Abstract
Xanthohumol (XN) is the major prenylated chalcone of the female inflorescences (cone) of the hop plant (Humulus lupulus). It is also a constituent of beer, the major dietary source of prenylated flavonoids. It has shown strong antitumorigenic activity towards various types of cancer cells. In the present study, we show the impact on human hepatocarcinoma cell line HepG2 cell and potential adverse effects on rat primary hepatocytes. Cell growth/viability assay (MTT) demonstrated that HepG2 cells are highly sensitive to XN at a concentration range of 25-100 μM. The primary mode of tumor cell destruction was apoptosis as demonstrated by the binding of Annexin Ⅴ-FITC, we show that XN at a concentration of 25 μM induced apoptosis in HepG2. Further evidence that XN kills HepG2 by inducing apoptosis was provided by the impact of XN on the cleavage of PARP-1 and caspases-3. In contrast, XN concentrations up to 100 μM did not affect viability of primary rat hepatocytes in vitro, meanwhile, XN did not induce the apoptosis of primary rat hepatocytes in vitro. In summary, our data provide a rationale for clinical evaluation of XN for the treatment of liver cancer.
Introduction
Hepatocellular carcinoma (HCC) is one of the most frequent malignant tumors worldwide with an incidence still rising. 1 According to global cancer statistics, in 2020, 905,677 new HCC cases were diagnosed, and 830,180 HCC-related deaths occurred. 2 Since HCC is almost asymptomatic in the early stage, most patients are diagnosed at the advanced stage, which current treatment modalities have limited efficacy on HCC.3,4 Hepatocellular carcinoma with poor prognosis is characterized by rapid cell proliferation and strong expression of antiapoptotic genes, which suggest that they are mainly due to incomplete cell cycle arrest and apoptosis resistance under conventional therapies. 5 Therefore, understanding the underlying mechanism by which HCC progresses is critical for exploring effective diagnostic and therapeutic targets for HCC.
An accumulation of evidence disclosed that xanthohumol (XN), compound of the female hop plant, Humulus lupulus, has potential biological activities, such as anti-infection against microorganisms including bacteria, viruses, fungi, and anti-inflammatory, 6 as well as the most commonly studied anticancer properties.7-9 Exposure of cancer cells to XN may inhibit their proliferation, migration, and invasion, as well as induce apoptosis and cell cycle arrest. Consequently, all the aforementioned mechanisms may be involved in the chemopreventive activity of XN. Moreover, while most of the research on XN were performed on cancer cell lines, very few have addressed its effects on non-tumorigenic cells. Hence, the aim of this study was to further investigate the effect of XN on HepG2 cells and rat primary hepatocytes, by studying its role in cell proliferation, induction of apoptosis, and provided a rationale for clinical evaluation of XN for the treatment of liver cancer.
Materials and Methods
Chemicals and Reagents
XN (Figure 1) was provided by Yumen Tuopu Scientific and Technological Development Co., Ltd., purity > 98% (Yumen, Gansu, China). The cell culture medium RPMI-1640 was purchased from Gibco Laboratories (Grand Island, NY, USA). Neonatal bovine serum was obtained from Lanzhou Minhai Bio-engineering Co., Ltd. (Lanzhou, China). Streptomycin, penicillin, L-glutamine, and dimethyl sulfoxide (DMSO) were all greater than 98% in purity (Sigma-Aldirch Corp.). The 3-(4,5-dimethylthiazo-2-yl) −2,5 -diphenyl-tetrazolium bromide (MTT) kit and Annexin V-FITC/PI apoptosis detection kit were obtained from KeyGEN Biotech (Nanjing, China). RIPA Lysis Buffer and Enhanced BCA Protein Assay Kit were purchased from Beyotime (Shanghai, China). Collagenase type II was purchased from Worthington Biochemical Corp. (Lakewood, NJ, USA). Williams’ medium E was obtained from Sigma-Aldrich Japan (Tokyo, Japan). Anti- Pro-caspase-3、Anti- PRAP-1、Anti-β-actin receptor rabbit monoclonal antibody and rabbit IgG monoclonal isotype control were purchased from Abcam (Cambridge, MA, USA). All other chemicals made in China were analytical grade. Chemical structure of Xanthohumol.
