Abstract
Background
Nitric oxide (NO) as a vaso- and cardio-protective agent could reduce vasomotor dysfunction in different cardiovascular diseases. One of the current therapeutics targeted at NO availability in the vascular wall are highly diluted antibodies to endothelial NO-synthase (eNOS). This drug has previously shown its endothelium-protective effect and effectiveness in reducing hypertension. Current study was dedicated to evaluate the direct impact of highly diluted antibodies to eNOS on the vessel constriction and dilation ex vivo.
Methods
For that purpose, we used thoracic aortas dissected from spontaneously hypertensive (SHR) rats. Endothelium-dependent relaxation in the presence of highly diluted antibodies to eNOS (1 mL) was examined after phenylephrine-induced pre-constriction of the aorta rings in response to gradually increased acetylcholine concentration (1 nM to 10 µM).
Results
Highly diluted antibodies to eNOS enhanced acetylcholine-induced relaxation in a statistically significant manner. Moreover, it was demonstrated that observed effect was similar to perindopril, a well-known angiotensin-converting-enzyme inhibitor, which works through relaxing and widening blood vessels.
Conclusions
Our findings indicate that highly diluted antibodies to eNOS restored impaired endothelium function, as demonstrated by increased relaxation of SHR rats aorta rings. The revealed results suggest beneficial effect of highly diluted antibodies to eNOS to ameliorate hypertension and related diseases.
Keywords
Introduction
Vascular endothelium is known as an active paracrine, endocrine, and autocrine organ that plays crucial role in the regulation of vascular tone 1 and the maintenance of vascular homeostasis. 2 Under physiological conditions, several relaxing and contracting factors are maintaining endothelial equilibrium. However, in pathological conditions such as hypertension, atherosclerosis, and diabetes balance of these factors is upset resulting in endothelial dysfunction and vascular damage. 3 One of the most studied relaxing factors is nitric oxide (NO), generated by endothelial NO synthase (eNOS). Nitric oxide plays a pivotal role in the maintenance of vascular tone and reactivity. 4 Disruption of NO production results in the intracellular energy deficit, vascular inflammation, and atherogenesis. 5 Therefore, targeting vascular (endothelial) dysfunction still represents attractive pharmacological approach due to its central involvement in the progression of most cardiovascular diseases. 6
Currently, there are several drugs for treatment of endothelial dysfunction. The main purpose of this therapy is the elimination of paradoxical vasoconstriction and creation of a protective environment in relation to pathological factors through the increased availability of NO in the vascular wall. The most commonly used medications with such mechanism of action are angiotensin-converting-enzyme (ACE) inhibitors and statins. 6
In addition to these classical drugs, new therapeutics that are based on the direct targeting of the NO-cGMP signaling cascade have been successfully used for years. For example, it was shown that phosphodiesterase (PDE) inhibitors prevent the enzymatic degradation of cGMP and are used in a wide range of diseases including the use of PDE 5 inhibitors to treat sexual dysfunction in females, cardiovascular disease, and pulmonary hypertension. 7 Activators and stimulators of the soluble guanylate cyclase (sGC) represent a novel class of “repair” drugs of a (oxidatively) damaged sGC enzyme. It has been demonstrated that they may fulfill a similar role for uncoupled/dysfunctional eNOS and only activate the damaged, oxidatively inactivated form of eNOS,8,9 which would represent a safe way to treat endothelial dysfunction without the risk of overdosing. These drugs are mostly intended to be used for the therapy of pulmonary hypertension and heart failure, but also in other indications, such as arterial hypertension, renal fibrosis/failure etc. 10
Another approach is the development of eNOS enhancers with upregulation of eNOS mRNA or protein as the mechanism of action. AVE3085 being an example of a potent eNOS enhancers normalized endothelial function in spontaneously hypertensive (SHR) rats, 11 reduced oxidative stress, and endothelial dysfunction in diabetic db/db mice, 12 attenuated cardiac remodeling in vivo, 13 and prevented endothelial dysfunction by homocysteine in human vascular tissue. 14 However, the potential of eNOS enhancers requires further exploration in clinical studies, maybe in combination with antioxidant therapy in order to prevent the uncoupling of overexpressed eNOS protein which might boost pre-existing oxidative stress. 15
Thus, a drug that would have to meet efficacy and safe requirements is still needed. The aim of this study was to test the effect of highly diluted antibodies (HD Abs) to еNOS on endothelium-dependent relaxation in animal model of hypertension, which exhibit many phenotypic characteristics observed in humans.
