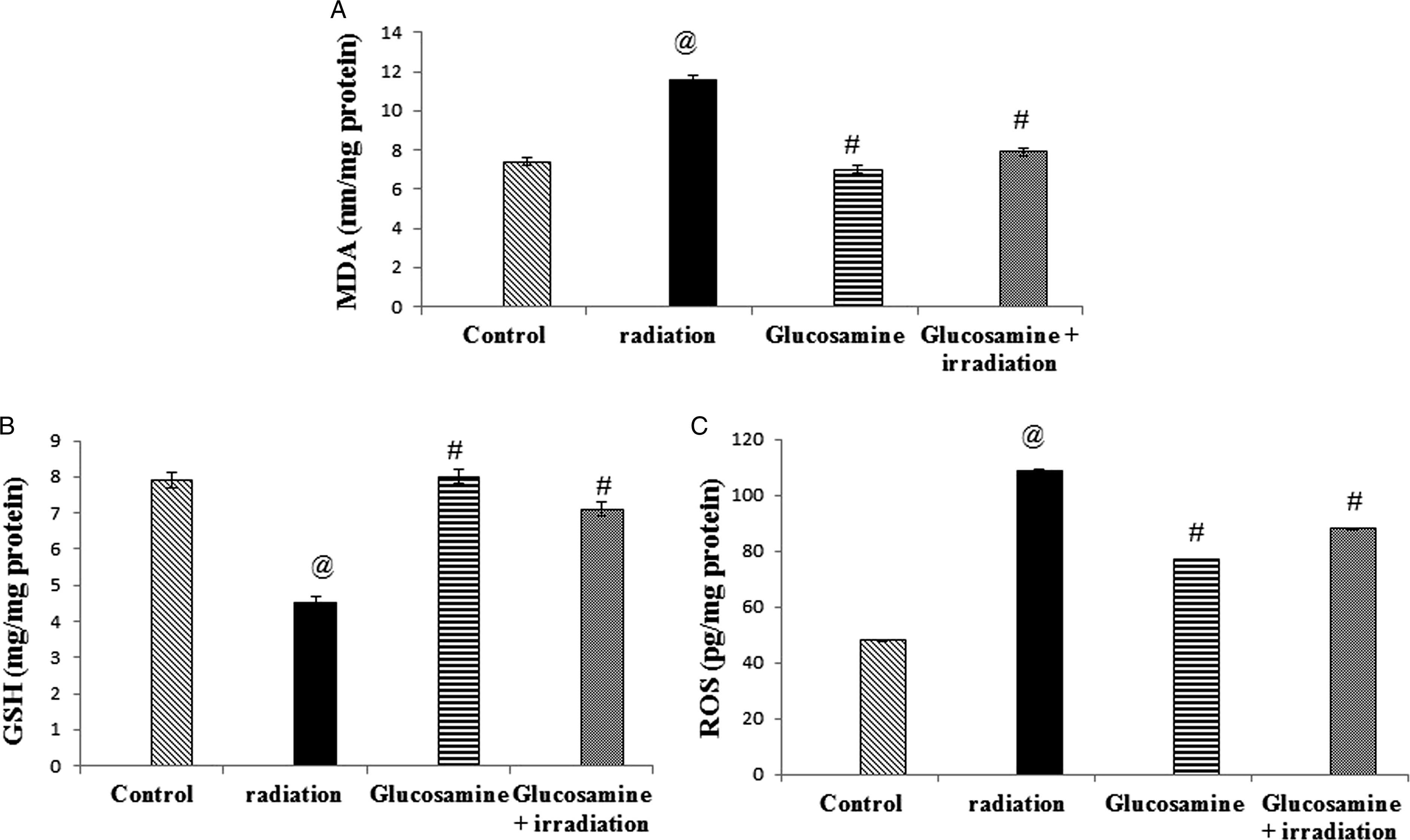
Effect of Glucosamine on Neurotransmitters (Serotonin [SE], Norepinephrine [NE], and Dopamine)
SE, NE, and dopamine contents significantly decreased (67%, 66%, and 90%, respectively) after radiation exposure compared to the control group (Figure 5(A)-(C)). The glucosamine + irradiation group showed a significant increase in the neurotransmitter’s concentration compared with the irradiated group by 250%, 150%, and 107%, respectively (Figure 5(A)-(C)). Consequently, this revealed an improvement in the mode after glucosamine treatment. Effect of glucosamine on serotonin (A), norepinephrine (B), and dopamine (C) contents in brain tissue of irradiated rats. @ Significantly different from the control group at P < 05. # Significantly different from the irradiated group at P < 05.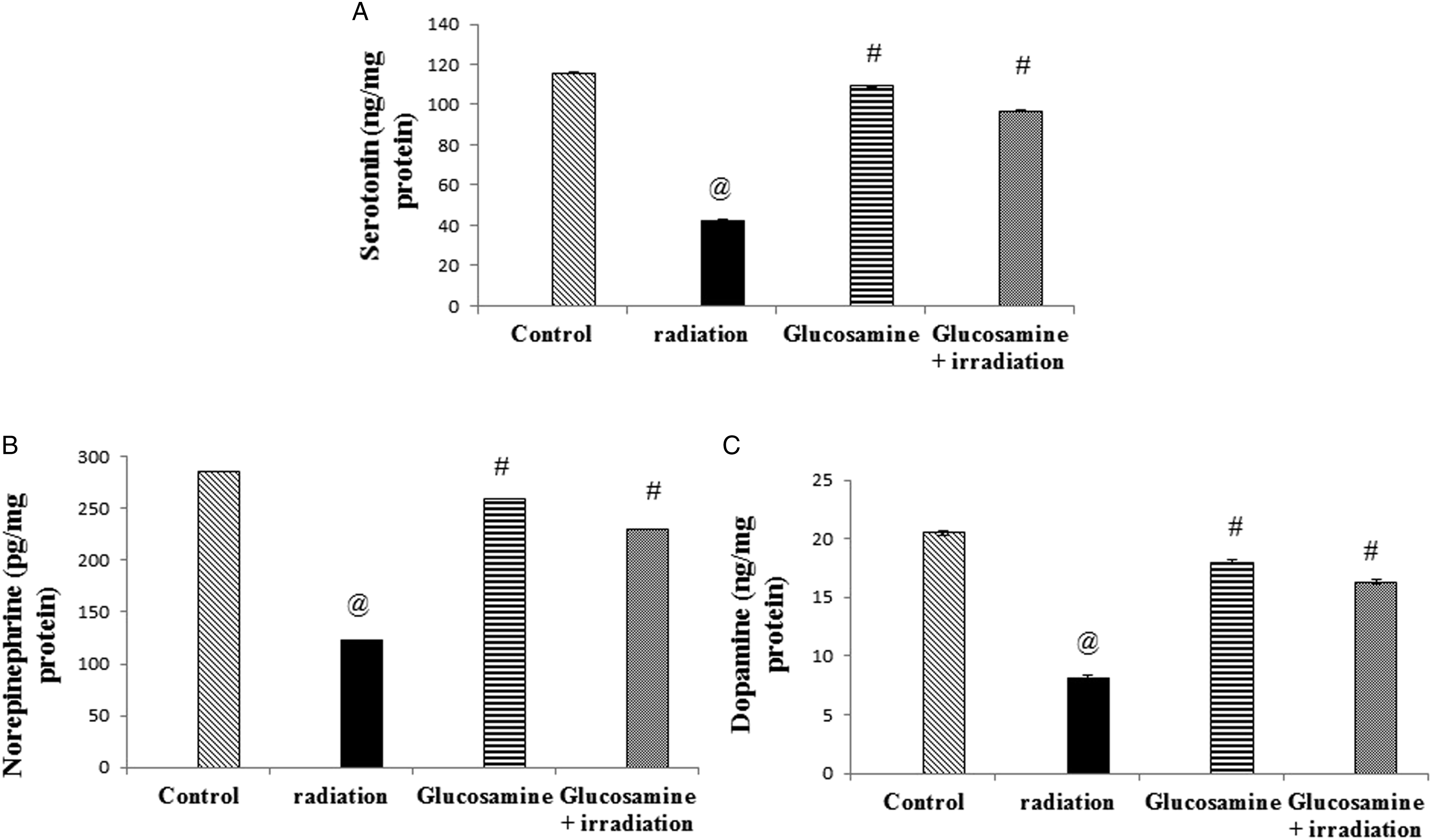
Effect of Glucosamine on Glycogen Synthase Kinase Pathway
Exposure to gamma radiation revealed a significant decline in PI3K and p-AKT contents (57% and 63%, respectively) compared with the control group. The glucosamine treatment pre-irradiation succeeded in returning the levels of PI3K and p-AKT to the normal values (101% and 102%, respectively) (Figure 6(A) and (B)). Rats exposed to gamma radiation showed a significant increase in p-GSK contents (175%) compared with the control group (Figure 6(C)). The glucosamine + irradiation group presented a significant reduction in p-GSK concentration compared with the irradiated group (64%). Pretreatment of irradiated rats with glucosamine improved the glycogen synthase pathway. Effect of glucosamine on P13K (A), AKT (B), and GSK (C) contents in brain tissue of irradiated rats. @ Significantly different from the control group at P < 05. # Significantly different from the irradiated group at P < 05.