Cell Culture
Human hepatoma cell line HepG2 was obtained from the cell bank of Chinese Academy of Science (Shanghai, China) and preserved in liquid nitrogen in our laboratory. Cells were routinely cultured in RPMI1640 medium supplemented with 10% (v/v) heat-inactivated fetal bovine serum (FBS), 100U/ml penicillin, 100 g/mL streptomycin, 1% non-essential amino acids, and 2 mmol/L l-glutamine, in a humidified atmosphere of 95% air and 5% CO2 at 37°C. The cells were cultured every 7 days at 1:3 split ratios and seeded in 12.5 cm2 cell culture flasks (1×106 cells/flask) in the experiments described below. The cells were used for experiments within eight passages to ensure cell line stability.
Animals and Primary Hepatocyte Isolation
Male Wistar rats (Lanzhou University School of Medicine) were used at the age of around 60 days (230g–300 g of body weight). Rats were anesthetized by intraperitoneal injection of 45 mg/kg sodium pentobarbital. Primary hepatocytes were isolated and enriched by a two-step in situ Seglen procedure10,11 with minor modifications as previously described. 12 Fresh hepatocytes (5.0 × 104 cells/cm2) were placed onto 12.5 cm 2 cell culture flasks coated with collagen type II and incubated in Williams’ medium E containing 10% fetal bovine serum.13,14
Cell Proliferative MTT Assay Dose Response
The MTT assay was used to evaluate the antiproliferative activities of XN on HepG2 cells and rat primary hepatocyte. The assay is based on the cleavage of the yellow tetrazolium salt MTT into purple formazan by metabolically active cells, which can be photometrically quantified. 15 An increase in the number of living cells results in an increase in total metabolic activity, which leads to a stronger color formation.
For the assays, cells (1×104) were seeded into each well of a 96-well plate in 100 μL of culture medium. After 24 h incubation to allow cells to adhere, cells were treated with XN at different concentrations for 24–48 h. The relative amount of viable cells was determined by measuring the reduction of MTT dye in live cells to blue formazan crystals at optical density at 540 nm.
Cell Morphology
Cells were seeded on 35 mm Petri dish at a density of 1×105 cells/ml with or without specified concentrations of the samples and the test groups. At each of the incubation periods (24 h, 48 h), the cells were observed under a microscope (IX71, Olympus Corporation).
Analysis of Apoptosis With Annexin-V FITC/PI Staining Assay
Apoptosis or necrosis was determined using an Annexin-v FITC/PI apoptosis detection kit as previously described. 16 HepG2 cells (5×105 cells/well) grown in 6-well plates for 24 h were used for Annexin-v FITC/PI experiments. Briefly, cells were resuspended in the binding buffer and reacted with 5 μL of annexin V-FITC reagent and 5 μL of propidium iodide (PI) for 30 min at room temperature in the dark. Stained cells were analyzed by fluorescent activated cell sorting (FACS) on a FACS flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA)
Western Blot
HepG2 cells (1.5 × 105 cells/ml) were seeded in 100 mm2 culture dish and incubated for 24 h to allow attachment. Various concentrations of XN were added to the dishes and incubated for 48 h. After treatment, cells were harvested and washed twice with PBS. Protein was isolated with lysis buffer and the total cellular protein concentration was determined by Bradford assay. Protein samples (50 μg) were separated on 10–14% SDS-polyacrylamide gels. Proteins resolved on the gels were transferred to nitrocellulose membranes and probed with protein specific antibodies to Pro-caspase-3 (1:2000) and PARP-1(1:3000) and β-actin (1:500). The membranes were washed with buffer and incubated with the appropriate horseradish peroxidase conjugated rabbit antibody as described previously. 16
Statistical Analysis
Data were expressed as mean ± standard deviation (SD) from at least three independent experiments. The two-tailed Student’s t test was used to determining the significant differences between the two groups. P values of less than .05 were considered as significant (*P < .05; **P < .01; ***P < .001; ****P < .0001; ns non-significant).
