Abstract
Hormetic-biphasic dose response relationships are reported herein for human endothelial progenitor cells involving estradiol, nicotine, the anti-diabetic agent pioglitazone, resveratrol, and progesterone. In general, these studies demonstrate the capacity of these agents to enhance EPC proliferation and angiogenesis functional applications, having a focus on repairing endothelial tissue damage due to acute injury (e.g., stroke), as well as damage from chronic conditions (e.g., atherosclerosis) and normal aging processes.
Introduction
Endothelial repair was originally seen as a local type of process involving the migration and proliferation of endothelial cells from a nearby uninjured cellular zone. However, it is now known that vascular healing requires not only cells located within vessel wall environments but also circulating 1 bone marrow–derived stem cells (BMSCs).2-5 A subset of these BMSCs, called endothelial progenitor cells, are present within the peripheral blood, with the potential to proliferate and differentiate into endothelial cells, 6 leading to the restoration of endothelial function, enhancement of angiogenesis and overall vascular repair, preventing atherosclerosis. The present paper provides an assessment of the occurrence of chemically induced hormetic dose responses in human endothelial progenitor cell functions, including cell proliferation, cell migration, cell adhesion, and the process of angiogenesis, their underlying mechanistic foundations and therapeutic applications.
Hormesis Overview
While hormesis has been substantially evaluated in the biological, toxicological, and biomedical literature, the use of the terms hormesis or hormetic within the stem cell literature is extremely limited, affecting the capacity for a standard key word search strategy, further suggesting the need to provide a brief overview of the hormesis concept for those researchers in the area of stem cells with particular interest in cell proliferation and cell differentiation and their dose response features.
Hormesis is a biphasic dose/concentration response, displaying a low dose/concentration stimulation and a high-dose/concentration inhibition.7-9 Its quantitative characteristics include a maximum stimulatory response typically between 30% and 60% greater than the control group (Figure 1) along with a stimulatory width that is usually in the 10–20-fold range but may show considerable variability, not uncommonly being greater than 50-fold.10-12 The hormetic response results from a direct subtoxic (hormetic) dose or a subtoxic (hormetic) preconditioning dose and a subsequent toxic dose.13,14 The hormesis concept shows considerable generality, being independent of biological model (e.g., microbes, plants, animal models, and humans), endpoint, level of biological organization (i.e., cell, organ, and organism), in vitro and in vivo evaluations, inducing agent, and mechanism.15-17 Comprehensively integrated evaluations of hormetic dose responses for both chemicals and ionizing radiation provide historical foundations of hormesis from first reporting in the 1880s concerning the effects of multiple chemical disinfectants on the growth/metabolism of yeast to the present.18-24 General representation of the hormetic dose response (modified from: Calabrese and Baldwin, 1998
25
).
As a result of the general lack of linkage of hormesis and stem cells in the literature, including EPCs, it was necessary to develop a broader and more general search strategy. Using principally PubMed and Web of Science databases, key words such as stem cells, cell proliferation, cell differentiation, EPCs, biphasic dose responses, low dose stimulation, as well as hormesis and hormetic and their combinations were used. In addition, all papers obtained were cross-referenced. Further, all relevant papers were assessed for each article that cited these papers (using Web of Science) and checked for relevance. Finally, all active research groups in the area were followed for all their relevant publications.
Estrogen
Estrogens provide protective roles in the vascular system, including functioning of the endothelial cell layer. Within this biological context, estrogens play an important role in the endothelial cell restoration process following damage.26-30 The capacity of estrogen to affect EPC proliferation was assessed by Foresta et al.
