Abstract
Objective
This study aimed to evaluate the efficacy of Corymbia citriodora, Melaleuca alternifolia (Myrtaceae), Mentha × piperita (Lamiaceae), and Schinus terebinthifolius (Anacardiaceae) essential oils as an alternative to manage Sitophilus zeamais (Coleoptera: Curculionidae) adults.
Methods
Acute contact toxicity, acute toxicity on treated maize grain, fumigation toxicity, repellency bioassays, and GC-MS analysis of the essential oils were carried out.
Results
Corymbia citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius oils were toxic at different levels to S. zeamais through residual contact, ingestion and via fumigation, and were also repellent to adults of this pest. Melaleuca alternifolia oil was the most active in contact (LC50 = 18.98 μL.mL−1), ingestion (LC50 = 1.03 μL.g−1), and fumigant (LC50 = 20.05 μL.L−1 air) bioassays. Citronelal (53.6% in C. citriodora), terpinen-4-ol (46.9% in M. alternifolia), menthol (44.8% in M. × piperita), and β-caryophyllene (16.2% in S terebinthifolius) are the major constituents of these oils.
Conclusions
Melaleuca alternifolia and M. × piperita essential oils can be used by residual contact, while those of C. citriodora, M. alternifolia, and M. × piperita by mixing with maize grains. Melaleuca alternifolia essential oil can be used as a fumigant, while those of C. citriodora and S. terebinthifolius as repellents for S. zeamais adults.
Introduction
The maize weevil, Sitophilus zeamais (Motschulsky), 1855 (Coleoptera: Curculionidae) is one of the main pests of maize, Zea mays L., rice, Oryza sativa L., sorghum, Sorghum bicolor (L.) Moench, and wheat, Triticum aestivum L. (Poaceae) and on stored cereal products such as pasta, cassava, Manihot esculenta Crantz (Euphorbiaceae), and milled grains.1-3 This insect is a major pest of maize grains and seeds in storage facilities in Brazil, Egypt, and the United States of America.4,5
Sitophilus zeamais adult body size ranges from 2.3 to 4.9 mm 6 depending on the food type of its larvae. 7 Sitophilus zeamais is similar to the rice weevil, Sitophilus oryzae (L., 1763), but with widely numerous and usually larger marked spots on the wing covers, 8 and its adults can fly which helps in the exploration of environments. 9 The female of this insect chews a hole and deposits 1 egg per grain covering it after removing its ovipositor with a waxy secretion forming a plug. This plug fast stiffens, and leaves a small elevated area on the seed surface. The hatched larva feeds and pupates inside the grain and the adult chews a circular exit hole to emerge.10,11 Each female may lay 300 to 400 eggs throughout its lifetime 12 for 5 to 8 months of its adult stage. 13
Integrated Pest Management (IPM) approaches to S. zeamais combine monitoring, prevention, and control methods.14,15 These methods include cultural control (hygiene, removal of infested residues, and aeration); host-plant resistance from phenolic acids in the grains, grain hardness, and ear coverage by straw; chemical control with synthetic insecticides including pyrethroids and botanicals with the first applied mainly by fumigation; and controlled atmospheres, irradiation, control of the environmental temperature, and biological control.16-18 Management measures such as gaseous synthetic insecticides and fumigants cause environmental pollution, impact non-target organisms, S. zeamais resistance, insecticide residue on grains, and worker fatality and can imperil human health.19,20
The properties of lemon-scented gum, Corymbia citriodora (Hook.) K.D. Hill and L.A.S. Johnson (Myrtaceae) essential oil include allelopathic (bioherbicide), anti-microbial, insect toxicity, and repellent effects, especially against S. zeamais and mosquitoes (Diptera: Culicidae) and perfumery.21-23 The tea tree, Melaleuca alternifolia (Maiden and Betche) Chee (Myrtaceae) essential oil when topically applied presents anti-microbial properties and has insecticidal action, especially against S. zeamais.24,25 The essential oil of peppermint, Mentha × piperita L. (Lamiaceae) is used for culinary, medicinal products, and agricultural, and domestic insecticides,26,27 while that of the Brazilian peppertree, Schinus terebinthifolius Raddi (Anacardiaceae) presents astringent, antibacterial, antiviral, diuretic, digestive stimulant, tonic, and wound healing properties. 28
The C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius essential oils were chosen for the present study because they are inexpensive, available in the market in several countries, and their toxicity and side-effect impacts are very low.29-31 Essential oils are an excellent alternative to conventional insecticides in IPM programs with low toxicity to non-target organisms determined by their structure and physico-chemical properties and short residual period in the environment.32,33 The objectives of this work were to investigate the chemical composition and the efficacy of the major components of C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius essential oils to S. zeamais adults for IPM of this pest.
