Abstract
The use of doxorubicin (DOX) as an anthraquinone antineoplastic agent is limited due to its cardiotoxicity. Our previous study suggested that low-dose radiation (LDR) could mitigate the cardiotoxicity induced by DOX via suppressing oxidative stress and cell apoptosis. However, the molecular targets and protective mechanism of LDR are not understood. In the present study, we sought to investigate the mechanisms underlying LDR’s cardioprotection. Balb/c mice were randomly divided into four groups: Control group (no treatment), DOX group, LDR group (75 mGy), and LDR-72 h-DOX group (LDR pretreatment followed by intraperitoneal injection of DOX). Electron microscopy, PCR, and Western blot analyses indicated that LDR pretreatment mitigated changes in mitochondrial morphology caused by DOX, upregulated activity of mitochondrial complexes, and restored ATP levels in cardiomyocytes that were decreased by DOX. Whole genome microarray and PCR analyses showed that mitochondrial-related genes were altered by LDR pretreatment. Thus, our study showed that LDR can protect cardiomyocytes against DOX through improving mitochondrial function and increasing ATP production. This research could inform DOX chemotherapy strategies and provide new insight into the molecule mechanisms underlying the cardioprotective effects of LDR.
Introduction
Doxorubicin (DOX) was first introduced in the 1970s and became one of the most commonly used anthracycline antibiotics for the treatment of hematological and solid tumors. 1 Anthracyclines are currently used to treat various cancers, including leukemia, lymphoma, melanoma, uterine, breast, and gastric cancers. 2 However, within one year of completing anthracycline chemotherapy, 9% of patients present with impaired left ventricular (LV) ejection fraction (EF% <50%). 3 Anthracycline-induced cardiotoxicity is caused by multiple molecular processes that converge to functionally alter cardiomyocytes, subsequently resulting in cell death. 2 Dox inhibits DNA and RNA syntheses through inhibiting topoisomerase II.4,5 While DOX can delay the development and progression of multiple malignant tumors, its eminent use is subdued by several side effects, primarily cardiotoxicity. 6
The cardiotoxic effects of chemotherapy initially manifest as subclinical myocardial damage and progressively result in early asymptomatic reduction of LVEF% and ultimately symptomatic, and often intractable, heart failure. 7 Some clinical studies have demonstrated that troponins can be used to assess the cardiotoxicity induced by chemotherapy, especially trastuzumab 8 and several tyrosine kinase inhibitors. 9 Similarly, natriuretic peptides including brain natriuretic peptide (BNP), atrial natriuretic peptide (ANP), and their N-terminal counterparts (NT-pro BNP and NT-pro ANP) can be used as biomarkers of heart failure and have been explored as predictors of chemotherapy-induced cardiotoxicity.10-12 Oxidative stress, mitochondrial impairment, increased apoptosis, dysregulated autophagy, and increased fibrosis have also been shown to play important roles in the process of DOX-induced cardiotoxicity.
Low-dose radiation (LDR), proposed in 1986 by the United Nations Committee on the effect of atomic radiation (UNSCEAR), refers to a radiation dose <.1 Gy within low LET (linear energy transfer) or a radiation dose <.05 Gy within high LET.13,14 In 2006, LDR was revised as 100 mSV.15,16 Although LDR can induce gene mutations and carcinogenesis, 17 it also exerts protective effects against type 1 diabetes-induced cardiomyopathy and resistance to diabetic renal damage.18,19 The biological effects of LDR mainly manifest as hormesis and adaptive responses. 20 Hormesis is a biphasic dose–response phenomenon, characterized by a low-dose stimulation and a high-dose inhibition. Hormesis exhibits a “wake-up” function to induce adaptive responses to prepare living organisms to cope more efficiently with high-dose challenges of ionizing radiation (IR) or other damaging agents. 21 The effects of hormesis include increasing longevity, enhancing growth, enhancing immune function, and increasing embryo production.22,23 Low doses of IR (<.1 Gy) tend to affect cell homeostasis by producing hormesis, adaptive responses, low-dose hypersensitivity, immunological effects, and genomic instability. 24 Adaptive responses induced by LDR are defined by the use of a very low priming dose of radiation to stimulate cellular processes that result in an enhanced resistance to a second but larger challenge dose of radiation, reducing the side effects of chemotherapy drugs. 25
