Abstract
The aim of the present study was to compare genotoxicity induced by high- versus very low dose-rate exposure of mice to γ-radiation within a dose range of 5 to 61 cGy using the single-cell gel electrophoresis (comet) assay and the micronucleus test. CBA/lac male mice were irradiated at a dose rate of 28.2 Gy/h (high dose rate) or 0.07 mGy/h (very low dose rate). The comet assay study on spleen lymphocytes showed that very low dose-rate irradiation resulted in a statistically significant increase in nucleoid relaxation (DNA breaks), starting from a dose of 20 cGy. Further prolongation of exposure time and, hence, increase of a total dose did not, however, lead to further increase in the extent of nucleoid relaxation. Doses of 20 and 61 cGy were equal in inducing DNA breaks in mouse spleen lymphocytes as assayed by the comet assay. Of note, the level of DNA damage by 20–61 cGy doses of chronic irradiation (0.07 mGy/h) was similar to that an induced by an acute (28.2 Gy/h) dose of 14 cGy. The bone marrow micronucleus test revealed that an increase in polychromatic erythrocytes with micronuclei over a background level was induced by very low-level γ-irradiation with a dose of 61 cGy only, with the extent of the cytogenetic effect being similar to that of 10 cGy high-dose-rate exposure. In summary, presented results support the hypothesis of the nonlinear threshold nature of mutagenic action of chronic low dose-rate irradiation.
Keywords
INTRODUCTION
All living organisms, including humans, are continuously exposed to ionizing radiation (IR) from natural sources. However, development of nuclear technologies and associated intentional (e.g., Hiroshima and Nagasaki) and accidental (e.g., Chernobyl) releases of radioisotopes have led to an increase in a background level of IR. This technogenic part of IR exposure has risen significantly over the past few decades.
Although there are no doubts about the negative biological effects of high-dose IR, debates about whether low-dose IR exposure is harmful or beneficial (hormetic) still continue in the scientific community. Analysis of available literature indicates that low-dose IR exposure induces a complex of biochemical and biophysical reactions in animals (Calabrese and Baldwin, 2000; Mothersill and Seymour, 2003). It is not clear, however, whether those changes are consequences of organism adaptation to increase in IR background, and whether low doses cause any significant genetic alterations.
The aim of the present study was to compare genotoxicity induced by high- versus very low dose-rate exposure of mice to γ-radiation within a dose range of 5 to 61 cGy using the single-cell gel electrophoresis (comet) assay and the micronucleus test.
MATERIALS AND METHODS
In chronic IR exposure experiments, 4–5-week-old CBA/lac male mice weighting 12–14 g (purchased from Pitomnik-Stolbovaya) were used. Mice were placed in plastic cages 14 days prior to IR exposure. Distribution of animals into control and experimental groups was random. Mice were given standard dry feed and water ad libitum. Experiments with chronic low dose-rate irradiation were carried out from 2000 to 2003. Two independent experiments with identical conditions were performed, utilizing a total of 280 mice (140 animals in each of the two experiments).
Experimental animals were chronically exposed to IR from a γ-ray unit “UOG-1” (VNIIFTRI, Russia) equipped with a 137Cs source (activity 7.2 × 108 Bq) mounted in a steel container and specifically designed for long-term irradiation of biological objects. The IR source was placed above exposed targets. Chronic irradiation of animals was performed at a dose rate of 0.07 mGy/h (distance from mouse bedding to the γ-radiation source was 64 cm, filter-lens #5). Variability of a dose rate within the area to be irradiated (1 m2) did not increase over 10%. Irradiation was continuous with 10–15 min daily break for hygiene procedures. Control dosimetry was performed using thermoluminescent detectors TLD-100 (Sweden) and DTG-4 (Russia). To deliver to animals total cumulative doses of 6.7, 13.4, 20.2, 35.3, 45.4, and 61.3 cGy, low dose-rate irradiation was performed for 40, 80, 120, 210, 270, and 365 days, respectively.
