Abstract
During radiotherapy, immune-modulatory effects of radiation doses should be taken into consideration, not only the anti-tumor radiation effects. Thus, our study aimed to study how γ-radiation modulates immune response in comparison to common immune-suppressive/stimulant agents. Animals were divided into two groups. Category A received Echinacea purpura extract (EP) or irradiated at low radiation doses 0, .25 or .5 Gray (Gy), whereas Category B received cyclophosphamide (CP) or irradiated at high radiation doses 1, 2, or 5 Gy. Serum levels of immunological mediators interleukin-10 (IL-10) and tumor necrosis factor (TNF-α), as well as redox-markers malondialdehyde (MDA) and nitric oxide (NO), hemoglobin (Hgb), white and red blood cells (WBCs, RBCs), and platelet counts were assessed following irradiation. Of the immune-stimulant category, .25 Gy dose offered EP-comparable effects in TNF-α, RBCs, Hgb, and platelet counts cases. As for the immune-suppressive category; 5 Gy irradiation dose induced inflammatory/immunosuppressive responses indicated (rise in NO, TNF-α, and IL-10), and an oxidative stress status (increase in serum MDA). However, 5 Gy γ-irradiation was not observed, herein, as a single immunosuppressive agent. To conclude, during radiotherapy, immunological impact(s) of the used radiation doses should be optimized and followed-up closely to assess the risk/benefit of their usage.
Keywords
Introduction
Ionizing radiation (IR) has been reported to induce local and systemic/bystander effects via various signaling pathways. However, the type and the magnitude of such effects depend on the radiation dose level. 1 A high dose (HD) of radiation is known to be large enough to effectively disrupt or injure protective mechanism(s), causing an observable, either immediate or latent, harm(s). 2 However, a low dose (LD) of IR induces a controllable state of reactive oxygen species (ROS) overproduction enough to activate one or more of the body’s defense mechanisms resulting in health benefits, yet not strong enough to initiate detectable adverse effects. Radiation-induced ROS overproduction initiates biochemical reactions, directly and indirectly, targeting sub-cellular components. As a result, radiation is likely to induce adaptive and innate immune responses, pro-inflammatory reactions, and oxidative stress with the consequent beneficial or harmful systemic impacts inside and outside the irradiation field/target. 3
Many studies on LD and its consequences reported different effects from HD’s. However, rare investigations have been focusing on comparing the immunological impacts of IR either as a stimulant or a suppressor. In the current work, we investigated the immune-modulatory effects of high and low doses of gamma irradiation. We compared them to the immune-stimulatory/anti-inflammatory effects of “Echinacea purpura” (EP) extract4-6 and to the immune-suppressor cytotoxic “cyclophosphamide” (CP) 7 as reference standards, respectively.
The macrophage-derived pro-inflammatory cytokine, tumor necrosis factor-alpha (TNF-α), has been reported to be implicated in a variety of immunological disorders, including psoriatic and rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel syndrome, and juvenile chronic arthritis. 8 On the other hand, the anti-inflammatory cytokine, Interleukin-10 (IL-10), is known to be secreted by both immune and non-immune cells and implicated in several experimental autoimmune models, 9 such as experimental rheumatoid arthritis, autoimmune encephalitis, and inflammatory bowel disorder. Furthermore, the oxidative stress biomarkers, Nitric Oxide (NO) and Malondialdehyde (MDA), have been reported as reliable markers to assess the IR-induced immunological responses.10-12 On the other hand, the effects of low-dose IR on the hematopoietic tissue, as in the case of occupational exposure of workers, have drawn attention to the possible risk of HD exposure. Yet, recent studies are limited to investigating the type rather than the magnitude of the IR dose-response effects on blood constituents. However, a recent study suggested that blood components might be biomarkers for low-dose IR impacts.13,14
During radiotherapy, not only the anti-tumor radiation effects are of concern, but also the immune-modulatory effects of different radiation doses should be taken into consideration. Thus, our study aimed to classify and quantify the potential immune response to various doses of IR on measured mediators involved in multiple immunological diseases.