Studies have shown that various factors, such as helium, benzopyran, and certain drugs, can be used as cardioprotective agents.26-28 Our team has been focusing on the biological effects of LDR. Our previous studies showed that LDR pretreatment can reduce DOX-induced cardiotoxicity through protecting cardiomyocytes. 29 Our previous studies have shown that LDR does not induce tumor cell proliferation in vitro and in vivo and that LDR pretreatment can improve the excitatory effect and adaptive response of normal cells, enhance the tolerance of normal tissues to radiotherapy and chemotherapy, and promote damage repair, without effecting tumor cells. 30 LDR pretreatment can reduce DOX-induced myocardial toxicity by preserving mitochondrial function. An expression profile microarray suggested that LDR pretreatment can reduce myocardial toxicity induced by chemotherapy drugs by regulating energy metabolism. Our team has also confirmed mitochondrial dysfunction in the myocardium in response to chemotherapy.31,32
Although we have evidence of LDR’s protective effects, the molecular targets and mechanisms underlying the cardioprotection conferred by LDR remain unclear. In this study, a DOX-induced cardiotoxicity mouse model was used to investigate the potential protective effects and underlying mechanism of LDR pretreatment. We further sought to identify the role of mitochondria in this process and confirmed that LDR pretreatment can protect myocardial mitochondria. Our results indicate that LDR pretreatment may attenuate DOX-induced mitochondrial damage and increase ATP synthesis by improving mitochondrial function, deepening our understanding of how LDR pretreatment can protect the heart.
Materials and Methods
Animals and Treatment
Eight-week-old Balb/c female mice (Experimental Animal Center of Norman Bethune Medical College, Jilin University) were housed under temperature-controlled (22 ± 2°C) and light-controlled (12-h light and 12-h dark cycles) conditions, with free access to food and water. The experimental protocol was approved by the Animal Ethics Review Committee of Basic Medical Sciences, Jilin University, in accordance with the regulations of the Institutional Committee for the Care and Use of Laboratory Animals. All mice were housed in specific pathogen-free conditions at a constant temperature and humidity, with free access to food and water. The bedding was replaced regularly. The mice were randomly divided into four groups: Control group, DOX group, LDR group, and LDR-72 h-DOX group, and each group included 12 mice. The groups were treated as follows: (1) Control group (vehicle treatment): mice were irradiated with 0 mGy and were treated with a .9% NaCl solution through intraperitoneal injection 72 h later; (2) LDR group: mice received whole body irradiation with LDR 72 h before injection of a .9% NaCl solution. A deep X-ray machine model was used to deliver the LDR whole-body irradiation at a dose of 75 mGy at a rate of 12.5 mGy/min; (3) DOX group: mice were sham irradiated with X-ray and then injected with 7.5 mg/kg DOX 72 h later ; and (4) LDR-72 h-DOX group: mice were irradiated with 75 mGy and injected with 7.5 mg/kg DOX at 72 h later. Five days after the injection, mice were anesthetized via injection of .1 mL pentobarbitone (1%) before harvesting the heart. The LV myocardium was separated for mitochondrial mRNA and protein isolation (Figure 1). Animal disposal process.
Transmission Electron Microscopy
Transmission electron microscopy was utilized to observe the ultrastructural changes of cardiomyocytes and mitochondria. Three mice were tested in each group. Briefly, the isolated heart apex was immediately fixed in 2% glutaraldehyde and embedded in resin. Ultrathin sections (50–70 nm) were made using the Leica EMUC Ultrathin microtome and stained with a 3% uranyl acetate and lead citrate double staining reagent. The ultrastructure of the cardiomyocytes and their mitochondria was shown in electron micrographs.33,34
Isolation of Mitochondria
Three myocardium samples were taken from each group. Cytoplasmic and mitochondrial proteins were isolated using the Tissue Mitochondrial Isolation Kit (Beyotime, C3606) according to the kit’s instructions. The kit contained a mitochondrial isolation reagent, trypsin digestion solution, mitochondrial storage solution, and mitochondrial lysis solution. Briefly, tissues were incubated in 1 mL ice-cold mitochondrial lysis buffer for 10 min and then transferred into a glass homogenizer to be homogenized for 10 strokes on ice. The homogenate was centrifuged at 800 r/min for 10 min at 4°C. The supernatant was collected and centrifuged again at 12 000 r/min for 25 min at 4°C to obtain the cytosolic (supernatant) and mitochondrial (deposition) fractions. Samples of mitochondria were dissolved in lysis buffer, and proteins were detected using Western blot analysis as described. For proteomics and Western blot assays, 150 to 200 μL of mitochondrial lysate was added to the isolated mitochondrial from 50 to 100 mg of the LV tissue. Protein concentrations were quantified using a Bio-Rad assay reagent.
