Abstract
We investigated the effects of low dose rate radiation (LDR) on M1 and M2 macrophages in an ovalbumin-induced mouse model of allergic airway inflammation and asthma. After exposure to LDR (1 Gy, 1.818 mGy/h) for 24 days, mice were euthanized and the changes in the number of M1 and M2 macrophages in the bronchoalveolar lavage fluid and lung, and M2-associated cytokine levels, were assessed. LDR treatment not only restored the M2-rich microenvironment but also ameliorated asthma-related progression in a macrophage-dependent manner. In an ovalbumin-induced mouse model, LDR treatment significantly inhibited M2, but not M1, macrophage infiltration. M2-specific changes in macrophage polarization during chronic lung disease reversed the positive effects of LDR. Moreover, the levels of cytokines, including chemokine (C-C motif) ligand (CCL) 24, CCL17, transforming growth factor beta 1, and matrix metalloproteinase-9, decreased in ovalbumin-sensitized/challenged mice upon exposure to LDR. Collectively, our results indicate that LDR exposure suppressed asthmatic progression, including mucin accumulation, inflammation, and Type 2 T helper (Th2) cytokine (interleukin (IL)-4 and IL-13) production. In conclusion, LDR exposure decreased Th2 cytokine secretion in M2 macrophages, resulting in a reduction in eosinophilic inflammation in ovalbumin-sensitized/challenged mice.
Introduction
Asthma is a chronic inflammatory airway disease that is recognized as a major public health problem and characterized by airway inflammation and mucus secretion. 1 Asthma is considered an inflammatory disease induced by T-helper 2 (Th2) cells. The activated Th2 response induces secretion of interleukin (IL)-4 and IL-13. These Th2 cytokines play a role in the recruitment of effector cells, resulting in eosinophil infiltration and histamine secretion, which contribute to asthma progression. These allergic asthma pathophysiological factors are involved in several signaling pathways, including the inflammatory (NF-kBm MAPKs, MMPs, etc.), fibrotic (TGF-1β), oxidative (GSH, HO-1, NRF-2, etc.), and mucoidal (MUC5AC) signaling pathways. In particular, the innate immune system is recognized to be important for the development of asthma 2 ; however, its role has been considerably undervalued and largely unexplored. 3 Macrophages are a diverse and dynamic group of cells that can perform a wide range of important functions. 4 Furthermore, macrophages can be classified into two major functional subsets: classically activated macrophages (M1), characterized by antimicrobial and cytotoxic properties in response to lipopolysaccharide or interferon gamma stimulation, and alternatively activated macrophages (M2), which exhibit anti-inflammatory and regulatory properties via Th2-specific cytokines, such as IL-4 and IL-13. 5
Our previous study revealed that chronic exposure to low dose rate ionizing radiation (LDR) reduced airway inflammation in a mouse model of asthma. 6 Although the inflammatory process in asthma is dominated by Th2 inflammation, accumulating evidence supports the parallel development and association of M1 and M2 macrophages in this disease. 7 M2 cells are induced by the archetypical asthma cytokines IL-4 and IL-13 and have been studied extensively in this disease. 8 An increase in the number of cluster of differentiation (CD)206+ M2 cells was associated with more severe asthma. 9 In fact, these cells were found to be involved in disease progression in mouse models of allergic lung inflammation. However, dynamic changes in M2 macrophages during asthma have not been defined using animal models or patients.
In this study, we aimed to determine the effect of LDR on disease development using a murine model of ovalbumin (OVA)-induced asthma. Our findings indicate that LDR represses the M2-rich asthma microenvironment and relieves asthma-related progression in a macrophage-dependent manner.
Materials and Methods
Animals
Six-week-old female C57BL/6 mice (Central Lab. Animal Inc, Seoul, Korea) were used in the study after 1 week of quarantine and acclimation. Briefly, mice were housed at a temperature of 23 ± 2°C and relative humidity of 50 ± 5%, with artificial lighting from 08:00 to 20:00 and approximately 13–18 air changes per hour. Mice were provided ad libitum access to standard laboratory diet and water. All experimental procedures were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023, revised 1978) and followed a protocol approved by the Institutional Animal Care and Use Committee of the Dongnam Institute of Radiological and Medical Sciences (DIRAMS; Permit Number: DI-2015-002 & DI-2021-003).
Experimental Procedure
The mice were randomly divided into the following four groups (n = 6 per group): control, OVA, LDR, and LDR+OVA. The experimental procedure for OVA-induced asthma is summarized in Figure 1, and the methods were performed as previously described.
