Abstract
Purpose:
To determine the hepatoprotective mechanisms of Heracleum candicans in rats with acute liver injury induced by carbon tetrachloride (CCl4).
Methods:
Rats were intragastrically administered H candicans twice a day for 14 consecutive days and were intraperitoneally challenged with CCl4. Alanine aminotransferase and aspartate aminotransferase were measured to indicate liver injury. Malondialdehyde antioxidant enzyme activity and tumor necrosis factor-alpha and interleukin 6 secretion were measured as liver injury indicators. Histopathological tests were conducted to determine whether H candicans ameliorated liver injury.
Results:
CCl4-induced liver injury led to significant increases in liver injury biochemical indicators transaminase and malondialdehyde activities. H candicans pretreatments inhibited these increases. Pathological sections in pretreated samples exhibited reduced vacuole formation, neutrophil infiltration, and necrosis.
Conclusion:
H candicans increases the antioxidant capacity of the liver and maintains hepatocyte function in the face of CCl4-induced injury.
Introduction
The liver is an essential homeostatic organ that is involved in detoxifying harmful drugs and chemicals. Liver injury may arise from excessive alcohol intake or exposure to toxins and can progress to severe liver diseases such as hepatitis, cirrhosis, and liver cancer. 1,2 Producing harmful intermediate products such as free radicals and redox-active reactants can induce metabolic pressure. 3 The excessive production of reactive oxygen species (ROS) induces oxidative stress and leads to degenerative and pathological diseases. 4 -6 Liver injury is closely correlated with oxidative stress.
Various toxic chemicals and drugs cause hepatocellular damage. 7 Carbon tetrachloride (CCl4) is one such potent toxin. CCl4-induced liver injury models have been used to evaluate the potential of medicines or foods to protect against hepatotoxicity. 8,9 CCl4 metabolism occurs via the cytochrome P450 system and leads to the formation of trichloromethyl radicals 7,10 that attacks lipids and proteins, causing lipid peroxidation of cell membranes and damaging liver tissue. 11 -13 Studies show that antioxidants protect oxidative stress-related liver pathologies against CCl4-induced injury by blocking deleterious lipid peroxidation reactions. 14 Some herbs and derived chemical components have proven to be good sources of antioxidants. 15 -17
The dry root of Heracleum candicans Wall. ex DC. is translated as “zhuga” in Tibetan. H candicans is distributed in Tibet, Qinghai, Yunnan, Sichuan, and other provinces in China. It grows at altitudes of 2000-4200 m and is commonly used as folk medicine. It contains coumarins, volatile oils, and other chemical compositions. 18,19 Pharmacological studies indicated that H candicans exhibits biological effects, including analgesic, anti-inflammatory, anti-bacterial, and cardiovascular protective activities. 18,20 However, studies have yet to report its hepatoprotective effects. Therefore, we investigated the hepatoprotective antioxidant effects of H candicans on CCl4-induced acute liver injury in rats.
Materials and Methods
Chemicals and Reagents
H candicans was purchased from Ganlu Traditional Medicine (Tibet, China) and was identified by the Shaanxi Chinese Medicine Institute. Bifendate (C20H18O10, DDB) (Cat#H44023176) was purchased from Baiyunshan Xingqun Pharmaceutical Co., Ltd. (Guangzhou, China). Aspartate aminotransferase (AST) kit (Cat# C010-2-1), alanine aminotransferase (ALT) kit (Cat# C009-2-1), superoxide dismutase (SOD) kit (Cat# A001-3-2), and malondialdehyde (MDA) kit (Cat# A003-1-2) were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). ELISA kits (Rat tumor necrosis factor-alpha (TNFα), Cat# ERA56RB; Rat interleukin 6 (IL-6), Cat# ERA31RBX5) were purchased from Thermo Fisher, USA.