Materials and Methods
Chemicals
Monoterpenes (α-pinene, β-pinene, α-terpinene, limonene, 1,8-cineole, terpinolene, linalool, citronellal, menthol, terpinen-4-ol, citronellol, and geraniol) and sesquiterpenes (α-copaene, β-caryophyllene, aromadendrene, germancrene D, bicyclogermacrene, and caryophyllene oxide) utilized for chemical component identification were obtained from Sigma-Aldrich (Jurubatuba, São Paulo State, Brazil; 97% purity).
Insects
Unsexed S. zeamais adults around 2 weeks old were obtained from a laboratory stock culture of the Laboratory of Agricultural Entomology, Department of Agronomy, Federal Rural University of Pernambuco (UFRPE) in Recife, Pernambuco State, Brazil. These insects were reared in 2 L glass jars and then put in an environmentally controlled room at 25 ± 2°C, 70 ± 10% RH and 12:12 (L:D) h photoperiod. Organic whole maize grains were the food media used.
Harvesting Plant Material
The fresh leaves of S. terebinthifolius were collected from 4 specimens located in a fragment of the Atlantic Forest in Recife in February 2020 (average location: 8°00' S × 34°57' W, 10 m above sea level). A single voucher was prepared from 4 specimens and deposited in the UFRPE Herbarium under number #49259. The essential oils of C. citriodora (batch: 119), M. alternifolia (batch: 213), and M. × piperita (batch: 204) were purchased on June 2021 from the Ferquima Indústria e Comércio Ltda. in Vargem Grande Paulista, São Paulo State.
Isolation of S. terebinthifolius Essential Oil
The S. terebinthifolius essential oil was isolated from the fresh leaves of each plant (100 g of leaves per plant specimen) using a Clevenger-type apparatus by hydrodistillation technique for 2 h. 34 Then, after separating the oil from water, an aliquot of anhydrous sodium sulfate (Sigma-Aldrich in Jurubatuba; 99% purity) was added into the oil to remove excess water, and after 15 minutes, the mixture was filtered to separate the oil from the water. The oil was put in hermetically sealed glass containers (1 L capacity) and stored at −5°C before the analysis and the bioassays. All experiments were done in triplicate.
Instrumental Analysis
The samples of oils were analyzed using gas chromatography with a flame ionization detector (GC-FID) on a PerkinElmer Clarus 500 GC equipped with a fused silica capillary column model DB-5 (30 m length × .25 mm inner diameter × .25 mm film thickness) (J&W Scientific, Folsom, CA, United States of America). The oven was programmed to heat from 60 to 240°C at a rate of 3°C min−1. The temperature of the injector and detector was 260°C. The carrier gas (H2) with 1 mL min−1 flow and 30 psi inlet pressure in split mode (1:30). The injection volume was 1 μL of diluted solution (1/100) of oil in n-hexane. The quantity of each compound was calculated from GC peak areas in a DB-5 column elution and expressed as a relative percentage of the total area of the chromatograms. Three replications were performed for each essential oil analyzed. The qualitative GC-MS analysis of the essential oils was carried out using a Varian 431 GC 220-MS system with a mass selective detector, mass spectrometer in EI 70 eV with a scanning interval of .5 seconds, and fragments from 40 to 550 Da fitted with the same column and temperature program as that for the GC experiments, with the following parameters: carrier gas = helium, flow rate = 1 mL min−1, split mode (1:30), and injected volume = 1 μL of diluted solution (1/100) of oil in n-hexane.
