Abstract
Pharmaceutical wastes are environmental micro pollutant and potential risk for the ecosystem. Therefore, the present study was planned to find out the effects of different pharmaceutical effluent (PE) regimes on growth, secondary metabolism, and oxidative defense in 2 carrot lines. The seeds of 2 carrot lines (DC-3 and T-29) were spread in plastic pots containing sandy loam soil. The design of experiment was completely randomized with 3 replicates per treatment. At vegetative stage, plants were irrigated with 5 different doses (control), 25%, 50%, 75% and 100%) of PE on every 3-day interval, while control plants were irrigated with canal water. The carrot roots were harvested after 25 days’ application of the treatments to determine various attributes. High concentration of PE caused a substantial decline in growth, beta carotenoids, anthocyanin, total soluble protein, free amino acids, total soluble sugar, phenolic and flavonoid contents and an increase in proline, levels of H2O2 and MDA, activities of antioxidant enzymes such as peroxidase (POD), superoxide dismutase (SOD), and catalase (CAT) in both lines. Moreover, PE caused significant reduction in the levels of essential nutrients (K+, Ca2+) and increased in Na+ content. However, T-29 line was found to be more PE tolerant because it had less H2O2, MDA and ascorbic acid contents. Thus, our findings showed that diluted PE (25%) could not be used for irrigation to increase the growth of plants in nutrients deprived environments without using bio filtration and biocarbon sorption technologies for treatments.
Introduction
Pharmaceuticals are the compounds used as medicine or drug to cure or control diseases. 1,2 Each pharmaceutical product contains active ingredients that have many beneficial effects on the health of animals and human. After absorption of the drug in the body, it undergoes the metabolic reactions by the aid of enzymes or microorganisms and produces many substances. These substances are removed from the body through urine or faeces, 3 but un-metabolized fraction of drug also excreted from the body through urine or faeces in the form of active drug. There is no specific system for the removal of pharmaceuticals from water through wastewater treatment systems as a result, contamination of the environment occurs. 4
PE may hazardously effect on several species present in ecosystems that eventually leads to a potential risk to human health. 5 Most of the administered therapeutic compounds or active metabolites that can enter the ecosystem via sewage. 6 PE are commonly discharged into the soil and water that could change the quality of water and affect the aquatic plants and animals. 7 In developing countries, pharmaceutical and other industries discharge their effluents without treatment into soil and water. These effluents can directly contaminate the surface and ground water. 8 Besides water, pharmaceutical compounds, including their metabolites are known to be found in the environmental systems such as water, wastewater, sediments and sludge. 9 PE when discharged into the nearby areas cause a serious threat to the semi-urban and rural populations. 10
Pharmaceutical substances like antibiotics, antiseptics, analgesics, anti-inflammatory drugs, beta-blockers, antiepileptic, lipid regulator agents, antihypertensive, hormones, contraceptives, antivirals, cancer therapeutics, cosmetic ingredients and other hormones and personal care products like disinfectants and antiseptics are recognized as common environmental micro-pollutants. 11,12 The most commonly used drugs against muscular pain and inflammatory disorders is IBP. IBP is a determined pollutant because of not destroyed in a municipal water treating station. 13 IBP in sewage effluent has pharmacological impacts on aquatic fauna, especially in rainbow trout. 14 PE contained organic compounds are toxic and carcinogenic. 15 Pharmaceutical compounds absorbed through the roots, accumulate in plants parts results inhibition of the seed germination, and ultimately yield in some crops such as rice, sweet pea and cucumber 16 as well as root vegetables like carrots. 17
Having of COD, BOD, TSS, large amount of chemical elements, pharmaceutical wastes is one of the abiotic stresses that upsets the whole life of plants and human. 18 Pharmaceutical compounds like antibiotics reduced the plant growth and performance of plants, soil respiration and enzymatic activities, 19 uptake of macro and micro-nutrients in plant tissues and cause ROS generation. 20
The inhibitory effects of pharmaceutical drugs and active metabolites are partly due to oxidative damage caused by too much accumulation of ROS. 21 ROS are highly reactive in nature altering normal cellular metabolism by inducing substantial damage to proteins, lipids, pigments and nucleic acid. 22 The accumulation of ROS in chloroplast is minimized by well-organized antioxidative system composed of enzymatic antioxidants as well as non-enzymatic antioxidant (phenolic and flavonoid compounds) to overcome stress injury. 11,23,24
Carrot is a biennial herbaceous species, is a member of the Apiaceae family. Carrot is widely used as a root vegetable in the globe and vast cultivated in the temperate regions.