Indeed, HD Abs to eNOS were shown to stimulate the “eNOS–NO–GC–cGMP” cascade that corrects vasomotor disturbance through regulation of regional blood flow. 16 In animal models of NO deficiency induced by L-NAME 17 or by lack of estrogen HD Abs to eNOS prevented endothelial damage. 18 In addition, HD Abs to eNOS decreased arterial pressure in hypertensive rats without affecting main hemodynamic parameters in normotensive rats. Moreover, the administration of HD Abs to eNOS as a part of complex drug Divaza® (OOO “NPF “MATERIA MEDICA HOLDING”, Russian Federation) in patients with chronic cerebrovascular disease (CBVD, NCT03485495) led to normalization of ischemia and inflammation biomarkers, such as fibrinogen and von Willebrand factor in plasma after 3 months of therapy. Moreover, statistically significant decreases in C-reactive protein and the monocyte chemoattractant protein-1 concentrations were shown, suggesting the endothelioprotective effect and the ability of HD Abs to eNOS as a part of this complex drug to reduce the severity of the inflammatory process in vessel walls. 19 The mechanism of action of HD Abs to eNOS is determined by the properties inherent in HD solutions. In particular, it was shown that these solutions possess a number of features typical for nonlinear system: solutions obtained during the dilution process acquire special long-lasting physicochemical and biological properties that are different both from the properties of the original substance and the properties of the solvent (water in this case).20-23 Second, despite the high dilution degree, the available data indicate the possibility of preserving molecules even in the highly diluted solutions, which may be explained by the flotation effect.24-27 At the same time, it is likely that spontaneously forming nanoassociates are the "carrier" of activity that determines special physicochemical and biological properties of HD solutions.28,29 And finally, the most important properties of HD solutions, in practical terms, are their ability to modify the activity of the target via conformational changes, 30 affecting to the hydration shells 31 which can be probably be due to the effect of HD solutions on the target without direct contact with it. 32 It is well-known that the activity of eNOS and consequently production of NO are regulated at the transcriptional, post-transcriptional, and post-translational levels; any modifications occurring at these levels induce changes in the eNOS. 33 Most likely that HD Abs to eNOS affect the conformation and hydration shells of eNOS and consequently influence target-mediated effects. Therefore, we suggest that HD Abs to eNOS might be used as a treatment option against a variety of cardiovascular pathologies.
Nevertheless, more data with regard to the role of HD Abs to eNOS on the vessel constriction and dilation are needed. Thus, the aim of this study was to investigate a direct effect of HD Abs to eNOS on endothelium-dependent vascular relaxation of aortic rings dissected from SHR rats ex vivo.
Material and Methods
Experimental Samples
Affinity-purified rabbit polyclonal antibodies to human eNOS (stock concentration 1.0 mg/mL) were manufactured in accordance with the current EU requirements for GMP for starting materials (EU Directive 2001/83/EC as amended by Directive 2004/27/EC) by Ab Biotechnology Limited (UK). The HD Abs to eNOS were produced by the GMP manufacturing facility of OOO “NPF “MATERIA MEDICA HOLDING” (Russian Federation) according to the technology described in the US patent 8 535 664. Briefly, antibodies to eNOS (1.0 mg/mL) were mixed with a solvent (ethanol-water solution) at a ration 1:100 and shaken intensively to produce the first centesimal dilution (i.e., 100-fold dilution). All subsequent dilutions comprised one part of the previous dilution and 99 parts of solvent (ethanol-water solution for intermediate dilutions and purified water for the preparation of the final dilutions), with intensive vibration treatment between the dilution steps. Thus, if we do not take into account the physical aspects mentioned in the Introduction, the theoretical concentration of initial Abs to eNOS might be 1.0 × 10−24 mg/mL. This value is mentioned here only to avoid misunderstanding regarding the dilution level of HD of Abs to eNOS. The solution of HD of Abs to eNOS was prepared in glass vials under sterile conditions and was stored at room temperature protected from direct intense light. Distilled water (hereafter, a vehicle) served as a negative control.
