Abstract
Background
Arsenic (As) is a highly toxic and carcinogenic pollutant commonly found in soil and water, posing significant risks to human health and plant growth.
Objective
The objectives of this study to evaluate morphological, biochemical, and physiological markers, as well as ion homeostasis, to alleviate the toxic effects of As in sunflowers through the exogenous application of salicylic acid (SA), γ-aminobutyric acid (GABA), and their combination.
Methods
A pot experiment was conducted using two sunflower genotypes, FH-779 and FH-773, subjected to As stress (60 mg kg−1) to evaluate the effects of SA at 100 mg L−1, GABA at 200 mg L−1, and their combination on growth and related physiological and biochemical attributes under As stress.
Results
The study revealed that As toxicity had a detrimental effect on various growth parameters, chlorophyll pigments, relative water content, total proteins, and nutrient uptake in sunflower plants. It also led to increased oxidative stress, as indicated by higher levels of malondialdehyde (MDA) and hydrogen peroxide (H2O2), along with As accumulation in the roots and leaves. However, the application of SA and GABA protected against As-induced damage by enhancing the enzymatic antioxidant defense system. This was achieved through the activation of superoxide dismutase (SOD), catalase (CAT), and peroxidase (POD) activities, as well as an increase in osmolytes. They also improved nutrient acquisition and plant growth under As toxicity.
Conclusions
We investigated the regulatory roles of SA and GABA in mitigating arsenic-induced phytotoxic effects on sunflower. Our results revealed a significant interaction between SA and GABA in regulating growth, photosynthesis, metabolites, antioxidant defense systems, and nutrient uptake in sunflower under As stress. These findings provide valuable insights into plant defense mechanisms and strategies to enhance stress tolerance in contaminated environments. In the future, SA and GABA could be valuable tools for managing stress in other important crops facing abiotic stress conditions.
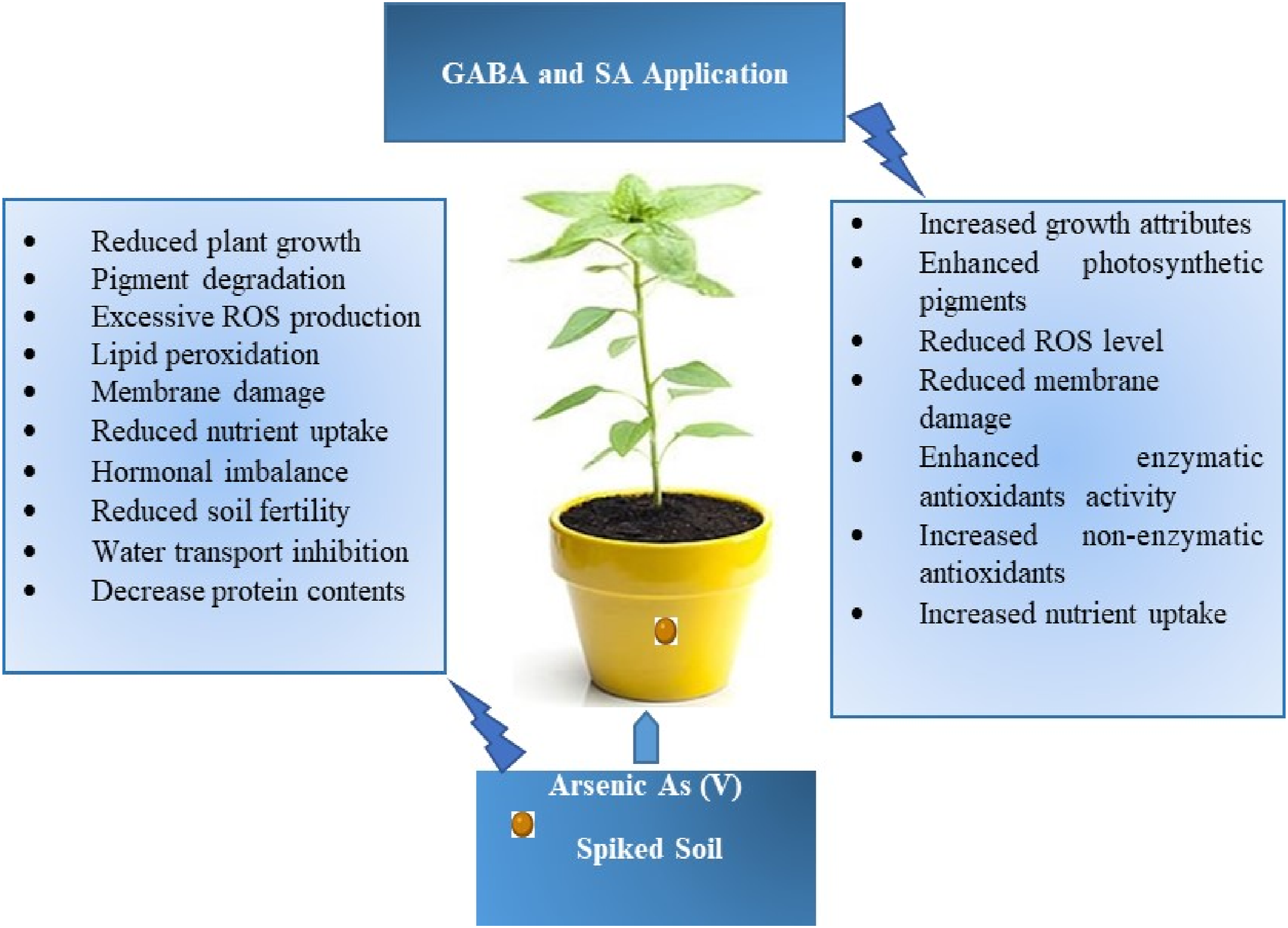
Highlights
• SA and GABA were used to enhance arsenic (As) tolerance in sunflower crops. • SA and GABA increased growth, photosynthesis, and nutrient uptake in As-stressed plants. • SA and GABA improved the antioxidant defense system and reduced ROS in plants under As stress. • SA and GABA influenced metabolic pathways and essential plant metabolites under As stress.
Novelty Statement
This study investigates the use of gamma-aminobutyric acid (GABA) and salicylic acid together to alleviate the effects of arsenic stress on sunflowers. This innovative approach reveals the complex mechanisms involved in the regulation mediated by SA and GABA, providing insights into their significant impact on growth, metabolite composition, and nutrient absorption in sunflower crops under challenging arsenic conditions. The specific functions of GABA and SA together in regulating plant defense responses to abiotic stresses, such as arsenic toxicity, are not well understood and require further research.
