Abstract
This paper provides an assessment of hormetic dose responses in epidermal stem cells (EpSCs) in animal models and humans, with emphasis on cell proliferation and differentiation and application to wound healing and aging processes. Hormetic dose responses were induced by several agents, including dietary supplements (eg, luteolin, quercetin), pharmaceuticals (eg, nitric oxide), endogenous agents (eg, growth/differentiation factor 5), and via diverse chemical means to sustain steaminess features to retard aging and disease onset. While hormetic dose responses have been extensively reported in a broad spectrum of stem cells, this area has only been explored to a limited extent in EpSCs, principally within the past 5 years. Nonetheless, these findings provide the first integrated assessment of hormesis and EpSC biology within the context of enhancing key functions such as cell proliferation and differentiation and resilience to inflammatory stresses. This paper assesses putative mechanisms of hormetic responses in EpSCs and potential therapeutic applications to prevent dermatological injury and disease.
Keywords
Introduction
Over the past year a series of papers have been published on the occurrence of hormesis with a broad spectrum of stem cells, including adipose, 1 neural, 2 embryonic, 3 dental pulp, 4 periodontal ligament, 5 muscle, 6 apical papilla, 7 endothelial progenitor cells, 8 and induced pluripotent stem cells (PSC). 9 The present paper represents an extension of this integrative, yet exploratory survey of hormesis and stem cells, with a focus on epidermal stem cells. The epidermis is derived from embryonic ectoderm cells, with these cells proceeding via a sequential process to form a complex arrangement of basal, spinous, and granular layers. 10 The skin epidermis includes sweat glands, hair follicles, and sebaceous glands that have immune regulatory, pigmentation and sensory functions. 11 The self-renewal and damage repair functions of the skin are highly contingent on the epidermal skin stem cells for compensatory proliferation and cell differentiation. 12 The present paper assessed the occurrence of hormetic dose responses in epidermal stem cells. This paper represents the first attempt to identify and assess experimental literature in this area. While a range of hormetic examples are presented, none of the identified papers assessed their biphasic dose response within the framework of hormesis. This lack of recognition of the hormetic concept in stem cell biology was also the case with other stem cells. Thus, prior to providing the assessment of hormetic effects of epidermal stem cells a brief overview of the hormetic concept will be presented.
Hormesis Overview
Since this assessment represents the first comprehensive paper on the role of hormesis in wound healing, it is necessary to provide a brief overview on the concept of hormesis. Hormesis is a biphasic dose/concentration response. It displays a low-dose/concentration stimulation and a high-dose/concentration inhibition.13-16 It exhibits specific quantitative characteristics with a maximum stimulatory response typically between 30% and 60% greater than the control group (100%) (Figure 1). The hormetic stimulatory dose/concentration range is approximately 10–20-fold but may show considerable variability, not uncommonly exceeding 50 fold. The hormetic response results from a direct subtoxic (hormetic) dose, a subtoxic (hormetic) preconditioning dose, and a subsequent toxic dose,17,18 or a modest overcompensation stimulation following an initial disruption in homeostasis.
19
The hormetic dose/concentration response exhibits broad generality, being independent of biological model (eg, microbes, plants, animal models and humans), endpoint, level of biological organization (ie, cell, organ, organism), in vitro and in vivo evaluations, inducing agent, and mechanism.20-23 Comprehensively integrated evaluations of hormetic dose responses for both chemicals and ionizing radiation provide historical foundations of hormesis from inception in the experimental literature, from the 1880s to the present.24-30 General representation of the hormetic dose response (modified from: Calabrese and Baldwin
32
).
Growth/Differentiation Factor 5 (GDF-5)
Growth differentiation factor 5 (GF-5), which is a member of the bone morphogenetic protein family, was shown by Schiefer et al
31
to enhance wound healing in a full thickness wound study. These findings led to the hypothesis that GDF-5 may have the potential to enhance the proliferation of EpSCs.