In an experiment with acute (high-dose-rate) γ-irradiation, 3–4-month-old CBA/lac male mice weighing 22–26 g were used. The total number of mice used for acute irradiation experiments was 120. Irradiation was performed using the Panorama-3C device equipped with four mobile 137Cs sources (4.5 × 1013 Bq each) producing a dose rate of 28.2 Gy/h.
Upon completion of chronic low dose-rate or acute irradiation, mice were sacrificed and spleen and red bone marrow were removed and processed for subsequent analysis. Suspension of spleen cells in phosphate-buffered saline (pH 7.4) containing 0.14 M NaCl, 2.7 mM KCl, and 3 mM NaN3, was filtered through nylon mesh at 4°C. Cell concentrations were counted using a hemocytometer.
Alkali single-cell gel electrophoresis was carried out as described by Singh et al. (1988). According to the assay, the number of alkali labile sites and single-strand breaks (SSBs) is proportional to the number of DNA fragments and to the distance that DNA migrated from the nucleus after alkali electrophoresis of agarose-immobilized single cells. Fluorescent dye Hoechst 33258 (Sigma Chemical Co, St. Louis, MO) was used to visualize DNA. Analysis was performed using the “Lumam I-2” fluorescent microscope (LOMO, Russia). One hundred comets were counted from each slide. Comets were divided into classes 0–4 (0 corresponded to no visible tail, 4 corresponded to total migration of DNA from the nucleus into the tail) depending on the shape (diameter, tail length, etc.). This method of visual damage is considered a valid means of DNA damage analysis (Kobayashi et al., 1995). Results of the visual classification were subsequently confirmed using the analytic package image analysis software (Kinetic Imaging, Liverpool, UK).
A number of comets in each class was recorded and the average comet index (ACI) was calculated as ACI = (1 · n1+2 · n2+3 · n3+4 · n4)/Σ, where n1 – n4 is a number of comets in classes 1–4, and Σ is the sum of counted comets, including comets in class 0.
The percentage of apoptotic cells was determined by the DNA diffusion assay described elsewhere (Singh, 2000).
For cytogenetic analysis of bone marrow cells by the micronucleus test, cytological slides were prepared using standard procedure (Schmidt, 1975). Polychromatic erythrocytes (PCEs) with micronuclei (MN) were recognized and counted as described by Zaichkina et al. (1998). Three thousand PCEs were analyzed from each mouse.
Statistical analysis of experimental results was performed using the student t-test. Results are presented as averages for a dose group of animals (20 mice per dose group for chronic irradiation, and 10 mice per dose group for acute irradiation) ± standard errors.
RESULTS AND DISCUSSION
Figure 1 shows the results of the ACI determination in spleen lymphocytes from mice exposed to acute and chronic (very low dose-rate) IR. Data are presented as differences of the ACI in irradiated and untreated control groups. A dose-response curve after acute IR exposure is perfectly fitted by a linear regression ACI = a + 0.049 * D, where a is the ACI value in the control group, and D is a dose in cGy Depending on cell type, and proliferative and metabolic activity rate, the ACI in control animals varied within 0.2–1.