Other chemicals and drugs used in experimental design were as follows: phenylephrine (hereafter PE; Sigma-Aldrich, P6126), acetylcholine chloride (hereafter Ach, Sigma-Aldrich, A6625) and perindopril (Sequoia Research Products Limited, SRP01317p). Perindopril was solubilized in distilled water, and then further diluted in Krebs buffer (to achieve final concentration of 1 µM in the bath (a standard effective dose which works very well in aorta rings)34,35 and utilized as a clinically approved ACE inhibitor, which eliminates vasoconstrictor activity of angiotensin II.
All reagents were of analytical grade and stored according to the manufactures prescriptions.
Animals
This study was carried out on 14-week-old SHR (SHR, n = 9) and normotensive Wistar–Kyoto (WKY, n = 6) male rats weighing 300-350 g. Animals were obtained from Janvier SAS (France). As WKY rats have normal vascular properties, six species was sufficient to have reproducible data. In contrast, the impairment in the SHR rats could be slightly different among animals, so it was decided to include more animals to reduce variability. Rats were housed in groups of 2 animals on a normal 12 hours light cycle (at 7
Organ Bath Assay
Isolation and preparation of aortic rings
After the 5-day acclimation period, rats were terminally anesthetized with pentobarbital sodium, (50 mg/kg, ip) thoracic aortas were excised, cleared of all adherent tissues and placed in cold modified Krebs buffer of the following composition (mM): 118.5 NaCl, 25 NaHCO3, 4.7 KCl, 1.2 MgSO4-7H2O, 1.2 KH2PO4, 11 glucose, and 2.5 CaCl2-H2O. The aortas were then cut into rings (4 to 5 mm in length) with their respective assignment to the experimental groups.
After being cut, the aortic rings were mounted on fixed segment support pins and two fine stainless-steel holders were placed through the lumen. Aortic segments have then been bathed in isolated organ chambers containing Krebs buffer (5 mL) at 37°C, continuously bubbled with 95% O2 and 5% CO2 to pH 7.4. Each ring was connected to a force displacement transducer for the measurement of isometric force, which was continuously displayed and recorded on-line on a computer via an eight-channel transducer data acquisition system (MP100, Biopac System Inc.).
The aortic rings were submitted to a tension of 2 g, which was readjusted every 15 min during a 60-min equilibration period. The organ bath fluid has been refreshed every 15 min during this period. Subsequently tissue was exposed to 60 mM KCl to check preparation viability and then washed with Krebs buffer.
Experimental procedure
The animal model used in experiment was described in literature previously.36,37
After equilibration, the rings were repeatedly stimulated with KCl solution (high K+, 80 mmol/L) until 2 consecutive equal contractions were reached. Following washout of high K+ responses, the aortic rings were incubated for 30 minutes with either HD Abs to eNOS (1 mL), vehicle (1 mL) or perindopril as positive control (1 µM). All of the tested substances were added directly to the organ bath. Each group had 8 to 12 evaluable aortic rings from at least 6 different animals. The difference in a number of rings can be explained by quality control standards used: some rings could be excluded due to insufficient KCl-induced contraction or observation of tissue damage. In addition to that, the length of the trachea varies from one animal to another.
Vascular relaxation study was performed by doing cumulative concentration–response curves to the endothelium-dependent relaxant agonists—acetylcholine (ACh; 1 nM, 3 nM, 10 nM, 30 nM, 100 nM, 300 nM, 1 µM 3 µM, 10 µM, and 30 µM) on tissues of SHR and WKY rats, precontracted with phenylephrine (PE, 1 µM).