Microarray Analysis
For microarray analysis, we included two mice from each group to achieve biological replicates. Total RNA was extracted from the LV using TRIzol (Sigma-Aldrich), and RNA integrity was analyzed using the Agilent RNA 6000 Nano assay. Qubit was used to determine RNA quality. The same amount of total RNA (100 ng) was used for each sample. Targets were prepared using an Eberwine-based amplification method with the One Array plus RNA amplification kit (Phalanx Biotech Group, CA San Diego, USA) to generate amino-allyl antisense RNA. Aminoallyl-RNA probes labeled with NGS-Cy5 were hybridized at 50°C for 16 h to the Mice Whole Genome One Array Version 2.0. The hybridized array was scanned with the Agilent Microarray Scanner (G2505 C) under 100% and 10% PMT, respectively, at a 10 μM resolution. After scanning, the raw intensity data were analyzed with Gene Pix™ 4 to determine mRNA levels. The data were pre-processed using the Rosetta Resolver® System (Rosetta Biosoftware). Differentially expressed genes were those with a log2 (fold change) ≥1 and P < .05.
PCR
RNA Extraction and Quantification
Myocardial tissue samples were homogenized in TRI Reagent (1 mL), and 200 μL chloroform was added. The sample was centrifuged at 12 000 g (4°C, 15 min), and the supernatant was transferred to a sterile centrifuge tube. Isopropanol (500 μL) was added to the supernatant, mixed, and then centrifuged again at 12 000 g (4°C, 10 min). The upper layer of transparent liquid was removed and 1 mL of 75% ethanol was added. This step was repeated 5 to 10 times. After the final ethanol wash, the sample was centrifuged at 7600 g (4°C, 5 min), placed at room temperature for 3 to 5 min, air dried, and dissolved in RNase-free water (30 μL).
mRNA was reverse transcribed to synthesize cDNA, followed by PCR. Each PCR assay used 1 μg RNA per 20 μL reaction. The experiment was performed according to the manufacturer’s instruction (PCR array kit: Mouse Mitochondrial Energy Metabolism, SABiosciences, Qiagen, PAMM-008Z). Briefly, every reaction was performed using 10 ng cDNA per 25 μL reaction volume. Quantitative PCR was performed under the following conditions: 95°C 10 min; 40 cycles of 95°C, 15 sec and 60°C, 1 min. The assays were performed on a Bio-Rad CFX ConnectTM Real-Time PCR Detection System. The qPCR master mix (QIAGEN, 330 523) was used for qPCR assay.
Quality control
We used PCR array reproducibility, RTC (reverse transcription controls), and PPC (Positive PCR Control) for quality control. B2m and Gapdh were used as the reference genes.
Mitochondrial complex assays
Three samples were collected for each group. Activities of mitochondrial complexes I–V were measured using enzyme assay kits according to the manufacturer’s protocols (Abcam: ab109721, ab109905, ab109911, and ab109714). Mitochondrial pellets were suspended in PBS supplemented with 10% detergent provided in the kits. Protein concentrations of the mitochondrial lysates were estimated, and 25 μg (for complexes I, III, IV, and V) or 100 μg (for complex II) mitochondrial protein was used per reaction. Enzyme activities were measured using a spectrophotometric method in duplicate and expressed as change of absorbance per minute per mg protein.
Measurement of Myocardial Mitochondria ATP Content
Total intracellular ATP content was measured using a mitochondrial ATP detection assay kit (Abcam, ab113849) according to manufacturer’s protocol. The absorbance was read using a Tecan Infinite M1000 Pro plate reader, and ATP content was calculated based on a standard curve generated from ATP standards on the same 96-well plate.