1
using EPCs from healthy adult donors over a 10,000-fold concentration range (Figure 2). These preliminary findings indicate that the estrogen treatment enhanced cell proliferation in a biphasic manner, with the response being optimized between 10–1000 pM, a 100-fold concentration range. The stimulatory response was mediated by the estrogen receptor-α (ER-α), with no involvement of the ER-β receptor. The estrogen concentration range that was associated with the enhanced EPC proliferation effect was similar to that reported for women under hyperstimulation with in vitro fertilization techniques. Effects of β-estradiol on human endothelial progenitor cell proliferation (modified from: Foresta et al., 2007) *= P ≤ .05.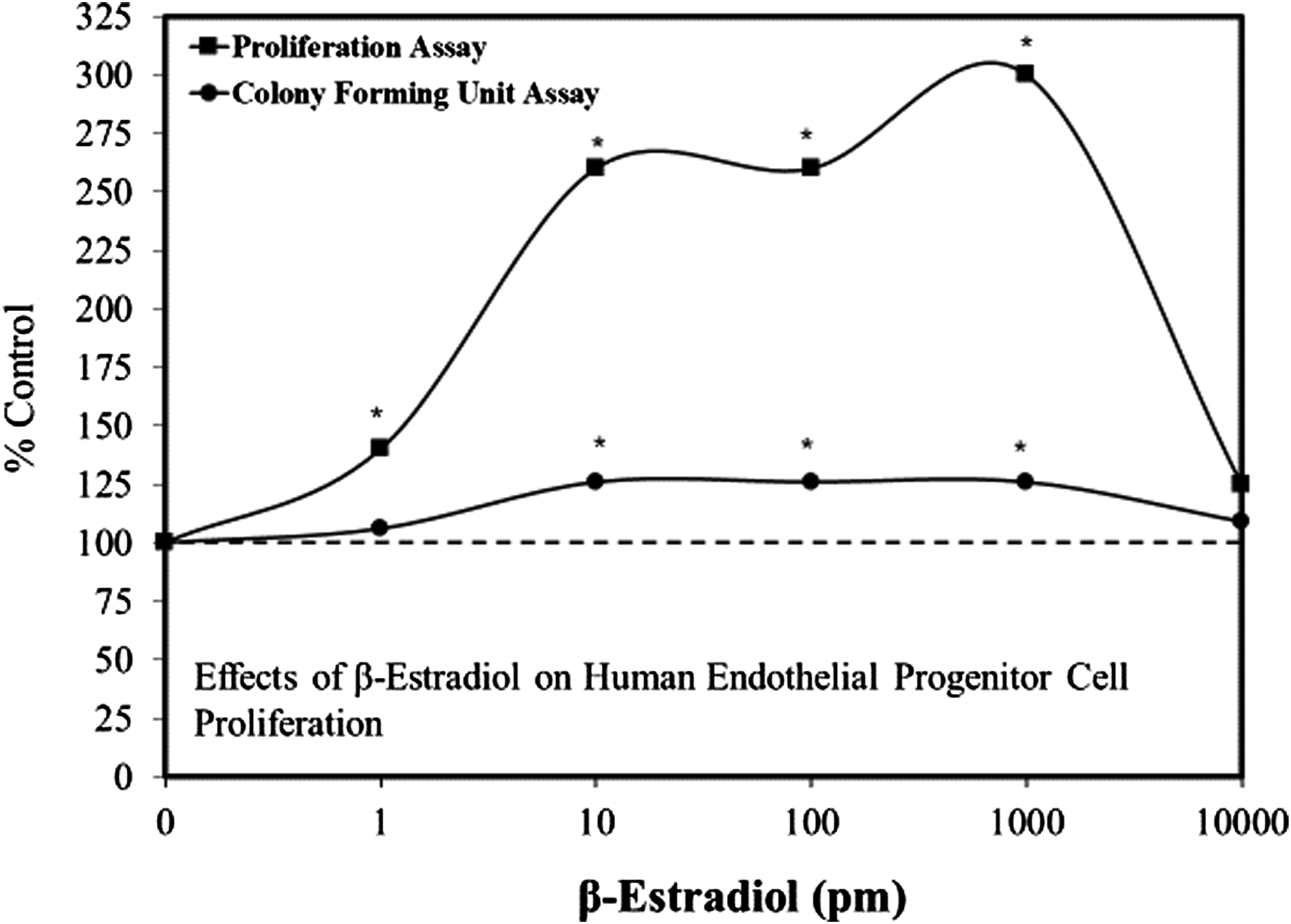
Progesterone
Progesterone, an endogenous sex hormone, has significant protective effects in the vascular system. Such protection is mediated, in part, due to its capacity to enhance EPC proliferation.
