Abstract
The aerobic organisms not only need oxygen for survival, but oxygen is also fundamentally malignant to the aerobic organism on the grounds of free radical generation and their affiliation with free oxidative stress. This study was done to evaluate the antioxidant and protective properties of P kurroa, S aromaticum, L inermis, R emodi, and C longa against lipid peroxidation induced by different pro-oxidants. The aqueous extracts of these medicinal plants showed inhibition against thiobarbituric acid reactive species (TBARS) induced by different pro-oxidants (10 mM FeSO4 and 5 mM sodium nitroprusside) in the brain and liver of mice. Moreover, the free radical scavenging activities of the extracts were evaluated by the scavenging of the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical. L inermis, S aromaticum, and R emodi showed higher inhibitory effects, which could be attributed to their significantly reduced ability and free radical scavenging activities. Therefore, the oxidative stress in the brain and liver could be potentially managed or prevented by the dietary intake of L inermis, S aromaticum, and R emodi plants, which justifies the use of these plants in various degenerative diseases. C longa and P kurroa showed relatively weak antioxidant activities.
Introduction
The aerobic organisms not only need oxygen for survival, but oxygen is also fundamentally malignant to the aerobic organism on the grounds of free radical generation and their affiliation with free oxidative stress. In the bodies of living organisms, there are several free radicals formed as general respiration by-products and from several biochemical reactions in cells, which are common physiological procedures that are necessary for living. For survival in the aggressive habitat of oxygen, antioxidant water is produced by these living organisms, which can counteract extremely volatile free radicals. 1 Physiological stress, that is, oxidative stress, should be stabilised by the body's antioxidant defence system. Biochemicals, for example, fats, proteins, carbohydrates, and nucleic acids, can be harmed by the overabundance of free radicals if the antioxidant system of the body is not working in favourable conditions. Free radicals can be generated as products of homoltica, heteroltica, or redox reactions and are usually composed of reactive oxygen species and reactive nitrogen species. 2 (S. Shaikh et al; Shiraz Shaikh et al.)
The human body also contains approximately 4.5 mg of iron, most of which is located in haemoglobin and other proteins. However, a small amount of iron, through the formation of complexes with a variety of small molecules, can react with hydrogen peroxide, resulting in the production of the Fenton reaction's hydroxyl radical. Normally, the oxygen radicals hydroxyl, nitrogen, superoxide, and radical alkoxy and peroxyl radicals are the liberated oxygen-centred radicals (activated oxygen generation) that offer an essential part in the oxidative stress initiation.
3
Nitric oxide is not synthesised from
There is a dual origin of free radicals, one being the exogenous forms and the other being the endogenous source. Radical hydroxyl, hydrogen peroxide, anion superoxide, radical peroxyl, singlet oxygen, and ozone are some of the many general reactive oxygen species.
The physiological process reviews the mitochondrial respiration process, essential to life, which generates radicals, superoxide anion. The autooxidation of many biological molecules (haemoglobin, Myoglobin, catecholamines, etc.) in the human body can produce free radicals. These radicals are some of the more dangerous free radicals found in Physiology.
4
Singlet oxygen is a form of electronically excited oxygen, but it is not a free radical. Nitric oxide (NO) is synthesised from
There are external sources of free radicals. For example, breathing polluted air can expose people to free radicals. These small particles in polluted air have radical activities that can cause lung inflammation and damage to the cells of the lung. 5 If possible, it is preferable to stay home as much as possible, especially when the air quality is bad on hot days, when free radicals containing particles may be at ground level.
Certain medications can also cause oxidative stress. Anticancer drugs such as methotrexate, Adriamycin, bleomycin, doxorubicin, inhibitors of topoisomerase, and menadione can cause oxidative stress. Immunosuppressive drugs are used to prevent organ rejection in transplant recipients.
Mycophenolic acid, cyclosporin A, and sirolimus may induce oxidative stress, while the immunosuppressant tacrolimus produces oxidative stress of a lesser magnitude. The antiretroviral drugs used in the treatment of patients with HIV infection, even common ones like aspirin, can induce oxidative stress. A deficiency of glucose-6-phosphate can put you at greater risk of toxicity from some of these medications. Excessive alcohol consumption can induce considerable oxidative stress, especially in alcoholics. 6
Many industrial solvents, such as the tetrachloride of carbon, chloroform, trichloroethylene, benzene, xylene, and toluene, may induce oxidative stress, and some of these chemicals are also carcinogenic. It has been proposed that these solvents can activate enzymes of cytochrome P450 (especially CYP2E1), which are responsible for the metabolism of most of the drugs in the liver and induce stress oxidative. 7 Vitamin D deficiency can lead to an increased risk of fractures and can also cause rickets. Proper exposure to sunlight is the best way to prevent a deficiency of vitamin D.