6
Experimental procedure for establishing the OVA-induced asthma model. (A) Low dose rate ionizing radiation (LDR) equipment at Dongnam Institute of Radiological and Medical Sciences. (B) Schematic of the irradiation and experimental procedures.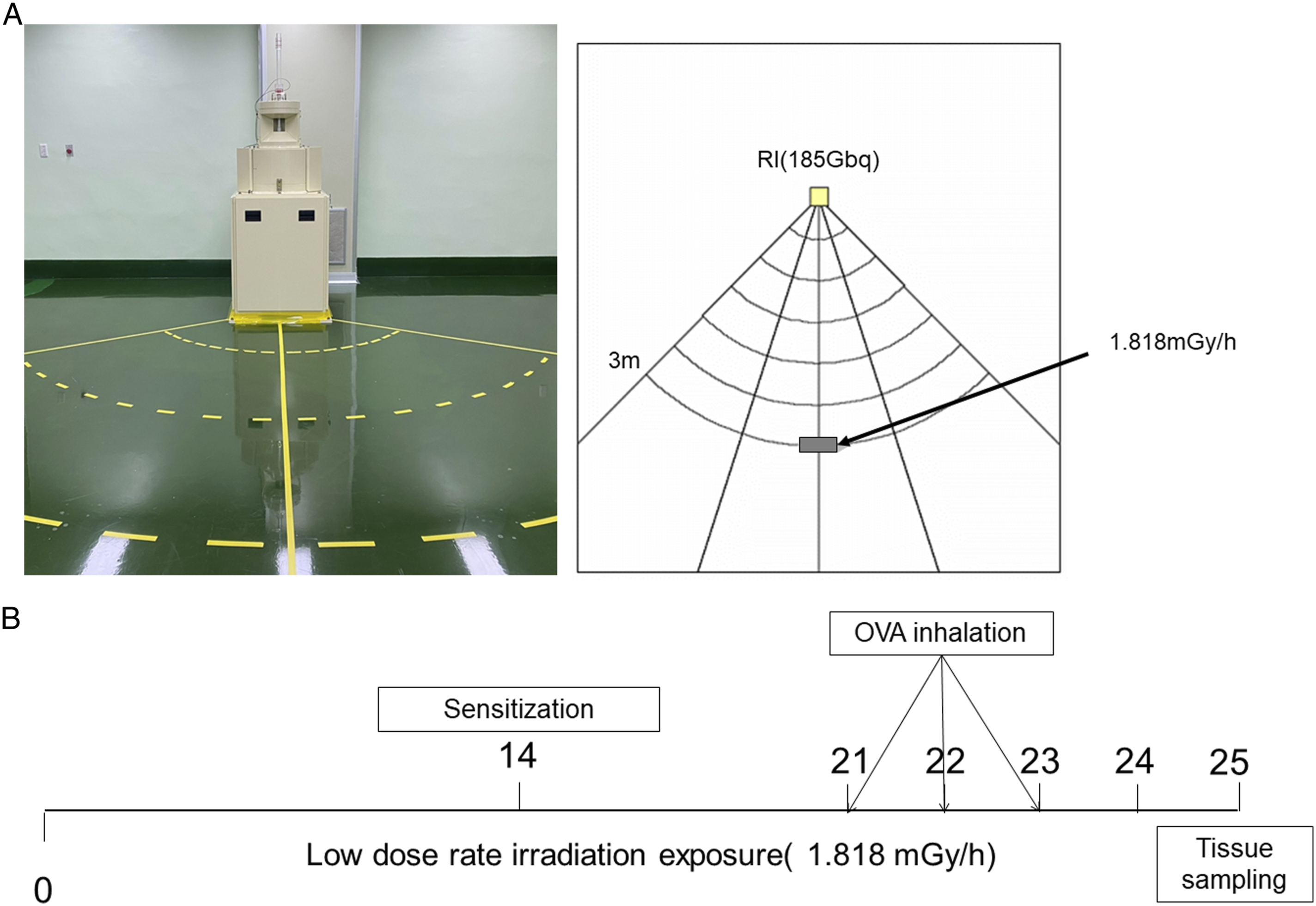
Briefly, mice were sensitized on days 0 and 14 via an intraperitoneal injection of 20 μg OVA (Sigma-Aldrich, Carlsbad, CA, USA) emulsified with 2 mg aluminum hydroxide in 200 μL phosphate-buffered saline (PBS) (pH 7.4). On days 21, 22, and 23, the mice received an airway challenge with OVA (1% (w/v)) for 1 hour using an ultrasonic nebulizer (NE-U12; Omron Corp, Tokyo, Japan). Radiation exposure was performed in a low dose rate irradiation facility at DIRAMS in a specific pathogen-free conditioned room equipped with a 137 Cs source (185 GBq). The mouse cages were placed on shelves located 3 m (1.818 mGy/h) from a source that provided either sham or 1 Gy exposure for 24 days. The animals were continuously exposed to radiation for 24 hours per day, except for 2 hours each week to allow for cleaning of the room, changing of the bedding, and replenishment of the food and water supply (Figure 1).
Animals were sacrificed 48 hours after the last challenge via intraperitoneal injection of tiletamine/zolazepam (Zoletil 50®; Virbac Korea, Seoul, Korea). Thereafter, tracheostomy was performed. Briefly, the trachea was cannulated and the left bronchi were tied for histological experiments. Bronchoalveolar lavage fluid (BALF) was obtained after instillation of ice-cold PBS (.5 mL) into the lungs; this procedure was repeated three times (total volume, 1.5 mL). BALF was centrifuged (200g, 10 minutes at 4°C), the supernatant was stored at -20°C to evaluate cytokines, and the cell pellet was used to determine the inflammatory cell count. Total inflammatory cell numbers were determined by counting the cells in ≤5 squares of a hemocytometer, following the exclusion of dead cells with trypan blue staining. Differential BALF cell counts were performed using Diff-Quik® staining reagent (IMEB Inc, San Marcos, CA, USA) according to the manufacturer’s instructions.