Western Blot Analysis
Mitochondrial and cytoplasmic protein markers were detected using Western blot analysis. Briefly, mitochondria were dissolved in lysis buffer, and mitochondrial protein concentrations were detected using a Bio-Rad detergent-compatible protein assay kit (Bio-Rad, USA). Cell lysates were separated by 10% SDS-PAGE and transferred onto nitrocellulose membranes. The membranes were blocked with 5% (w/v) non-fat dry milk in TBS-T-buffered saline for 1 h at room temperature and incubated with primary antibodies overnight at room temperature. After being washed, the bound antibodies were detected with an HRP-conjugated secondary antibody and visualized using the ECL detection reagent (Thermo Fisher Scientific) according to the manufacturer’s instruction. β-tubulin (1:1000; Proteintech) was used as a cytoplasmic protein marker, and ATP5A (1:1000; Proteintech) was used as a mitochondrial protein marker.
Statistical Analysis
Data are presented as means ± standard error (SE). Statistical analyses were performed with SPSS 17.0. Differences among groups were compared with a one-way ANOVA and the Newman–Keuls test. P < .05 was considered statistically significant.
Results
LDR Pretreatment Reduces DOX-Induced Mitochondrial Morphological Alterations and Injury
Hao et al have demonstrated that the mechanism of DOX–induced cardiotoxicity is related to mitochondrial injury.
35
To further explore whether LDR could mitigate the mitochondrial injury caused by DOX, we used electron microscopy to observe changes in cardiomyocyte ultrastructure. In the Control group, the cardiac muscle fibers were arranged regularly and the cell morphology was normal. Compared with the Control group, the cardiac muscle fibers in the DOX group became disordered, mitochondria were swelled, and the number of mitochondrial cristae was decreased. Compared with the DOX group, the cardiac muscle fibers in the LDR-72 h-DOX group were relatively regularly arranged, and the mitochondria were only slightly swelled. The representative micrographs in Figure 2 suggest that LDR pretreatment could lessen the mitochondrial structural damage caused by DOX and increase the number of mitochondria in the myocardium. Transmission electron micrograph of myocardial mitochondria (×7500) in different groups. (A) Cardiomyocytes from the Control group showing normal myocardial ultrastructure with regularly arranged mitochondria. (B) Cardiomyocytes from the LDR group showing normal myocardial ultrastructure, and the number of mitochondria was increased. (C) Cardiomyocytes from the DOX group showing large vacuoles and damaged mitochondrial structures. (D) Cardiomyocytes from the LDR-72 h-DOX group showing relatively normal myocardial ultrastructure and slight injury of mitochondria.