Components’ Identification
The identification of compounds was initially performed with data obtained by the analysis GC-MS retention indices with the retention time provided by injecting a series of C8-C40 n-alkanes calculated using the Van der Dool and Kratz equation 35 and by computer matching against the mass spectral library of the GC-MS data system (NIST 14) and co-injection with authentic standards as well as comparing them with default values provided by Adams. 36 Area percentages were obtained from the GC-FID response without the use of an internal standard or correction factors.
Acute Contact Toxicity Bioassay
The acute contact toxicity of essential oils and a deltamethrin-based insecticide was tested following described methods5,37 with slight modifications as follows. Deltamethrin is a pyrethroid ester insecticide registered by the Ministry of Agriculture, Livestock and Supply (MAPA) of Brazil to control S. zeamais in stored maize grains and seeds. 5 The C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius essential oils at 40 to 56, 16 to 24, 18 to 24, and 100 to 160 μL mL−1, respectively, and a deltamethrin-based insecticide (K-Obiol 25 CE as a positive control) at .8 to 4.8 μL mL−1 were used in this bioassay, following methods for dilution using a logarithmic series 20 or pure acetone as a negative control. The active ingredients of the K-Obiol 25 CE are deltamethrin @ 25 g L−1 (2.5% m v−1) and technical piperonyl butoxide (PBO) at 250 g L−1 (25% m v−1); inert components are diluents, solvents, and emulsifier stabilizers at 684 g L−1 (68.4% m v−1). Deltamethrin is currently classified as a highly hazardous pesticide by Forest Stewardship Council (FSC). A total of 1 mL was applied using a precision micropipette on the surface of a glass Petri dish (9 cm diameter; 63.6 cm2 inner surface area). Each dish was left out of direct sunlight for 15 minutes; then, 20 unsexed S. zeamais adults around 2 weeks old were put in each one. The concentration was repeated 4 times. The dishes were covered with a glass cover and placed in an environmental room at 25 ± 1°C, 70 ± 10% RH and 12:12 (L:D) h photoperiod. The weevil mortality (%) was evaluated 24 h after the bioassay started.
Acute Toxicity on Treated Maize Grain Bioassay
The acute toxicity of the essential oils and the deltamethrin-based insecticide (K-Obiol 25 CE) mixed with maize grains was evaluated by applying .5 mL of C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius oils and K-Obiol 25 CE at 1.5 to 3, .75 to 1.5, 1 to 2, 2 to 5, and .01 to .05 μL g−1, respectively, in a logarithmic series. 20 The same volume of pure acetone was used as the negative control in 12 grams of maize grains using a precision micropipette. A glass jar of .2 L capacity with each parcel was shaken for 10 seconds to blend uniformly the tested solutions or the acetone with the maize grains. The treated grains were kept out of direct sunlight for 15 minutes to evaporate the solvent. Twenty unsexed S. zeamais adults around 2 weeks old, separated 24 h before starting the bioassay, were left to feed on maize grains, treated or not, in an environmentally controlled chamber at 25 ± 1°C, 70 ± 10% RH and 12:12 (L:D) h photoperiod. The weevil mortality was counted after 24 h as previously reported.5,37
Fumigation Toxicity Bioassay
Glass jars of .2 L capacity with covers were utilized as exposure chambers for the acute fumigation toxicity bioassay of the essential oils 38 with slight modifications in an environmentally controlled chamber at 25 ± 1°C, 70 ± 10% RH and 12:12 (L:D) h photoperiod. Each essential oil was applied to a 2 cm diameter filter paper disk (Whatman Nº 1). The concentrations of C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius oils were 60 to 120, 17.5 to 27.5, 20 to 35, and 100 to 250 μL L−1 air, respectively, in a logarithmic series. 20 Each paper filter disk was air dried for 2 minutes in the chamber and put to the undersurface of the lid of the glass jars. Twenty insects were placed in each jar containing 5 g of whole maize grains. The jars were hermetically closed with their respective lids. Each solution and respective control were replicated 4 times. Weevil mortality was evaluated after 24 h.