Fluorescence-Activated Cell Sorting Analysis
After blocking the Fc receptors, isolated BALF lung cells were washed and resuspended in PBS supplemented with 1% fetal bovine serum. Cells were stained with FITC-conjugated anti-CD206 (BioLegend, San Diego, CA, USA), PE-conjugated anti-CD11b, and PE-conjugated anti-major histocompatibility complex (MHC) class II (BD Biosciences, San Jose, CA, USA) for 30 minutes at 4°C.10,11 Flow cytometry was then performed using a Navious flow cytometer (Beckman Coulter, Miami, FL, USA) for quantification.
Measurement of Cytokine Levels in Bronchoalveolar Lavage Fluid Supernatant
The levels of IL-4 (DY404, R&D Systems, Minneapolis, MN, USA) and IL-13 (DY413, R&D Systems) in the BALF supernatant were determined using sandwich ELISA kits, according to the manufacturer’s protocols. Absorbance was measured at 450 nm after incubation for 10 minutes in the dark. ELISA was performed in triplicate.
Western Blot Analysis
The left lung tissue samples were solubilized in sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis sample buffer, and the protein concentration in each sample was determined using a Bio-Rad protein assay kit (Bio-Rad Laboratories, Hercules, CA, USA), with bovine serum albumin used as the standard. Equal amounts of total protein for each sample (70 μg protein per lane) were then separated using 10% SDS-polyacrylamide gel and electrophoretically transferred to an Immobilon-PSQ transfer membrane (Roche Diagnostics, IN, USA). The membrane was immediately placed in blocking solution (5% non-fat milk) at room temperature for 1 hour. The membrane was incubated with a diluted primary antibody specific for mucin-5 (ab24071, 1:1000; Abcam, Cambridge, UK), IL-4 (12227, 1:1000; Cell Signaling, Danvers, MA, USA), IL-13 (4730, 1:1000; Cell Signaling), chemokine (C-C motif) ligand 24 (CCL24) (NBP2-12109, 1:1000; Novus Biologicals), CCL17 (ab182793, 1:1000; Abcam), transforming growth factor beta 1 (TGF-β1) (ab92486, 1:1,000, Abcam), matrix metalloproteinase (MMP)-9 (4730, 1:1000; Cell Signaling), or β-actin (A5441, 1:10,000; Sigma-Aldrich) in TBS-T buffer (Tris-HCl-based buffer with .2% Tween 20, pH 7.5) at 4°C overnight. After washing four times with TBS-T buffer for 10 minutes, the membrane was incubated with secondary polyclonal anti-rabbit antibody or monoclonal anti-mouse antibody (1:10,000; Sigma-Aldrich) in TBS-T buffer at room temperature for 1 hour. Horseradish peroxidase-conjugated secondary antibody labeling was detected by enhanced chemiluminescence and exposure to a radiographic film. Pre-stained blue markers were used for molecular weight determination. The bands were quantified using Scion Image Beta 4.0.2 software for Windows XP (Scion, Frederick, ME, USA).
Histology
The BALF samples and left lung tissues were subjected to histopathological examination as described previously. 6 After BALF isolation, the left lung tissues were fixed with 4% (v/v) buffered formalin and embedded in paraffin wax. Lung tissue sections were prepared for hematoxylin-eosin and periodic acid-Schiff (PAS; IMEB, San Marcos, CA, USA) staining to estimate inflammation and mucus production, respectively. Images of lung sections were obtained using a digital camera mounted on a microscope (Nikon Eclipse 80i; Nikon Corporation, Tokyo, Japan). Quantification was performed using a previously described protocol for image analysis. 6
For immunohistochemistry, lung tissue sections were subjected to antigen retrieval with 1 M Tris-EDTA buffer for 15 minutes. The slides were blocked, incubated with anti-CCL24 (NB92-12109, 1:100; Novus) and anti-CCL17 (ab182793, 1:100; Abcam) antibodies, and assessed with a specific anti-rabbit secondary antibody (4730, 1:100; Abcam). Antigen-antibody complexes were visualized using an avidin-biotin-peroxidase complex kit (Elite kit, Vector Laboratories, Burlingame, CA, USA). The sections were counterstained with hematoxylin and mounted on coverslips for microscopic examination. For immunofluorescence, lung tissue sections were treated with 1 M Tris-EDTA buffer for 15 minutes for antigen retrieval. The sections were then blocked with 1% goat serum in TBS-T solution for 1 hour at room temperature, incubated with the corresponding CD11b antibody (NB600-1327, 1:100; Novus Biologicals) and the appropriate secondary antibody (Invitrogen, Carlsbad, CA), and counterstained with Hoechst 33342 (4ʹ,6-diamidino-2-phenylindole; Invitrogen). Slides were monitored and photographed using a Carl Zeiss LSM 700 confocal microscope and processed using ZEN 2009 (Carl Zeiss, Germany).
The isolated BALF cells were cytocentrifuged on slides, fixed with 4% (v/v) buffered formalin, permeabilized on ice for 10 minutes using .2% Triton X-100 in PBS, and washed with PBS. Thereafter, the sections were treated with the corresponding CD11b antibody (NB600-1327, 1:100; Novus Biologicals) and the appropriate secondary antibody (Vector) and then incubated with 4 μg/mL Hoechst 33342 at room temperature for 15 minutes. All sections were mounted with .05 mL PBS containing 10% glycerol (Wako, Osaka, Japan), analyzed using Carl Zeiss LSM 700 confocal microscope, and processed using ZEN 2009 (Carl Zeiss).