Radiation exposure during airport security checks is generally considered safe, while radiation therapy to treat cancer induces oxidative stress. 8 Free radicals are extremely volatile in nature, due to which they can destroy nearly every biomolecule, including carbohydrates, lipids, proteins, and nucleic acids. Preoxidized lipids might result in destroying the membrane of the cell by modifying the membrane's fluidity and penetrability. Free radicals, when induced by lipid peroxidation, form a series of chain reactions that inaugurate, propagate, and terminate. The initiation of lipid peroxidation due to the presence of free radicals can contain radical peroxyl, radical hydroxyl, alkoxyl radical, and peroxynitrite that are highly reactive. Metal ions can also initiate the chain, for example, ferrous and cuprous irons. There are some secondary oxidation by-products that are products of a series of elements called isoprostane prostaglandins, also referred to as monocyclic and cyclic peroxides of series. Isoprostanes have remarkable characteristics of endogenous oxidative stress and lipid peroxidation, especially F2-isoprostane. 9
The carbonyl group can enter the protein by the reaction with aldehydes (malondialdehyde and 4-hydroxy-2-nonenal) or by reaction with cetoaminas, cetoaldehdos, and deoxyosones as a result of the reaction of sugars or their oxidation, which generates products with protein lysine residues. 10 Advanced glycation end products are formed due to the reaction between the carbohydrates and proteins amino groups. Haemoglobin A1c, a marker of glucose in the blood, is a product of haemoglobin glycation. The by-products of advanced glycation are produced through several intermediary outcomes, such as Schiff base, Maillard, and Amadori. Mutagenesis, ageing, and carcinogenesis are causes of hydroxyl radicals that can damage the DNA too. Oxidation of DNA can directly modify apurinic sites and nucleotide bases due to reactive nitrogen and oxygen species. The hydroxyl radical may interrelate with the C4, C5, and C8 positions under the guanine imidazole ring as all of the nucleotides are capable of oxidative damage. So the production of 8-hydroxyguanine (8-dehydroalanine, also known as 8-oxo-7) may also be used as an attribute of DNA damage. Peroxynitrite is observed as a reason for DNA damage; this can also react with guanine to make 8-nitroguanidine. 11
Oxidative stress can also cause damage to mitochondrial DNA, and such damage has been associated with ageing and the development of cancer in different tissues, including human skin. 12 RNA oxidation by reactive oxygen species has also been reported, but it has not been studied as much as the oxidation of DNA. Both ribosomal RNA and messenger RNA can be oxidised by reactive oxygen species. Mitochondrial DNA is more vulnerable to oxidative damage than nuclear DNA due to mitochondrial respiration, which generates reactive oxygen species. In the ageing process, the accumulation of mitochondrial DNA mutations and the deterioration of oxidative phosphorylation, as well as an imbalance in the expression of antioxidant enzymes, result in the additional production of reactive oxygen. This overproduction of mitochondrial dysfunction caused by reactive oxygen species is the basis of the mitochondrial free radical theory of ageing. 13
It is known that low concentrations of ROS are able to induce the expression of anti-oxidant enzymes and other defense mechanisms. The basis to explain this phenomenon refers to the concept of hormesis, 14 which is a dose–response relationship in which a given substance is stimulatory at low dose while at high doses exerts inhibitory effects. Radical active species may be beneficial since they act as signals to enhance cellular defenses but become deleterious when present within cells at high levels. ROS when in excess over long-term disrupt redox homeostasis, cause oxidative stress, loss of molecular fidelity which underlie accumulation of unfolded or misfolded proteins in brain. Alzheimer’s, Parkinson’s, Huntington’s, amyotrophic lateral sclerosis, and Friedreich ataxia all belong to the so called “protein conformational diseases” affecting several thousands of aged people all over the world. 15 Unfolded protein response put in motion by chaperons actively rescue misfolded proteins, breaking up aggregates, and assisting the refolding process. Those proteins that cannot be rescued by refolding are, however, delivered to the proteasome by other chaperones and recycled. 15 In general, an unfolded protein response occurs within conformational diseases characterized by dysfunctional aggregation of proteins accumulating in a non-native conformation. Under these physiopathological conditions, multiple metabolic derangements generally take place in association to excessive production of ROS and oxidative stress. 15