Repellency Bioassay
The repellent effect followed the previously described method. 39 Petri dishes (9 cm diameter, 63.6 cm2 inner surface area) with the filter papers (Whatman N° 1, 9 cm diameter) inside the bottom of the dishes. Essential oil solutions at 10, 20, 30, and 40 μL mL−1 concentration were used. A total of 200 μL per concentration of each essential oil was applied uniformly on one half of the filter paper, and pure acetone as a negative control on the other half. The treated and control half-discs were left out of direct sunlight for 15 minutes for solvent evaporation. Twenty unsexed S. zeamais adults around 2 weeks old were released in the center of each dish. The treatments were repeated 4 times. The repellency bioassay was conducted in an environmentally controlled room at 25 ± 2°C, 70 ± 10% RH and 12:12 (L:D) h photoperiod. The number of weevils in the control (NC) and the treated (NT) dish halves was accounted after 2 and 4 hours.
Statistical Analysis
The LC50 was calculated by Probit analysis 40 using PROC PROBIT. 41 Data taken were corrected using Abbott’s formula 42 when necessary. The data of the repellent test were compared by the paired t test at 5% probability using SAS Institute software. 41 Percentage repellency (PR) was counted as follows: PR = [(Nc − Nt) ÷ (Nc + Nt)] × 100, Nc is the number of insects on the untreated area after the exposure interval, and Nt is the number of insects on the treated area after the exposure interval. PR was classified into the repellency classes of 0, I, II, III, IV, or V, where class 0 (PR ≤.1%), class I (PR = .1–20%), class II (PR = 20.1–40%), class III (PR = 40.1–60%), class IV (PR = 60.1–80%), and class V (PR = 80.1–100%).5,43
Results
Chemical Composition of the Essential Oils
Compounds (%) of Corymbia citriodora (Ccit) (Myrtaceae), Melaleuca alternifolia (Malt) (Myrtaceae), Mentha × piperita (Mpip) (Lamiaceae), and Schinus terebinthifolius (Ster) (Anacardiaceae) Essential Oils and Identification Methods (Iden. Met.).

aRI, retention indices calculated from the retention times in relation to those of a series C8-C40 of n-alkanes on a 30 m DB-5 capillary column.
bRI, retention indices from the literature; RI, retention indices; MS, mass spectroscopy and CI; co-injection with authentic compounds; SD, Standard Deviation; -, compound not detected.
Acute Contact Toxicity Bioassay
LC50 Calculated (Mean ± Data Variation) for Contact, Ingestion, and Fumigant Toxicities of Corymbia citriodora (Ccit) (Myrtaceae), Melaleuca alternifolia (Malt) (Myrtaceae), Mentha × piperita (Mpip) (Lamiaceae), and Schinus terebinthifolius (Ster) (Anacardiaceae) Essential Oils to Sitophilus zeamais (Coleoptera: Curculionidae) Adults After 24 h of Exposure.

aPositive control (a deltamethrin-based insecticide), N, total number of weevils tested; df, degrees of freedom; Slope, the slope of the toxicity line; SE ¼ standard error, Х2, chi-square; P value, probability; confidence interval, 95%.
Acute Toxicity on Treated Maize Grain Bioassay
The mortality rates of S. zeamais by the 4 essential oils differed. The Probit analysis, according to the lack of overlap in 95% confidence limits, demonstrated that S. zeamais is more susceptible to the C. citriodora (LC50 = 1.70 μL g−1), M. alternifolia (LC50 = 1.03 μL g−1), and M. × piperita (LC50 = 1.44 μL g−1) essential oils than to that of S. terebinthifolius (LC50 = 4.17 μL g−1) (Table 2).