Preparation of Plant Extract
We extracted 50 g H candicans with 600 ml ddH2O at 100°C for 1 h twice, followed by filtration. The filtrates were concentrated using a vacuum rotary evaporator to 133 ml. The extracts were stored at 4°C until further use. According to the
Animals and Treatments
Specific pathogen-free Sprague Dawley rats were obtained from the Xian Jiaotong University Animal Center (Xian, China). Rats were housed in the Xizang Minzu University Laboratory Animal Center with a 12 h-12 h light-dark cycle and regular chow and water provided ad libitum.
CCl4 was dissolved in olive oil (50% V/V, 1 mL/kg). Bifendate (C20H18O10, DDB) was used as a positive control. Rats were randomly divided into 6 groups with 10 rats in each group: (1) control with ddH2O; (2) CCl4; (3) 2.7 mg/kg body weight DDB + CCl4; (4) 0.42 g/kg body weight H candicans + CCl4; (5) 1.25 g/kg body weight H candicans + CCl4; and (6) 3.75 g/kg body weight H candicans + CCl4. Rats in the control and CCl4 groups were orally administered distilled water, and those in the DDB group were orally administered DDB in distilled water. Rats in the H candicans group were intragastrically administered H candicans twice a day for 14 consecutive days. Two hours after the final administration, all rats except those in the control group were intraperitoneally challenged with CCl4. Rats were anesthetized with an intraperitoneal injection of 1% pentobarbital sodium at 50 mg/kg body weight 24 h after CCl4 injections. Blood was collected by orbit puncture in EDTA, serum was separated, and liver tissues were removed for analysis.
Hematology Analysis
Blood samples collected by orbit puncture were analyzed using a hematology analyzer (BC-2800Vet, mindray, Shenzhen, China) immediately after rats were sacrificed.
Serum Biochemical Analysis
Blood samples were collected by eyeball puncture and centrifuged at 3000 rpm/min at 4°C for 30 min to separate the serum. Serum AST, ALT, and SOD levels were measured using spectrophotometric diagnostic kits.
Hepatic Antioxidant Analysis
We collected 150 mg liver tissue samples, rinsed them in cold phosphate-buffered saline, and centrifuged at 12,000 rpm/min for 15 min at 4°C to separate the supernatants for measurement of ALT, AST, MDA, SOD, nitric oxide (NO), and inducible nitric oxide synthase (iNOS) activity.
Cytokine Analysis
According to the manufacturers’ instructions, the concentrations of TNFα and IL-6 in liver tissue supernatant were measured using ELISA kits. The absorbance was recorded at 450 nm.
Histochemistry Analysis
Liver tissues were rapidly dissected and fixed in 4% paraformaldehyde (v/v) for 24 h before being embedded in paraffin. We then sectioned and stained samples using standard histological methods. Sections were stained with hematoxylin and eosin (H&E), and pathological changes were examined using light microscopy (Nikon, Tokyo, Japan).
Statistical Analysis
Data were expressed as means ± SEM. One-way ANOVA with Bonferonni’s post-test was used for multiple comparisons. P < 0.05 was considered statistically significant.
Results
H candicans Attenuates CCl4-Induced Increases in Inflammatory Cells
Leukocyte exudation is a typical feature of inflammation that initiates immune responses. In response to acute injury or bacterial infection, many neutrophils are produced. We performed hematology analysis to determine the composition of inflammatory cells. Hematology analysis was conducted immediately after collection by an orbital puncture. White blood cells, especially neutrophils, were significantly higher than those of the control group. H candicans pretreatment inhibited these increases (Figure 1). These findings suggest that H candicans attenuates inflammatory cell infiltration in CCl4-induced acute liver injury.