Introduction
The rising pollution levels in the environment are disrupting ecosystem biodiversity and threatening ecological balance. 1 Environmental stress factors negatively impacts plant growth, development, and productivity.2,3 Plants use defense mechanisms like antioxidant enzyme activity, hormonal signaling, and gene expression changes to cope with stress.4,5 However, prolonged stress can result in higher plant mortality. 6
Arsenic, a naturally occurring metalloid found in the earth’s crust, is widely distributed in the environment, from soil to groundwater. 7 The main sources of As in the environment are sulfide minerals like As-rich pyrite (FeS2) and arsenopyrite (FeAsS). 8 Arsenic enters farming systems through natural geochemical processes, 9 As-containing pesticides, mining operations, irrigation with As-contaminated groundwater, and fertilization with municipal solid waste. 10 It significantly impacts plant growth and development by disrupting biochemical processes and altering nutrient uptake. 11 Arsenic exposures can lead to health issues such as cancer, high blood pressure, arsenosis, and diabetes in humans. 12 In plants, As disrupts physiological, biochemical, and metabolic pathways, impairs nutrient absorption, negatively affects plant growth and the photosynthetic apparatus, disrupts plant water status, and interacts with enzyme functional groups.11,13 Additionally, As toxicity can result in oxidative damage to essential macromolecules such as lipids, proteins, carbohydrates, and DNA, ultimately leading to cell death.12,14 Plant cells have efficient enzymatic and non-enzymatic antioxidant activity to counteract oxidative stress. 15 Other stress markers, such as H2O2 levels, indicate the degree of damage and its prevention.
Arsenic compounds, such as arsenate (As5+) and arsenite (As3+), are highly toxic to humans and other organisms.16,17 Inorganic As species are more toxic than organic ones. 18 Arsenite binds to biological ligands with sulfur groups, leading to an increase in ROS and suppression of the antioxidant defense system, causing oxidative injury to cells. 19 Plants can absorb higher concentrations of As5+ than As3+, which can accumulate in plant tissue and be consumed by animals and humans. 11 Arsenate is the main form of As in aerobic soils and is similar to phosphate. Plants can absorb As5+ instead of phosphate, but they are unable to utilize it for their regular metabolic processes, resulting in adverse effects on their growth and development. 16 Arsenite is often found in anaerobic environments, where it can undergo various chemical reactions depending on ecological conditions. 20 In anaerobic environments, certain bacteria can convert As3+ to more toxic methylated As compounds, which can accumulate in aquatic organisms and humans.21,22 Arsenic in plants can disrupt their metabolism and growth, leading to iron deficiency. 13 Iron deficiency can reduce chlorophyll production, photosynthesis, and other physiological processes in plants. 13
Sunflower (
Salicylic acid (SA) is a beneficial compound for plant growth, enhancing resilience and productivity under stressful environmental conditions. 29 It also acts as a signal molecule in plant defense responses to various stresses. 30 Salicylic acid regulating photosynthesis and serving as a precursor for growth regulators and nitrogen metabolism. 31 SA also improves antioxidant defense, membrane stability, and osmoprotectants. 32 Additionally, SA detoxifies ROS by inducing gene expression in ROS scavenging and antioxidant defense mechanisms.33,34
Gamma-aminobutyric acid (GABA) is a 4-carbon amino acid present in animals, plants, and microorganisms.35,36 Research suggests that GABA plays a vital role in plant growth and development under abiotic stresses. 37 It acts as a signaling molecule to improve plant stress tolerance and defense responses. 38 GABA has been found to alleviate the adverse effects of As on plants by regulating gene expression and antioxidant activity.39,40 A recent study has found that GABA has the ability to enhance the detoxification and antioxidant functions in white clover. 41 Furthermore, nano materials of these plant growth regulators have the ability to regulate phytohormone levels, thereby altering plant growth and defensive mechanisms.3,42,43 However, research regarding the effects of SA and GABA supplementation on plant growth and As toxicity has been rare to date. Arsenic toxicity has emerged as a significant contaminant and threat to agriculture and environmental sustainability around the globe. Therefore, it is crucial to investigate the changes in plant metabolism and physiological indices in response to As contamination. Sunflower, an important oilseed crop, has the ability to tolerate various environmental conditions but is susceptible to damage from As toxicity. The underlying regulatory mechanisms that enable sunflower to tolerate As toxicity are not fully understood. This study aims to address this knowledge gap by examining the interactive roles of GABA and SA, two essential signaling molecules in plant defense responses. Therefore, the objective of present study to evaluate the morphological, physiological, biochemical markers and ion homeostasis for alleviating As toxicity in sunflower plants through exogenous application of SA and GABA. These findings may provide insights into how GABA and SA influence sunflowers under As toxicity.
Materials and Methods
Plant Material and Growth Conditions
Physio-Chemical Properties of Soil Used for Experiment.

Growth Attributes
The sunflower plants were removed from the pots, and the soil was gently shaken off the roots, followed by washing with distilled water and drying with a muslin cloth. The plants were then separated into root and shoot portions, and their height and root length were measured. The fresh weights of the roots and shoots were recorded, and then they were placed in an oven at 80°C until a constant weight was achieved to determine the dry weight.
Chlorophyll Pigments
The Lichtenthaler 47 procedure was employed to measure the photosynthetic pigments (Chl a, Chl b, and carotenoids) in sunflower leaves (1987) . A .25 g fresh leaf sample was ground in 80% acetone and then centrifuged at 4000 r/min for 10 minutes. Chlorophyll levels were determined by measuring absorbance at 480, 663, and 645 nm, with acetone serving as a blank, using a spectrophotometer.
Measurement of Relative Water Content
Relative water content (RWC) was measured following the protocol of González and González-Vilar.
48
The fresh weight (FW) of the leaves was recorded, and then the leaves were floated in distilled water to determine the turgid weight (TW). Subsequently, the leaves were dried in an oven at 70°C to obtain the dry weight (DW). The RWC was calculated using the formula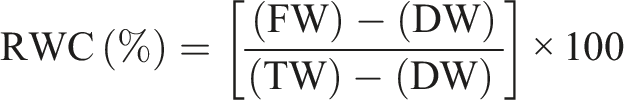
Determination of H2O2 and MDA Contents
Using the protocol of Velikova et al
49
the content of H2O2 in fresh leaf biomass was measured (2000) . Heath and Packer method was used to assess the MDA level (1968),
50
with the optical density of the mixture measured at 532 and 600 nm. Malondialdehyde contents were determined using an absorption coefficient of 155 000 nmol mol1.