Material and Methods
Experimental Animals
In this experiment, adult male albino rats of the Wistar strain, weighing 150 ± 10 g, were used. They were all kept at our institutional animal house and left to acclimatize for seven days before the experiment. They were fed standard pellets for rats and allowed free access to fresh drinking water. The detailed handling procedures during the experimental period have been pre-approved by the National Centre for Radiation Research and Technology Research Ethics Committee (NCRRT-REC) and given the Approval number: 72A/21.
Irradiation Process
Animals irradiation was carried out at the NCRRT; using the biological experimental gamma irradiator GammaCell 40 (Nordion®; Canada), whose irradiation chamber’s dimensions are 10 cm (height) X 40 cm (diameter), which accommodated a whole animal group at a time. The radiation source is Cs-137 delivering the doses at a rate of .66 cGy/Sec. At exposure time, six groups were exposed to gamma IR doses of 0, .25, .5, 1, 2, and 5 Gy, respectively. The dosimetry dose validation has been carried on by the Dosimetry Department members at the National Center for Radiation Research and Technology on scheduled basis to ensure the dose rate of the source, the absorbed dose received by the animals, and the uniformity of dose via dose mapping measurements.
Experimental Design
Sample size calculations for each group have been carried out using the G-power analysis software sample size feature (version 3.1.9.4); α = .05 and power = .8. Animals were randomly assigned into eight groups falling into two main categories, A and B, each consisting of four experimental groups (n = 5). Below are the details of our experimental design:
Category A : “Immune-Stimulant Radiation Effects Assessment”
Group I (0 Gy): sham irradiated rats that served as control, Group II (.25 Gy): rats were irradiated at a dose level of .25 Gy as a single dose; Group III (.5 Gy): rats were irradiated at a dose level of .5 Gy as a single dose, and Group IV (EP): non-irradiated rats that received oral EP extract (Falcon Group-Sigma Industries) daily at a dose of 500 mg/kg body weight for seven successive days. 15
Category B : “Immune-Suppressive Radiation Effects Assessment”
Group V (1 Gy): rats were irradiated at a dose of 1 Gy and served as a control; Group VI (2 Gy): rats were irradiated at a dose level of 2 Gy as a single dose, Group VII (5 Gy): rats were irradiated at a dose level of 5 Gy as a single dose, and Group VIII (CP): non-irradiated rats that received CP (Endoxan® 1gm-Baxter company) by intraperitoneal route daily at a dose of 50 mg/kg body weight for seven successive days. 16
Irradiation was carried out 24 hours following the last dose of each EP and CP, respectively. For all groups, animal sacrifice was performed under deep urethane anesthesia 24 hours post-irradiation after blood samples were collected from the retro-orbital plexus.
Samples Collection and Analysis
The blood samples from the retro-orbital vein were collected in heparinized tubes and used for blood count automatic hematology analyzer at Cairo University’s Biochemistry Unit to determine hemoglobin content (HB), platelet, white, and red blood cells count (Plat, WBCs, and RBCs). Another aliquot of blood was collected in non-heparinized tubes and centrifuged after clotting to separate the serum, which was stored at −80°C for further determination of serum IL-10 and TNF-α using ELISA kits, Mybiosource® (USA), using a microplate reader: AWARENESS Technology INC (stat Fax 2100). Serum aliquots were also used for the colorimetric determination of MDA and NOx using TECH COMP® UV-Spectrophotometer, according to the methods of Ohkawa et al 17 and Montgomery and Dymock, 18 respectively. MDA and NOx colorimetric kits were purchased from Biodiagnostics® Co., Egypt.
Statistical Analysis
The current work used R Software (version 4.1.0) for the statistical data analysis. Data normality was tested according to the methods described by Kolmogorov–Smirnov 19 and Shapiro–Wilk. 20 Then, Kruskal–Wallis rank-sum test 21 was used to determine whether there was a statistically significant difference in mean values within Categories A and B since our observed data were not normally distributed. As a post-hoc test, the Dunn test 22 with Benjamini–Hochberg correction was used to determine the mean differences among groups. Moreover, Mann–Whitney test 23 was applied within both categories to compare immune stimulants and suppressors to the irradiated groups. Also, Spearman correlation coefficient 24 was used to fit the relations between each pair of variables. A significance level of α = .05 (P < .05) was chosen for statistical analysis and the data were presented in the “Results” section as means and standard errors of means (SEM).