Statistical Analyses
The data are expressed as mean ± standard error of mean (m±SEM). For substances that elicit contraction of aortic rings (KCl and PE), results are expressed as an increase in isometric tension from the resting level. For Ach that elicit relaxation, the response is expressed as percent of reversal of the PE-induced pre-constriction and was calculated for each concentration as relaxation = [(maximal tension induced by PE—tension induced by Ach)/(maximal tension—basal tension)] x 100.
Statistical analysis was performed using R language version 3.4.0 (RStudio, ViennaS). Data are presented as relative values in the range of 0% to 100%, so logit transformation was performed for its analysis.
After transformation, linear mixed effect model followed by Tukey’s Post Hoc Test was used to assess intergroup differences.
Differences were considered statistically significant when p-value was less than .05.
Results
The tension induced by KCl (60 mM) or PE (1 µM) was significantly higher for SHR compared to WKY rats, the values were 2.5±.2 vs 2.07±.15 and 2.3±.13 vs 1.7±.18, respectively, (P < .05).
We observed a marked endothelial dysfunction in SHR vascular ring preparations, which was exhibited in the lowest rates of aortic relaxation. The Ach-induced relaxation in this group was 37% lower compared to a group of WKY rings (p< .05; Figure 1A) (A) Concentration-response curves to acetylcholine (1 nM to 30 µM) in aortic rings from Wistar–Kyoto and spontaneously hypertensive rats. *P < .05 difference vs vehicle-treated rings dissected from spontaneously hypertensive. Data presented as m ± SEM. (B) Effect of HD Abs to endothelial NO-synthase or perindopril treatment on Ach-induced relaxation in spontaneously hypertensive rats. Data presented as m ± SEM. *P < .05 difference between a group of HD Abs to endothelial NO-synthase vs vehicle-treated rings dissected from spontaneously hypertensive rats. +P < .05 difference between a group of perindopril vs vehicle-treated rings dissected from spontaneously hypertensive rats.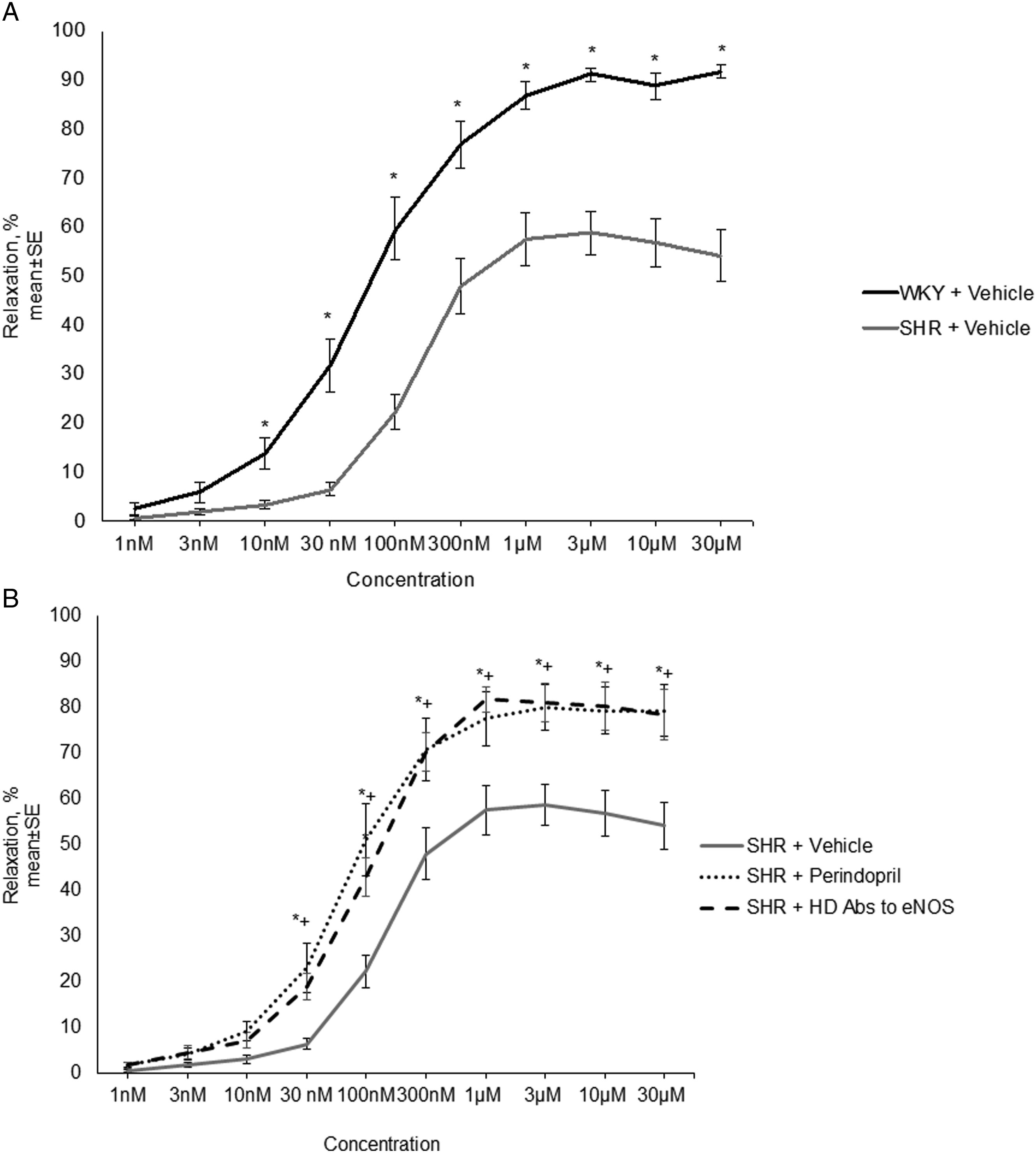