Estimation of Ascorbic Acid
A .5 g leaf sample was ground in a 10 mL solution of 6% trichloroacetic acid. The homogenate was filtered, and 2 mL of the filtrate was mixed with 1 mL of 2% 2,4-dinitrophenylhydrazine and a drop of 10% thiourea in ethanol. The samples were then heated in a water bath at 95°C for 30 minutes, cooled in an ice bath, and 2.5 mL of 80% H2SO4 was added. The optical density (OD) of the samples was measured at 530 nm. 51
Total Soluble Protein Determination
The total soluble protein (TSP) of fresh leaf tissue (.5 g) was measured by homogenizing samples in 10 mL of 50 mM KP buffer (pH 7.5) using the protocol of Bradford. 52 The optical density (OD) of the supernatant was measured at 595 nm using a spectrophotometer. Bovine serum albumin was used as the standard protein.
Anthocyanin Content Measurement
The leaf tissue was homogenized in methanol with 1% HCl and then centrifuged to obtain the supernatant. The absorbance of the supernatant was measured at 530 nm and 657 nm using the method described by Hodges and Nozzolillo.
53
The content of anthocyanin was calculated using the following formula
Flavonoids Content Estimation
Zhishen et al 54 method was used to determine the flavonoid content in the leaf sample. A 0.5 g sample was homogenized in 10 mL of 80% aqueous ethanol. The resulting solution was mixed with 300 μL of NaNO2 and allowed to incubate at room temperature for 5 minutes. Afterward, 300 μL of AlCl3 and 2 mL of 1M NaOH were added. Finally, the absorbance was measured at 510 nm with the use of a spectrophotometer.
Phenolic Contents Measurement
The total phenolic content was determined using the method described by (1985). 55 Firstly, a .25 g fresh leaf sample was ground in 10 mL of 80% acetone and then centrifuged at 10 000 g for 10 minutes. The resulting supernatant was used for the reaction solution, which included 1 mL of Folin and Ciocalteau’s phenol reagent, 2 mL of distilled water, and 5 mL of 20% Na2CO3. The optical density (OD) of the reaction solution was measured at 765 nm using a spectrophotometer.
Cytosolutes Determination
Using the method of Bates et al 56 the free proline was estimated from leaf samples (1973). First, .25 g of fresh leaf material was ground in 80% ethanol. The resulting extract was then mixed with the anthrone reagent to determine the total amount of soluble sugars present using the method of Yemm and Willis. 57 The total free amino acids present in the .25 g leaf samples were analyzed by grinding them in KP buffer (pH 7.8) and following the procedure given by Hamilton and Van Slyke. 58
Enzymatic Antioxidants Assay
The antioxidant activity in 0.5 g fresh leaf samples was determined by homogenizing them in 10 mL of pre-chilled 50 mM tris buffer (pH 7.5). After that, centrifuge the mixture at 10 000 g for 20 minutes at 4°C. The resulting supernatant were used to estimate the activity of the following antioxidants: Using the method of Zhang et al the SOD activity was measured (2008),
59
recording the capacity of each unit to inhibit 50% photochemical reduction of nitro-blue tetrazolium chloride (NBT). Using the method of Upadhyaya et al.
60
POD activity was measured using the guaiacol method . The absorbance of the solution was noted at 470 nm on a spectrophotometer for POD activity. The Aebi
61
method was used to measure the
Determination of Arsenate Reductase (AR) Activity
Using the method of Ellis et al
62
the
Determination of Total As (V), As (III), and Elemental Uptake in Root and Leaf of Plants
The roots and leaves (1g) were ground and then digested in glass test tubes using an acid digestion procedure that involved 65% nitric acid (4 mL), 30% hydrogen peroxide (1 mL), and 45% hydrogen fluoride (3 mL). Once digested, the samples were diluted with dH2O2 up to 50 mL, filtered, and used to measure the levels of Fe, Zn, Mg, and ca The analysis was performed using an atomic absorption spectrophotometer (Hitachi, Z-2000, Tokyo, Japan) with a fuel-rich air-acetylene flame, a 10 cm burner head, .2-.7 nm slit width, and a 10-mA lamp current. The contents of As, Mg, Ca, Zn, and Fe were measured at 193.7, 285.2, 422.7, 213.9, and 248.3 nm, respectively. Using the method of Abdel-Lateef et al 63 arsenite content was determined with modified procedure (2013).
Statistical Analysis