Isolation of Bronchoalveolar Lavage Fluid and Lung Cells
Bronchoalveolar lavage fluid samples were prepared as described previously. 6 The total number of inflammatory cells was determined by counting the cells using at least 5 squares of a hemocytometer following the exclusion of dead cells identified by trypan blue staining. Differential cell counts in the BALF were quantified using the Diff-Quik staining reagent (B4132-1A; IMEB, CA, USA), according to the manufacturer’s instructions. The number of inflammatory cells, including macrophages and eosinophils, was calculated by multiplying the percentage obtained with the total yield. Images of the slides were analyzed using a digital camera mounted on a microscope (Nikon Eclipse 80i; Nikon Corporation, Tokyo, Japan).
Statistical Analysis
Data are expressed as mean ± standard error of the mean (SEM). The normality distribution of quantitative variables was tested using the Shapiro–Wilk test, and all subgroups were tested to determine the homogeneity of variances. Statistical significance was determined using one-way ANOVA followed by Tukey’s post hoc test. Comparative analytical graphs were obtained using the Statistical Package for the Social Sciences (SPSS) statistical software for Windows (version 18.0; SPSS Inc, Chicago, USA). Statistical significance was set at P < .05.
Results
Low Dose Rate Radiation Exposure Decreases the Number of M2 Macrophages in the Bronchoalveolar Lavage Fluid of Asthmatic Mice
The pan-macrophagic CD11b marker is expressed in both macrophages and CD11b+ dendritic cells.10,12 However, consistent with previous literature, common markers used to identify M1 and M2 macrophages in the BALF or mouse lung are MHC class II
13
and CD206,11,13,14 respectively. The number of MHC class II/CD11b+ macrophages and CD206+CD11b+ macrophages was markedly increased in the BALF of OVA-challenged mice compared to that of normal controls (control M1: 6.75 ± .54%, OVA M1: 44.37 ± 1.57%, P < .001, control M2: 2.58 ±.47%, OVA M2: 34.95 ± 1.85%, P < .001; Figure 2B), despite no statistically significant difference in the BALF between the control and LDR groups. LDR exposure did not affect the number of MHC class II/CD11b+ macrophages (LDR+OVA M1: 41.20 ± 1.52%; Figure 2B) in LDR-treated mice compared with that found in OVA-challenged mice; however, such exposure significantly decreased the number of CD206+CD11b+ macrophages in the LDR-treated mice compared with that found in the OVA-challenged mice (LDR+OVA M2: 7.60 ± .54%, P < .001; Figure 2B). By confirming the presence of CD206+CD11b+-positive BALF macrophages, LDR exposure was found to selectively deplete M2 macrophages in the BALF of OVA-challenged mice (Figure 2C). Effect of low dose rate ionizing radiation (LDR) on M1 and M2 macrophages in the bronchoalveolar lavage fluid (BALF) of mice with asthma. FACS (A, B) and immunofluorescence analysis of MHC class II/CD11b+ and CD206+CD11b+ cells (C) in BALF samples. A significant increase in the number of MHC class II/CD11b+ and CD206+CD11b+ cells was observed in the BALF of ovalbumin (OVA)-sensitized/challenged mice. However, exposure to LDR significantly decreased the number of CD206+CD11b+ macrophages. Data are presented as mean ± SE (n = 6 per group); *P < .05 vs OVA-sensitized/challenged mice.
Increased CD206 expression and a consequent increase in M2 macrophages were found in the lung samples of OVA-sensitized/challenged mice (control M1: 6.53 ± .68%, OVA M1: 31.67 ± 1.61%, P < .001, control M2: 4.73 ± .39%, OVA M2: 31.80 ± 2.37%, P < .001; Figure 3B). LDR exposure did not affect the number of MHC class II/CD11b+ macrophages in LDR-treated mice compared with that found in OVA-challenged mice (LDR+OVA M1: 27.07 ± 1.35%; Figure 3B); however, such exposure significantly decreased the number of CD206+CD11b+ macrophages in the LDR-treated mice compared with that found in OVA-challenged mice (LDR+OVA M2: 5.53 ± .38%, P < .001; Figure 3B). By confirming the presence of CD206+CD11b+-positive lung macrophages, LDR exposure was revealed to selectively deplete M2 macrophages in the lung (Figure 3C); however, there was no statistically significant difference in CD206 expression between the control and LDR groups. These responses were consistent with the results obtained for lung tissues (Figure 3C). Altogether, LDR exposure selectively depleted M2 macrophages in lung tissue samples. Effect of irradiation (IR) on M1 and M2 macrophages in the lungs of asthmatic mice. FACS (A, B) and immunofluorescence analysis of MHC class II/CD11b+ and CD206+CD11b+ cells (C) in the lung tissue samples. A significant increase in the number of MHC class II/CD11b+ and CD206+CD11b+ cells was observed in the lungs of ovalbumin (OVA)-sensitized/challenged mice. However, exposure to low dose rate ionizing radiation (LDR) significantly decreased the number of CD206+CD11b+ macrophages. Data are expressed as mean ± SE (n = 6 per group); *P < .05 vs OVA-sensitized/challenged mice.