25
The carrot has rich source of nutrients, vitamin A and C, Beta-carotene, mineral nutrients (K
Materials and Methods
Plant Material and Growth Conditions
Seeds of DC-3 and T-29 lines were obtained from vegetable section, AARI, Faisalabad, Pakistan. The study was conducted at the Department of Botany, Government College University, Faisalabad. The seeds were sown at a depth of 0.5 cm in plastic pots (15 cm diameter and 20 cm height) filled with sandy loamy soil (pH = 7.1) containing very good nutrients. The PE was collected in plastic containers from disposal point of the Saffron Pharmaceuticals (Pvt.) Limited located in Gatwala, Sheikhupura Road, Faisalabad. After germination, 5 seedlings were maintained in each pot. The physico-chemical properties of the PE were determined. At vegetative stage, plants were irrigated with 5 different doses ((control-0%), 25%, 50%, 75% and 100%) of PE on every 3-day interval. The control plants were irrigated with canal water. The design of experiment was completely randomized with 3 replications. Carrot roots were harvested after 25 days application of the treatments. Following data for different attributes were measured at the vegetative stage.
Physico-Chemical Properties of Pharmaceutical Effluents (PE)
The physico-chemical properties of the PE such as temperature, color, odor, pH, BOD, COD, TSS, TDS, total Cl-, total alkalinity, total SO4 -2, PO4 3-, total NO3, Ca2+, Cu, Fe, Ni, Zn, Mn, Ba, Cd and Pb were measured using the standard methods 28 and are shown in Table 1.
Mean Values of Physico-Chemical Parameters of PE.

Growth Attributes
After harvest, root length, root width was measured in centimeters. Fresh and dry weights of roots were recorded in grams with the help of weighing balance.
Total Soluble Proteins Assays
The fresh root material (0.15 g) of carrot homogenized with 10 mL of chilled 50 mM potassium phosphate buffer (pH 7.5). Centrifuge material at 4°C at 10,000 rpm for 15 min. The supernatant was used for the determination of total soluble proteins according to the Bradford, 29 method. The absorbance was determined after 2 min at 595 nm.
Total Free Amino Acids Contents
Total free amino acids were measured by the ninhydrin assay. 30 Fresh root (0.15 g) were homogenized in the potassium buffer solution. The homogeneous material (1 mL) was shifted to test tubes (25 mL), then mixed 10% ninhydrine solution (0.5 mL) and 2% pyridine solution (0.5 mL). After this material was boiled for 30 min at 100ºC. Then, distilled water was added in each test tube made the volume 25 milliliter. The optical density was measured at 570 nm with a spectrophotometer.
Proline Content
Fresh carrot root (0.15 g) was grinded in 10 mL aqueous sulfo-salicylic acid (3%) and filter the mixture by filter paper. Root extract (2 mL) was mixed with 2 mL acid ninhydrin and glacial acetic acid and boiled for 1 hour at 100°C and then terminated the reaction by bathing the tubes with ice. Then toluene (4 mL) was added and shaken for 10-20 seconds. Then chromophore which contained toluene was extracted. Then OD was measured at 520 nm and toluene was used as a blank. 31
Total Phenolic Contents
Fresh carrot root (0.15 g) were grinded in 80% acetone (5 mL), then centrifuged the material for 10 min at 10,000 rpm. The aspirated material (100 µL) was added in distilled H2O2 (2 mL) 20% sodium carbonate (5 mL) and Folin-Phenol Ciocalteus reagent (1 mL). Then added the distilled water to make the volume up to 10 mL. In carrot root, total phenolics measured by using the Folin-Phenol Ciocalteus reagent with the help of spectrophotometer at 750 nm. 32
Malondialdehyde (MDA) Contents
Malondialdehyde contents were measured according to the method of Dhindsa et al. 33 Fresh carrot root (0.15 g) were grinded in 5% TCA (1.5 mL). Then centrifuged the material at 12,000×g for 15 min. The supernatant material (1 mL) was mixed with 1 mL TBA (0.5%) in 20% TCA (w/v). The reaction mixture was heated at 95 oC for 30 min, then cooled it and for clarification of material centrifuged at 7500×g for 5 min. The OD was measured with the help of a spectrophotometer at 600 nm and 532 nm.