The experiment followed a completely randomized design (CRD) with 3 replications for each treatment. Analysis of variance was conducted using Statistix software (Version 8.0). Differences among treatment means were compared using the Tukey test at
Results
Growth-Related Traits
Effect of Salicylic Acid (SA) and Gamma-Aminobutyric Acid (GABA) on Growth Characteristics in Two Sunflower Genotypes at the Flowering Stage Under Arsenic Stress.
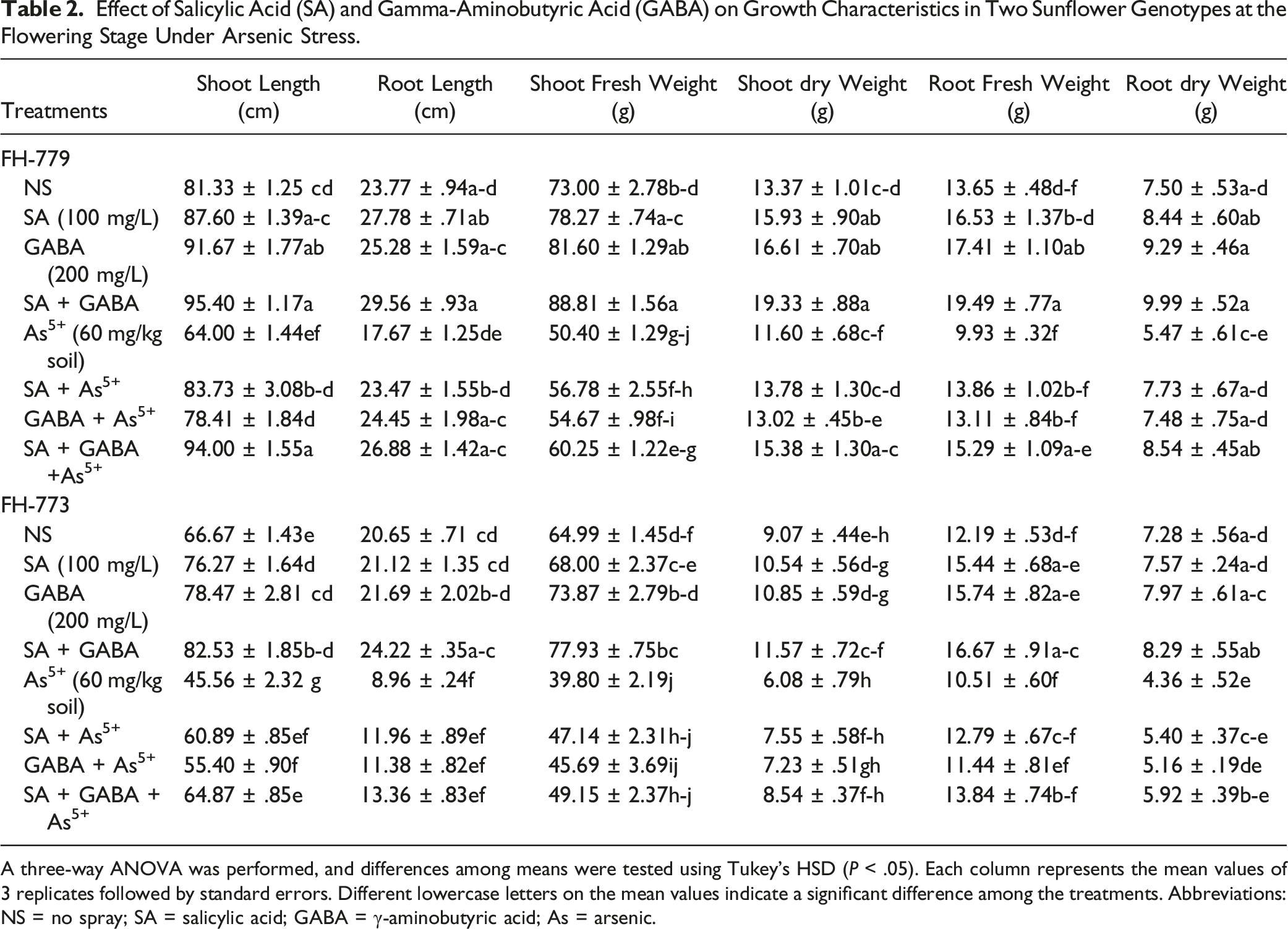
A three-way ANOVA was performed, and differences among means were tested using Tukey’s HSD (
Photosynthetic Pigments, and Relative Water Contents
Effect of SA and GABA on Physiological Characteristics in Two Sunflower Genotypes at the Flowering Stage Under Arsenic Stress.

Each column represents the mean values of 3 replicates with standard errors. A three-way ANOVA was performed, and differences among means were tested using Tukey’s HSD test (
Furthermore, As toxicity resulted in a significant decrease in the RWC of FH-779 by 31.92% and FH-773 by 23.44%. However, when exogenous SA, GABA, and a combination of both were applied, resulted in a notable improvement in the RWC of FH-779 (30.49%–43.87%) and FH-773 (17.02%–25.87%) under As toxicity (Table 3 and Table S1). Moreover, under control conditions, the exogenous application of SA, GABA, and their combination increased the levels of chlorophyll
Secondary Metabolites, Osmolytes and Ascorbic Acid Contents
Effect of SA and GABA on Metabolites, Osmolytes, and Ascorbic Acid Characteristics in Two Sunflower Genotypes at the Flowering Stage Under Arsenic Stress.

Each Column Represents the Mean Values of Three Replicates With Standard Errors. A Three-Way ANOVA was Performed, and Differences Among Means Were Tested Using Tukey’s HSD Test (
Under As stress, TFAA levels increased by 132.38% in FH-779 and by 62.18% in FH-773. Additionally, spraying SA, GABA, and SA + GABA significantly increased TFAA levels in FH-779 (18.42%-31.95%) and FH-773 (21.79%-36.59%) compared to unsprayed plants (Table 4 and Table S1).
The results indicate a notable decrease in TSP content under As toxicity in FH-779 (24.41%) and FH-773 (26.54%). However, supplementation with SA, GABA, and SA + GABA increased TSP levels in FH-779 (26.39% and 47.64%) and FH-773 (12.86% and 36.62%) compared to non-sprayed plants under As toxicity (Table 4 and Table S1).
A recent study found that the levels of ascorbic acid in the leaves of the FH-779 and FH-773 genotypes (60.51% and 83.01%) increased significantly under As stress. The increase in the content of ascorbic acid was higher in FH-773 compared to FH-779. Additionally, the application of SA, GABA, and a combination of SA + GABA increased the ascorbic acid content in FH-779 (4.72%–16.35%) and FH-773 (14.89%–26.14%) compared to unsprayed plants under As stress (Table 4 and Table S1).
Exposure to As also increased the phenolic content of FH-779 (84.38%) and FH-773 (42.48%). Spraying with SA, GABA, and a combination of SA + GABA enhanced phenolic levels in both FH-779 (33.38%-51.1%) and FH-773 (47.73%–78.68%) genotypes under As toxicity (Table 4 and Table S1).
Furthermore, the study showed that As toxicity significantly increased flavonoid content in both FH-779 (31.67%) and FH-773 (60.04%). Foliar spray with SA, GABA, and a combination of SA + GABA enhanced flavonoid content in both FH-779 (20.76%–47.51%) and FH-773 (13.45%–30.21%) under As stress compared to control plants (Table 4 and Table S1).
Our results show that under As toxicity, there was a significant increase in anthocyanin levels in FH-779 (40.10%) and FH-773 (70.41%). Additionally, the application of SA, GABA, and a combination of SA + GABA further increased anthocyanin levels in FH-779 (16.06%-38.95%) and FH-773 (21.59%-36.99%) compared to unsprayed plants (Table 4 and Table S1).
In control conditions, we observed a similar increasing trend in TSS (39.54%–71.49% and 22.91%–83.08%), TSP (17.03%–34.05% and 9.35%–25.59%), TFAA (15.13%–26.09% and 23.33%–41.11%), ascorbic acid (15.62%–25.42% and 10.96%–15.65%), total phenolic (44.65%–85.12% and 80.69%–112.41%), flavonoid (23.99%–48.61% and 41.19%–75.16%), and anthocyanin content (16.06%–38.95% and 21.60%–30.99%) in both FH-779 and FH-773 genotypes with the application of SA, GABA, and SA + GABA at the flowering stage. Importantly, the combined application of SA + GABA resulted in a significant increase in these parameters in both FH-779 and FH-773 genotypes (Table 4 and Table S1).