The antioxidant defence body is made up of both endogenous and exogenous compounds derived from the diet, which can be classified into three broad categories: antioxidant enzymes, anti-oxidants from breakage of chains, and metal-binding proteins. The main antioxidant enzymes are superoxide dismutase (SOD), catalase, and peroxidases, which are endogenous in origin. 16 As an antioxidant, probably the most important antioxidant enzyme is superoxide dismutase, which neutralises harmful superoxide anion radicals by converting them to hydrogen peroxide. There are three forms of superoxide dismutase. 17 SOD1 requires both copper and zinc for its function and is located in the mitochondria as well as the cytosol. Superoxide dismutase (SOD2) and manganese occur primarily in mitochondria. The third form of superoxide dismutase (SOD3) is located outside the cells. SOD3 also requires zinc and copper as cofactors. The concentrations of these enzymes are regulated by appropriate genes. 18 Glutathione peroxidase (GPx) belongs to a family of enzymes related phylogenetically, comprising eight Glutathione peroxidases (GPx1–GPx8). Collectively, GPx containing selenium (GPx1, 4, and -6) as well as fellow nonselenium (GPX5, -7, and -8) play an important role beyond detoxifying hydroperoxides. In humans, GPx isoforms 1-4, and 6 contain selenium residues that are important for their catalytic activities, and all the members of this group use reduced glutathione to catalyse the reduction of hydrogen peroxide in water. 19 GPx1 was the first identified selenoprotein. It is a homotetramer and one of the most abundant and ubiquitous forms of expressed selenoproteins that play an important role in the total recovery of cells after oxidative stress. GPx1 can react with hydrogen peroxide and low-molecular-weight hydroperoxides that are low-molecular-weight soluble, such as tert-butyl, acids hydroperoxy fatty, and lisofosfatidos hydroperoxide, but not with more complex lipids.
However, GPx can react with these more complex lipid hydroperoxides. In the process of the hydroperoxide reaction, glutathione becomes glutathione disulfide. The glutathione reductase converts the new glutathione to a reduced form for an additional reaction. GPx1 plays a role in the protection of individuals from cancer and neurodegenerative diseases and is expressed in the gastrointestinal tract as well as the liver. 20 GPx2 is a homotetramer and is closely related to GPx1. GPx2 is mainly expressed in the gastrointestinal system and liver. GPx3 is a tetramer and is located in plasma. You can play a role in the regulation of the bioavailability of nitric oxide produced by platelets and vascular cells. GPx4 is a monomer but may react with hydroperoxides in lipid complexes such as phospholipids, cholesterol, and cholesterol hydroperoxides, even when inserted in membranes and lipoproteins. GPx exists in three different isoforms: a Cytosolic isoform, a Mitochondrial isoform, and a nuclear isoform of spermatozoa.
However, the three isoforms are derived from the same gene. GPx4 seems to play a protective role in cardiovascular diseases, together with GPx1 and GPx3. It is believed that GPx6 is restricted in its expression in the development of the embryo and the olfactory epithelium of adults. 21 GPX5 glutathione is a specific cysteine of the epididymis in mice. It is the closest GPx3 counterpart and seems to play a role in fertility. GPX7 was first described as a new peroxidase glutathione with cysteine rather than selenium in the catalytic centre. GPx8 is the glutathione peroxidase of mammals that was most recently discovered. GPx8 is a protein from the membrane of the endoplasmic reticulum. 22 Thioredoxin is a ubiquitous redox protein, and human thioredoxin is a protein with two reactive cysteine residues and 105 amino acid redox on the Active Centre. This protein plays an important role in cellular responses, including cell growth, cell cycle, gene expression, and apoptosis, to maintain redox balance. 23
Description of Herbs
(Pharmacological Effect of Herbs).

Materials and Methods
Materials
All of the materials under consideration, which are Folin-Ciocalteus reagents, Thiobarbituric acid (TBA), Malonaldehyde-bis-dimethyl acetal (MDA), gallic acid, 2, 2-diphenyl-1-picryl-hydrazyl (DPPH), and gallic acid, were purchased from Sigma (St. Louis,MO,USA). Sodium nitroprusside (SNP) was bought from Merck (Germany), while iron (II) sulphate was obtained from Biochemical, Lahore.
Preparation of Plant Extract
• We purchased the plant from a nearby local market and got it authenticated by a qualified botanist who works at the University of Poonch Rawalkot, situated in Azad Kashmir, Pakistan. Only those parts of the plant were dipped in boiling water that we wanted to use in our experiment. Going forward, we soaked .1 grammes of dried powder in boiling hot water for 15 minutes, which was 100 mL in quantity. 29 Then it was kept at room temperature to cool off. When the underlying water reached room temperature, using Wattman filter paper, we extracted the residues twice. The next step involved the use of a rotary evaporater in order to concentrate the residues. We used an oven to dry the collected filtrates. We set the temperature 2 hours at 160°C (320 °F), which hovered between 40 and 500°C, giving a yield of 21 to 23% and giving the filtrates the desired powdered form. It should be noted that in order to get the desired concentration, we made serial dilutions.