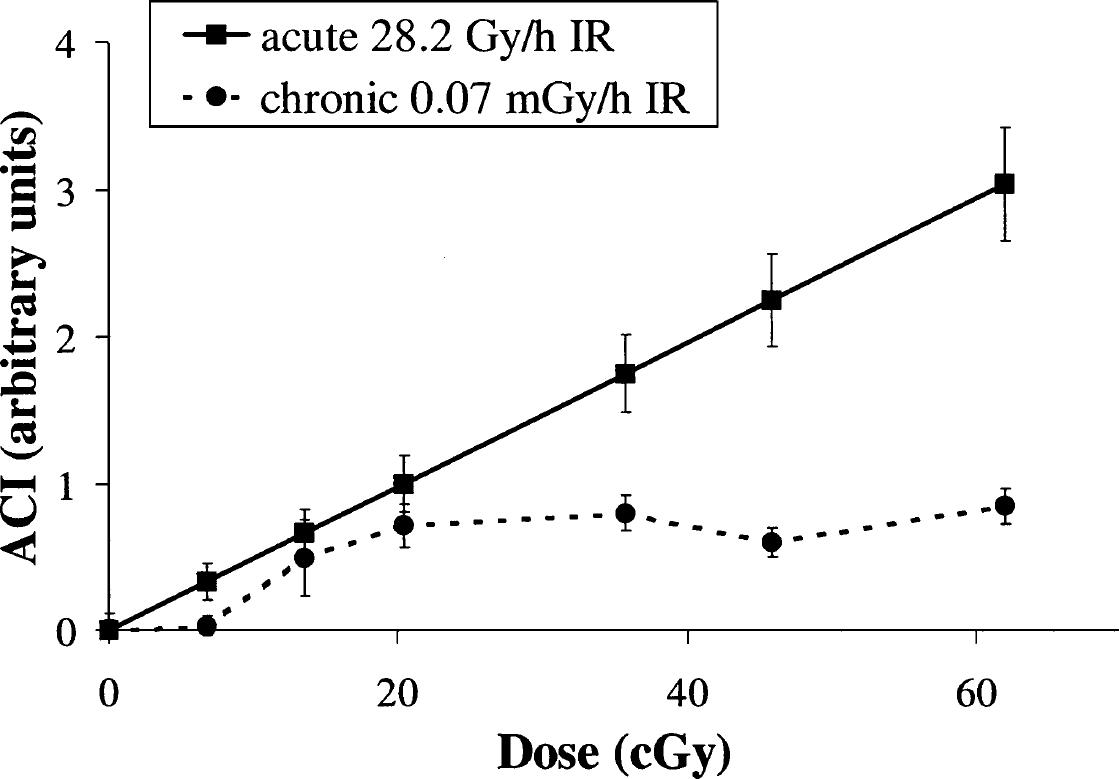
Dose-response curves of the ACI of comet DNA of spleen lymphocytes of mice exposed to acute (28.2 Gy/h) or chronic very low dose-rate (0.07 mGy/h) IR. Data are presented after subtraction of control (untreated) values from experimental (irradiated) values.
Using the BALB/c mouse strain, we have previously shown that the dose-response curve of initially induced DNA SSBs in mouse spleen lymphocytes after acute IR exposure is linear in the range 5–50 cGy with the similar ACI = a + 0.042 · D (Mayzin et al., 2002).
In contrast to acute IR exposure, chronic low dose-rate irradiation resulted in a nonlinear dose-response curve (Figure 1). In the range of about 7 to 20 cGy (40–120 days), the ACI increased linearly. Further accumulation of a dose did not, however, lead to further increase in the ACI. Its value did not significantly change within a dose range of 20 to 61 cGy. These results suggest that the dose-response curve of DNA SSBs formation in mouse spleen lymphocytes after in vivo chronic γ-irradiation (0.07 mGy/h) contains a “plateau” section that is not observed in cases with acute γ-irradiation.
Because long periods of irradiation time can significantly change the ACI value in a control group, interfering with accurate evaluation of the parameter in an exposure group, the ACI values were recorded throughout all experimental time (Table 1). The ACI value decreased with age of animals, probably due to decrease in proliferation rate of spleen lymphocytes. Nevertheless, starting from 40 and up to 270 days of the experiment (the main experimental points), ACI values did not significantly change, confirming that our way of presenting the results is accurate.
Changes in the ACI Values of Spleen Lymphocytes of Control Untreated Mice During Chronic Irradiation Experiments

It is well known that some background level of DNA breakage is always present in mammalian cells due to normal metabolism. These breaks can be divided into two classes:
breaks resulting from DNA damage by free radicals produced by cellular oxygen metabolism; and
breaks resulting from DNA replication, repair, transcription processes, as well as chromatin condensation and decondensation.