Hydrogen Peroxide (H2O2) Contents
Hydrogen peroxide contents in fresh root of carrot were measured according to Velikova et al. 34 Fresh carrot (0.15 g) root were grinded with 2 mL of 0.1% TCA (w/v) by using the mortar and pestle on the ice. The material was centrifuged at 12,000×g for 15 min. Supernatant (0.5 mL) was mixed with 0.5 mL potassium phosphate buffer (pH 7.0; 10 mM) and 1 mL of potassium iodide (1 M). Then the mixture was shaken for a few minutes. After that OD was recorded at 390 nm.
Ascorbic Acid (ASA) Contents
Ascorbic acid content was determined by the method of Mukherjee and Choudhuri. 35 Fresh root material of carrot (0.15 g) was grinded in 6% trichloroacetic acid solution (10 mL). Two mL of dinitrophenyl hydrazine (2%) was added in test tube contained 4 mL extract and, then added 1 drop of thiourea (70% ethanol). The reaction mixture was heated for 15 minutes at 100°C and chilled in an ice bath. After that 5 mL of sulfuric acid 80% (v/v) was added to the reaction mixture. The OD of the reaction mixture was measured at 530 nm by using a spectrophotometer.
Total Anthocyanin Contents
Total anthocyanin content was recorded with the method of Hodges and Nozzolillo. 36 Fresh carrot root (0.15 g) were grinded in 2 mL of acidified methanol (methanol+1% HCI) by using the mortar and pestle. The grinded material was moved to the test tube and boiled at 100°C in the water bath for 30 min. Then the material was centrifuged for 15 min at 12000×g. The OD was measured with the help of the spectrophotometer at 536 nm and 600 nm.
Total Soluble Sugars Contents
Total soluble sugar contents were estimated by the method of Yemm and Willis. 37 For total soluble sugars, 0.15 g fresh root material was homogenized in 5 mL of ethanol (96%) and then washed with 5 mL ethanol (60%). The homogenate was centrifuged for 10 min at 3500 × g. The supernatant (0.1 mL) stored at 4°C was added to freshly prepared anthrone. The mixture was cooled and used for the estimation of total soluble sugars and determine its absorbance at 625 nm by using a spectrophotometer (Hitachi U–1800).
Beta-Carotenoids Contents
Carotenoid extraction was performed according to the procedure of Rodriguez et al. 38 Carrot root samples (5 g) were grinded in 80% chilled acetone and then filtered by using the Buchner funnel. This method was repeated until residue becomes colorless and then pigments were moved to test tube contained ether, then washed with distilled water for complete removal of acetone from the pigments. After extraction, total carotenoid content was extracted and absorbance of carotenoid was determined at 449 nm by using the spectrophotometer. A standard curve relating the total carotenoid content (beta-carotene). Beta-carotene were measured using its absorption coefficient of 2592. 39
Antioxidant Enzymes Activities
The fresh carrot roots were grinded in 50 mM phosphate buffer with pH (7.0) and dithiothreitol (1 mM). Then centrifuged for 15 min at 15,000 rpm at 4 oC and saved it at cooled place for measuring the activities of superoxide dismutase (SOD), catalase (CAT) and peroxidase (POD) enzymes. 40 Superoxide dismutase (SOD) activity of fresh carrot root was estimated with the standard procedure proposed by the help of Giannopolitis and Ries 41 was followed. Briefly, the enzyme extract (20 mL) was mixed with a reaction mixture containing phosphate buffer (50 µM pH 7.8), NBT (50 µM), riboflavin (1.3 µM), EDTA (75 µM), and methionine (75 µM). A light source was used to irradiate the reaction mixture in the test tube and then absorbance was measured at 560 nm using a spectrophotometer (Hitachi U–1800). Accordingly, 1 Unit SOD activity was equal to the amount of enzyme sufficient to inhibit 50% NBT production. Chance & Maehly method 42 was followed for the estimation of CAT and POD activities. Accordingly, the CAT solution mixture (3 mL) was composed of enzyme extract (0.1 mL), phosphate buffer (50 mM, pH 7.0), and H2O2 (5.9 mM). Similarly, POD solution mixture (3 mL) contained phosphate buffer (50 mM, pH 5.0), enzyme extract (0.1 mL), H2O2 (10 mM) and guaiacol (20 mM). Both the solution mixtures (CAT and POD) were irradiated separately and absorbance after a 20 second interval was measured at 240 and 470 nm, respectively using a spectrophotometer (Hitachi U–1800). Per-minute change in absorbance of 0.01 Units was equal to 1 Unit of each CAT and POD activities.