Results
Hepatocyte Isolation and Cell Viability
Primary hepatocytes were isolated and enriched by a two-step in situ Seglen procedure with minor modifications as previously described.
14
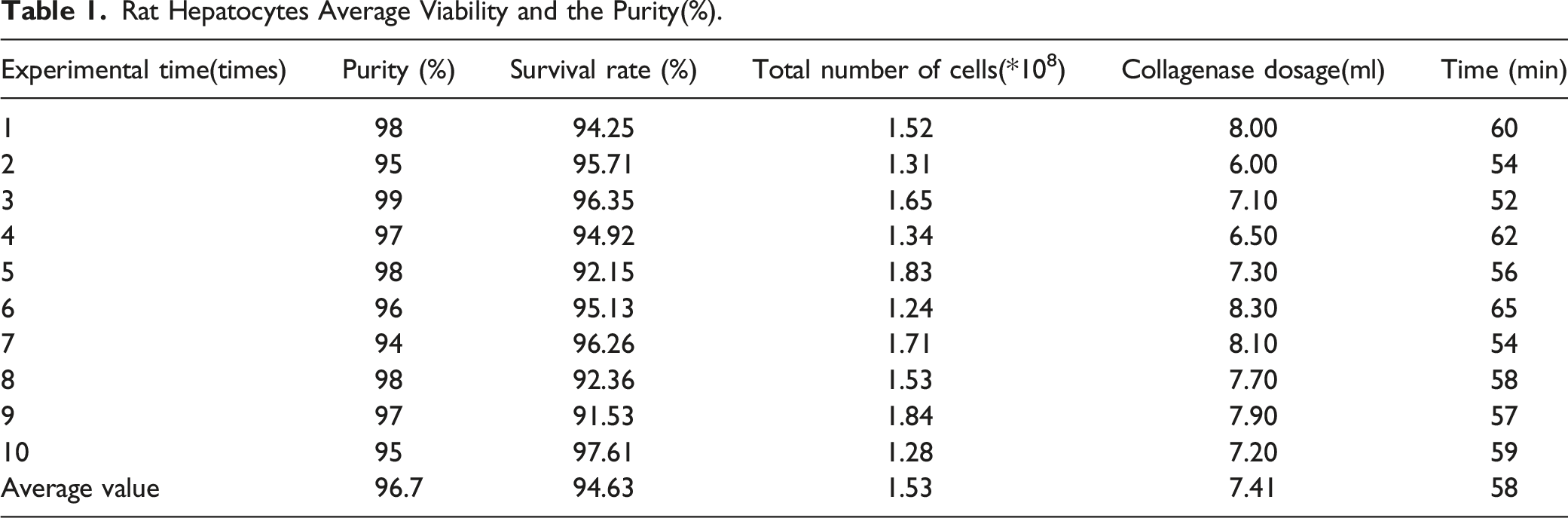
A total of 13 Wistar rats were used for primary hepatocytes isolation, of which 10 rats were successful, and the other 3 rats died due to accidental operation (the puncture failed when the portal vein was cannulated with D-Hanks solution), with a success rate of 76.9% (Figure 2, Figure 3). In most cases, typical hepatocyte yield is (1.53 +.31)×108 viable cells per mouse and the viability is 94.63% (Table 1). Anatomy the form of rats and separation of the rat liver. The rat primary hepatocytes after HE stain (×200). Rat Hepatocytes Average Viability and the Purity(%).