Oxidative Markers and Proline Content
Both FH-773 and FH-779 genotypes showed a significant increase in ROS levels, specifically H2O2 and MDA, under As stress. The increase in H2O2 levels was 103.15% and 265.01% in FH-779 and FH-773, respectively, while MDA levels increased by 16.09% and 98.87% in the same genotypes. However, when both genotypes were treated with SA, GABA, and a combination of SA + GABA, there was a decrease in MDA levels by 3.67%–10.46% and 6.75%–26.94%, respectively. Additionally, there was a reduction of 4.70%–26.21% and 9.00%–25.18% in H2O2 levels for FH-779 and FH-773, respectively. Similar reductions were observed for H2O2 (10.09%–31.11% and 19.49%–32.51%) and MDA levels (5.59%–12.02% and 2.03%–16.68%) in control plants of both FH-779 and FH-773 genotypes with SA, GABA, and combined SA + GABA during the flowering stage (Figure 1 and Table S1). Influence of GABA and SA spray on oxidative stress indicators and proline levels in sunflower at the flowering stage under arsenic stress. The bars graphs represent the mean values of 3 replicates followed by the standard error. Tukey’s HSD (
Under As stress, both FH-779 and FH-773 genotypes had an increased proline content. FH-779 showed a 178.93% increase, while FH-773 exhibited a 73.78% increase in proline content compared to the control. However, when treated with SA, GABA, and combined SA + GABA during the flowering stage, both genotypes showed an increased proline accumulation. FH-779 and FH-773 genotypes showed a 12.42%–43.08% and 19.24%–40.95% increase in proline content, respectively. Control plants of both genotypes also showed similar increases in proline content when treated with SA, GABA, and combined SA + GABA during the flowering stage (Figure 1 and Table S1).
Enzymatic Antioxidants
Our study revealed that exposure to As toxicity led to a significant increase in the activity of SOD in FH-779 (32.84%) and FH-773 (29.29%) plants, respectively. Applying foliar supplements of SA, GABA, and SA + GABA also led to a significant increase in SOD activity in both FH-779 (11.14%–23.69%) and FH-773 (10.99%–23.53%) under stress conditions compared to the control treatment. Moreover, FH-779 and FH-773 genotypes subjected to As toxicity showed a significant increase in POD activity, with increases of 55.09% and 40.70%, respectively, compared to control plants. Foliar application of SA, GABA, and SA + GABA intensified POD activity in both FH-779 (24.70%–39.05%) and FH-773 (32.59%–65.36%) relative to non-sprayed plants under As toxicity. This study also found that As toxicity caused a significant increase in Influence of GABA and SA spray on enzymatic antioxidants of sunflower at the flowering stage under arsenic stress. The bars graphs represent the mean values of 3 replicates followed by the standard error. Tukey’s HSD (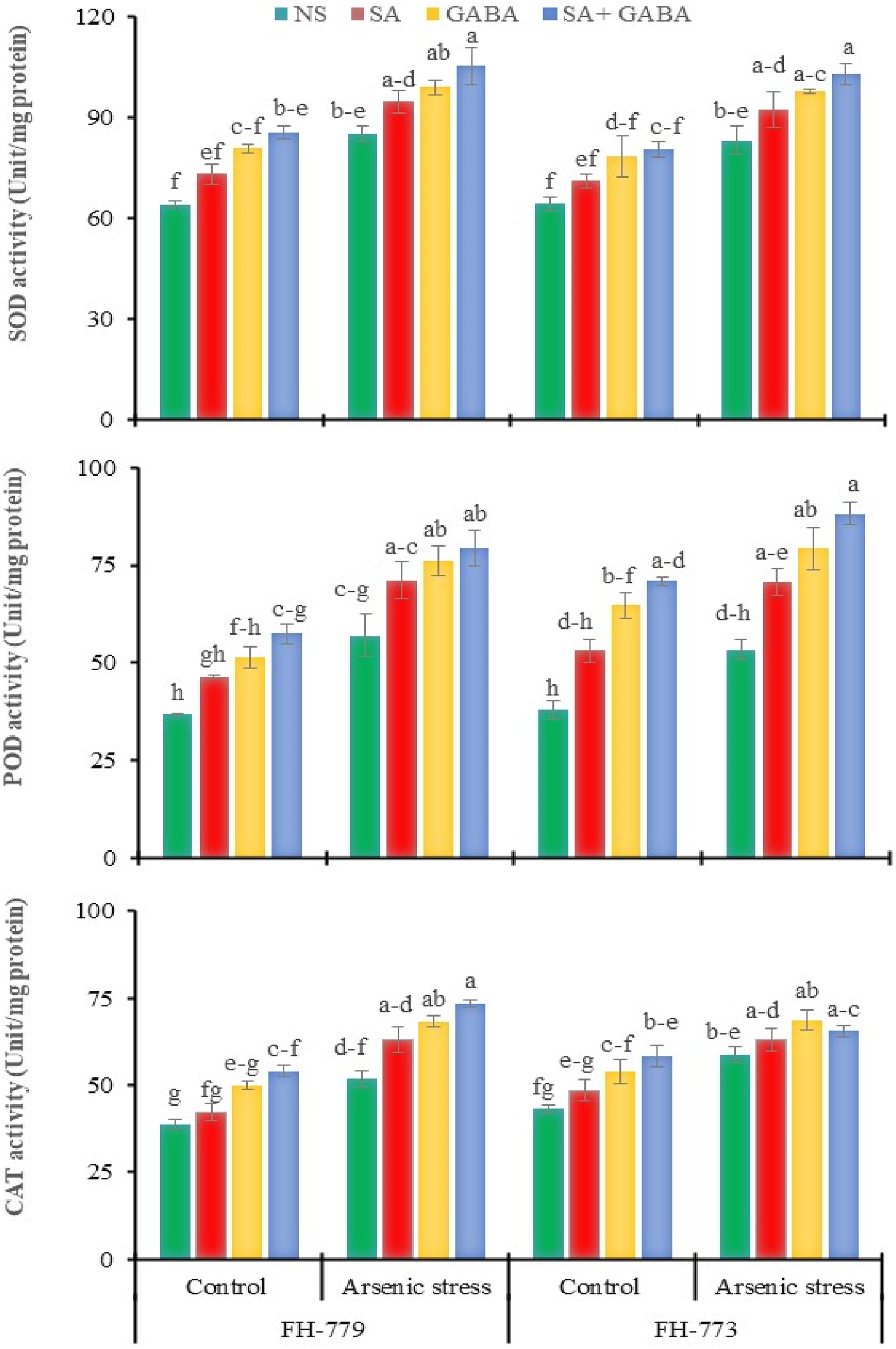