LDR Affects the Expression of Mitochondria-Related Genes
To further determine the molecular targets of LDR pretreatment, we performed whole genome expression microarray to detect differentially expressed genes. The dashed red and green lines in Figure 3, respectively, represent the thresholds for P-values and multiple screening. Compared with the Control group, the LDR group had 35 upregulated genes and 490 downregulated genes (Figure 3(A)). Compared with the Control group, DOX-treated mice showed a total of 343 differentially expressed genes (Figure 3(B)). Gene ontology (GO) analysis revealed that these differentially expressed genes were assigned to the cellular component process and were mainly involved in mitochondria (77 differentially expressed genes) and endoplasmic reticulum (63 differentially expressed genes) (Figure 4(A)). Compared with the LDR group, 258 genes were upregulated and 204 genes were downregulated in the LDR-72 h-DOX group (Figure 3(C)). Compared with the DOX group, the LDR-72 h-DOX-treated mice showed a total of 204 differentially expressed genes (Figure 3(D)). GO analysis revealed that these differentially expressed genes were mainly involved in plasma membrane (79 differentially expressed genes) and mitochondria (42 differentially expressed genes) (Figure 4(B)). These data confirmed that LDR confers its protective effects by altering mitochondrial gene expression. Gene expression profile. (A) Differential gene expression between LDR and Control groups. (B) Differential gene expression between DOX and Control groups. (C) Differential gene expression between the LDR-72 h-DOX and LDR groups. (D) Differential gene expression between LDR-72 h-DOX and DOX groups. GO analysis in different groups. (A) GO analysis of differential genes between Control and DOX groups. (B) GO analysis of differential genes between DOX and LDR-72 h-DOX groups.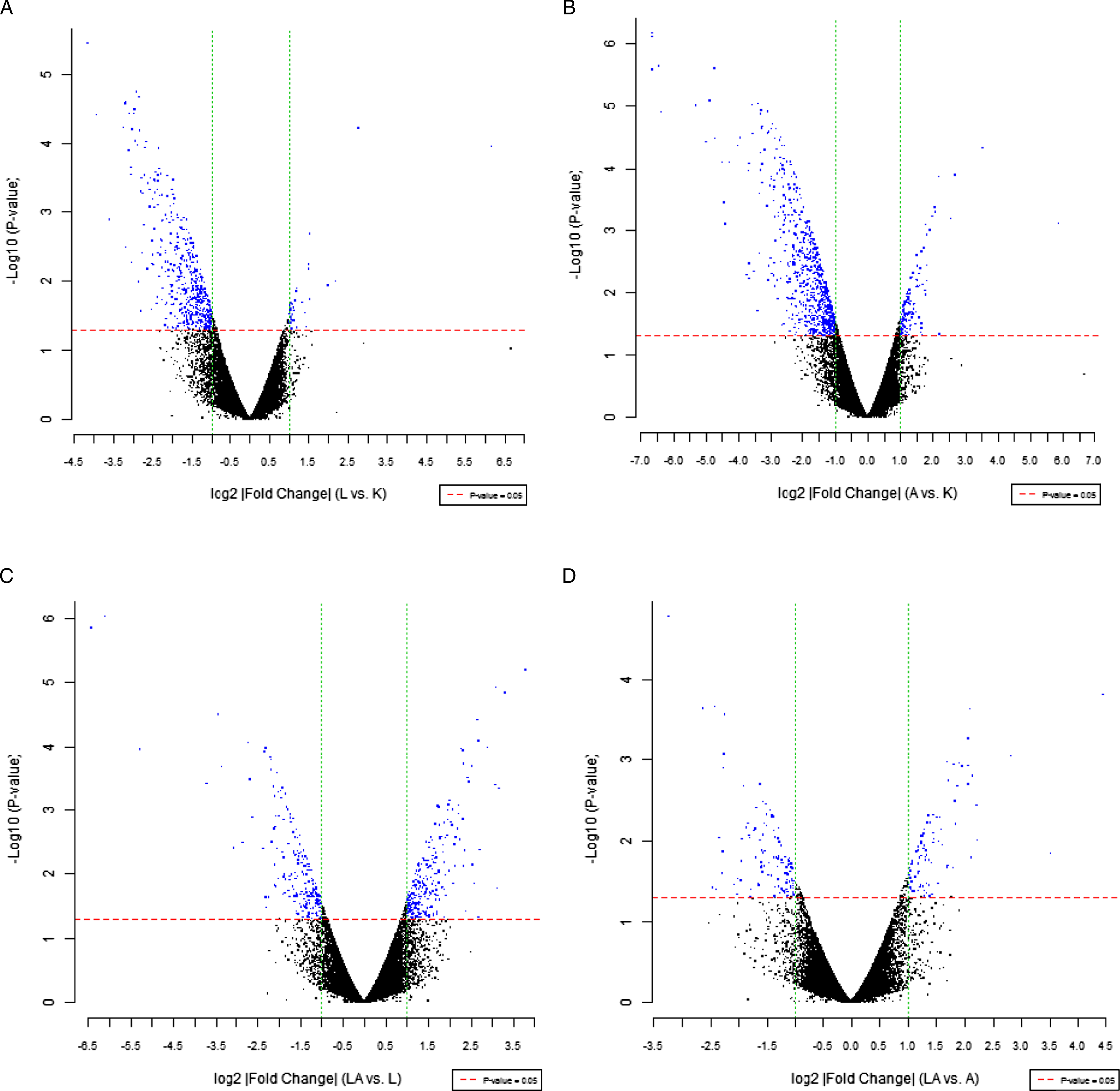

Expression of mitochondrial-related genes in the LDR-72 h-DOX group comparing with the DOX-treated mice.

DOX group: doxorubicin administration to induce myocardial damage.