Results
Effects on Redox Status Biomarkers
Effects on the Malondialdehyde Serum Levels in Categories A and B
According to the results shown in Figure 1, Malondialdehyde (MDA) levels in serum after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V–VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05).
According to the data in Figure 1,
Effects on the Nitric Oxide Serum Levels in Categories A and B
The results presented in Figure 2 Nitric oxide (NO) levels in serum after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V-VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05).
The data presented in Figure 2
Effects on Immunological Mediators
Effects on Tumor Necrosis Factor-Alpha Serum Levels in Categories A and B
In the current study The immune response of tumor necrosis factor-alpha (TNF-α) levels in serum after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V-VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal Cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05).
Regarding the effects of the radiation doses 2 and 5 Gy on the serum levels of TNF-α, Figure 3
Effects on Interleukin-10 Serum Levels in Categories A and B
In the current study, .25 and .5 Gy radiation doses had no significant (P ≥ .05) immune-modulatory effects as reflected by the lack of change in serum levels of IL-10 as compared to the control un-irradiated group. On the other hand, the administration of EP induced a significant rise (P < .05) in serum levels of IL-10 compared to all groups.
Regarding the effects of the high radiation doses on the serum levels of IL-10, results shown in the effectiveness of the 2 and 5 Gy groups showed non-significant change (P ≥ .05) serum IL-10, as compared to the control group (V) of The immune response of interleukin-10 (IL-10) levels in serum after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V–VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05).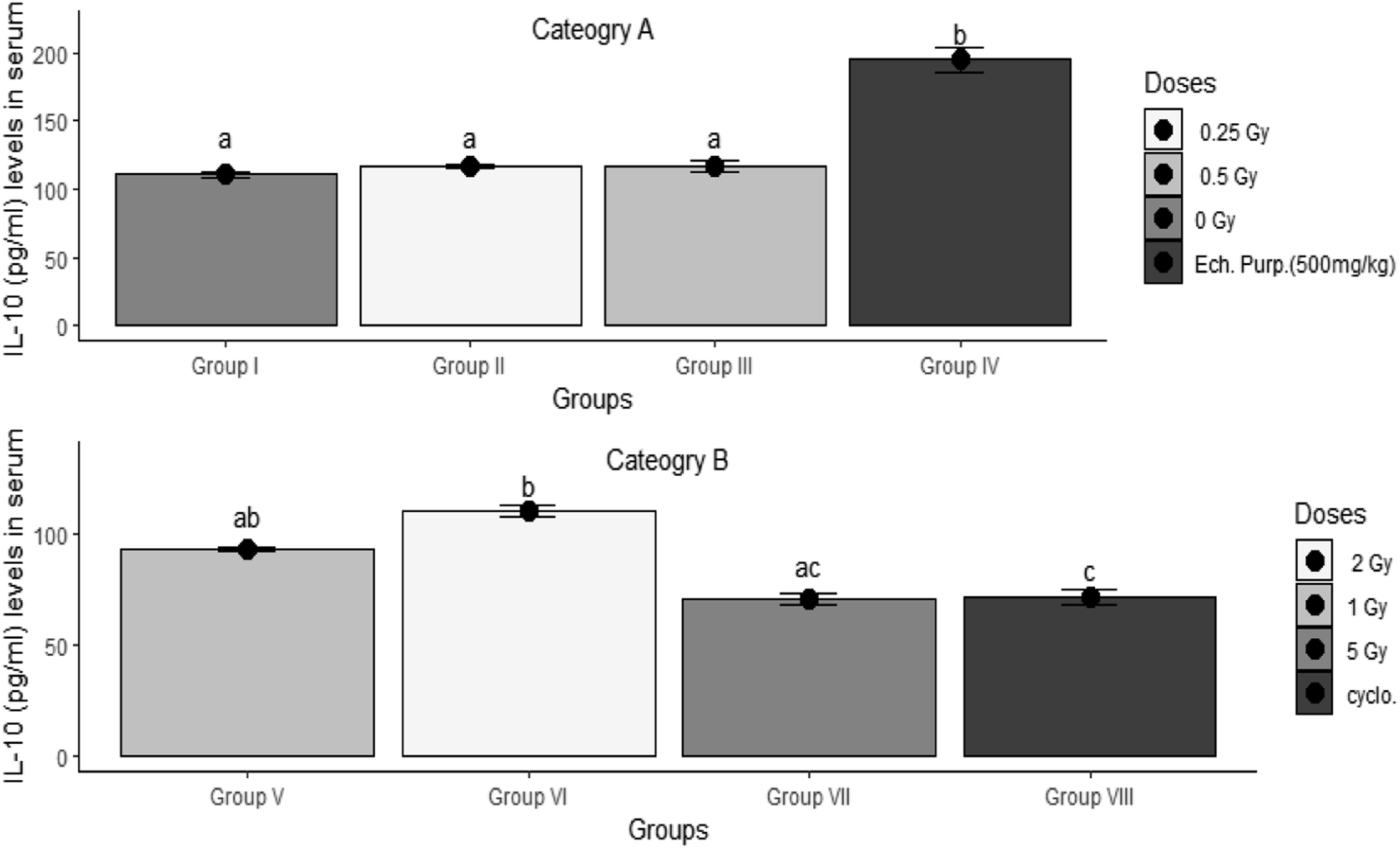
Effects on Blood Components
Effects on Hemoglobin Content (Hgb) in Categories A and B
Results of the present work, in Hemoglobin content (Hgb) after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V-VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05).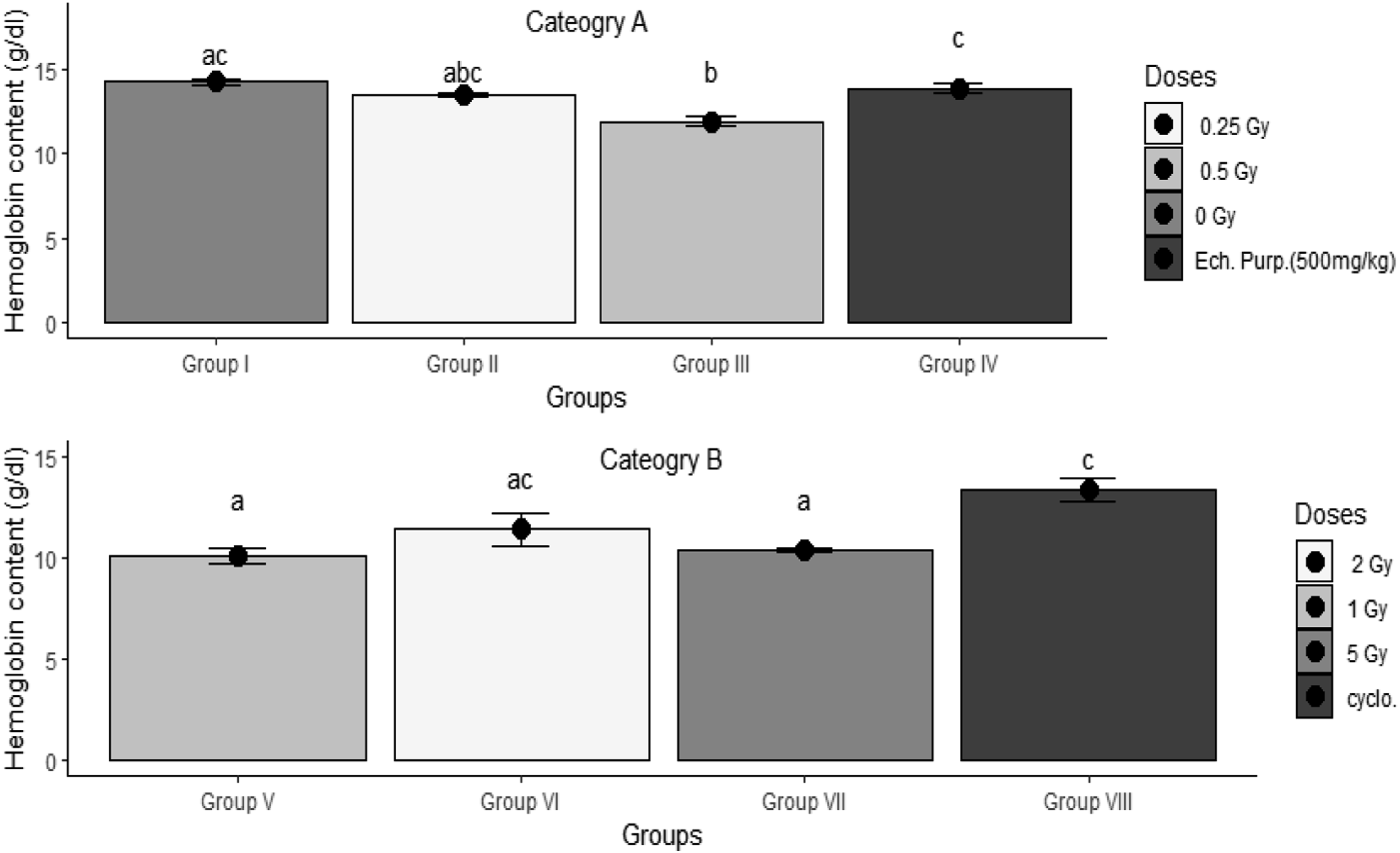
Interestingly, neither of the radiation doses, 2 and 5 Gy, induced any significant changes (P ≥ .05) in rats’ blood constituents, Figure 5
Effects on Platelet Cell Counts in Categories A and B
In
Interestingly, in Platelet (Plat) cell counts after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V–VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05). White blood cell counts (WBCs) after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V–VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05). Red blood cell counts (RBCs) after whole-body gamma radiation doses, one-day post-exposure in Category