Estimation of Mineral Nutrients From Carrot Root
Root sample (500 mg) of each carrot line were digested with HNO3 and HCLO3 (3:1) at hot plate for 2-3 hours. Digested material was used to measure the nutrients with the procedure of Yoshida et al. 43 Ca2+, K+ and Na+ contents in the root of carrot were measured using atomic absorption spectrophotometer (Hitachi, Z-2000, Tokyo, Japan).
Statistical Analysis
All variables studied in this experiment were analyzed by using Statistics 8.1 software. Analyzed data is the mean value of 3 replications per treatment and assayed in Two-way ANOVA at P < 0.05 at LSD.
Results
Physico-Chemical Properties of PE
Physico-chemical properties of PE were recorded to be above the tolerable level according to WHO Standards (Table 1). The analysis of PE exhibited that it is basic in nature with whitish in color. It contained a great quantity of TDS and TSS. The PE had higher value of BOD, COD, and DO, considerable amounts of total chloride, calcium, nitrate, sulphate, iron and manganese.
Growth Attributes
The PE (25%) cause a significant improvement (P ≤ 0. 001) in root lengths, root, fresh and dry weights and root width in DC-3 and T-29. The effect of PE on different growth attributes was dose dependent. Maximum decline was observed in 100% of PE in both carrot lines. The maximal decrease in these variables was recorded in the plants of DC-3 as compared to T-29 (Figure 1; Table 2). While higher concentrations of PE relatively reduced total soluble protein and free amino acid contents.

Effect of PE on growth attributes (root length, root fresh weight, root dry weight and root width) in 2 carrot lines (n = 3; means ± S.D).
Mean Square Values From ANOVA of Data for Growth, Biochemical Attributes, Oxidative Defense System, and Nutrient Contents in Carrot (Daucus carota L.) Grown Under Different PE Regimes.

Abbreviations: df = Degree of freedom; RL = Root length; RFW = Root fresh weight; RDW = Root dry weight; TSP = Total soluble protein; TFAA = Total free amino acids; TSS = Total soluble sugar; H2O2 = Hydrogen peroxide; MDA = Malondialdehyde; SOD = Superoxide dismutase; POD = Guaiacol peroxidase; CAT = catalase; Ca2+ = Calcium; K+ = Potassium; Na+ = Sodium.
***, **, *, significant at 0.001, 0.01 and 0.05 levels, respectively, ns = non-significant.
Total Soluble Protein, Total Free Amino Acids, Proline Contents and Phenolic Contents
Exposure of carrot plants to PE in growth medium significantly (P ≤ 0.001) decreased total soluble protein and total free amino acid contents. A substantial increase and decrease (P ≤ 0.001) in proline contents and total phenolic contents were observed in both lines under PE stress, respectively. However, PE induced more accumulation of total free proline contents in T-29 carrot line as compared with line DC-3 (Figure 2; Table 2). The increase in free proline contents was exhibited in plants of both lines under 75% and 100% PE stress (Figure 2; Table 2).

Effect of PE on biochemical attributes (total soluble protein, total free amino acids, free proline and total phenolic) in 2 carrot lines (n = 3; means ± S.D).
Total Anthocyanin and Total Soluble Sugar Contents
A significant increase (P ≤ 0.001) in root total anthocyanin and total soluble sugar contents were noted in both carrot lines under PE stress. The response of 2 carrot lines was not the same with respect to this attribute. However, maximum decline was recorded at 75% and 100% of PE in both carrot lines (Figure 3; Table 2). A marked increase (P ≤ 0.001) in β-carotene was noticed in plants when different concentrations (0%, 25%, 50%, and 75%) of PE were added to the growth medium. Of different doses of PE, 100% revealed a significant decreased in this attribute. However, the improvement in this attribute was greater in T-29 than that in DC-3 (Figure 3; Table 2).