Fumigation Toxicity Bioassay
The M. alternifolia essential oil was the most toxic in the fumigant toxicity bioassay. The LC50 (176.34 μL L−1 of air) of S. terebinthifolius essential oil was the highest with a low fumigant effect on the pest (Table 2), being about 8.8, 6.8, and 1.75 times higher than those of M. alternifolia (20.05 μL L−1 of air), M. × piperita (25.87 μL L−1 of air), and C. citriodora (100.64 μL L−1 of air), respectively.
Repellency Bioassay
The C. citriodora essential oil (P = .01, .001, .001, and .008) repelled S. zeamais adults after 2 h of exposure at all concentrations tested (Figure 1A-D) and, the C. citriodora [(P = .01) and (P = .03)] and S. terebinthifolius [(P = .03) and (P = .01)] oils repelled the S. zeamais individuals after 4 h, respectively, at 30 and 40% concentrations (Figure 2C and D). The M. × piperita essential oil did not repel S. zeamais 4 h after exposure at any of the concentrations tested (P = .62, .89, .07, and .94) (Figure 2A-D). Repellency (%) of Sitophilus zeamais (Coleoptera: Curculionidae) adults for a half filter paper treated or not with Corymbia citriodora (Myrtaceae), Melaleuca alternifolia (Myrtaceae), Mentha × piperita (Lamiaceae), and Schinus terebinthifolius (Anacardiaceae) essential oils with 200 mL of concentrations 10 μL mL−1 (A), 20 μL mL−1 (B), 30 μL mL−1 (C), and 40 μL mL−1 (D) after 2 h, in free choice test. *Significant values at 5% probability by t-paired test (P < .05). Repellency (%) of Sitophilus zeamais (Coleoptera: Curculionidae) adults for a half filter paper treated or not with essential oils from Corymbia citriodora (Myrtaceae), Melaleuca alternifolia (Myrtaceae), Mentha × piperita (Lamiaceae), and Schinus terebinthifolius (Anacardiaceae) with 200 mL of concentrations 10 μL mL−1 (A), 20 μL mL−1 (B), 30 μL mL−1 (C), and 40 μL mL−1 (D) after 4 h, in free choice test. *Significant values at 5% probability by t-paired test (P < .05).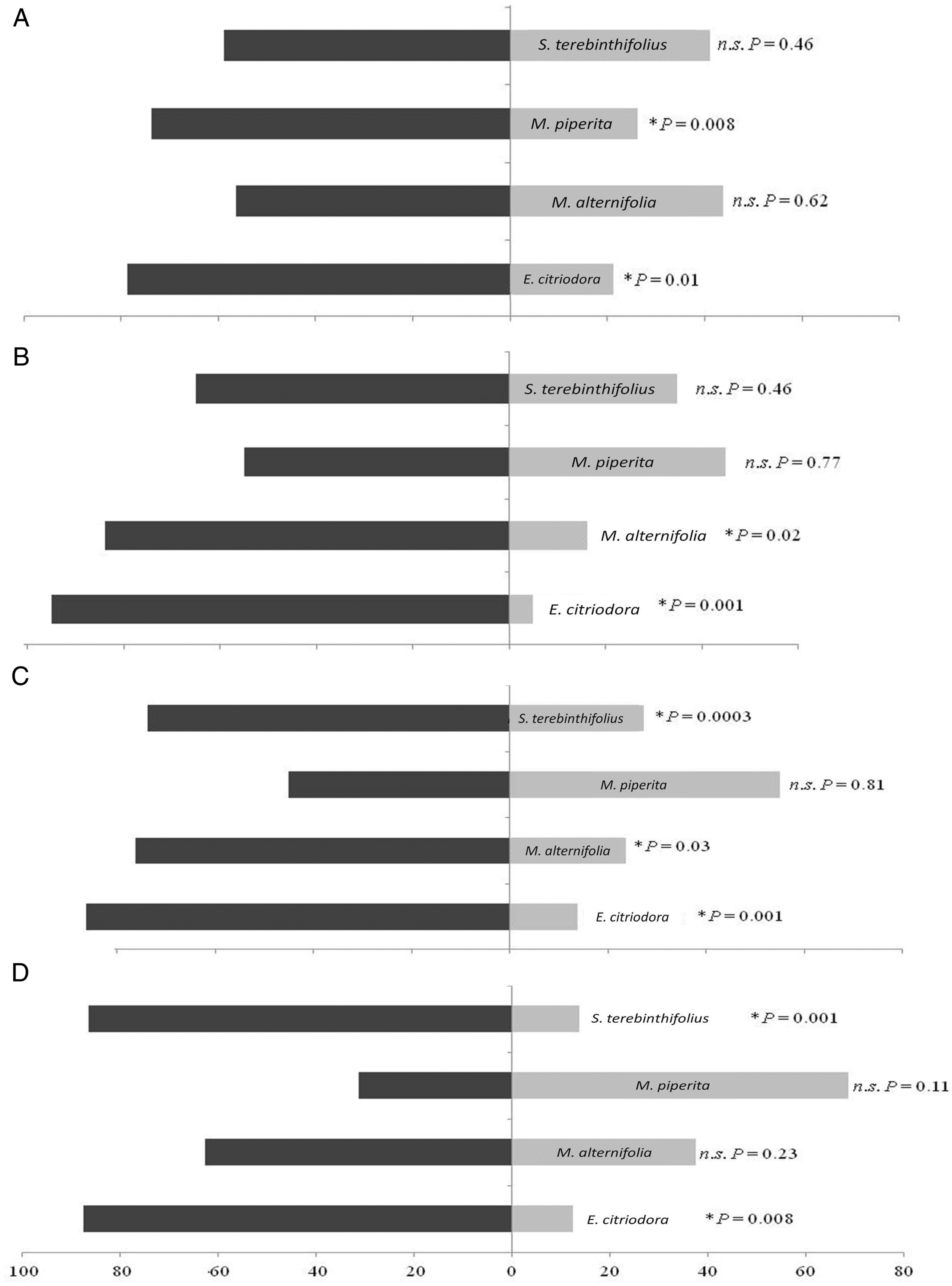

Repellency (PR) Values (%) for Corymbia citriodora (Ccit) (Myrtaceae), Melaleuca alternifolia (Malt) (Myrtaceae), Mentha × piperita (Mpip) (Lamiaceae), and Schinus terebinthifolius (Ster) (Anacardiaceae) Essential Oils to Sitophilus zeamais (Coleoptera: Curculionidae) Adults in Free-Choice Test.