A, [Group I served as a control, Group II and III rats were irradiated at dose levels of .25 and .5 Gray, respectively, and Group IV rats received oral Echinacea purpura extract at a dose of 500 mg/kg daily for seven days] and Category B [Group V–VII were irradiated at dose levels of 1, 2, and 5 Gray, respectively, and Group VIII rats received intraperitoneal cyclophosphamide at a dose of 50 mg/kg daily for seven days]. Data represented as a Mean ± SEM. Significant difference at α = .05 (P < .05). Average values marked with the same letters are insignificant at α = .05 (P ≥ .05).


Effects on White Blood Cell Counts in Categories A and B
In
Interestingly, neither of the radiation doses, 2 and 5 Gy, induced any significant changes in rats’ white blood cells (P ≥ .05); furthermore, CP induced a significant decrement (P < .05) in the counts of WBCs.
Effects on Red Blood Cell Counts in Categories A and B
In the present work, irradiation at .25 Gy dose did not affect (P ≥ .05) the RBCS of rats compared to the un-irradiated ones. However, the .5 Gy irradiation dose induced significant decrements (P < .05) in the RBC count. In EP-treated group (IV), the counts of RBCs remained unchanged (P ≥ .05) in comparison to the control group.
Interestingly, neither of the radiation doses, 2 and 5 Gy induced significant changes (P ≥ .05) in rats’ RBC counts. Furthermore, CP induced no changes (P ≥ .05) observed in the RBCs count.