Effect of PE on biochemical attributes (anthocyanin, total soluble sugar and Beta carotenoids) in 2 carrot lines (n = 3; means ± S.D).
Oxidative Damage and Ascorbic Acid Contents
The damage to membrane lipid bilayer measured in the form of MDA contents and H2O2 was markedly (P ≤ 0.001) enhanced in the carrot plants grown under PE stress. The response of the lines was dose dependent, being the highest at 100% level of PE with respect to this MDA and H2O2. DC-3 line accumulated higher MDA and H2O2 as compared with T-29 under all levels of PE (Figure 4; Table 2). Imposition of PE stress significantly (P ≤ 0.001) affected ASA contents in plants. The pattern of decrease or increase in ASA level was variable in both carrot lines. While increase in this variable was more evident in DC-3 than T-29 under PE stress (Figure 4; Table 2).

Effect of PE on biochemical attributes (H2O2, MDA and ascorbic acid contents) in 2 carrot lines (n = 3; means ± S.D).
Antioxidant Enzymes Activities
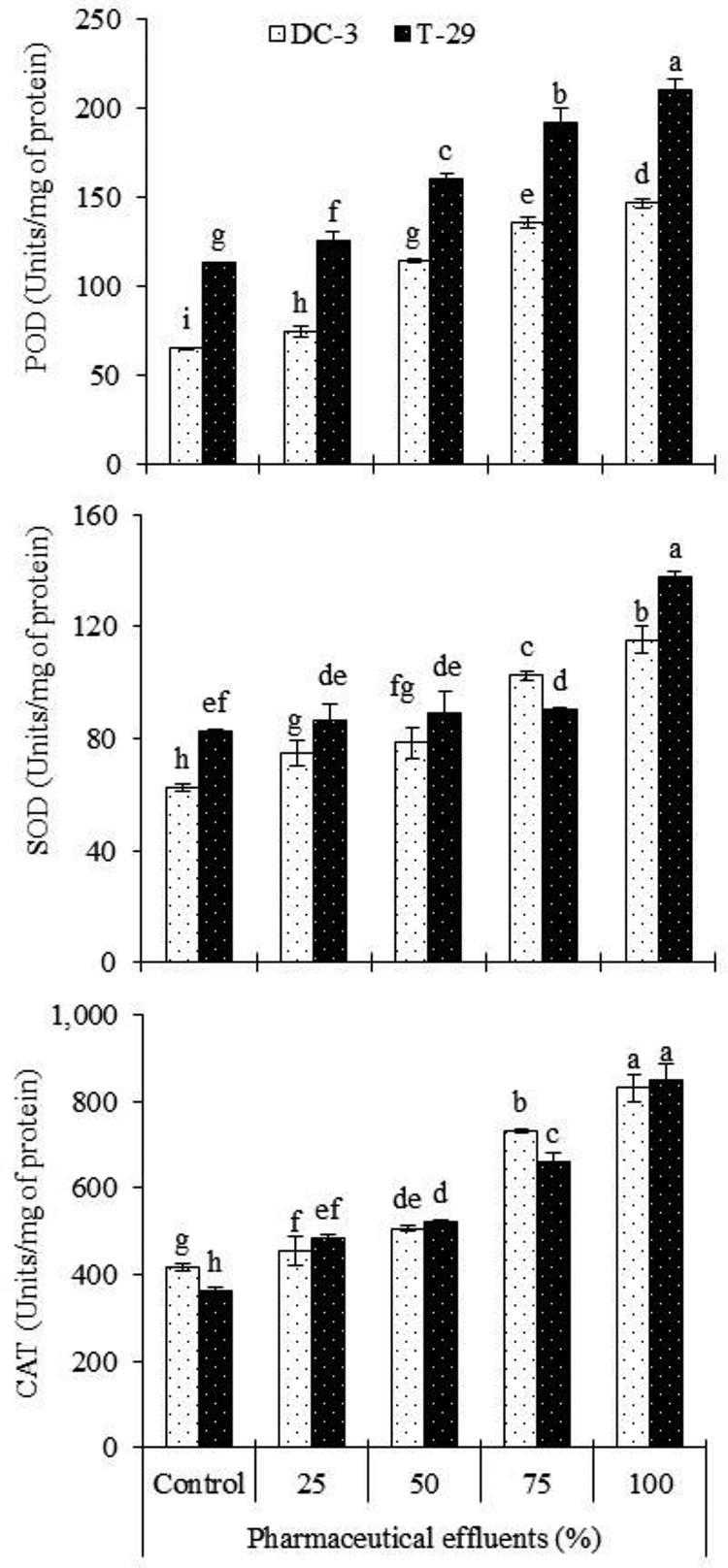
Activity of antioxidant enzymes, viz, POD, SOD, and CAT increased in both carrot lines under different levels of PE stress. Addition of PE to the growth medium caused a consistent increase (P ≤ 0. 001) in the activities of POD and SOD. Higher activities of POD and SOD were documented in T-29 when compared with the DC-3 line (Figure 5; Table 2). PE-induced consistent rise (P ≤ 0.001) in the activity of CAT was noticed in both lines. However, the increase in the activity of CAT was more in T-29 than that in DC-3. The PE-induced increase in this attribute was dose dependent, being maximal at 100% level of PE (Figure 6; Table 2).