Low Dose Rate Radiation Exposure Decreases M2 Macrophage Chemokine Levels in the Lungs of Asthmatic Mice
M2 macrophages express several chemokines, including CCL24 and CCL17. The levels of these molecules were found to be significantly higher in OVA-sensitized/challenged mice than in normal controls (control CCL24: 92.36 ± 5.51%, OVA CCL24: 386.25 ± 65.84%, P < .01; Figure 4B, control CCL17: 100.91 ± 2.19%, OVA CCL17: 235.18 ± 27.13%, P < .001; Figure 4C). Furthermore, CCL24 and CCL17 levels were significantly lower in LDR-irradiated mice than in OVA-sensitized/challenged mice (LDR+OVA CCL24: 173.85 ± 8.01%, P < .01; Figure 4B, LDR+OVA CCL17: 160.29 ± 19.76%, P < .05; Figure 4C); however, there was no statistically significant difference in CCL24 and CCL17 levels between the control and LDR groups. This change was confirmed by histological examination, which indicated that LDR markedly decreased the expression of CCL24 and CCL17 in irradiated mice relative to OVA-sensitized/challenged mice, despite no statistically significant difference in expression between the control and LDR groups. Effect of irradiation (IR) on CCL24, CCL17, TGF-β1, and MMP-9 levels in the lungs of asthmatic mice. (A-C), Western blot analysis of CCL24 and CCL17 in lung tissues. Immunohistochemical analysis of CCL24 (D) and CCL17 (E) in lung tissues. CCL24 and CCL17 levels were significantly increased in the lung tissues of ovalbumin (OVA)-sensitized/challenged mice. Exposure to low dose rate ionizing radiation (LDR) resulted in a marked reduction in CCL24 and CCL17 levels in the lung tissues of OVA-sensitized/challenged mice. (F–H) Western blot analysis of TGF-β1 and MMP-9. A significant increase in TGF-β1 and MMP-9 was observed in the lung tissues of OVA-sensitized/challenged mice. LDR resulted in a marked reduction in TGF-β1 and MMP-9 levels in the lung tissues. Data are presented as mean ± SE (n = 6 per group); *P < .05 vs OVA-sensitized/challenged mice.
Ovalbumin sensitization/challenge resulted in markedly increased levels of TGF-β1 and MMP-9 in the challenged mice relative to those in normal controls (control TGF-β1: 28.32 ± 2.23%, OVA TGF-β1: 51.68 ± 3.07%, P < .001; Figure 4G, control MMP-9: 107.21 ± 7.57%, OVA MMP-9: 260.55 ± 21.58%, P < .001; Figure 4H); however, these levels had a non-significant tendency to be decreased in LDR-irradiated mice compared with OVA-sensitized/challenged mice (LDR+OVA TGF-β1: 43.04 ± 1.54%, P = .156; Figure 4G, LDR+OVA MMP-9: 217.51 ± 22.36%, P = .317; Figure 4H); no statistically significant difference was found in TGF-β1 and MMP-9 levels between the control and LDR groups.
Low Dose Rate Radiation Exposure Decreases the Levels of Th2 Cytokines (IL-4 and IL-13) in Bronchoalveolar Lavage Fluid Supernatants and Lung Tissues of Asthmatic Mice
The levels of IL-4 and IL-13 were significantly higher in the BALF supernatant of OVA-challenged mice than in normal controls (control IL-4: 24.13 ± 3.67 pg/mL, OVA IL-4: 39.31 ± 5.08 pg/mL, P < .05; Figure 5A, control IL-13: 51.48 ± 4.42 pg/mL, OVA IL-13: 113.85 ± 22.57 pg/mL, P < .05; Figure 5B). A non-significant reduction in IL-4 and IL-13 levels was also observed in irradiated mice compared to OVA-sensitized/challenged mice (LDR+OVA IL-4: 33.40 ± 3.17 pg/mL, P = .692; Figure 5A, LDR+OVA IL-13: 69.22 ± 14.52 pg/mL, P = .136; Figure 5B), despite no statistically significant difference between the control and IR groups in IL-4 and IL-13 levels. Similar to these results, a significant elevation in IL-4 and IL-13 expression was observed in the lung tissues of OVA-sensitized/challenged mice compared to normal controls based on western blotting (control IL-4: 94.79 ± 6.38%, OVA IL-4: 252.10 ± 29.39%, P < .001; Figure 5D, control IL-13: 94.38 ± 5.57%, OVA IL-13: 168.71 ± 24.73%, P < .05; Figure 5E). Furthermore, western blotting revealed a substantial reduction in IL-4 levels in irradiated mice compared with OVA-sensitized/challenged mice (LDR+OVA IL-4: 163.63 ± 21.79%, P < .05; Figure 5D); however, a non-significant tendency for a decrease in IL-13 levels was found between irradiated mice and OVA-sensitized/challenged mice (LDR+OVA IL-13: 137.71 ± 19.12%, P = .534; Figure 5E). Effect of irradiation on IL-4 and IL-13 levels in the bronchoalveolar lavage fluid (BALF) and lung in a mouse model of asthma. (A–B) ELISA results for IL-4 and IL-13 levels in the BALF samples. (C–E) Western blot analysis of IL-4 and IL-13 in the lung tissue samples. A significant increase in IL-4 and IL-13 levels was observed in the BALF of OVA-sensitized/challenged mice. Exposure to low dose rate ionizing radiation (LDR) resulted in a significant reduction in IL-4 and IL-13 levels in exposed mice compared with OVA-sensitized/challenged mice. Western blotting for IL-4 and IL-13 expression and relative expression ratios (compared with β-actin expression). IL-4 and IL-13 levels were significantly increased in the lung tissues of ovalbumin (OVA)-sensitized/challenged mice, based on western blot analysis. LDR resulted in a marked reduction in IL-4 and IL-13 levels in the lung tissues of OVA-sensitized/challenged mice. Data are presented as mean ± SE (n = 6 per group); *P < .05 vs OVA-sensitized/challenged mice.