Additionally, FH-779 and FH-773 genotypes showed a similar increase in SOD (14.08%–33.30% and 10.64%–25.25%), POD (26.09%–56.49% and 40.11%–87.18%), and
Arsenate Reductase (AR) Activity
During the flowering stage, FH-779 and FH-773 genotypes showed an increase in ascorbate reductase ( Influence of GABA and SA spray on root and leaf arsenate activity of sunflower at the flowering stage under arsenic stress. The bar graphs represent the mean values of 3 replicates followed by the standard error. Tukey’s HSD (
As5+ and As3+ Uptake and Accumulation
FH-779 and FH-773 genotypes showed a significant increase in the accumulation of As5+ in their roots (516.55% and 632.95%, respectively) and leaves (557.52% and 617.47%, respectively) when exposed to As toxicity. However, when treated with SA, GABA, and SA + GABA, the As5+ content in their roots decreased by 24.53%–73.25% and 20.28%–74.04% and in their leaves by 33.60%–57.23% and 33.78%–55.66%. The same plant genotypes also exhibited an increase in As3+ accumulation in their roots (275.30% and 174.39%, respectively) and leaves (273.66% and 281.38%, respectively) under As toxicity. But, when treated with SA, GABA, and SA + GABA, the As3+ content in their roots decreased by 11.95%–55.22% and 7.62%–52.34% and in their leaves by 22.74%–40.68% and 13.30%–44.85%. The reduction of As5+ and As3+ levels followed the order: SA + GABA > SA > GABA in the roots and leaves of FH-779 and FH-773 during the flowering stage under both conditions (Figure 4 and Table S2). Influence of GABA and SA spray on total arsenic uptake in roots and leaves of sunflower at the flowering stage under arsenic stress. The line graphs represent the mean values of 3 replicates followed by the standard error. Tukey’s HSD (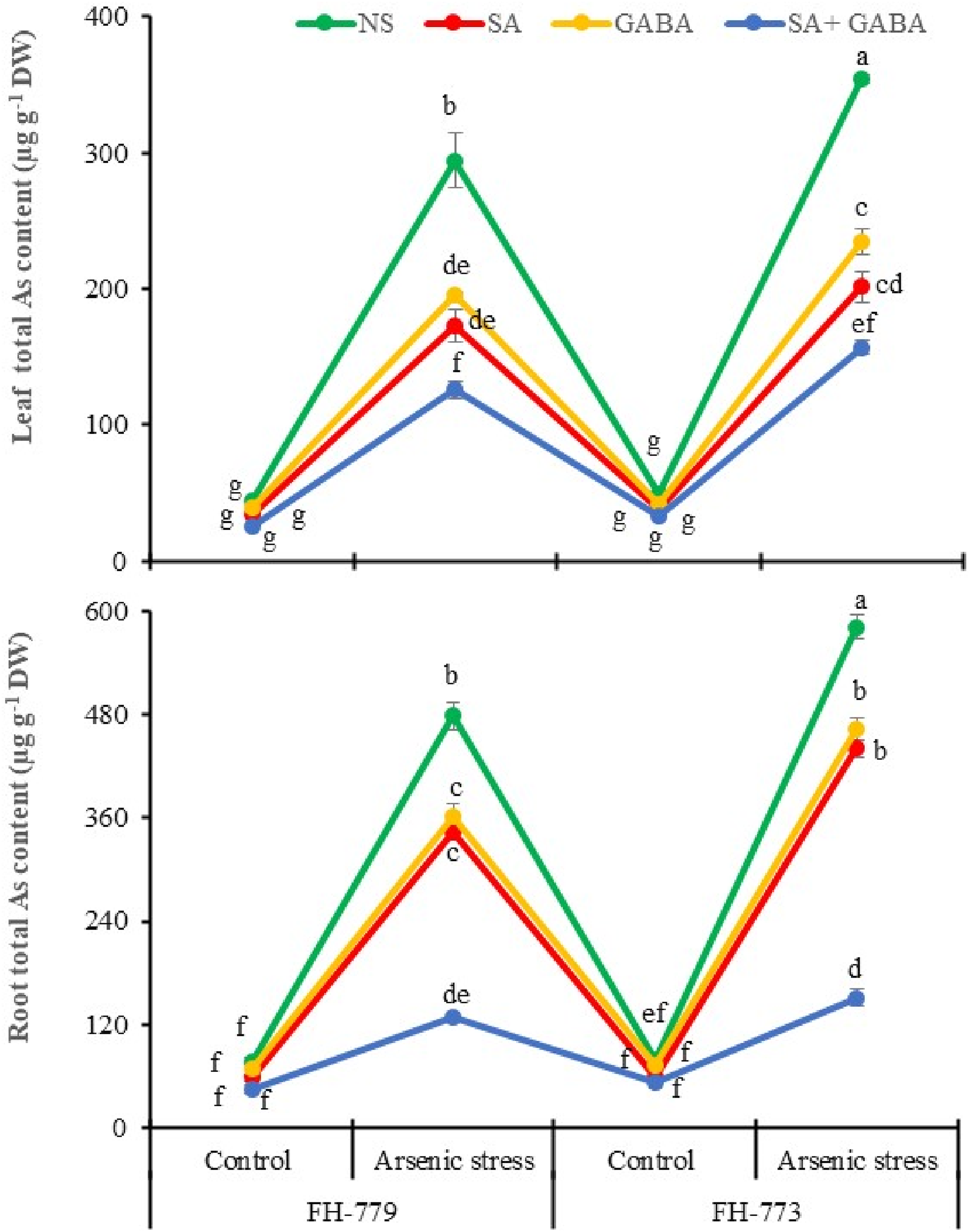
Nutrient Acquisition
Effect of Salicylic Acid (SA) and Gamma-Aminobutyric Acid (GABA) Application on Elemental Uptake in Two Sunflower Genotypes at the Flowering Stage Under Arsenic Stress.

Each column represents the mean values of 3 replicates with standard errors. A three-way ANOVA was performed, and differences among means were tested using Tukey’s honestly significant difference (HSD) test (
Similarly, there was a significant decrease in magnesium (Mg) content in the roots (5.84% and 8.17%) and leaves (10.61% and 14.18%) of FH-779 and FH-773 under As toxicity. However, spraying with SA, GABA, and SA + GABA led to a noticeable improvement in root (3.05%–9.25% and 2.25%–6.95%) and leaf Mg (8.67%–16.51% and 6.89%–13.03%) levels of FH-779 and FH-773 under As toxicity (Table 5 and Table S2).
A significant decrease in zinc (Zn) content was observed in As-stressed plants of FH-779 and FH-773, both in the roots (23.67% and 15.36%) and leaves (5.17% and 5.34%). However, treatment with SA, GABA, and SA + GABA substantially improved Zn content in the roots (8.65%–19.99% and 8.15%–16.58%) and leaves (2.27%–9.84% and 2.31%–12.53%) of FH-779 and FH-773 (Table 5 and Table S2).