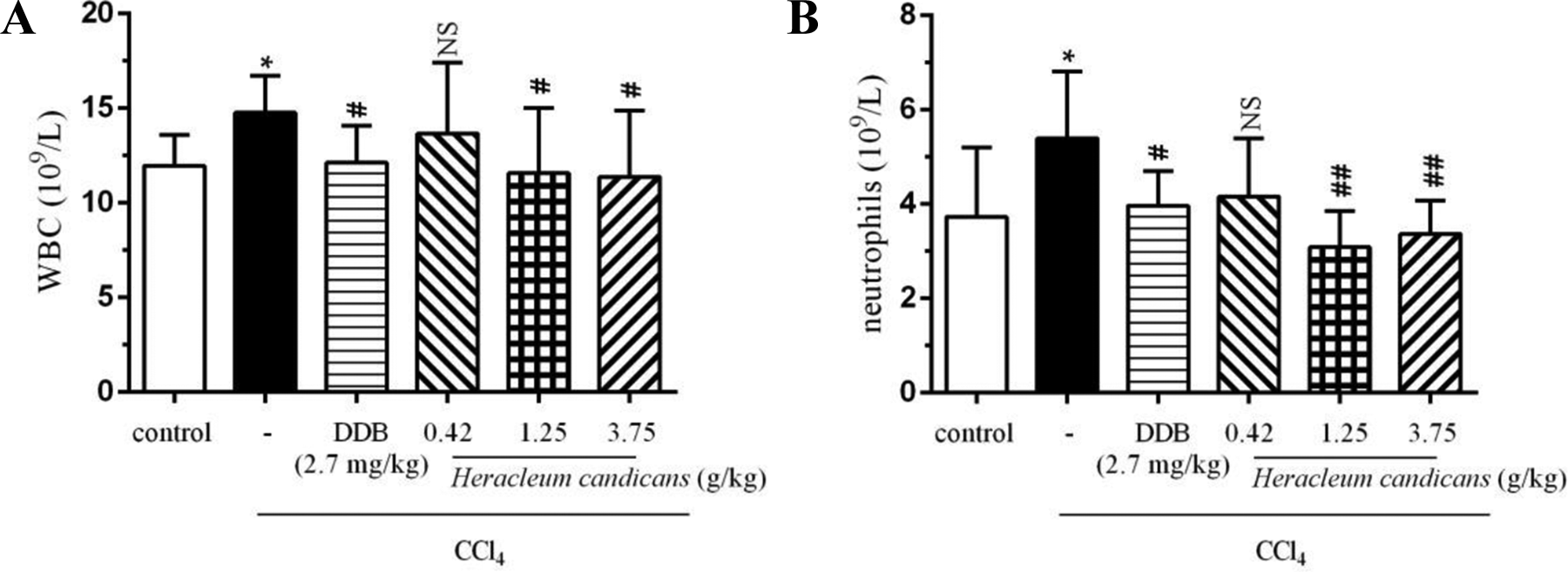
H candicans attenuates the increase in inflammatory cells induced by CCl4. Rats were randomly divided into 6 groups: (1) control with ddH2O, (2) CCl4, (3) 2.7 mg/kg body weight DDB + CCl4, (4) 0.42 g/kg body weight H candicans + CCl4, (5) 1.25 g/kg body weight H candicans + CCl4, (6) 3.75 g/kg body weight H candicans + CCl4. (A) WBC and (B) neutrophils concentration were determined by routine blood test. *P < 0.05 vs control group, NS, not significant, #P < 0.05 and ##P < 0.01 vs CCl4 group.
Effects of H candicans on ALT, AST, and SOD Activity
ALT and AST are liver function markers. After treatment, the liver functions biochemical index changes in the serum of rats in each group were examined. As shown in Figure 2A and B, ALT and AST levels in the CCl4 group were significantly higher than in the control group. By contrast, the H candicans appeared to prevent these increases.

Effects of H candicans on blood ALT, AST, and antioxidative parameter SOD activities. Effects of H candicans on serum (A) ALT, (B) AST, and (C) SOD levels in rats treated with CCl4. *P < 0.05 and ***P < 0.001 vs control group, NS, not significant, # P < 0.05, ## P < 0.01 and ### P < 0.001 vs CCl4 group.