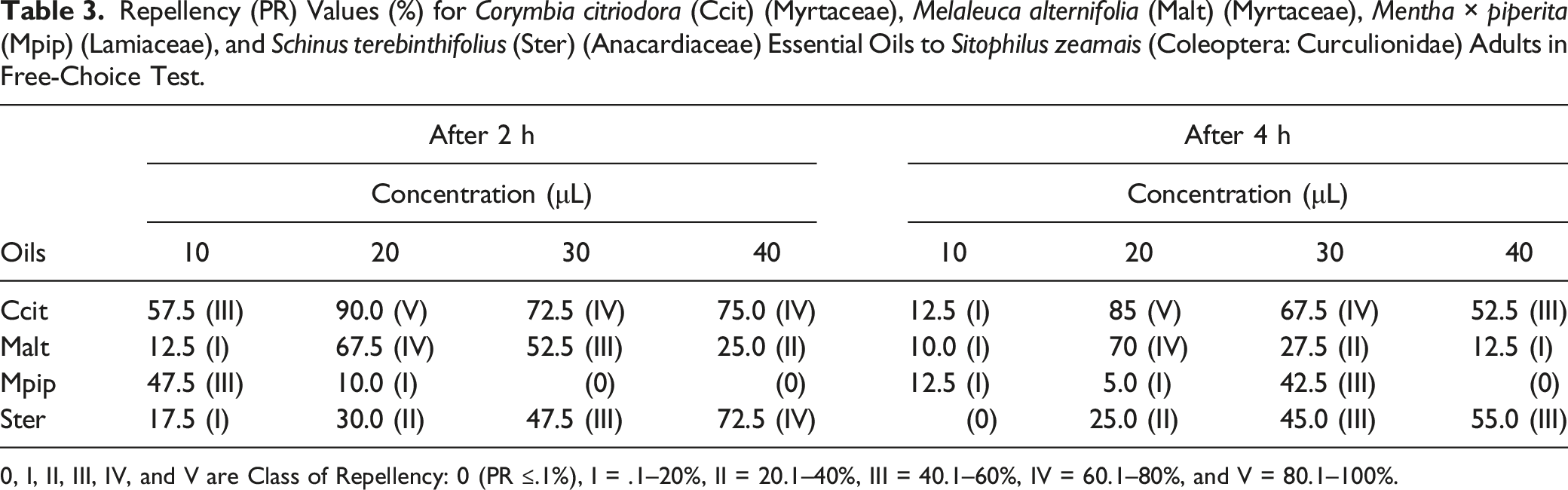
0, I, II, III, IV, and V are Class of Repellency: 0 (PR ≤.1%), I = .1–20%, II = 20.1–40%, III = 40.1–60%, IV = 60.1–80%, and V = 80.1–100%.
The repellent effect of C. citriodora and S. terebinthifolius essential oils was higher on S. zeamais adults than that of M. alternifolia > M. × piperita 2 and 4 h after exposure. The repellency effect followed the order: C. citriodora > S. terebinthifolius > M. alternifolia > M. × piperita. The values of the LC50 of the deltamethrin-based insecticide (positive control) and the essential oils on S. zeamais showed that this pyrethroid insecticide was more toxic to this insect.
Discussion
High percentages of citronellal and geraniol in C. citriodora; terpinen-4-ol, α-terpinene, and 1,8-cineole in M. alternifolia; menthol, menthone, and iso-menthone in M. × piperita; and β-caryophyllene and aromadendrene in S. terebinthifolius essential oils have been reported from Argentina, Australia, Brazil, Canada, China, Egypt, Morocco, New Zealand, the Galápagos Islands, and the United States of America.44-47 The quantitative and qualitative composition of secondary metabolites depends on genotypes and on the environmental factors of the area where the plant is growing as found for variations in C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius essential oils from different localities.48-51 Corymbia citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius are cultivated at low cost and sustainability in several countries with manual mechanized labor, period of planting, and adequate spacing and fertilization.52,53
The highest mortality of S. zeamais achieved after treatments with the essential oils of M. alternifolia, M. piperita, and C. citriodora might be attributed to their major components, especially terpinen-4-ol, α-terpinene and 1,8-cineole, menthol, menthone, all relatively toxic to S. zeamais.54,55
The high contact activity (LC50 estimated in 18.98 μL mL−1) of M. alternifolia essential oil might be attributed to its major components, especially terpinen-4-ol. This compound was also relatively toxic to the black bean aphid, Aphis fabae Scopoli, 1763 (Hemiptera: Aphididae) and the cotton leaf worm, Spodoptera littoralis (Boisduval, 1833) (Lepidoptera: Noctuidae). 56 The symptoms on insects by contact bioassay to M. alternifolia essential oil include convulsion and tremors followed by paralysis. This response may be because of activation of octopaminergic receptors by terpenes of essential oils in different medicinal plants, including M. alternifolia by its absorption through the insect tarsus and cuticle. 57
The higher toxic effect of the M. alternifolia essential oil in the ingestion bioassay (LC50 estimated in 1.03 μL g−1) agrees with the results of a pronounced antifeedant effect 24 h after its consumption by the larvae and a 97.8% antifeedant rate at 40 mg mL−1 on the third instar corn earworm, Helicoverpa armigera (Hübner, [1808]) (Lepidoptera: Noctuidae). The deterrent-feeding activity on H. armigera larvae by the major constituent of the M. alternifolia essential oil, terpinen-4-ol was high. 8
The greatest fumigant toxicity of M. alternifolia essential oil (LC50 estimated in 20.05 μL L−1 air) confirms that insecticide fumigation is among the most widely practiced control methods to protect stored products from insect infestations. The fumigant activity of its most abundant constituent, terpinen-4-ol, was high against coleopteran pests of stored products, including S. zeamais by penetrating as vapor into the airways (fumigation) of this insect. 58