Treatment with perindopril (1 µM) significantly improved Ach-induced relaxation compared to a vehicle-treated rings from SHR. Percent of maximal pre-constriction was 79.9±5.1% (P < .05). Similarly, to perindopril, treatment with the HD Abs to eNOS normalized relaxation starting from 30 nM Ach concentration (P < .05; see Figure 1B). Maximal relaxation to Ach was 81,7±2.77% and significantly differed from vehicle-treated SHR group (P < .05)
Discussion
Mechanisms underlying endothelial dysfunction observed in animal models of hypertension and in clinical practice include an increase in the levels of cyclooxygenase-generated vasoconstricting factors, a decreased release of vasorelaxant agents (such as NO and bradykinin), and a reduced bioavailability of NO which may be due to a higher production of oxygen free radicals. 38
In earlier studies, HD Abs to eNOS had an effect on the eNOS-NO system.17,18 However, an involvement of HD Abs to eNOS in a direct vasorelaxant effects ex vivo has not been previously reported.
In the present study, which did address this question we have shown that HD Abs to eNOS restored the impaired endothelium-dependent relaxation in the aortae of SHRs suggesting its antihypertensive effect. We link the determined effect to the properties of HD solutions and primarily, their ability to modify the activity of the target via conformational changes. Indeed, Tarasov et al 30 using high-resolution solution nuclear magnetic resonance spectroscopy, on the example of HD Abs to IFN-γ have demonstrated that HD Abs induced conformational changes in the targeted protein. These molecular changes shown to be crucial for the function of the protein, as evidenced by an observed therapeutic effect induced by HD Abs. eNOS being a strong vasodilator with a key role in the regulation of systemic vascular resistance and responsible for NO production seems a promising therapeutic target for management of endothelial dysfunction.