10
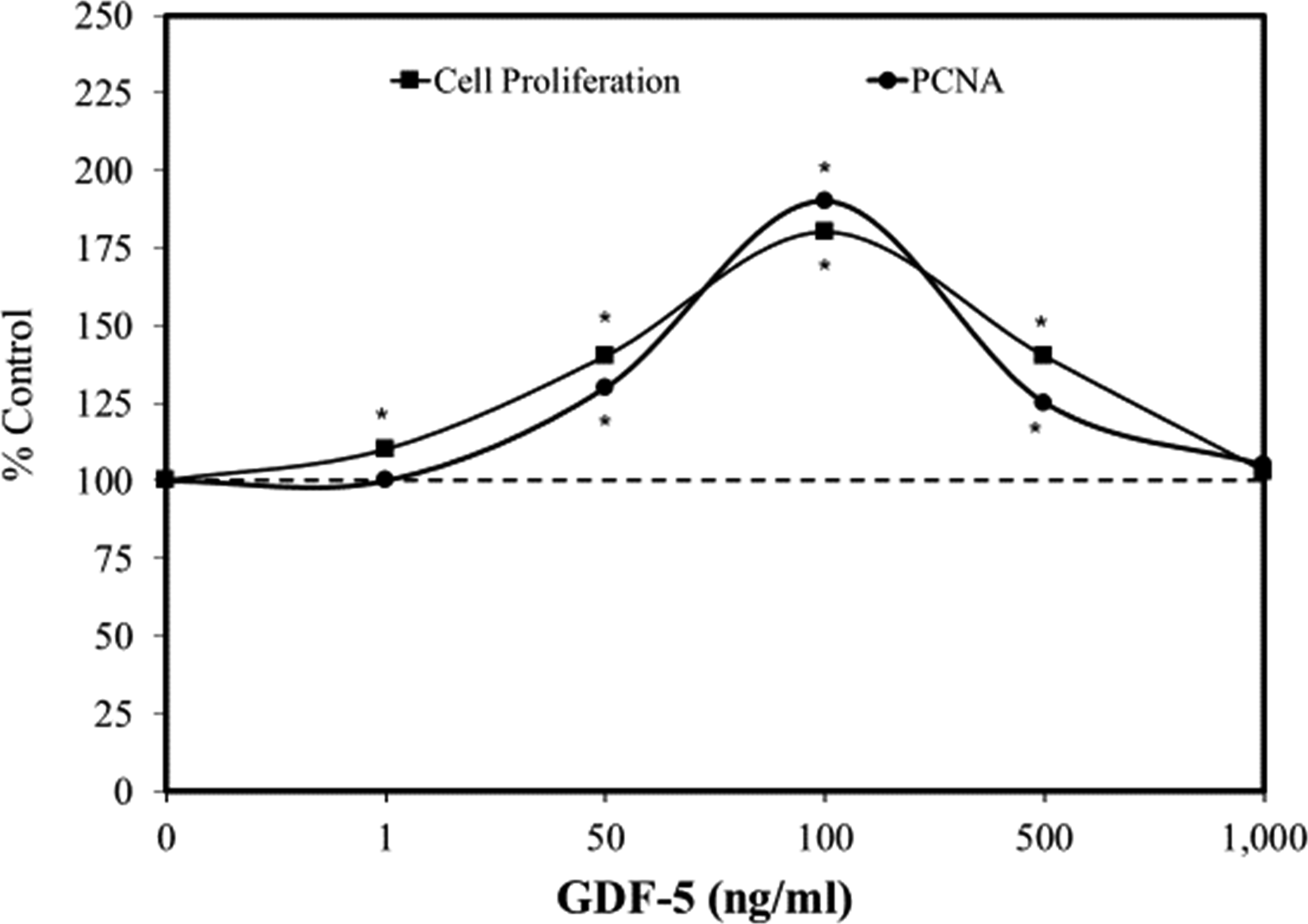
GDF-5 is a bone morphogenetic protein, which belongs to the TGF-β family, further supporting its potential likelihood to enhance cell proliferation. A series of experiments showed that GDF-5 induced proliferation of mouse epidermal stem cells at 24, 48, and 72 hour exposures, each in an hormetic manner
1
(Figure 2). Other experiments, likewise, showed that GDF-5 enhanced the production of PCNA protein, with the stimulatory pattern closely matching that of the cell proliferation assay.