An IR dose of 1 Gy induces approximately 1000 DNA SSBs per cell (Billen, 1990). Simple calculations based on these data show that irradiation with a dose rate of 0.07 mGy/h (∼0.17 cGy/day) will induce about two DNA SSBs per cell per day, whereas a number of endogenous SSBs resulting from normal oxygen metabolism is ∼1.2 × 105 per cell per day (Billen, 1990). It is, therefore, obvious that low dose-rate IR-induced DNA SSBs can hardly influence a total number of DNA breaks. The nature of spontaneous and induced DNA breaks is rather similar. IR-induced primary lesions could lead to genotoxic effects only if the spatial distribution of the lesions along the chromatin and effectiveness of their repair is different from those spontaneously induced. Studies of the past decade suggest that particular areas within chromatin possessing hypersensitivity to IR do exist (Oleinick et al., 1994). Double-stranded DNA clusters composed of multiple lesions on opposing DNA strands within a few helical turns are of particular danger to cells, because it is hard for DNA repair machinery to repair them (Goodhead, 1994). These clusters are thought to be crucial IR-induced DNA lesions leading to double-stand breaks (DSBs), and eventually to mutagenesis and cell death (Ahnstrom and Bryant, 1982). Sutherland et al. (2002) demonstrated that as low as 10 cGy IR caused an increase in clustered DNA damage level in human monocytes. Consistent with our results, nondividing primary human fibroblasts exposed to 1 mGy of IR were not able to repair DNA DSBs for several days, whereas effectiveness of DSB repair after higher doses was much better (Rothkamm and Lobrich, 2003).
A minor part of cells that have an extremely high level of DNA damage (e.g., apoptotic cells) would supposedly contribute substantially to an overall DNA damage level within an entire cellular population. To take into account the contribution of an apoptotic cell subpopulation to a final readout of DNA breaks in our experiments, we measured the percentage of apoptotic spleen lymphocytes from mice exposed to very low dose-rate IR or untreated animals using the “DNA diffusion” assay. At days 120, 270, and 365 of the chronic irradiation (20, 45, and 61 cGy, respectively), an approximately twofold increase over a control level in the apoptotic cell fraction was observed (Table 2). As expected, a correlation (r = 0.86; p < 0.05) between an overall level of DNA damage and percentage of apoptotic cells was noticed. These observations prompted us to recalculate overall DNA damage levels (the ACI coefficient) in irradiated versus untreated groups. When performed without counting highly damaged cells (comets within classes 3 and 4), the comet assay yielded less, but still statistically significant, difference in DNA damage levels between irradiated (20–61 cGy) and untreated mice (data not shown). The appearance of apoptotic cells with highly fragmented DNA in irradiated animals cannot, therefore, have a direct impact on overall level of DNA strand breaks. An indirect mechanism, also known as a “bystander effect,” a response of unirradiated cells due to signaling originating from irradiated cells (Hall, 2003), can possibly be involved in the effects observed in this study. In this scenario, lesions within supersensitive chromatin regions in a minor, apoptotic cell population appear to trigger a cascade of metabolic processes in different cell populations on both organ and organism levels. Bystander effects have been demonstrated after both low-linear energy transfer (LET) and high-linear energy transfer (LET) IR exposures (Mothersill et al., 2002). A signal from irradiated cells can be transmitted by direct intercellular contacts (gap junction communications), as well as by cytokines and/or oxygen species secreted by irradiated cells (Lorimore and Wright, 2003). In our previous study, using mammalian cultured cells, we showed that low IR doses induced production and secretion of the ApoJ lipoprotein involved in the bystander effect by regulating expression of genes downstream of the TFG-beta signaling pathway (Klokov et al., n.d.). A variety of changes has been reported to occur in bystander cells, including overproduction of free radicals (Narayanan et al., 1997; Leach et al., 2001); induction of stress-related kinases such as JNK, ERK1/2, and others (Little et al., 2002); and cytokines Beta1-integrin and IL-1alpha (Osterreicher et al., 2003). In addition, reactive oxygen species (ROS) can act as signal molecules to propagate and regulate a particular cellular response such as proliferation, differentiation, and apoptosis (Lehnert and Iyer, 2002). It is well known that actively transcribed DNA sequences are much more susceptible to DNA damage than those in compact chromatin regions due to unlimited accessibility of them for ROS (Chiu et al., 1982; Warters et al., 1987). An increase in actively transcribed genes, together with an increase in ROS production can, therefore, lead to elevated DNA damage. On the other hand, DNA damage within active genes is repaired faster and more efficiently compared to that in silent genes (Oleinick et al., 1984; Bohr, 1987).