The Effects of Xanthohumol on Cell Viability
To investigate the effect of XN on cell proliferation, MTT assays were executed with different concentrations of XN. As shown in Figure.4A, treatment with XN significantly inhibited the growth of HepG2 cells in a dose dependent manner with an IC50 value of 68 ±0.3 μM, 52 ± 1.98 μM at 24h and 48h, respectively. The cytotoxicity of XN was also enhanced in a time-dependent manner. Extensive inhibition of cell growth was observed in the cells treated with high concentration (100 μM) of XN after 48 h of treatment. While the higher concentrations treatments exhibited no inhibitory effect on rat primary hepatocyte proliferation (Figure 4B). The maximum inhibition ratio is 11% at 120 μM of XN. Taken together, these results demonstrated that XN could significantly inhibit the growth of HepG2 cells, however, had no inhibitory effect on rat primary hepatocyte. Effects of XN on proliferation of HepG2 and rat primary hepatocyte cells. HepG2(A) and rat primary hepatocyte (B) were treated with increasing concentrations of XN for 24 and 48 h, and cell viability was determined by the MTT assay. Results are expressed as percentages of MTT absorbance with respect to the untreated vehicle control wells. Treatment with 0 μM XN was used as control. Data are expressed as mean ± standard error of the mean. n=3. *P<.05. **P<.01 vs control vs control (C) Photomicrographs of HepG2 (above) and Rat primary hepatocyte(down) from different treatment groups (magnification,×100).
The Effects of Xanthohumol on Apoptosis With Annexin-ⅤFITC/PI Staining
Whether XN destroys HepG2 cells by inducing apoptosis was investigated by the annexin V-FITC with flow cytometry. As shown in Figure 5A, HepG2 cells in early or late stages of apoptosis after XN treatment of 48 h. At 25 μM of XN, the apoptotic rate was approximately 46.8% (initial apoptosis, 18.2 ± 1.4%; late apoptosis, 38.6 ± 1.6%). In addition, an increase in levels of apoptosis was detected with the increase of the concentration of XN (Figure 5A.). A large number of necrosis and fragments appeared in HepG2 cells, up to 81.8%, while the 100 μM XN treated (Figure 5A.). All test groups produced a significantly greater percentage of apoptosis rate compared with the control (P < .001). While the same concentrations treatments exhibited no effect on rat primary hepatocyte apoptosis (Figure 5B). Inhibitory effects of XN on HepG2 cells (A) and rat primary hepatocyte cells (B) via apoptosis. HepG2 and rat primary hepatocyte were treated with increasing concentrations of XN for 24h, and the apoptosis percentage was analyzed by flow cytometry using Annexin V-FITC/PI staining. Results were summarized (right). Controls contained test cells and culture medium, but no test compounds (mean ± standard deviation, n=3, ***P<.001 vs control.).
The Effects of Xanthohumol on the Protein Level of Procaspase-3, PRAP-1
For elucidating the molecular mechanisms of XN in HepG2 cells, the expression of protein related to apoptosis was explored. The level of Procaspase-3 and PARP-1 proteins in HepG2 cells was investigated using Western blot assay with specific antibodies against the enzymes XN diminished the levels of Procaspase-3 and PARP-1 proteins in HepG2 cells. The levels of Procaspase-3 and PARP-1 were markedly reduced at 25 μM to 100 μM XN (Figure 6). There was a decrease in Procaspase-3 and PARP-1 with increasing doses of XN suggesting that the cells went through the caspase cascade–mediated apoptotic pathway. HepG2 cells were treated with XN at concentration of 25 to 100 μM for 48 h, and PARP-1 and procaspases-3 was probed by Western blotting. β-Actin served as the loading control. Each experiment was repeated at least two times. After treatment with different concentration XN, PARP-1 and procaspases-3 levels decreased. ***P<.001 vs control.
Discussion
Many dietary constituents of plant origin have been identified to exhibit protective effects against tumor and mutation-related diseases. In the past few years, the potential tumor chemopreventive properties of XN, an ingredient in beer, have been extensively researched. XN inhibited cancer cell proliferation and induced apoptosis in different human cell lines, including hepatocellular carcinoma. 17 Nevertheless, most studies on XN have been performed on cancer cell lines, and very few have addressed its effects on non-tumor cells. The aim of this study was to investigate the anti-cancer properties of XN on HCC HepG2 cells and rat primary hepatocytes, by studying its role on cell proliferation, induction of apoptosis, and caspase activities.