Test Animals
We strictly abide by the guidelines laid down by the NIH guide regarding the use of animals in experimentation. We used Swiss Albino male and female mice weighing between 200 and 250 g and ageing around 2 to 2.5 years. It should be noted that we procured these mice from the National Institute of Health in Islamabad, which has an animal breeding colony. We kept those animals in tidy cages but provided them with unrestricted access to libitum, water, and food. We kept those cages in a room that had a managed temperature of 22°C (3). We also provided a 12-hour light and dark schedule to the subject animals (light was provided from 7 a.m. to 7 p.m.).
In Vitro Assays
Production of Thiobarbituric Acid Reactive Species From Animal Tissues
We used the modified method of Moral et al. (1983) 30 in order to produce the thiobarbituric acid reactive species (TBARS). Then we anesthesized the mice mildly using ether and then placed a Perti Dish on ice on which we then put the collected tissues of brain and liver. We used Teflon glass homogenizer in order to homoginized the samples of these tissues, in cold strokes. It should be mentioned that samples weighed 1°gm individually. For 10°minutes, we centrifuged these tissues at 1400°rev/min in order to form a precipitate. We discarded this precipitate subsequently using a low speed supernatant tissues. We incubated the homoginized tissues both with and without the 50 μl of various oxidants, namely iron and nitroprusside and different concentration levels of plant aqueous extracts alongwith a volume of deionized water. This process was performed in order to obtain 300 μl of total volume at 37°C. It should be duly noted that these oxidants were freshly prepared. By adding 200 and 500 L each of the acetic acid buffer (pH 3.4), 8.1% sodium dodecyl sulphate (SDS), and .6% TBA, the colour reaction was performed. We incubated the reaction mixtures for 60°minutes at 97°C with the ones from serial dilutions of .03 mM standard MDA. After cooling the tubes, the reading of absorbance was taken using a wavelength of 532 nm in a spectrophotometer. We cooled the underlying tubes with the help of a wavelength of 532 nm in a spectrophotometer in order to get the readings of absorbance.
DPPH Radical Scavenging
According to the method stated by Senguttuvan et al,
31
we performed DPPH radical activity was measured using the technique described. 500 L of the various concentrations of vitamin C (
Phenolic Content
2.5 mL of 10% Folin-Ciocalteau’s reagent (v/v), 2.0 mL of 7.5% sodium carbonate, and .5 mL of the aqueous extract were missed for the determination of total phenolic content. At a temperature of 45°C, we incubated the mixture for 30 to 40°minutes. Second, at 765 nm, we measured the absorption in the spectrophotometer. As a standard phenol, Gallic acid was used. 32 Three readings were used to calculate the mean value and all of the phenolics in order to obtain milligrammes of gallic acid equivalents per gramme.
Total Antioxidant Assay
The extracts of the plants were applied in order to form a green phosphate/Mo(V) complex at the acidic pH. Aqueous extracts of plants (each of .1 mg/mL) were mixed with 3 mL of the reagent solution (28 mM sodium phosphate, .6 M H2SO4, and 4 mM amonium molybdate) were mixed. These tubes were kept in incubator at 95°C for 1.5 hours. At room temperature, we cooled the mixture, which was then withdrawn, and at 695 nm, the absorbance was measured later.
Statistical Analysis
We used standard deviations in order to mention the collected results. We used one-way ANOVA in order to statistically analyse the data at hand and the Duncan multiple range test in order to compare groups of different means. P < .05 was the significance level used in all of the cases. For the purpose of data analysis, we used a software package by the name of Statistica.
Result and Discussion
The effects of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum were determined to induce lipid peroxidation with sodium nitroprusside and iron in brain homogenates and livers of mice. These plants were chosen as they are important ingredients in daily life, both in the kitchen and in many herbal formulations.
Inducing Lipid Peroxidation in Liver of Mice with Sodium Nitroprusside and Iron
It has been shown in graph 1 the effects of C longa, L inermis, Picrorhizha kurroa, R emodi and S aromaticum in mice liver induced with iron. The liver tissue increased the production of TBARS after treatment with iron. However, treatment with aqueous extracts of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum caused a significant (P < .05) decrease in lipid peroxidation. On the other hand, less antioxidant activities have been shown against iron lipid preoxidation inducing with comparison to sodium nitroprusside in liver of mice inducing lipid peroxidation. But the order of reducing antioxidant activity was the same as above L inermis > S aromaticum > R emodi > Picrorizha kurroa > C longa. Graph 1 The effect of aqueous extracts of Curcuma longa, Lawsonia inermis, Picrorhizha kurroa, Rheum emodi, and Syzygium aromaticum against lipid peroxidation in mice liver induced by iron.