In the liver, collagen synthesis participates liver fibrogenesis, which is triggered by lipid peroxidation caused by oxidative stress, suggesting that liver cell damage is correlated to oxidative stress. 21,22 Figure 2C shows that SOD activity was reduced in the CCl4 group. H candicans administration appeared blunt this effect.
Effects of H candicans on Inflammatory Cytokines and Antioxidant Enzymatic Activities in the Liver
The hepatoprotective effects of H candicans in rats with CCl4-induced liver injury are presented in Figure 3A and B. CCl4 treatments significantly increased ALT and AST levels. Pretreatment with H candicans blunted these effects.

Effects of H candicans on inflammatory cytokines and antioxidant enzymatic activities in liver. Effects of H candicans on the liver (A) ALT, (B) AST, (C) SOD, (D) MDA, (E) NO, (F) iNOS, (G) TNFα, and (H) IL-6 levels in rats treated with CCl4. *P < 0.05 and **P < 0.01 vs control group, NS, not significant, # P < 0.05, ## P < 0.01 and ### P < 0.001 vs CCl4 group.
Figure 3C shows that the activity of hepatic SOD was reduced in the CCl4 group. H candicans administration appeared to improve SOD activity, though not significantly. We measured MDA levels to evaluate the degree of hepatic lipid peroxidation. We found that MDA levels were significantly higher in the CCl4 group, and those in the H candicans groups were significantly lower (Figure 3D). Hepatic NO levels and iNOS activity were lower in the CCl4 group. H candicans treatment blunted both effects (Figure 3E and F).
Figure 3G and H shows the levels of inflammatory cytokines TNFα and IL-6 in CCl4-induced acute liver injury. The levels of these cytokines in the CCl4 group were significantly higher than those in the control group. Treatment with H candicans significantly reduced these levels, suggesting that H candicans protects against CCl4-induced liver damage.
H candicans Relieves CCl4-Induced Hepatotoxicity
Histopathological examinations revealed that the liver tissue of the normal control group had normal liver architecture, including distinct hepatocytes, sinusoidal spaces, and clear central veins. The liver tissue of the CCl4-treated group showed a high number of necrotic cells, hepatocyte degeneration, and inflammatory cell infiltration. Rats administrated H candicans showed reduced hydropic degeneration and fewer necrotic and inflammatory cells (Figure 4 and Table 1).

H candicans improves hepatotoxicity induced by CCl4. Hepatic histological analyses of H candicans on CCl4-induced acute liver damage. Hepatocyte degeneration were indicated by green arrow and inflammatory cell infiltration were indicated by blue arrow. Liver tissues were subjected to H&E staining (400×).
Quantitative Summary of the Protective Effects of H candicans on CCl4-Induced Hepatic Damage.
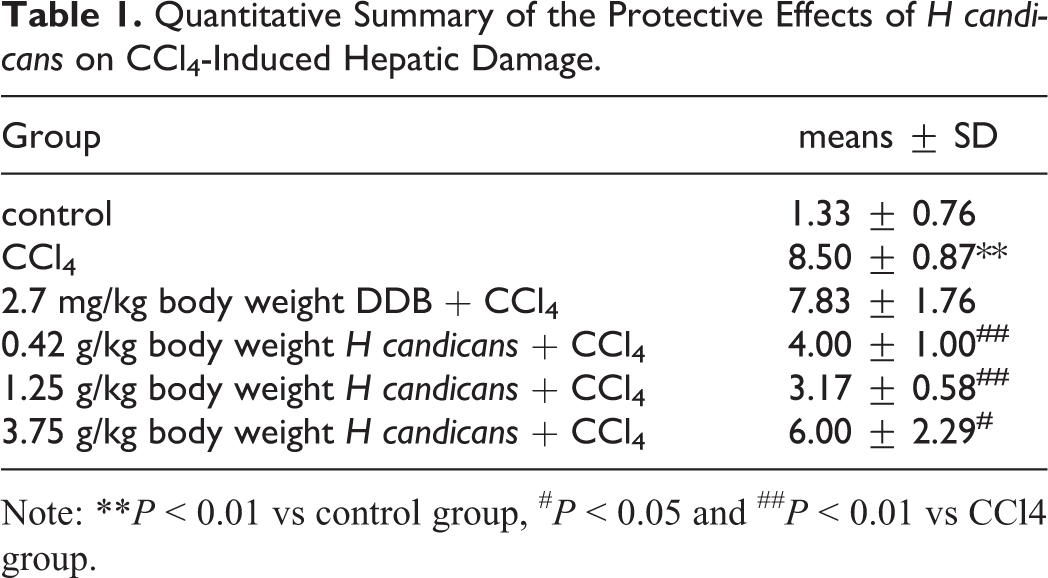
Note: **P < 0.01 vs control group, # P < 0.05 and ## P < 0.01 vs CCl4 group.