The strong repellent activity of C. citriodora essential oil against S. zeamais and citronellal as its main compound (53.6%) are consistent with the high repellency of the cowpea weevil, Callosobruchus maculatus (F., 1775) (Coleoptera: Chrysomelidae) and S. zeamais at all doses tested (1–64 μL) of this essential oil, especially from −.16 to −.60. The overall repellencies of C. maculatus and S. zeamais at all doses of citronellal were 67.50 ± 7.0% and 92.12 + 3.9%, respectively. 59
The relatively strong and moderate toxicity of M. alternifolia and M. × piperita oils, respectively, and its low repellent effect against S. zeamais indicate that toxicity is not directly attached to the repellent or attraction–inhibitory effect, but it is a complex combination of different mechanisms. 60 The biological activities of the C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius essential oils may be due to differences in their chemical compositions. The chemicals of essential oils are generally monoterpenes, such as limonene, myrcene, pinene, p-cymene, phellandrene, and terpinene.61-63 These compounds act as neurotoxins, with several proposed modes-of-action, for example, as octopamine agonists or antagonists, as acetylcholinesterase inhibitors, or as GABA antagonists.64-66
The C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius essential oils were toxic to S. zeamais adults through contact, fumigation, ingestion, and repellency at different levels. These methods are widely used to control stored product pests reducing the use of conventional insecticides; their constituents can become more important in the IPM of stored products reducing the risks associated with synthetic insecticides. The essential oils of C. citriodora, M. alternifolia, M. × piperita, and S. terebinthifolius can be used as botanical insecticides to control S. zeamais adults.
Footnotes
Acknowledgments
Thanks to Irineu Lorini (Embrapa Soybean, Londrina, Paraná State, Brazil) and Paulo Roberto Valle da Silva Pereira (Embrapa Wheat, Passo Fundo, Rio Grande do Sul State, Brazil) for providing insects to initiate our laboratory colony. Thanks also to Maria Rita Cabral Sales de Melo (Biology Department, Federal Rural University of Pernambuco, Recife, Pernambuco State, Brazil) for identifying S. terebinthifolius species name.
Author Contributions
Hany Ahmed Fouad, Cláudio Augusto Gomes da Câmara, Marcílio Martins de Moraes designed the study; Hany Ahmed Fouad, Wagner de Souza Tavares conducted the literature search, collected, interpreted the data. Hany Ahmed Fouad, Cláudio Augusto Gomes da Câmara, Marcílio Martins de Moraes analyzed and researched the data; Hany Ahmed Fouad, Wagner de Souza Tavares, Cláudio Augusto Gomes da Câmara drafted the manuscript. Hany Ahmed Fouad, Wagner de Souza Tavares, Cláudio Augusto Gomes da Câmara, Marcílio Martins de Moraes literature search, analysis and interpretation of data and wrote the manuscript; Jesusa Crisostomo Legaspi, José Cola Zanuncio revised the manuscript; all authors have read and approved the final version of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the thanks to The World Academy of Sciences (TWAS) and the Brazilian institution Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for their scholarship and financial support.