According to previous study, sought to assess the ability of the aqueous extract of some Nigerian spices [Monodora myristica (Africa nutmeg), Xylopia aethiopica (Ethiopian pepper), S aromaticum (Tropical Cloves), Piper guineense (Black pepper, Aframomum danielli (Bastered melegueta), Afromomum melegueta (Alligator pepper/grains of paradise), and Clerodendrum volubile (Locally known as ‘Obenetete’)]; to inhibit Fe2+-induced lipid peroxidation in rat’s brain (in vitro) using Thiobarbituric acid reactive species (TBARS). The total phenol, total flavonoid, and vitamin C contents, as well as ferric reducing antioxidant property, Fe2+- chelating ability, and the ability of the spice extracts to prevent Fe2+/H2 O2 - induced decomposition of deoxyribose were also determined. The results of the study revealed that incubation of the brain tissues in the presence of 25 µM Fe2 + caused a significant increase (P < .05) in malondialdehyde (MDA) production in the rat’s brain (173.4%) when compared with the basal (100%). However, the spice extracts caused a significant decrease (P < .05) in the MDA production in both the basal and the Fe2+- induced lipid peroxidation in the Rat’s brain, with Alligator pepper showing the highest inhibitory properties while Bastered melegueta had the least inhibition. Conclusively, the inhibitory effect of the spice extracts on lipid peroxidation (basal and Fe2+- induced) could be attributed to high total phenol, total flavonoid and vitamin C contents, as well as ferric reducing antioxidant property, Fe2+- chelating ability and inhibition of Fe2+/H2 O2 - induced decomposition of deoxyribose, with Alligator pepper showing the most promising potentials. 33
Graph 2 shows the antioxidant effect of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum in mice liver induced with sodium nitroprusside. The liver tissue after treatment with sodium nitroprusside increased the production of TBARS. However, treatment with aqueous extracts of C longa, L inermis, Picrorhizha kurroa, R emodi and S aromaticum caused a major (P < .05) decrease in lipid peroxidation. Here again the impact of reducing the TBARS of L inermis and S aromaticum are good as compare to the other plant extracts. The order of decreasing lipid per-oxidation was L inermis > S aromaticum > R emodi > Picrorizha kurroa > C longa. The effect of aqueous extracts of Curcuma longa, Lawsonia inermis, Picrorhizha kurroa, Rheum emodi, and Syzygium aromaticum against lipid peroxidation in mice liver induced by sodium nitroprusside.
According to previous study, turmeric extracts were obtained from two lots of raw material (M and S) using various techniques: hydrodistillation, low pressure solvent extraction, Soxhlet, and supercritical extraction using carbon dioxide and cosolvents. The solvents and cosolvents tested were ethanol, isopropyl alcohol, and their mixture in equal proportions. The composition of the extracts was determined by gas chromatography−flame ionization detection (GC-FID) and UV. The largest yield (27%, weight) was obtained in the Soxhlet extraction (turmeric (S), ethanol = 1:100); the lowest yield was detected in the hydrodistillation process (2.1%). For the supercritical extraction, the best cosolvent was a mixture of ethanol and isopropyl alcohol. Sixty percent of the light fraction of the extracts consisted of ar-turmerone, (Z)-γ-atlantone, and (E)-γ-atlantone, except for the Soxhlet extracts (1:100, ethanol), for which only ar-turmeronol and (Z)-α-atlantone were detected. The maximum amount of curcuminoids (8.43%) was obtained using Soxhlet extraction (ethanol/isopropyl alcohol). The Soxhlet and low pressure extract exhibited the strongest antioxidant activities. 34
Inducing Lipid Peroxidation in Brain of Mice with Sodium Nitroprusside and Iron
Graph 3 presents the effect of anti-oxidants from C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum in the brains of mice. This time, the TBARS with 10 M iron was induced. Outcomes showed a significant (P < .05) increase in TBARS as compared to after 10 M iron basal treatment. Five separate controls were used for C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum. Treatment with different concentrations of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum significantly reduced lipid peroxidation. These outcomes revealed that these extracts have about the same effects on reducing lipid peroxidation in mice's brains as iron compared to sodium nitroprusside. L inermis and S aromaticum showed a high percentage reduction in lipid peroxidation compared with the other three plant extracts. The effect of aqueous extracts of Curcuma longa, Lawsonia inermis, Picrorhizha kurroa, Rheum emodi, and Syzygium aromaticum against lipid peroxidation in mice brain induced by iron.