Discussion
CCl4-induced liver injury models are commonly used to investigate the hepatoprotective effects of natural medicine. Changes in transaminase levels have been attributed to liver damage. 23 -25 We demonstrated that treatment with H candicans prevented increased AST and ALT levels in the model, demonstrating that the extract prevents hepatocellular damage.
Lipid peroxidation in liver cell membranes is an important oxidative stress indicator. We detected increased levels of MDA after CCl4 administration. This suggests liver cell damage and the failure of antioxidants to handle excessive levels of free radicals. 26,27 By contrast, H candicans significantly reduced MDA levels, suggesting that H candicans protects against free radicals, protecting hepatocytes from hepatopathy.
Inflammation is initiated by CCl4-induced hepatotoxicity, followed by the release of inflammatory cytokines, including iNOS, TNFα, and IL-6. NO is an inflammatory mediator that reacts with superoxide anions to form a robust cytotoxic oxidant, causing lipid peroxidation and cellular damage. 28,29 TNFα and IL-6 are critical cytokines in inflammatory responses, and their levels are elevated during liver damage. 30 We found that the production of TNFα and IL-6 were significantly inhibited by treatment with H candicans.
Histological analyses indicated that hepatic cell injury was accompanied by hydropic degeneration, inflammation, and necrosis. The increasing permeability of the liver cell membrane causes metabolic disorders, inhibits protein synthesis, and produces necrosis-inducing cellular degradative enzymes. The observed liver damage was significantly improved by treatment with H candicans. These results suggest the hepatoprotective effect of H candicans on CCl4-induced hepatotoxicity.
The liver plays a vital role in metabolism. 31,32 Liver detoxification converts toxic materials into harmless metabolites that are then excreted. 33 Liver diseases can be caused by toxic chemicals, drugs, and viral infiltration through ingestion or infection. These toxins induce the production of ROS and cause severe injuries. 5 Many medicinal herbs express a high antioxidant activity that reduces lipid oxidation and protects hepatocytes against chemically-induced liver carcinogenesis. Our findings suggest that H candicans displays goods hepatoprotective effects. Nevertheless, additional studies should be conducted to determine the mechanisms underlying these effects.
Footnotes
Authors’ Note
Jie Li and Dan Song contributed equally to this work.
Acknowledgments
This work was supported by grants from the Natural Science Foundation of Tibet Autonomous Region (Grant No. XZ2018ZRG-86(Z)), the Young Scholar Cultivation Project of Xizang Minzu University (Grant No. 20MDX02), the Research Found for Advanced Talents of Xizang Minzu University (Grant No. RCYJ602111) and by the National Natural Science Foundation of China (Grant No. 81660722).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Natural Science Foundation of Tibet Autonomous Region (Grant No. XZ2018ZRG-86(Z)), the Young Scholar Cultivation Project of Xizang Minzu University (Grant No. 20MDX02), the Research Found for Advanced Talents of Xizang Minzu University (Grant No. RCYJ602111) and by the National Natural Science Foundation of China (Grant No. 81660722).