Discussion
Based on radiation protection aspects, a low radiation dose has been defined as one below 100 mGy. However, clinically, low radiation doses are practically the intermediate ones of ranges between .3 to 1.0 Gy, applied as single radiation doses for managing degenerative, benign, and inflammatory disorders.25,26
NO is a biologically active molecule involved in several physiological and pathological reactions in the body. 27 In the current work, we observed a significant rise in serum NO levels following high and low IR doses. Such effects had been previously attributed to two distinct mechanisms. HD-IR directly activates macrophages, resulting in NO generation; LD-IR activates NO production indirectly via a paracrine cytokine-dependent pathway. 28 In a similar context, .5 and 5 Gy doses increased MDA serum levels. Being one of the multiple end products of lipid peroxidation, 29 MDA overproduction is commonly observed in vivo following exposure to oxidative stress inducers such as IR and CP. Moreover, IR and pro-inflammatory cytokines were proven to produce ROS and metabolites.30-32 On the same line, CP-induced lipid peroxidation has been reported to raise MDA levels in rat erythrocytes and tissues and suggested to increase NO production is a crucial step in its main cytotoxic action,7,33,34 where NO acts as an inflammatory mediator together with being an oxidative stress pathways’ intermediate.11,35
Regarding the modulation of the IR-induced cytokine, our results showed a rise in serum levels of TNF-α following .5 and 5 Gy irradiation and a similar rise in IL-10 following 2 Gy irradiation. Interestingly, EP induced an increment in both TNF-α and IL-10 as a part of its anticipated immune-stimulant 36 and anti-inflammatory 9 effects. Nevertheless, high radiation doses induced a rise in serum IL-10 levels; in the case of 2 Gy and TNF-α levels; in the case of 5 Gy.35,37 However, CP administration decreased the serum levels of both TNF-α and IL-10, reflecting its immune-suppressive effects. 38 Earlier studies have observed and confirmed the IR-induced TNF-α production to act in collaboration with CD8+ T-cells and M1 macrophages to eliminate intracellular viruses, germs, and cancers. 29 On the other hand, in comparison to other Category B groups, Group VIII showed the lowest serum TNF-α level, implicating that HD-IR alone cannot be considered an immunosuppressive tool. Earlier investigations have concluded that high and low IR can activate the transcription and the secretion of IL-2, IFN-γ, and TNF-α; 39 nevertheless, this activation appeared stronger with high IR doses than with low IR doses.40,41
As for the anti-inflammatory cytokine, IL-10 has been reported to modulate T-cell responses and NK cell activities indirectly via IL-10’s direct action on monocyte-macrophages. 42 In addition, IL-10 activated the regulatory T cells and boosted their dynamic equilibrium with Th17 cells, a mechanism through which IL-10 is involved in the pathophysiology of several chronic inflammatory and autoimmune diseases. 43
The impact of LD-IR in eliciting changes, most commonly a shift toward a Th2 phenotype, is reported in earlier literature, 3 where monocytes release IL-10 to activate various immune cells, including macrophages, some dendritic cell subsets, and granulocyte subsets such as eosinophils and mast cells. 9 Furthermore, non-immune cells such as keratinocytes and epithelial cells 29 have been reported to produce IL-10, which might have contributed to the increase seen in Group VI.
Regarding the effect of different IR doses on blood pictures in our study, the .5 Gy group was the only low-dose group that showed decrements in Hgb concentration and RBC count, with no effects on other blood components. On the contrary, only the 2 Gy group showed a decrement in platelet counts of high IR doses. In a different context, EP administration significantly increased WBCs count, consistent with its immune-stimulant outcome. On the contrary, the CP-treated group showed an increment in Hgb concentration and decrements in platelets and WBC counts. In an earlier study, workers’ chronic exposure to IR induced a significant dose-dependent reduction in RBC count. 44 Another recent study reported an LD-IR-induced sharp rise in radiation workers’ platelet counts followed by a significant platelet count fall. 14
.25 Gy had no significant effect on the immunologic and hematological parameters measured compared to the control group. It had the same effect on serum TNF-α levels as the immune stimulant. On the other hand, CP was characterized by a decrease in all immunological parameters tested and increased lipid peroxidation damage. The only thing IR high doses, particularly 5 Gy, had in common with CP was the increased lipid peroxidation.
Conclusion
Future investigations are highly recommended aiming to understand the precise immune-stimulant/modulatory effects and mechanisms of low and high IR doses. Finally, it could be concluded that during the radio-therapeutic tumor management, the immunological impact(s) of the used radiation doses should be taken into consideration and followed-up closely in order to assess the risk/benefit ratio of their usage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The experiment was approved and carried out following the National Center for Radiation Research and Technology (NCRRT) Research Ethics Committee (Serial number of the protocol: 72 A/21).