Percent of Apoptotic Spleen Lymphocytes in Control Mice and Mice Exposed to Chronic Very Low Dose-Rate (0.07 mGy/h) IR

Significantly increased compared to control (p < 0.05).
Our speculation is supported by results of monitoring reparative and replicative DNA syntheses in mouse bone marrow cells reported by Mazurik et al. (2002), within a collaborative effort with our group, performed on the same mice used in our present study. The authors demonstrated that chronic low dose-rate irradiation of mice substantially induced reparative and replicative DNA syntheses in bone marrow cells (60 and 67% increase; p < 0.01, p < 0.01, respectively). As mentioned earlier, activation of DNA replication and repair is associated with increase in the DNA strand-breaks level. Besides, significant positive correlation (r = 0.87; p < 0.01) between DNA strand breaks and superoxide anion-radical content in bone marrow cells of the irradiated mice was shown, indicating additional production of DNA damage by ROS due to the loss of a part of structural proteins and conformational changes in expression sites of the chromatin during gene expression (Mazurik et al., 2002).
It was suggested that the bystander effect has an alternative protective feature due to elimination of highly damaged, potentially dangerous cells from a cell population (Belyakov et al., 2002; Prise et al., 2002). In accordance with this line of evidence, an adaptive response, an effect of increased radioresistance to high IR doses acquired after exposure to low doses, was associated with overproduction of ROS (Lehnert and Iyer, 2002). In our previous study, we demonstrated that an increase in DNA breaks level by days 120–365 of low-level IR exposure is accompanied by elevated radioresistance to hydrogen peroxide treatment (Osipov et al., 2002). It was suggested that the elevated cell radioresistance could be explained by either activation of DNA repair or elimination of a supersensitive cell population (Osipov et al., 2002).
Thus, overall increase in the level of DNA breaks in mouse spleen lymphocytes as a result of chronic low dose-rate IR exposure can possibly be explained by structural rearrangement of the chromatin during gene expression activation, free radical overproduction, and DNA repair activation. Although insignificant, a contribution of apoptotic cells to an overall level of DNA damage was also recorded, providing further support for the proposed mechanisms of low dose-rate radiation-induced cytogenetic effects observed in this study.
The mutagenic effect in PCEs was determined using the micronucleus test. MN are acentric chromosomal fragments or whole chromosomes lost during mitosis as a result of DNA damage. As expected, an increase in the portion of PCEs with MN in aging mice was observed, which correlates with a well-established age-related accumulation of mutations in bone marrow cells (Table 3).
Changes in the Frequency of Bone Marrow PCE with MN of Control Untreated Mice During Chronic Irradiation Experiments

Dose-response curves for frequency of PCEs with MN in mouse bone marrow upon IR with two different dose rates are presented in Figure 2. The dose-response curve upon acute IR exposure is described by a linear function and is consistent with results obtained by other investigators using different mouse strains (Uma Devi and Sharma, 1990; Jagetia and Ganapathi, 1994; Sudheer Kumar et al., 2003). The genetic background of the mice seems to be an important factor for cytogenetic studies and interpretations. Our previous data generated in experiments with SHK mice indicated that the dose-response curve for cytogenetic damage in bone marrow cells was described by a step function and consisted of two linear regions and a “plateau” between them in the range 10–35 cGy (Zaichkina et al., 1998). In contrast, chronic low dose-rate exposure did not result in an increase in cytogenetic damage up to 61 cGy, with the damage induced by 61 cGy being comparable with that induced by acute IR doses of 10 cGy. Using this assay, therefore, mutagenic activity of chronic IR exposure with a dose rate of 0.07 mGy/h was at least six-fold lower than that of acute IR exposure with a dose rate of 28.2 Gy/h. In addition, our results indicate a threshold in the dose-effect relationship after chronic IR exposure with a dose rate of 0.07 mGy/h. The threshold was observed to extend up to 35–61 cGy.