Low Dose Rate Radiation Exposure Decreases Macrophage Recruitment to the Bronchoalveolar Lavage Fluid and Lung Tissues in Asthmatic Mice
Mucus production in the bronchial airways was evaluated based on the PAS staining intensity of lung tissue sections from OVA-sensitized/challenged mice. Mucus production was lower in the lung tissues of LDR-exposed mice than in those of OVA-sensitized/challenged mice (Figure 6A). These findings were similar to those obtained via western blotting: mucin-5 expression was increased in the lung tissues of OVA-sensitized/challenged mice (control: 32.60 ± 5.90%, OVA: 106.88 ± 11.19%, P < .001; Figure 6B) but was significantly decreased in irradiated mice compared with OVA-sensitized/challenged mice (LDR+OVA: 72.87 ± 6.88%, P < .05; Figure 6B); there was no statistically significant difference in mucin-5 expression between the control and LDR groups. Effect of irradiation on lung inflammation in a mouse model of asthma. (A) Histological analysis of mucus production in lung tissues stained with periodic acid-Schiff. B, Western blot for the detection of mucin-5 expression and relative expression ratio (compared with β-actin expression). Exposure to low dose rate ionizing radiation (LDR) resulted in a marked reduction in mucus production and mucin-5 expression in the lung tissues. (C–D) Histological examination and quantitative analysis of airway inflammation after staining with hematoxylin-eosin. LDR reduced airway inflammation in the lung tissues. Data are expressed as mean ± SE (n = 6 per group); *P < .05 vs OVA-sensitized/challenged mice.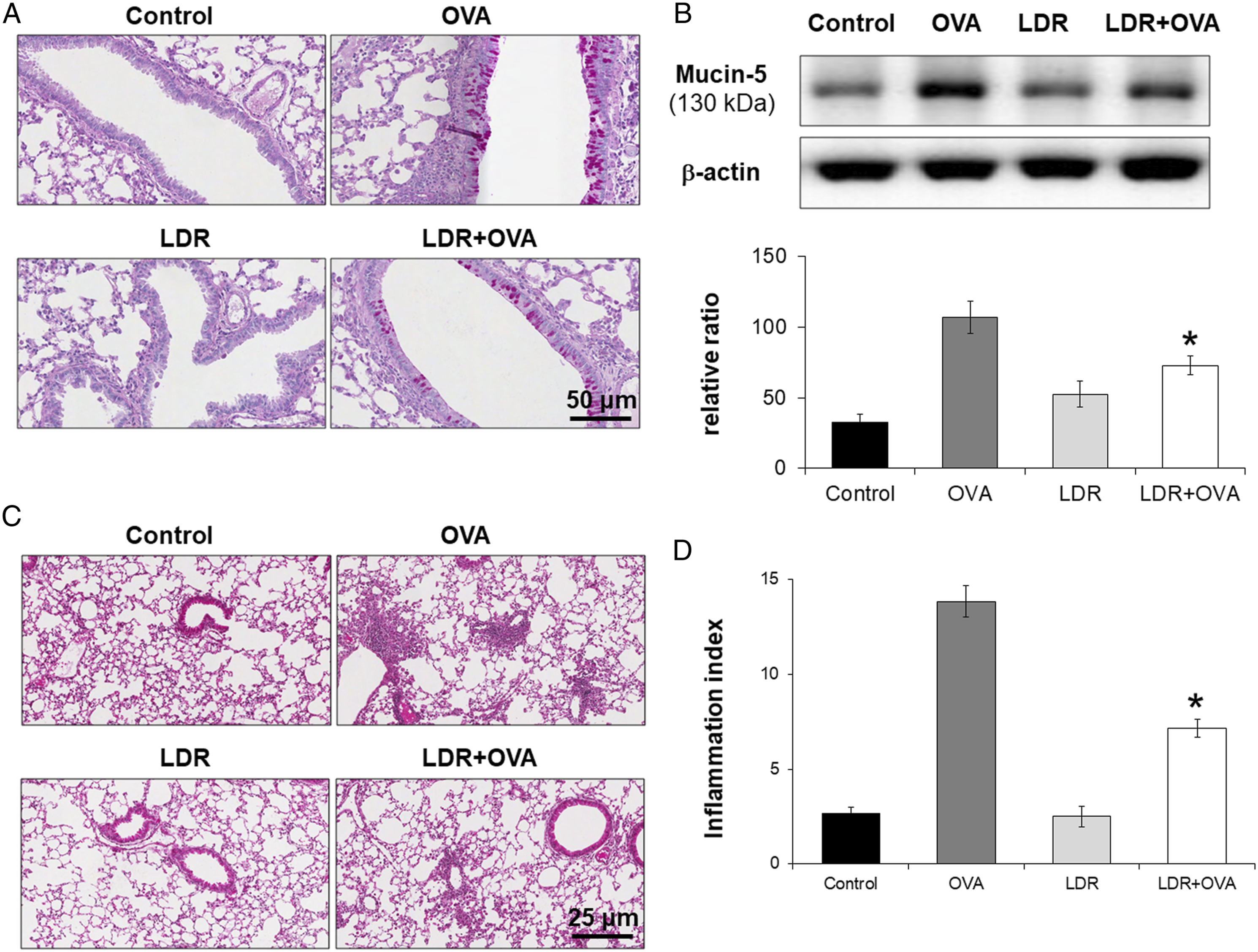
Inflammatory cells infiltrated the peribronchial and perivascular lesions of OVA-sensitized/challenged mice (control: 2.67 ± .33, OVA: 13.83 ± .83, P < .001; Figure 6C and D). However, after LDR exposure, cell infiltration in the exposed mice was lower than that in OVA-sensitized/challenged mice (LDR+OVA: 7.17 ± .48, P < .001; Figure 6D). Notably, there was no significant decrease in cell infiltration between the control and LDR groups.