Discussion
The present study was carried out to investigate the possible antidepressant effect of glucosamine on the depressive-like consequence resulting from gamma irradiation in female rats. To achieve this aim, FST, LDBT, and OFT were performed. The obtained data showed that gamma irradiation significantly increased the immobility time of FST, while glucosamine given before irradiation significantly reduced the immobility time. This test was conducted by Slattery and Cryan 38 who stated that the FST is the most extensively used test for evaluating antidepressant effectiveness and also used to distinguish the antidepressant impact from the stimulatory effect. 39 According to Gorlova et al, 40 the disturbance in behavioral symptoms in neuropsychiatric diseases was attributed to the elevation in oxidative stress. The antidepressants exert their therapeutic role by quashing the production of ROS and increasing antioxidant protection.
The data obtained from LDBT showed that the number of entries/exits, the number of squares crossed, the number of jumps, and the time spent in the dark decreased after irradiation. However, glucosamine administration reversed these actions. The LDBT was based on the nature of rodents, that rodents favor dark environments, and avoid lighted ones and on their proclivity for exploratory behavior. 32 Additionally, the increase of transitions is an index of the locomotive activity and tendency of rats to explore. 41 Likewise, glucosamine acted in the present study. This natural behavior has been used to determine the degree of anxiety in animals and depression-like effect. 42
In the current study, an increase of rearing and latency time in OFT occurred after irradiation, reversely glucosamine acted. The rearing activity is a critical indicator of depression-like behavior. 43 It has been proposed that rearing is a good indicator of environmental novelty and that hippocampal realizing is an important component of the system that controls rearing in novel situations. 44 These findings confirmed the results of previous studies indicating that persistent tetrahydrocannabinol therapy in rats reduces the rearing activity.45–47 The current results showed a remarkable improvement in all behavioral measurements of the irradiated rats pretreated with glucosamine, confirming the glucosamine’s neuroprotective effects, which is also supported by the biochemical parameter estimation.
The pathophysiology of depression and other brain illnesses, such as Parkinson’s disease, Alzheimer’s disease, bipolar disorder, major depression, and schizophrenia, has been linked to oxidative stress,48–52 which appears with radiotherapy. Furthermore, there is evidence that raising brain GSH levels (via the GSH precursor N-acetylcysteine) can help people with such diseases.53,54 The present study revealed that irradiation exposure significantly increased the MDA and ROS levels and significantly decreased the GSH content in the hippocampus of brain tissues which is in line with what was reported by Said et al. 55 It was found that a whole-body gamma irradiation (5 Gy) caused an imbalance between oxidant and antioxidant species in the cerebral hemispheres of rats. Moreover, an impairment of hippocampal function after a 4 Gy dose of gamma irradiation exposure was reported. 56 Oxidative stress-induced ROS is the main cause of radiation side effects. 57 However, brain tissue lipid content is 60%, and it consumes a lot of energy. 58 Because of its high O2 usage rate, a high content of polyunsaturated fatty acids formed (which are prone to lipid peroxidation) and a high content of iron, which increases the generation of free radicals via Fenton reactions. Furthermore, compared with other organs, the brain is deficient in antioxidants. 59 Therefore, the brain has been considered a radiosensitive organ. 60 A reduction in GSH, the most common cellular non-protein thiol in the brain, plays a critical role in protecting cells from the damaging effects of radiation-induced ROS. 61
It was noticed that no significant differences were detected in any of the tested markers between control animals and those given glucosamine without being irradiated, indicating that glucosamine administration is safe. Likewise, glucosamine administration before irradiation improved the antioxidant and oxidative stress parameters. Glucosamine has a regulatory function in the activation of the hexosamine biosynthesis pathway. 62 The ability of glucosamine to block the activation of nuclear factor kappa B (NF-κB) was largely responsible for its anti-inflammatory actions. 16 Glucosamine helps in managing the stress response and reducing inflammation, hence preventing tissue damage. 63 The study of Hwang et al 6 64 supported the present findings; they reported that glucosamine or its derivatives have been suggested as potential neuroprotective or anti-inflammatory drugs. Moreover, Chen et al 65 proved that glucosamine could protect retinal ganglion cells against oxidative stress-induced injury by modulating protein O-GlcNAc glycosylation.