Spontaneously hyperintensive rats are a standard model of NO-deficient hypertension similar to the one observed in human. The causes of pathological changes observed in SHR rats may include: the predominance of vasoconstrictors, impaired endogenous NO production, and reduced endothelial-dependent vasorelaxation in certain arteries, hyperproduction of superoxides, which is another mechanism of reduced NO bioavailability.36,39,40
Thus, we suppose that improved dilatation in response to Ach observed in a group with HD Abs to eNOS treated animals may be the result of ameliorated endothelium-dependent vasodilation, changes in signaling properties, increased production, release and consequently diffusion of NO.41,42 Indeed, in the previous study we have shown that application of HD Abs to eNOS enhanced NOS activity, increased the content of NO derivatives and cGMP16. Additionally, the efficacy of HD Abs to eNOS (as a part of Impaza) has been demonstrated in a model of L–NAME-induced (a non-selective NO synthase inhibitor) endothelial dysfunction.18,43 The endothelial protection activity was assessed according to the coefficient of endothelial dysfunction. This parameter indicates a change in the reactivity of the vascular bed in the model of NO deficiency and allows us to assess the extent of correction of endothelial dysfunction 44
For a group of HD Abs to eNOS (single administration or in combination with the other drugs) the coefficient of endothelial dysfunction was close to the values obtained for the intact animals and was comparable to that of Mildronate® and furostanol glycosides from the cell culture of the Dioscorea deltoidea (laboratory code DM-05). 17
Moreover, in the frame of the above-mentioned experiments a cardioprotective effect has also been shown, which consisted in a decrease in adrenoreactivity and in systolic blood pressure in the left ventricle during a resistance load test compared to the control animals.
What is more important, the results for HD Abs to eNOS observed in our study were comparable with the results published for other NO enhancers. For instance, AVE3085 significantly improved Ach-induced endothelium-dependent relaxations in the aortae of SHRs by 25% after a 4-weeks treatment. 11 In our study this parameter equaled to 24%, but unlike to AVE3085, the effect of HD Abs to eNOS has been realized in a shorter time, an additional pretreatment of rats was not required.
The results of preclinical studies are in agreement with the data observed in clinic. For example, on patients with endothelial and erectile dysfunction HD Abs to eNOS contributed to increase in NO 45 and NO metabolites level 46 in serum by the end of the therapy.
It is also known that endothelial dysfunction is associated with insulin resistance, contributes to the progression of type 2 diabetes (T2D) and its vascular complications. Indeed, high glucose in diabetes can undergo autoxidation and generate hydroxyl radicals that reduce the bioavailability of NO. The HD Abs to eNOS also being a part of complex hypoglycemic drug Subetta® (OOO “NPF “MATERIA MEDIC HOLDING,” Russian Federation) revealed sufficient glycemic management in subjects with T2D and led to normalization of microcirculatory system conditions through the markers related to oxidative metabolism. 47 These findings suggest that HD Abs to eNOS may increase the level of NO also via antioxidant property of this drug, allowing normal NO signaling and relaxations. However, to confirm this statement and to further investigate mechanisms underlying HD Abs to eNOS action future research using a set of antioxidant assays are necessary. Additionally, it would be interesting to estimate the effect of HD Abs to eNOS in other animal models, like STZ-induced diabetic rats 48 and examine an opportunity of combined with standard antihypertension therapy application.
In conclusion, the present results provide the evidence of the beneficial vascular protective effect of HD Abs to eNOS in vivo, however, additional experiments aimed at studying the effect of HD Abs to eNOS on quantitative NO production in this particular model are required. We suggest that the application of HD Abs to eNOS may help to restore the normal endothelial function and be useful in the treatment of hypertension and related diseases.
Footnotes
Acknowledgments
The authors thank employees of Сardiomedex (France) for carrying out the experiments, using validated procedures and in accordance with the respective SOPs of the company.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sergey A. Tarasov, Nataliya V. Petrova and Oleg I. Epstein are employees of OOO “NPF “MATERIA MEDICA HOLDING” (partly). Oleg I. Epstein is a founder and a shareholder of OOO “NPF “MATERIA MEDICA HOLDING. OOO “NPF “MATERIA MEDICA HOLDING” sponsored the study, performed statistical analysis, made a decision to publish the work, and covered the current article processing charges, took part in the design of the experiments and the manuscript writing. HD Abs to eNOS is the substance (single, or one among other components) for commercial drugs Impaza, Divaza, Subetta, Afalaza produced by OOO “NPF “MATERIA MEDICA HOLDING”. Patents on this substance and the drugs belong either to OOO “NPF “MATERIA MEDICA HOLDING”, or Oleg I. Epstein. The authors have disclosed those interests fully to Dose-Response Journal.
Funding
OOO “NPF “MATERIA MEDICA HOLDING” sponsored the study.