31
In an effort to explore further the effects of progesterone on EPC, Yu et al.
32
assessed its capacity to affect multiple functions including tube formation and length, cell adhesion, cell migration, and protection against induced damage due to cortical impact injury. The progesterone treatment biphasically affected multiple endpoints including cell tube formation, cell adhesion, and cell migration, with all showing the optimal concentration at 10−9 M (Figure 3). Follow-up experiments with UPA, a progesterone receptor antagonist, blocked the stimulatory response for multiple endpoints at the optimal 10−9 M concentration. This was also the case for the effect of progesterone on the secretion of vascular epithelial growth factor (VEGF). These findings suggested that low concentrations of progesterone enhanced EPC angiogenesis activities via the progesterone receptor B (PR B) while the inhibitory effect at the higher concentrations was hypothesized to be mediated by PR A. Effects of progesterone on endothelial progenitor cell adhesion and migration (modified from: Yu et al., 2017) * = P < .05.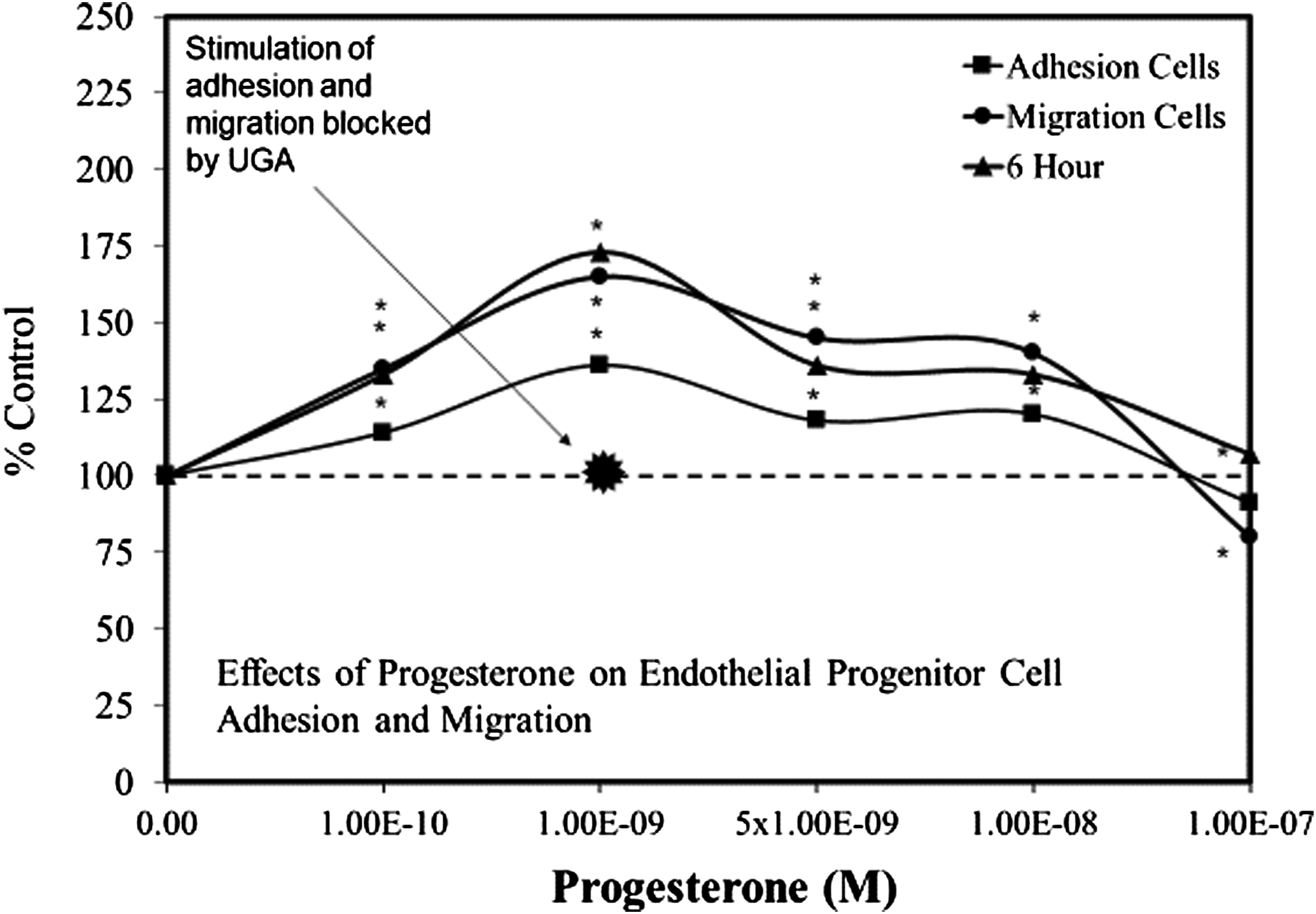
Nicotine
While high concentrations of nicotine can cause endothelial injury, lower concentrations can increase endothelial proliferation, reduce apoptosis, and enhance capillary network formation.