LDR-72 h-DOX group: doxorubicin was intraperitoneally injected 72 h after 75 mGy LDR irradiation; LDR: A deep X-ray machine model was used to deliver LDR whole-body irradiation at a dose of 75 mGy at a dose rate of 12.5 mGy/min.
LDR Pretreatment Protects Mitochondrial Function and ATP Production in DOX-Treated Mice
Since mitochondrial metabolism is expected to change with altered mitochondria-related gene expression, we next measured mitochondrial oxidative phosphorylation (OXPHOS) in the myocardium tissue. We found that the activity of complexes I, II, and IV decreased in the DOX group compared with the Control group, and these changes were recovered in the LDR-72 h-DOX group. These results indicate that DOX decreases mitochondrial NADH dehydrogenase, cytochrome oxidase, and cytochrome reductase activities, resulting in disordered ATP production disorder and subsequent myocardial injury. These data also indicate that pretreatment with LDR could mitigate the mitochondrial dysfunction induced by DOX (Figure 5(A)-(D)). Compared with the Control group, the activity of complex III in the DOX group was increased, while the activity in the LDR-72 h-DOX group was decreased. These data confirm that LDR pretreatment could mediate the generation of reactive oxygen species (ROS) as a protective complex III signaling molecule (Figure 5(C)). We also measured mitochondrial ATP concentrations in different groups. DOX treatment significantly decreased the ATP concentration while pretreatment with LDR increased the ATP concentration compared with the DOX-treated group (Figure 5(F)). LDR pre-treatment could mitigate the mitochondrial deficiency induced by DOX in the hearts of mice. (A) Mitochondrial complex I activity. (B) Mitochondrial complex II activity. (C) Mitochondrial complex III activity. (D) Mitochondrial complex IV activity. (E) Mitochondrial complex V activity. (F) ATP concentration (*P < .05, **P < .01).
LDR Pre-treatment Altered DOX-Induced Alterations in the Expression of Mitochondrial Complex-Associated Proteins in Cardiocytes
To assess the effects of LDR on mitochondria, we measured expression of mitochondrial complex-associated proteins using Western blot analysis. We previously found that the downregulation of NDUFV1, ATP5f1, and CYC1 was associated with mitochondrial complex I, III, and V functions, respectively. Therefore, after confirming changes in mitochondrial complex function and the protective effect of LDR pretreatment, we focused on the expression of these three proteins. Both LDR and DOX treatment decreased the expressions of NDUFV1, ATP5f1, and CYC1 compared with the Control group. However, expressions of these proteins did not differ between the LDR-72 h-DOX group and the Control group, indicating that LDR pretreatment protects against DOX-induced changes of mitochondrial functional proteins (Figure 6). Expression of mitochondrial complex-relative proteins NDUFV1, ATP5f1, and CYC1 (A) Western blot images. (B) Relative optical density (*P < .05, ***P < .005).
Discussion
Mitochondria play an important role in energy production, programmed cell death, cell division, cell metabolism, and proliferation. 36 The abundance of mitochondria in cardiomyocytes closely links mitochondrial bioenergetics with myocardial function and viability, and mitochondrial dysfunction has recently been recognized as a pivotal factor in the development of DOX-induced cardiotoxicity. 37 In our previous study, we observed that pretreatment with LDR could reduce DOX-induced oxidative damage (ROS formation, protein nitrification, and lipid peroxidation) and increase antioxidant activity (superoxide dismutase and glutathione peroxidase) in the hearts of LDR/DOX mice. DOX-induced cardiac apoptosis (TUNEL-stained and cleaved caspase-3) and mitochondrial apoptotic pathways (increased expressions of p53, Bax, and caspase-9; decreased Bcl2 expression; and δψM dissipation) were also reduced. These results suggest that LDR attenuates DOX-induced cell death, possibly by inhibiting mitochondrial oxidative stress and apoptotic signaling. 38 It has been reported that DOX treatment leads to mitochondrial swelling, fusion, dissolution, and pronounced disorientation of the cristae and vacuoles within the cytoplasm, 39 which is consistent with the findings in the current study. Here, we further demonstrated the benefit of LDR pretreatment for mitochondrial function, respiratory chain complex activity, and ATP production, deepening our knowledge of how LDR protects against DOX-induced cardiotoxicity. Our results also showed that mice pretreated with LDR had fewer and less extensively swollen mitochondria with less disorientation of the cristae and vacuoles within the cytoplasm. These findings suggest that the protective effects of LDR are related to preserving mitochondrial function (Figure 7).