Effect of PE on changes in nutrient contents (Ca2+, K+ and Na+) in 2 carrot lines (n = 3; means ± S.D).

Effect of PE on enzymatic antioxidant attributes (POD, SOD and CAT) in 2 carrot lines (n = 3; means ± S.D).
Mineral Composition
Pharmaceutical effluents resulted in a mark (P ≤ 0.01) decreased in Ca2+ contents in the root of carrot plants. Plants grown in a PE growth medium having different levels (0%, 25%, 50%, 75% and 100%) showed a significant reduction in Ca2+ contents. The root Ca2+ contents were significantly lower in DC-3 under 75% and 100% PE stress. The maximal values of root Ca2+ content was apparent in the control and 25% PE plants (Figure 5; Table 2). K+1 contents decreased (P ≤ 0.05) in the root of carrot plants when subjected to PE (50%, 75% and 100%) in growth medium. This reduction was more visible in plants treated with 75% and 100% PE. The other treatment of PE (50%) did not impose a significant impact on this attribute. However, the improvement in this attribute was greater at 25% PE in T-29 than that in DC-3 (Figure 5; Table 2). Na+1 contents increased conspicuously (P ≤ 0.001) in root of carrot plants subjected to PE in growth medium. This increase was more noticeable when 75% and 100% PE were applied in the growth media. The imposition of PE stress caused a substantial increase in root Na+1 in both carrot lines (Figure 6; Table 2).
Discussion
The PE plays a key role in the contamination of environmental systems because its effluent contained a large concentration of chemical elements, TSS, TDS, BOD, COD, DO, Na+, Ca2+, Cl-, PO4 3- and NO3. Furthermore, any considerable amount of metals such as Mn, Zn, Fe, Pb, and Cd are also present in the PE (Table 1). The physico-chemical properties of PE are in agreement with the earlier results of James et al. 44 and Gadipelly et al. 45
Pharmaceutical effluent is known to be one of the abiotic stresses that limit seed germination and yield. 15,16 Nevertheless, in the present study, root length and root fresh and dry masses of both carrot lines increased at lower and reduced at higher level of the PE. The lower level of PE contained optimal levels of nutrients that might have enhanced the growth parameters of plants. 44 However, toxic effects of PE on various growth attributes were more evident in T-29 than in DC-3 line. PE-induced reduction in various growth parameters has been described in a number of crop plants, e.g. rice, sweet pea and cucumber Tanoue et al. 16 and carrots. 17 There are few possible reports for this, wastewater caused harmful effects on the soil property and plant growth, including unbalanced the water relations due to Mg2+, K+1 and Ca2+ resulting in the wilting of plant seedlings. 46 In our study, excess amount of total alkalinity, Cl-, NO3, Mg2+, Ca2+, SO4 2- and PO4 3- in the PE the growth of carrot lines was extremely influenced the growth of carrot line. Lower concentration of PE increased Beta carotenoid contents in carrot plants. This might be due to promising effects of organic elements nitrogen, and NO3 - present in the effluent. 47 Additionally, effluent contain Zn2+, Cu2+, Fe2+, Mg2+ and K+1, that are essential for synthesis of carotenoid pigments. 48 In the present study, PE stress resulted in a marked increase of Beta-carotenoid contents in both lines. However, PE-induced synthesis of this attribute was more evident in T-29 than DC-3.