33
These findings lead Wang et al.
33
to explore the effects of nicotine on circulating human EPCs. This study provided the first report that nicotine induced biphasic concentration responses on cell proliferation, migration, adhesion, and vasculogenesis at physiologically relevant concentrations, being maximal at 10–8 mol/L (Figure 4). This concentration of nicotine is comparable to those found in the blood of smokers (i.e., ∼60 to 100 nmol/L).
34
Follow-up studies by Heeschen et al.
35
suggested that the mechanism for the low concentration effects was related to the capacity of nicotine to stimulate the endothelial nicotinic acetylcholine receptor (hACHR) which induces angiogenesis. Effects of nicotine on endothelial progenitor cells (modified from: Wang et al., 2004) * = P ≤ .05.
Pioglitazone: Anti-Diabetic Agent
Circulating EPCs have a critical role in the process of endothelial regeneration following arterial damage.36,37 The capacity to repair such damage is affected by multiple factors such as age, gender, and physical training status. Coronary artery disease is the leading cause of mortality of those with type 2 diabetes, a condition associated with significant deficits in EPC functions (e.g., altered proliferation and adhesion). Within the context of treating diabetic patients, considerable attention has been directed toward the thiazolidinediones (e.g., rosiglitazone and pioglitazone), peroxisome proliferator-activated receptor (PPAR)-γ agonists, and insulin-sensitizing agents. Further interest in these agents emerged from studies showing that they offered benefits beyond affecting only glycemia. Such additional benefits include the capacity to enhance the differentiation of EPCs, an effect mediated by PPAR-γ. Further, these agents affect potential benefits via the activation of transforming growth factor (TGF)-β1, enhancing the proliferation of EPCs in an hormetic-like biphasic manner.
38
These observations led Redondo et al.
39
to assess the effects of pioglitazone on human EPC function while elaborating the roles and interactions of PPAR-γ and TGF-β1. While evaluating the effects of pioglitazone over a 100-fold concentration range (.1 to 10 μM) these authors reported that pioglitazone induced a biphasic concentration effect, with the optimal response at 1.0 μM (Figure 5). Since the pioglitazone did not significantly affect proliferation and apoptosis, the increase of EPC number may be due to enhanced adhesion and differentiation, conclusions that were supported by experiments blocking PPAR-γ and TGF-β1 by antagonists. An integrated mechanistic assessment suggests that the pioglitazone low concentration stimulatory effect was mediated by PPAR-γ whereas TGF-β1-mediated the inhibition. Effects of pioglitazone on human endothelial progenitor cell adhesion on fibronectin (modified from: Redondo et al., 2007) * = P ≤ .05.
Resveratrol
Since endothelial cell injury/death can play an important role in the pathogenesis of intimal hyperplasia in response to vascular injury, a therapeutic strategy that stimulates early re-endothelialization of the damage area would prevent intimal lesion development while compromising long-term patency. Since there was a strong relationship of moderate consumption of red wine with a decreased occurrence of coronary artery disease in experimental models and in epidemiological studies, Gu et al.
40
assessed the effects of resveratrol on the angiogenesis activation and eNOS expression of isolated human endothelial cells in vitro and the effects of resveratrol on the mobilization of endothelial cells from bone marrow. Employing 4 concentrations (1, 5, 15, and 60 μM), Gu et al.