Graph 4 shows the antioxidant effects of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum in mice's brains. Outcomes prove an increase with a significant (P < .05) level of thiobarbituric acid reactive substances (TBARS) compared to after basal treatment with 5 M sodium nitroprusside. Five separate controls were used for C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum. On the other hand, extract treatments with various concentrations (50-200 g/mL) lead to a significant reduction in lipid peroxidation. L inermis and S aromaticum showed a higher level of reduction in lipid peroxidation than other plant species. Picrorizha kurroaproved an effect of pro-oxidant at 100 g/mL above that further proved the effect of antioxidant by decreasing lipid peroxidation. All the other plant extracts showed a gradual decrease in lipid peroxidation with increasing concentrations (50-200 g/mL). The effect of aqueous extracts of Curcuma longa, Lawsonia inermis, Picrorhizha kurroa, Rheum emodi, and Syzygium aromaticum against lipid peroxidation in mice brain induced by sodium nitroprusside.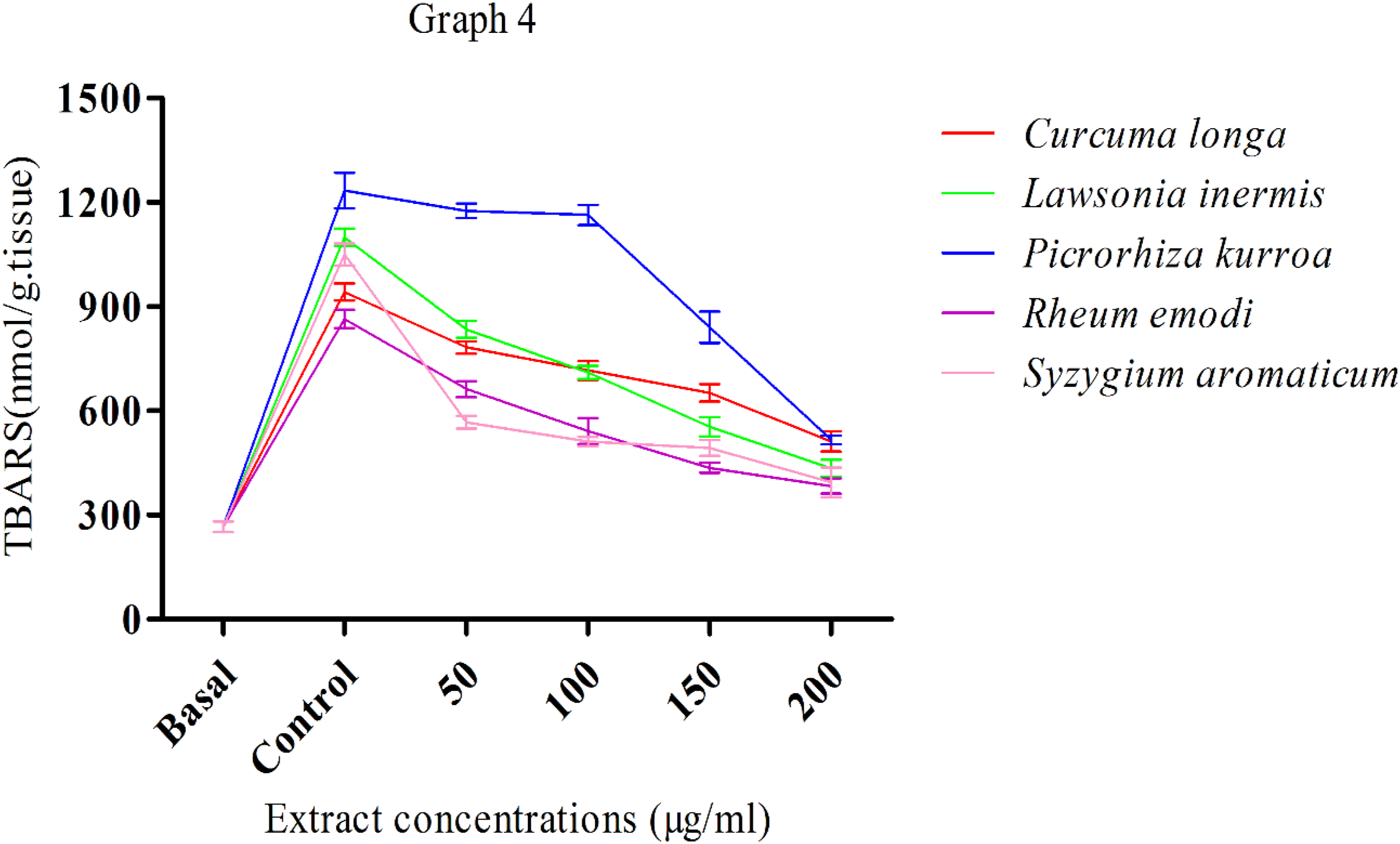