The number of inflammatory cells was significantly higher in the BALF of OVA-sensitized/challenged mice than that of normal controls (control total: 301.67 ± 20.56 104 cells, OVA total: 753.33 ± 60.59 104 cells, P < .001; Figure 7A, control macrophage: 31.67 ± 14.81 103 cells, OVA macrophage: 105.83 ± 22.89 103 cells, P < .05; Figure 7B, control eosinophil: 15.00 ± 10.08 104 cells, OVA eosinophil: 130.00 ± 16.98 104 cells, P < .001; Figure 7C). However, the total number of cells, including macrophages and eosinophils, in the BALF of irradiated mice was lower than that in OVA-sensitized/challenged mice (LDR+OVA total: 515.00 ± 44.93 104 cells, P < .05; Figure 7A, LDR+OVA macrophage: 39.17 ± 11.93 103 cells, P < .05; Figure 7B, LDR+OVA eosinophil: 85.00 ± 11.03 104 cells, P < .05; Figure 7C). There was no significant difference in the number of neutrophils and lymphocytes in the BALF between the irradiated and OVA-sensitized/challenged mice. Effect of irradiation on inflammatory cells in the bronchoalveolar lavage fluid (BALF) in control mice and in low dose rate ionizing irradiated (LDR) mice with asthma. Inflammatory cells in the BALF decreased after LDR-irradiation in ovalbumin (OVA)-sensitized/challenged mice. (A) Total cell count. (B) Macrophage count. (C) Eosinophil count. (D) Neutrophil count. (E) Lymphocyte count. Data are expressed as mean ± SE (n = 6 per group); *P < .05 vs OVA-sensitized/challenged mice.
Discussion
Previously, we demonstrated that chronic LDR decreased OVA-induced asthma without causing other adverse effects. LDR exposure was found to suppress airway inflammation and mucus production in the lung tissues of OVA-sensitized/challenged mice by decreasing MMP-9 expression. 6 In this study, we confirmed the attenuation of OVA-induced asthma after LDR treatment. LDR not only decreased M2 macrophages in the microenvironment but also alleviated asthma-related progression in a macrophage-dependent manner.
Asthma is an inflammatory condition of the airway characterized by reversible airway obstruction, increased mucus production, and eosinophil infiltration caused by exacerbated responses to inhaled allergens. 15 In the present study, the number of inflammatory cells, cytokine release, and OVA-specific IgE levels were increased in OVA-induced asthmatic mice. However, LDR-irradiation significantly reduced the number of inflammatory cells, including eosinophils, macrophages, and neutrophils, and decreased the levels of IL-4, IL-5, and OVA-specific IgE in OVA-induced asthmatic mice. Histological changes in the lung tissue were consistent with these findings as OVA-induced asthmatic mice had an increased number of inflammatory cells in the lung, whereas LDR-treated mice had significantly reduced inflammatory responses. In a previous study, fractionated irradiation at 5 Gy aggravated the progressions of asthma, 16 whereas 2 Gy irradiation ameliorated this condition.17,18 The gap between these previous reports and the present findings could be attributed to differences in the irradiation dosage and dose rate. Furthermore, 1 Gy irradiation, which is considered an acute high-dose irradiation dose, might cause myelotoxocity 6 ; however, the dose and dose rate used in this study were not found to reduce peripheral blood counts. Previous research has revealed that 1 Gy irradiation has no toxic effects on various organs, except the reproductive organs.19,20 Existing studies on irradiation toxicology provide strong evidence that low dose irradiation does not promote the development of asthma.
In this study, LDR reduced the number of M2 macrophages in a murine model of asthma, thereby preventing allergic airway inflammation. Although the negative effects of radiation exposure on other organs are unknown, the findings of this study indicate that LDR does not have a negative impact on asthma but instead may have a positive impact.