Our study demonstrated that mitochondria-related genes such as Cox5b, Cox7a2, Cox8c, Atp4a, Atp6v0d2, Atp6v1c2, Atp6v1e2, and Atp6v1g3 were downregulated by DOX and that LDR pretreatment reversed the effects of DOX, further validating that the cardioprotective effects of LDR are related to mitochondrial function. Complex IV (CcO), encoded by Cox5b, Cox7a2, and Cox8c, is the last enzyme in the respiratory electron chain of mitochondria and might be responsible for the mitochondrial dysfunction leading to myocardial damage. 40 H+/K+-ATPase, encoded by Atp4a, maintains calcium transfer and exchange of potassium with hydronium; DOX treatment has been shown to cause calcium overload and ion disturbance in cardiomyocytes. 41 Vacuolar-type H+-ATPase (V-ATPase), encoded by Atp6v0d2, Atp6v1c2, Atp6v1e2, and Atp6v1g3, is a highly evolutionary conserved enzyme that can aggravate cardiomyocyte autophagy. 42 As suggested above, LDR exerted protective effects on cardiomyocytes by lessening mitochondrial morphological injury and regulating the expression of mitochondria-related genes. Both mRNA levels and protein expression of mitochondria-related genes were affected by DOX in our study. The levels of mitochondrial complex-related proteins, NDUFV1, ATP5f1, and CYC1, were downregulated by DOX, which was consistent with previous studies.43,44
The mitochondrial respiratory chain consists of four enzyme complexes: complex I (NADH dehydrogenase), complex II (succinate dehydrogenase), complex III (cytochrome c reductase), and complex IV (cytochrome c oxidase). These complexes are responsible for generating 90% of the energy needed for proper heart function. 45 ATP synthesis via the respiratory chain involves two coupled processes: electron transport and OXPHOS machinery. Both processes are highly dependent on the function of the mitochondrial complexes. Thus, any mitochondrial complex-related damage could have a big impact on heart function. 46 In the present study, DOX inhibited the activity of complexes I, II, and IV and reduced ATP synthesis significantly, while LDR pretreatment restored these DOX-related effects. DOX also increased the activity of complex III, and LDR pretreatment decreased its activity, consistent with other studies.47-49 Activity of the mitochondrial complex in the LDR-72 h-DOX group was higher than that in DOX and LDR groups, indicating that LDR pretreatment can initiate adaptive responses of mitochondria, and when there is no secondary stimulation, activity of the mitochondrial complex decreased (LDR-alone group). However, when DOX was used as a second stimulus, the complex was excited (hormesis) and the activity increased (LDR-72 h-DOX group).
There are some limitations of our study that should be noted. We acknowledge that the mice used in this study were healthy and we did not include a tumor mouse model. To address this, we will establish a DOX-induced myocardial injury model in breast cancer tumor-bearing mice to demonstrate the synergistic anti-tumor effects of LDR and DOX and the myocardial protective effect of LDR in future studies. Protective mechanism of LDR pretreatment on DOX via mitochondrial pathways.
In conclusion, the results in the present study demonstrated that LDR pretreatment protected against DOX-induced myocardial injury, and this protection might be related to changes in mitochondrial gene regulation and protein expression, activity of mitochondrial complexes, and overall mitochondrial function. Therefore, our data suggest that LDR pretreatment might be a promising candidate to prevent the cardiotoxic side effects of DOX during chemotherapy. Our findings provide new insights into the underlying protective mechanisms of LDR.
Footnotes
Author Contributions
Conceptualization: Hongyu Jiang; data curation: Xinxin Meng; data analysis: Yanwei Du; funding acquisition: Hongyu Jiang and Dandan Liu; supervision: Lijing Zhao and Hongyu Jiang; writing—original draft: Xin Jiang; and writing—review and editing: Di Zhao.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Department of Science and Technology of Jilin Province (Nos. 20190902012TC and 20190304129YY) and Department of Finance of Jilin Province (JLSWSRCZX20210-084 and JLSCZD2019-077).