Activity of DPPH Radical Scavenging
The DPPH radical scavenging activity of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum has been shown in Graph 5. All the aqueous extracts of these plants have shown scavenging DPPH radical ability of more than 50%, which is evident from their high antioxidant activities. On the other hand, the order of their antioxidant activity is L inermis > S aromaticum > R emodi > Picrorizha kurroa > C longa. DPPH radical scavenging activity (antioxidant activity) of Curcuma longa, Lawsonia inermis, Picrorhizha kurroa, Rheum emodi, and Syzygium aromaticum. Values are Mean ± SD (n = 3).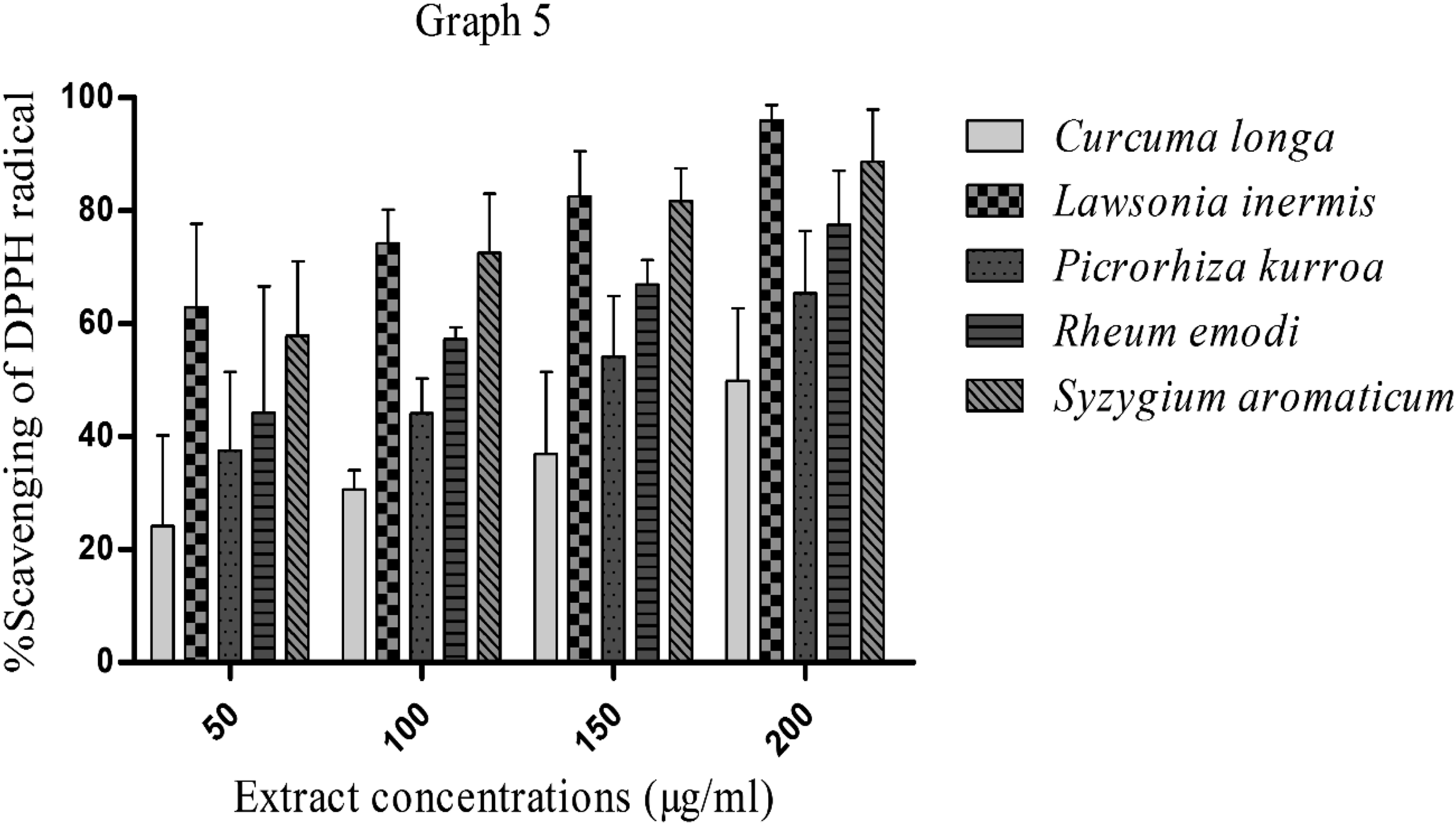
Total Activity of Antioxidant by Phosphomolybdenum Assay
The Graph 6 presents the total antioxidant activity of different extracts of plants explained as equal to ascorbic acid equivalent. All extracts of aqueous of these medicinal plants proved reduce in activity. However, the order of their reactivity was L inermis > S aromaticum > R emodi > Picrorizha kurroa > C longa. Total antioxidant activity measured by phosphomolybdenum reduction method. Values are Mean ± SD (n = 3).