40
reported the occurrence of an hormetic-like biphasic concentration response for multiple endpoints: endothelial cell proliferation, cell migration, cell adherence, and eNOS expression/concentrations. These findings were striking in their qualitative and quantitative consistency along with the optimal stimulatory response for each endpoint being 1.0 μM. Likewise, the maximum stimulatory response for each endpoint was in the 40 to 60% range. The follow-up findings of Gu et al.
40
were supported in follow-up in vivo experimental investigation using a rat model. These experiments indicated that low doses of resveratrol increased the mobilization of endothelial cells, facilitated re-endothelialization, and diminished the occurrence of neointimal formation and up-regulation of eNOS following an induced balloon injury. The Gu et al.
40
findings were strongly supported by Xia et al.
41
who also reported hormetic-like biphasic dose response of resveratrol on endothelial progenitor cell proliferation and cell migration (Figures 6(a) and (b)). They also reported that resveratrol induced hormetic effects by altering teleromerase activity. A Effects of resveratrol on cell proliferation of endothelial progenitor cells (modified from: Xia et al., 2008) * = P ≤ .05. B Effects of resveratrol on migration of endothelial progenitor cells (modified from: Xia et al., 2008) * = P ≤ .05.
Discussion
This paper documents the occurrence of hormetic dose responses of endothelial stem cells. The nature of research in this area has focused on the capacity of EPCs to enhance the repair of damaged endothelial cells due to traumatic injury such as from a stroke or in the course of chronic disease such as type 2 diabetes, which can damage endothelial cells, and contributes to the development of atherosclerosis. The quantitative features of the hormetic dose response (N=18) reported herein are consistent with those reported in the general hormesis literature with the median maximum stimulation 162.5% and with median stimulatory range of 100-fold.
The present analysis demonstrates that the hormetic potential of EPCs may affect either a post-trauma beneficial or undesirable response. For example, in the case of estrogens, there is the capacity to enhance tissue repair of damaged vasculature. However, it is well known that estrogen may have a tumor promotional effect.42,43 One of the mechanisms by which this occurs could be via the enhancement of angiogenesis in developing tumors. While the negative features of nicotine have been emphasized in the literature for tumor promotion via enhanced angiogenesis 44 it may also be the case for other agents with EPC proliferation potential. Thus, the role of hormetic effects on endothelial cells has the potential to promote health by repairing damaged endothelial cells due to normal aging and enhance some disease processes or other conditions or to play a role in tumor promotion.
The present findings complement and extend a recent extensive effort to discover, document, and assess the occurrence and biological/biomedical significance of hormetic dose responses in stem cell biology. Hormetic dose responses have been now reported to be commonly reported for adipose derived stem cells (ADSCs), 45 apical papilla stem cells (APSCs), 46 bone marrow stem cells (BMSCs), 47 dental pulp stem cells (DPSCs), 48 embryonic stem cells (ESCs), 49 neuronal stem cells (NSCs), 50 and periodontal ligament stem cells (PDLSCs). 51 Based on the substantial occurrence of hormetic dose responses affecting cell viability, cell survival, cell proliferation, cell differentiation, and a capacity to enhance resilience to a broad spectrum of inflammatory conditions, a broad spectrum of stem cell types use adaptive strategies that conform to the quantitative features of the hormetic dose response, displaying a substantial degree of generality. However, despite these widespread occurrences of hormesis within the stem cell literature these findings are generally unrecognized as general dose response concept clinical implications. However, the recent spate of papers on the role of hormesis in stem cell functioning is likely to enhance the recognition of the significance of hormetic-biphasic dose responses and their role in stem cell biology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: EJC acknowledges longtime support from the US Air Force (AFOSR FA9550-19-1-0413) and ExxonMobil Foundation (S18200000000256). The U.S. Government is authorized to reproduce and distribute for governmental purposes notwithstanding any copyright notation thereon. The views and conclusions contained herein are those of the author and should not be interpreted as necessarily representing policies or endorsement, either expressed or implied. Sponsors had no involvement in study design, collection, analysis, interpretation, writing, and decision to and where to submit for publication consideration.