Asthma is an inflammatory disorder characterized by airway narrowing, increased influx of inflammatory cells to the lungs, and excessive production of signature cytokines released by Th2 cells. 21 In this study, LDR exposure decreased the Th2 response, including the expression of the cytokines IL-4 and IL-13. These molecules are abundantly present in the lungs of asthmatic individuals, indicating an association between M2 macrophages and asthma. A previous study reported increased IL-13 content in the BALF of asthmatic patients, produced from M2 macrophages. 22 An increase in M2 macrophages is positively correlated with the severity of asthma progression in murine models. 23 These clinical and animal study findings indicate a correlation between asthma and M2 cells. However, only a few studies have investigated the role of M2 macrophages in asthma. In the presence of Th2 cytokines, including IL-4 and IL-13, these cells produce type 2 cytokines that play key roles in allergic responses, including asthma. 18
Lung macrophages are associated with an innate adaptive immune response to allergic inflammation. M1 macrophages augment tissue damage, whereas M2 cells discard necrotic fibers and deliver survival signals to myogenic precursors, supporting healing. M2 macrophages have been confirmed to play an important role in the pathogenesis of asthma by promoting cellular proliferation, tissue recovery, and angiogenesis.16,24,25 Alveolar macrophages are the most abundant resident immune effector cells in the airspace and may play a role in the pathogenesis of asthmatic inflammation. 26
Following radiation exposure in OVA-sensitized/challenged mice, a decrease in CD206+ lung macrophages was observed. In asthmatic animals, M2 macrophages are characterized by high levels of cell surface receptors such as CD206. This marker has an important function in phagocytosis. 27 In this study, an increase in M1 and M2 cells was confirmed in OVA-sensitized/challenged mice. Radiation-induced macrophage depletion was found to be mainly caused by the inhibition of M2 and not M1 cells. A previous study showed that polarization of the M2 phenotype in macrophages is associated with asthmatic inflammation; however, the role of M2 macrophages in asthmatic inflammation and airway remodeling has not been established. 28
M2 macrophages are characterized by the expression of high levels of IL-13, a cytokine critically involved in allergic immune responses and mucus production. 29 Recent findings have revealed that these mediators and M2-specific transcription factors are responsible for lung tissue remodeling and fibrosis. IL-13 might also increase the expression of mucin-5 and TGF-β. 30 IL-4 and IL-13 specifically induce the expression of CCL17 and CCL24 in M2 macrophages. 12 These chemokines facilitate Th2 responses and eosinophil infiltration into the lung. A previous study demonstrated that BALF macrophages in allergen-treated mice exhibited reduced expression of chemokines, including CCL24 and CCL17, in response to radiation exposure compared with cells of untreated mice.27,31,32 Thus, increased M2 macrophage infiltration might be mediated by the upregulation of CCL17 and CCL24 expression. 27
A previous study identified a key biological role of TGF-β1 signaling in promoting M2 macrophage recruitment. Significant experimental evidence has indicated that eosinophils in asthma primarily influence airway remodeling via the secretion of the important profibrotic cytokine TGF-β1.33,34 A simultaneous decrease in the markers of the alternative macrophage activation pathway and eosinophil infiltration indicated decreased bronchial collagen deposition and epithelial mucus secretion. TGF-β1 also activates the extracellular signal-regulated kinase (ERK) 35 and c-Jun N-terminal kinase (JNK) 36 pathways, resulting in a unique pattern of gene expression and corresponding physiological functions. Our previous study demonstrated that LDR markedly decreased the phosphorylation of ERK and JNK in exposed mice compared to OVA-induced asthmatic mice. This decrease was also found to be accompanied by a significant reduction in cytokine levels. 6
Results from previous studies indicate that M2 macrophages can contribute to lung damage through the excessive production of protease-like MMP-9, 37 which has a key role in various pathophysiological processes. MMP-9 is considered important for the development of asthma. 38 In addition, this protein induces the degradation of the extracellular matrix, resulting in airway remodeling during asthma. 39 MMP-9 expression was demonstrated to be significantly higher in asthmatic patients than in healthy controls. 40 Thus, MMP-9 is considered an important therapeutic target for the control of asthma. Previously, we demonstrated that LDR attenuates airway inflammation and mucus production in lung tissues by decreasing MMP-9 expression and activity. 6
Asthma is a heterogeneous, allergic lung disease. Most studies aimed at reveal the effects of ionizing radiation on macrophages have been performed using mouse models. Furthermore, previous studies have sought to establish the relationship between radiation exposure and asthma development; however, contradictory results have been reported.6,16 According to other studies, asthmatic reactions are attenuated by radiation exposure.16,18
Conclusions
Low dose rate radiatio prevents allergic airway inflammation by reducing the number of M2 macrophages in a mouse model of asthma. Although the detrimental effects of radiation exposure on other organs remain unclear, the present results indicate that LDR does not adversely affect asthma, and may instead be associated with positive effects.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Dongnam Institute of Radiological & Medical Sciences (DIRAMS) grant funded by the Korea government (MSIT), grant number 50492-2017 and 50491-2021. This work was also supported by grants from the National Research Foundation (NRF-2020M2C8A2069337 and NRF-2020R1A2C1004272) funded by the Ministry of Science and ICT (MSIT), Republic of Korea.
Ethics approval
All Experimental procedures were approved by the institutional Animal Care and Use Committee of Dongnam Institute of Radiological and Medical Sciences (DIRAMS; Permit Number: DI-2015-002). Animals were handled in accordance with the NIH Guide for the Care and Use of Laboratory Animals.