According to previous study, the anti-oxidant activity of extracts from 36 vegetables was evaluated by using a model system consisting of β-carotene and linoleic acid. The total phenolics of the extracts was determined spectrophotometrically according to the Folin–Ciocalteau procedure and ranged from 34 to 400 mg (100 g)−1 on a fresh weight basis. Mint, aonla, black carrots, chenopodium, fenugreek, kachnar and ginger had high phenolic contents. The anti-oxidant activity expressed as per percent inhibition of oxidation ranged from a high of 92% in turmeric extracts to a low of 12.8% in long melon. Other vegetables found to have high anti-oxidant activity (>70%) were kachnar, aonla, ginger, fenugreek, mint, beetroot, black carrots, Brussels sprouts, broccoli, lotus stem, yam, coriander, and tomato. Anti-oxidant activity correlated significantly and positively with total phenolics (r2 = .6578, P<.05). The results indicate that vegetables containing high phenolics may provide a source of dietary anti-oxidants. 35
Total Phenolics Content
Total Phenolic Content Among Aqueous Extracts of Curcuma longa, Lawsonia inermis, Picrorhizha Kurroa, Rheum emodi, and Syzygium aromaticum. The results are Expressed as Means ± SD.

According to previous study, proximate analysis and total phenolic contents of ethanol, methanol and water extracts of turmeric were also carried out. The total phenolic content (TPC) means related to three solvents at their different concentrations, that is, Ethanol (60%), Ethanol (80%), methanol (60%), methanol (80%), and aqueous extract were 678.76 mg GAE/100 g, 745.76 mg GAE/100 g, 523.87 mg GAE/100 g, 682.43 mg GAE/100 g and 496.76 mg GAE/100 g, respectively. Free radical scavenging activities of three solvents of turmeric at their different concentrations, that is, Ethanol (60%), Ethanol (80%), methanol (60%), methanol (80%) and aqueous extract were 46.16%, 52.19%, 35.41%, 49.83% and 31.33% respectively. Results indicated that ethanolic extract showed better phenolic acids and DPPH scavenge activity than methanol and water extracts. Proximate composition showed that turmeric contained moisture (13.02%), crude protein (6.47%), crude fat (5.33%), crude fibre (4.80%), ash (3.49%), and NFE (69.89%). 36
Limitations
• This study focused only on the effect of Picrorhiza kurroa (kutki), S aromaticum (loung), L inermis (henna), R emodi (revand chini), C longa (haldi) against lipid per-oxidation in mice brain and liver. So, further research studies must be done on severe form of lipid per-oxidation in order to validate of P kurroa (kutki), S aromaticum (loung), L inermis (henna), R emodi (revand chini), C longa (haldi)’s effective role. • In the current study, the effect of P kurroa (kutki), S aromaticum (loung), L inermis (henna), R emodi (revand chini), and C longa (haldi) was measured lab tests to determine disease activity. • Rats are relatively inexpensive to purchase and maintain, and they are easy to handle and train. • Rats have a well-characterized genome and a similar set of organs and physiological processes as humans, such as a nervous system. Rats can also be bred and genetically modified to mimic human diseases and traits, such as cancer, diabetes, addiction, depression, and anxiety. • Rats are highly reproducible in terms of their behaviour, physiology, and genetics. This means that experiments using rats can be repeated with high accuracy and reliability, which is important for validating scientific findings • Rats have a faster metabolism than humans, which may lead to overestimation of drug efficacy or toxicity. This means that drugs that are effective in rats may not be as effective in humans, or may have different side effects. Researchers need to account for these differences when interpreting results from rat studies. • Rats have a shorter lifespan than humans, which may prevent the study of long-term effects of drugs or diseases. This means that some diseases or conditions that develop slowly in humans may not be accurately represented in rat models. • Limited ability to capture human.
Conclusion
The effects of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum were determined to induce lipid peroxidation with sodium nitroprusside and iron in brain homogenates and livers of mice. These plants were chosen as they are important ingredients in daily life, both in the kitchen and in many herbal formulations. Total phenolic content among aqueous extracts of C longa, L inermis, Picrorhizha kurroa, R emodi, and S aromaticum.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