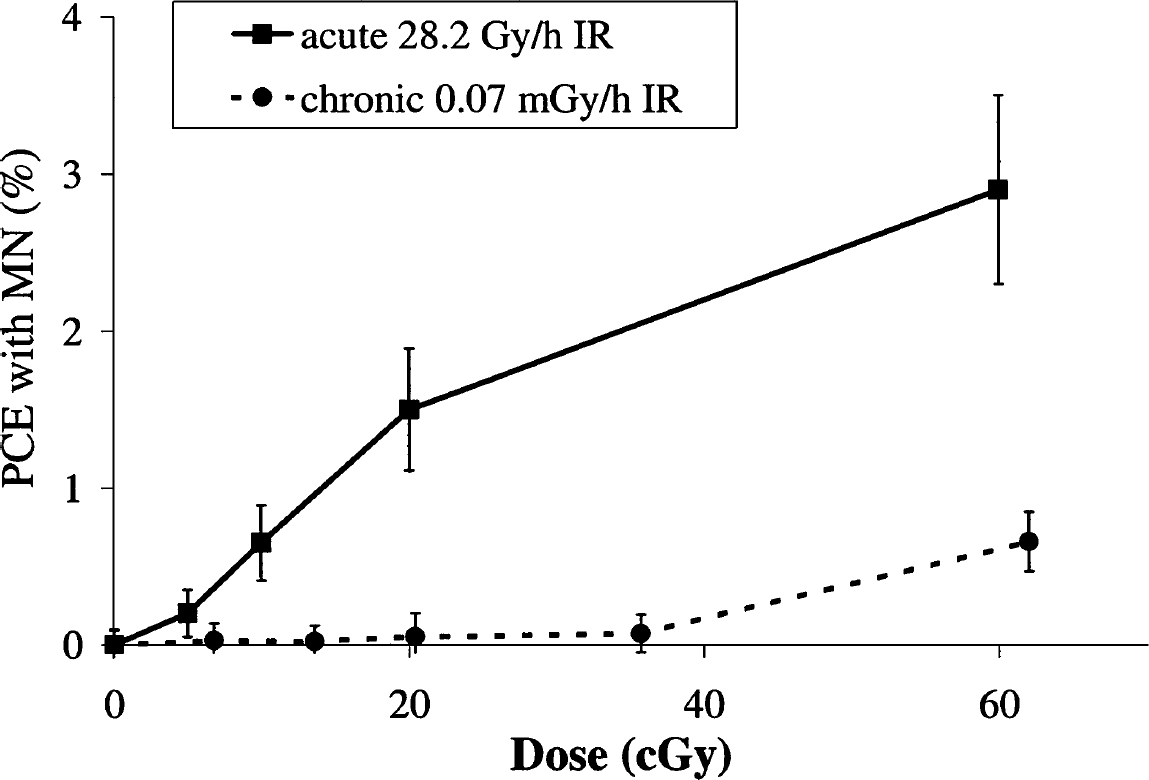
Dose-response curves for MN induction in bone marrow PCE from mice exposed to acute (28.2 Gy/h) or chronic very low dose-rate (0.07 mGy/h) IR. Data are presented after subtraction of control (untreated) values from experimental (irradiated) values.
To summarize, chronic IR exposure of three orders over a normal background level leads to a statistically significant increase in the number of DNA strand breaks within a 20–61-cGy dose range. More probably, this increase can be associated with the chromatin rearrangement accompanied by gene overexpression, increase in ROS production rate, and DNA repair activation, processes known to be triggered after low doses of IR. Six times higher dose of chronic 0.07 mGy/h IR exposure was required to induce cytogenetic damage in bone marrow cells comparable to that after acute 28.2 Gy/h exposure. Based on these results, we can suggest that mutagenic activity of chronic irradiation of three orders over the normal background level is at least six times lower than that of acute irradiation. Data on MN induction support the hypothesis of the nonlinear threshold nature of mutagenic action of chronic low dose-rate irradiation.