Antidepressant medicines enhance the reduced GSH levels, which are essential for depression treatments because lowered GSH levels have been documented in the brains of people with bipolar disorder, schizophrenia, and mental disorders. 52 The effects of antidepressant medications on lipid peroxidation appear to be linked to increases in the reduced GSH levels. The lower dose of fluoxetine, which also raised the GSH levels, reduced the increase in MDA levels in mice brains caused by lipopolysaccharide endotoxin, suggesting an antioxidant impact. 66 As well, glucosamine could be used to improve heart and brain damage induced by gamma radiation exposure or aluminum chloride due to its antioxidant effect. 4
Normally, ST and NE are important modulators of pain perception and depression; therefore, it is logical to assume that impairments in both functions are the result of abnormalities in SE and NE metabolism and transmission.
67
The current results showed a significant decrease in monoamine levels after irradiation (i.e., SE, dopamine, and NE); however, glucosamine administration attenuated all of them. These observations came by those of Said et al.
55
They described that radiation-induced oxidative stress and monoamine oxidation could decrease monoamine levels in the cerebral hemispheres of irradiated rats. This drop in monoamines could be due to the decrease in their synthesis because of radiation-induced injury to the ileal mucosa, or the reduction in net ilea absorption, where the decrease in tryptophan absorption reduces SE synthesis, and the decrease in
AKT (protein kinase B)-mediated phosphorylation of glycogen synthase kinase-3 (GSK-3) on serine-9 is one of the primary mechanisms that regulate GSK-3 activation. For AKT to be phosphorylated and activated, phosphoinositide 3-kinase (PI3K) must be activated which allows the phosphorylation of GSK-3 and suppression of its function.23,24 Otherwise, the 5-hydroxytryptamine (5-HT) (ST) receptor stimulation is thought to stabilize the dephosphorylated form of GSK-3, resulting in enhanced kinase activity. Agents that induce inhibitory serine-9 phosphorylation of GSK-3 have been demonstrated to improve the serotonergic activity by blocking 5-HT re-uptake transporter, inhibiting 5-HT metabolism, or increasing 5-HT release from the presynaptic neuron. According to these findings, serotonergic neurotransmission may play a key role in controlling GSK-3 activity in the brain via serine-9 phosphorylation-mediated inhibition. The lack of serotonergic neurotransmission seen in depression would corroborate this idea by contributing to impaired GSK-3 activity.23,69
The therapeutic effects of various psychiatric medicines are mediated in part by their ability to suppress the PI3K/AKT/GSK3 signaling. The mood stabilizer lithium, for example, competitively reduces the Mg2+-adenosine triphosphate-dependent catalytic activity of GSK3, and it has been used as a treatment for schizophrenia, depression, and other mental diseases. 70 The antipsychotic medication haloperidol has been associated with AKT signaling suppression. 71 The present study showed that, irradiation activated PI3K/p-AKT/p-GSK3 signaling, whereas glucosamine administration before irradiation inactivated PI3K/p-AKT/GSK3 signaling. In the same line, the neuroprotective effects of curcumin could be mediated by managing oxidative stress regulating PI3K/AKT/GSK3 signaling pathway.72,73 Various plants or fruit components may also be beneficial. Likewise, liquiritigenin and kaempferol are plant constituents that may be effective in protecting neurons and treating major depression by modifying PI3K/AKT/GSK3 signaling.74,75 Therefore, the antioxidant effect of glucosamine was established by inhibiting the PI3K/p-AKT/p-GSK3 signaling pathway, decreasing oxidative stress markers, and increasing monoamines.
Conclusion
It could be concluded that glucosamine has a potent anti-depressant-like effect by regulating the PI3K/p-AKT/p-GSK3 signaling pathway and decreasing ROS as well as managing the behavioral status. Thus, it could be utilized as a promising drug for some neuropsychiatric disorder treatments or a controlling agent against the side effects of radiation during radiotherapy. Further studies are recommended to identify different mechanisms of glucosamine as an antidepressant drug on a molecular basis. In addition, clinical trials are most important to figure out the beneficial effect of glucosamine on depressive patients exposed to radiotherapy.
Footnotes
Acknowledgments
The authors are appreciative of the staff members of the gamma irradiation unit of the NCRRT for their generous support in performing the experimental irradiation.
Authors Contributions
All authors have similar contributions in conceptualization, methodology, formal analysis, investigation, data analysis, writing, reviewing, and editing the manuscript. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The authors confirmed that all needed data and materials are included inside the manuscript, and any raw data are available upon request. Please contact the corresponding author for data availability.