Plants under effluent show oxidative damage reflected in the form of metal-induced reduction in antioxidant potential alongside a substantial increase in the cellular levels of H2O2 and MDA. 49 In our investigation, PE stress triggered a significant increase in MDA levels in both carrot lines. The effluent induced increase in MDA contents has been described in cucumber, 16 Typha, 50 and soybean. 51 H2O2 is a ROS as it prevents the C-3 cycle and therefore it decreases the carbon fixation and are lethal for various metabolic reactions in cells. In the present investigation, both carrot lines accumulated higher levels of H2O2 under PE stress. It has been reported that ROS causes a significant damage to nucleic acid, pigments, proteins, and lipids. 11,52 Phenolic, flavonoids, and ascorbic acids are important non-enzymatic antioxidant compounds that significantly mediate the stress injury. 24,53 Our results are in agreement with Islam et al. 54 and Malik et al. 24
Plants have an antioxidant defense system to combat with metal-induced excess ROS production 55 and the combined action of SOD, POX and AsA-GSH cycle is essential for scavenging of ROS levels in chloroplasts. 56 In the present investigation, it is observed that an increase in SOD, POD and CAT activities was noted in carrot lines at higher effluent level. Our results correlate with the results of Baskaran et al. 57
Anthocyanins worked as a defensive yardstick against effluent induced stress in plants. 58 In the present study, a substantial reduction in anthocyanin was found in both carrot lines at 75% and 100% levels of effluent. Flavonoids act as an antioxidant by donating hydrogen atoms and thereby significantly enhance plant metal tolerance. Higher flavonoids are positively associated with plant stress tolerance. 54 Our results indicated effluent induced an increase in flavonoids in rice. 59 Plants subjected to effluent enhanced proline accumulation. The proline accumulation in plants used as a bio-chemical marker for tolerance of stress. 60 Furthermore, proline as chelator scavenging the H2O2 and 1O2 has been reported. Therefore, providing a shield against ROS-induced cell injury. 61 Free amino acids, which act as signaling molecules, regulation of ion transport, and radical scavenging. 62 Plants subjected to effluent resulted in the reduction of amino acid contents. Decline of total free amino acid and total soluble protein contents in the cell has been noticed due to acidic pH and higher values of Mg2+ in the effluent. 63 The decline in free amino acid contents can be attributed at higher levels of effluent because of protease activity inhibition. 64 In plants, phenolics not only function as the metal chelator, but also enhance the removal of the ROS. 65 Our results are in agreement with Tripathi et al. 66 and reported that metal-induced decrease in total phenolic in rice crop.
Conclusions
It is inferred from the results that physico-chemical properties of the pharmaceutical effluent (PE) such as chemical oxygen demand (COD), electrical conductivity (EC), pH, calcium (Ca2+) chloride (Cl-), sulphates (SO4 -2) and total dissolved solids (TDS) were comparatively raised in the PE and upset the plant growth rigorously. However, the effluent enhanced the plant growth at lower levels of effluent. The effluent reduced the Beta-carotenoids pigment in both carrot lines at a higher level of PE (100%). The results exhibited that the PE enhanced the values of malondialdehyde (MDA), hydrogen peroxide (H2O2), ascorbic acid (ASA) contents, and increased the activities of peroxidase (POD), superoxide dismutase (SOD), and catalase (CAT) enzymes. Moreover, PE also enhanced the free proline in carrot root. At lower concentrations of PE (25%), minimal decreased in the growth attributes, amino acid, total soluble proteins, and phenolic contents were recorded in both carrot lines. Generally, the line T-29 was more tolerant than DC-3. These results suggest that carrot is armed with an efficient antioxidant mechanism against PE-induced oxidative stress, which defends the plant’s photosynthetic pigments from injury. This suggests that the diluted PE (25%) may be used for irrigation purpose carefully after using efficient technologies (bio filtration and biocarbon sorption) for treatments. This study contributes to enhance the knowledge about how PE can exert physical or chemical toxicity on the plant depending upon its chemical composition, amount and species?
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