Arsenic toxicity also resulted in a significant decrease in iron (Fe) content of FH-779 and FH-773 in the roots (16.42% and 17.51%) and leaves (20.68% and 24.22%). However, SA, GABA, and SA + GABA applications led to a considerable improvement in the root (5.33%–17.20% and 5.12%–10.17%) and leaf (5.57%–16.27% and .45%–14.36%) Fe content in FH-779 and FH-773 genotypes. Under both conditions, SA, GABA, and SA + GABA increased Ca, Mg, Zn, and Fe in roots and leaves in the following order: SA + GABA > GABA > SA in FH-779 and FH-773 (Table 5 and Table S2).
Pearson’s Correlation
Figure 5 A Pearson’s correlation graph was used to analyze the relationship between growth attributes and As uptake in sunflower plants treated with GABA and SA in As-contaminated soil, as shown in Figure 6. The concentration of As in the plants’ underground parts directly correlated with the As contents in the leaf and root tissues, as well as oxidative stress markers (H2O2 and MDA), osmolytes, and antioxidants. This relationship was also negatively associated with growth attributes, photosynthetic pigments, RWC, and nutrient acquisition (Ca, Mg, Zn, and Fe) in sunflowers. These results emphasize the importance of using SA, GABA spray, and combined SA + GABA to promote sunflower growth in As-contaminated soil. Influence of GABA and SA spray on As-III uptake in roots and leaves of sunflower at the flowering stage under arsenic stress. The line graphs represent the mean values of 3 replicates followed by the standard error. Tukey’s HSD ( The study investigated the correlation between various morpho-physiological and biochemical attributes of two sunflower genotypes (FH-779 and FH-773) grown in As-contaminated soil. Foliar sprays of GABA (200 mg/L) and SA (100 mg/L) were applied. Various abbreviations used in the figure are as follows: SOD, superoxide dismutase; POD, peroxidase; CAT, catalase; RAR, root arsenate activity; LAR, leaf arsenate activity; RTAs, root total arsenic content; LTAs, leaf total arsenic content; RAs.III, root arsenite content, LAs.III, leaf arsenite content; MDA, malondialdehyde; H2O2, hydrogen peroxide; TSS, soluble sugar; Chla, chlorophyll a; Chlb, chlorophyll b; TChl, total chlorophyll; Caro, carotenoid; Antho, anthocyanin; SL, shoot length; AsA, ascorbic acid; RWC, relative water content; RDW, root dry weight; RFW, root fresh weight; SDW, shoot dry weight; TSP, total soluble protein; TFAA, total free amino acids; RL, root length; SFW, shoot fresh weight; RCa, calcium in root; LCa, calcium in leaf; RMg, magnesium in root; LMg, magnesium in leaf; RZn, zinc in root; LZn, zinc in leaf; RFe, iron in root; LFe, iron in leaf.

Principal Component Analysis
The loading plots of PCA in Figure 7 reveal the effects of SA and GABA on As toxicity in sunflower. The most significant impact was observed in Dim1 and Dim2, which explained 84.2% of the total variance. Dim1 contributed 60%, and Dim2 contributed 24.2%. The attributes studied were distributed well in the database, indicating that As toxicity had a significant impact on the morphological and physio-chemical characteristics of sunflower genotypes. Negative correlations were found between As content in leaves and roots, oxidative markers (H2O2 and MDA), arsenate activity, and morphological attributes, chlorophyll pigments, RWC, and nutrient uptake (Ca, Mg, Zn, and Fe). On the other hand, positive associations were observed between phenolic, flavonoids, TSS, proline, anthocyanin, AsA content, antioxidants (SOD, POD, and CAT), and As concentrations in roots and leaves with the additional studied variables. The loading plots show the results of principal component analysis for two sunflower genotypes (FH-779 and FH-773) grown in sandy loamy soil with exogenous application of GABA and SA, with or without toxic concentrations of arsenic. Various abbreviations used in the figure are as follows: SOD, superoxide dismutase; POD, peroxidase; CAT, catalase; RAR, root arsenate activity; LAR, leaf arsenate activity; RTAs, root total arsenic content; LTAs, leaf total arsenic content; RAs.III, root arsenite content, LAs.III, leaf arsenite content; MDA, malondialdehyde; H2O2, hydrogen peroxide; TSS, soluble sugar; Chla, chlorophyll a; Chlb, chlorophyll b; TChl, total chlorophyll; Caro, carotenoid; Antho, anthocyanin; SL, shoot length; AsA, ascorbic acid; RWC, relative water content; RDW, root dry weight; RFW, root fresh weight; SDW, shoot dry weight; TSP, total soluble protein; TFAA, total free amino acids; RL, root length; SFW, shoot fresh weight; RCa, calcium in root; LCa, calcium in leaf; RMg, magnesium in root; LMg, magnesium in leaf; RZn, zinc in root; LZn, zinc in leaf; RFe, iron in root; LFe, iron in leaf.
Discussion
Arsenic, which is a non-essential element for plant metabolism, has become more common in soil due to human activities such as agriculture and industry.
64
It disrupts enzymes and inhibits plant growth, which leads to reduced photosynthesis, crop yield, nutrient uptake, and even plant death in severe cases.
11
The present study have shown that As negatively impacts sunflower growth and biomass production, especially in As-sensitive genotypes.
65
Arsenic accumulation in plants disturbs physiological processes, inhibiting growth and reducing biomass. Our study has demonstrated a significant decrease in shoot and root length, as well as fresh and dry mass under As treatment. The FH-779 genotype exhibited greater tolerance to As stress compared to the FH-773 genotype, possibly due to lower oxidative stress and higher photosynthetic activity (Table 2). Arsenic can interfere with cellular processes in various crops such as
It has been reported that As reduces the photosynthetic pigments in plants by increasing the activity of chlorophyll-degrading enzymes such as chlorophyllase and decreasing the activity of chlorophyll-synthesizing enzymes like δ-aminolaevulinic acid dehydrogenase. This leads to the build-up of ROS and oxidative stress, which can impact plant growth negatively. 72 In a recent study, it was found that the FH-773 genotype sunflowers showed a more significant reduction in chlorophyll levels under As treatment compared to the FH-779 genotype (Table 3). This difference in pigment levels may be attributed to As disrupting chloroplasts and inhibiting chlorophyll synthesis due to ROS production. 73 Similar effects of As stress on pigment reduction have been observed in rice 74 and spinach plants. 75 Under heavy metal stress, rice plants and other plants also show decreased chlorophyll content. 76 However, the application of SA and GABA has been found to help mitigate chlorophyll degradation in As-stressed plants (Table 3). Studies have shown that in lettuce plants under metal stress, the application of SA and GABA enhances photosynthetic activity and increases photosynthetic pigments, leading to an overall improvement in photosynthetic activity. 77
Metal toxicity can negatively impact the water relations in plants. This study found that As stress reduced the RWC in leaves, consistent with previous research by Ahmad et al.