The assessment of cytotoxicity, in the initial series of experiments, disclosed that XN led to a stronger reduction in the viability of HepG2 cells in comparison to normal rat primary hepatocytes. At the concentration of 20 μM, XN was highly toxic to HepG2 cells, while the higher concentrations treatments exhibited no inhibitory effect on rat primary hepatocyte proliferation. Similar observations were described by Dorn et al 18 where XN at a concentration of 25 μM induced apoptosis in two HCC cell lines, HepG2 and Huh7, while at concentrations up to 100 μM did not affect the viability of primary human hepatocytes. Normal murine hepatocyte cell line resistance to XN was also described by Ho et al. 19 Overall, these findings indicate that normal rat primary hepatocytes were more resistant to the cytotoxic effects of XN than carcinoma HepG2 cells. This is a significant observation, as the differential cytotoxic response between normal and tumor-derived liver cells may have important implications for both the therapeutic and chemopreventive application of XN.
In order to further to explore the possible effects of XN on two cell lines, HepG2 and rat primary hepatocyte, we assessed the apoptosis by flow cytometry analysis. Apoptosis, essential in regulating cell number in many developmental and physiological settings, has been found to be impaired in many human tumors, indicating that disruption of apoptotic function contributes substantially to the transformation of a normal cell into a tumor cell. Apoptosis is an important phenomenon in the chemotherapy-induced killing of tumor cells. Our data show that XN resulted in apoptosis with some necrosis of treated HepG2 cells in a dose-dependent manner. In contrast to rat primary hepatocytes, XN did not affect the treated cells in apoptosis.
Apart from cell proliferation, our studies indicated that XN elicited apoptosis via the caspase-3–related pathway. Caspases are part of a growing family of cysteine proteases that have been demonstrated to be involved in many forms of apoptosis. 20 Both activation of caspase-3 and proteolytic cleavage of PARP have been considered to be crucial events in apoptosis.21,22 Procaspase-3 has been found to have elevated concentrations in cells from various cancerous tissues including certain neuroblastomas, lymphomas, leukemias, melanomas, and liver cancers. 23 This allows for a therapeutic index to be achieved for caspase-3-directed agents. Due to the focal role of active caspase-3 in successfully inducing apoptosis, the use of a small molecule to directly activate procaspase-3 has been suggested as a tactical approach in targeted cancer therapy. This protease exists as dimers of inactive proenzymes and has to be cleaved at specific aspartic acid residues to be activated. Active caspase-3 cleaves numerous substrates that eventually lead to cell death. During apoptosis, active caspase-3 leads to a self-amplifying cascade of proteolysis and cleavage of numerous effector proteins, including PARP. The growth inhibitory effect of XN was attributed to the induction of apoptosis as determined by the increased binding of annexin V-FITC to treated cells. In addition, XN induced the cleavage of PARP-1. The growth inhibitory and apoptosis inducing effects of XN in HepG2 cells observed in this study are consistent with those previously reported in other tumor models, including Melanoma and colon carcinoma.24,25 Collectively, these studies indicate that induction of apoptosis is part of the mechanism by which XN inhibits the growth of cancer cells, including Hepato- cellular carcinoma. As shown in Figure 6, proteolytic cleavage of caspase-3 and PARP was observed in XN-treated HepG2 for 48 h, which indicated that XN induced apoptotic cell death. In addition, the native PARP-1 protein was almost completely cleaved at 75 and 100 μM XN with the appearance of cleavage product in HepG2 cells. Together, these data demonstrate that XN kills HepG2 cells by inducing apoptosis.
Conclusions
In this paper, we have shown that XN treatment is associated with reduced growth of HepG2 cells, but no effect on normal rat primary hepatocyte by MTT assay. Furthermore, we have demonstrated that part of mechanism of XN inhibits the growth of HepG2 cells by induction of apoptosis and caspase pathway involved in this process. Our data provide a rationale for clinical evaluation of XN for the treatment of liver cancer. Further studies in other cancer cells and animal models are necessary to explore the mechanism of xanthohumol.
Footnotes
Acknowledgments
We thank Mr. Lianqing Ma, the president and general manager of Yumen Tuopu Science & Technology Development Co., Ltd., for kindly providing xanthohumol for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Science-Technology Foundation for Middle-Aged and Young Scientists of Gansu Province, China, no.1170RJY031.