78
This decrease in RWC may be due to increased oxidative stress and disrupted root growth from As toxicity, as shown by Vezza et al.
79
However, the application of SA and GABA spray improved RWC by potentially reducing oxidative stress and enhancing osmolyte uptake in As-exposed plants (Table 3). Similar results were seen in studies on
Malondialdehyde is a byproduct of lipid peroxidation resulting from oxidative stress induced by As toxicity.
85
FH-779 has been found to have lower levels of MDA, suggesting that it is better able to tolerate As stress. This is consistent with previous studies on
Plants have developed mechanisms to counteract harmful ROS that are generated during oxidative stress. These mechanisms include both enzymatic and non-enzymatic antioxidants that help to reduce oxidative damage in plants. 68 The present study has shown that, in response to As stress, plants increase the production of metabolites such as flavonoids, phenolics, anthocyanins, and ascorbic acid, which act as non-enzymatic antioxidants to scavenge ROS and protect plant cells (Table 4). The accumulation of these compounds significantly increases under As stress compared to control plants, which is consistent with previous research. 15 Furthermore, the application of SA and GABA leads to a notable increase in these antioxidant compounds, which may enhance their protective effect on plants. 87
Proline is an osmolyte in plants that plays a crucial role in reducing metal-induced toxicity.
88
It is a well-known osmoprotectant that maintains cell wall flexibility, optimal hydration levels, and protects plants from damage by ROS.
11
Recent study suggests that under As stress, the FH-779 and FH-773 genotypes accumulated more free proline, with FH-779 having higher levels than FH-773. FH-773 was more susceptible to As toxicity, leading to higher proline concentrations (Figure 1). However, excessive proline accumulation can be detrimental, as it may degrade specific proteins or result in the de novo synthesis of amino acids.
89
Similar results have been observed in other plants like
The results of this study showed a significant decrease in TSP content under As stress (Table 4), which is consistent with previous research on metal stress in plants. 11 This decline may be caused by As-induced oxidative stress, leading to protein degradation or increased protease enzyme activity. 14 On the other hand, SA and GABA increased TSP levels in As-exposed plants by enhancing the antioxidant system and reducing oxidative stress.29,87 Additionally, As stress increased the TSS and TFAA contents in both sunflower genotypes. The application of SA and GABA also elevated TSS and TFAA levels in As-stressed plants (Table 4). TSS accumulation protects cellular membranes from oxidative damage and maintains redox balance, while TFAA and TSS contribute to water uptake and cell maintenance. 94 Higher levels of TSS and TFAA indicate greater tolerance to metal stress in plants. 95
Plants have defense mechanisms to protect themselves from oxidative damage caused by ROS when they are exposed to As stress.
83
Antioxidative enzymes such as SOD,
In this experiment, we observed an increase in As and As III levels in various plant tissues following As exposure. The roots showed the highest levels, followed by the leaves (Figure 3). This increase is attributed to the enhanced uptake of As facilitated by Pi transporters in the roots, leading to a potential disruption of plant metabolism. The enzyme arsenate reductase transforms As V to As III. 85 Treatment with SA and GABA led to a reduction in total As and As III content, as well as in arsenate reductase activity in sunflower genotypes. Higher concentrations of SA and GABA resulted in decreased As accumulation in both leaves and roots of sunflower plants, indicating the protective role of these compounds in reducing As uptake. This reduction may be attributed to decreased As uptake or reduced arsenate reductase activity, as observed in rice, 97 and soybean. 78
Several studies have shown As has a negative impact on plant growth by causing disruptions in ionic balance, metabolism, and nutrient uptake. 78 Arsenic toxicity can lead to reduced photosynthesis, stomatal regulation, and transpiration rates in plant roots, affecting the uptake of water and ions.79,98 It can also impede nutrient absorption, leading to changes in nutrient concentration and accumulation. Some plants, such as sunflowers, have developed mechanisms to restrict the transfer of As to above-ground parts by absorbing it in their roots. 98 In soils containing organic acids, As movement is restricted as these acids bind to form insoluble compounds. The presence of As was found to decrease the levels of Mg, Ca, Zn, and Fe in sunflower roots and leaves (Table 5). A negative correlation was observed between the As content and growth characteristics, which was more pronounced in FH-773 than in FH-779 (Figure 7). Pearson’s correlation analysis revealed a positive correlation between As content and H2O2 and MDA levels (Figure 6). Certain substances such as SA, GABA, and their combination can enhance nutrient uptake, reduce As uptake, and decrease As accumulation in plants under arsenic toxicity.
Conclusion
The study on sunflower plants exposed to As toxicity found that it inhibits growth, biomass production, photosynthetic pigments, and protein content due to oxidative damage and reduced nutrient uptake. Two sunflower genotypes, FH-773 and FH-779, showed varying susceptibility to As toxicity, with FH-773 being more severely affected. However, the addition of SA, GABA, or their combination improved plant health by enhancing photosynthetic pigments, reducing As accumulation, regulating osmotic pressure, and minimizing oxidative damage, resulting in decreased As translocation and toxicity. SA and GABA application also boosted the accumulation of secondary metabolites, antioxidant systems, osmolytes, and nutrient uptake while reducing As accumulation in leaves and roots. Foliar application of SA and GABA could enhance biomass and yield in sunflowers under stress conditions. Further research in field settings is recommended to gain a better understanding of stress tolerance mechanisms. SA and GABA could be effective stress mitigation strategies for other crops facing abiotic stress in the future.
Supplemental Material
Supplemental Material - Salicylic Acid and Gemma-Aminobutyric Acid Mediated Regulation of Growth, Metabolites, Antioxidant Defense System and Nutrient Uptake in Sunflower (Helianthus annuus L.) Under Arsenic Stress
Supplemental Material for Salicylic Acid and Gemma-Aminobutyric Acid Mediated Regulation of Growth, Metabolites, Antioxidant Defense System and Nutrient Uptake in Sunflower (
Footnotes
Acknowledgments
This study is part of Muhammad Nawaz’s PhD dissertation. The authors would like to acknowledge the Government College University, Faisalabad, Pakistan for their support through GCUF-RSP (Project Code: 48-Bot-7).
Author Contributions
Iqbal Hussain and Mahmood-ur-Rahman: conceptualization, visualization. Iqbal Hussain: Writing-original draft, supervision, and methodology. Muhammad Nawaz: Investigation, methodology, visualization and writing-original draft. Muhammad Arslan Ashraf and Rizwan Rasheed: Review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