10
Follow-up studies revealed that GDF-5 induced a dose dependent profile for Foxg1/cyclin D1 protein expression, with both tracking closely with the cell proliferation and PCNA biphasic concentration responses (Figure 3). Cell proliferation at the optimal concentration of GDF-5 was reduced to control group values using several means to silence Foxg1/cyclin D1. Follow-up in vivo studies confirmed the in vitro findings preventing the induction of cell proliferation via silencing methods. Since GDF-5 enhances EpSC proliferation it suggested possible application for wound healing. Effects of growth differential factor 5 (GDF-5) on cell proliferation and PCNA in mouse epidermal stem cells (modified from: Zhao et al.
10
). Effects of growth differential factor 5 (GDF-5) on key protein expressions that track with cell proliferation in mouse primary epidermal stem cells (modified from: Zhao et al.
10
).

Nitric Oxide—Wound Healing
Nitric oxide (NO) has a critical role in wound healing and in the support and maintenance of homeostasis. The first report of an explicit hormetic effect of NO on EpSCs in the wound healing process was given by Zhan et al
33
concerning cell proliferation. Using the NO donor, SNAP (S-nitroso-N-acetyl penicillamine), an hormetic-biphasic concentration response occurred at 12 and 24 hours. Subsequent cellular evaluations concerning the expression of integrin B and cytokeratin-19 (CK19) characterized the response of isolated cells as EpSCs. While the EpSCs showed a stimulatory concentration response at low exposure levels (10 and 100 µMol/L), inhibition was reported at 500 µMol/L; the SNAP treatment was stimulatory for cell migration at this inhibitory concentration for the above noted endpoints (eg, cell proliferation). These preliminary investigations led to a more detailed series of investigations several years later by this research team which explored the effects of both SNAP and spermine on cell proliferation using the CCK-8 and thymidine uptake endpoints, as well as their impact on Fog1 and c-Myc protein expression.
34
The follow-up study confirmed the hormetic concentration response with the optimal stimulation at 100 µM for both cell proliferation estimation parameters (CCK-8 and thymidine uptake) (Figure 4). Spermine also induced an hormetic response showing the same quantitative features of SNAP. However, the optimal stimulation was at a concentration 1/10 that of SNAP, due to the enhanced NO release capacity of spermine. In addition, they reported that L-arginine, a nitric oxide synthesis substrate, also significantly enhanced EpSC proliferation but only one concentration was assessed. Foxg1 and c-Myc were also biphasically affected by the NO treatment. When the expression of these proteins was silenced, the cell proliferation was blocked. It was found that NO-induced transcription of c-Myc was regulated by Foxg1-mediated c-Myc promoter activity. A similar hormetic pattern for EpSC cell migration was also reported by Zhan et al,
35
with the optimal response at 100 µM (Figure 5). However, there was inhibition at 300 µM whereas these concentrations were stimulatory for cell proliferation. The mechanisms by which NO promoted EpSC migration was measured via cGMP-Rho GTPase signaling. The small GTP-binding protein of the Rho family, such as RhoA, Rac1, and CdC42, regulate actin-based structure formation which is strong determinants of cell migration, affecting cytoskeletal dynamics, and cell motility. Blocking of cGMP-Rho-GTPase signaling prevented NO-induced EpSC migration. Effects of S-nitroso-N-acetylpenicillamine (SNAP) on primary human epidermal stem cells (modified from: Zhan et al.
35
). Effects of S-nitroso-N-acetylpenicillamine (SNAP) on human epidermal stem cell migration in vitro (modified from: Zhan et al.
35
).

Epidermal Stem Cell and Human Hair Growth
The role of polyamines (spermidine, putresence, and spermine) in human hair growth is an area of active research. Polyamines have long displayed an essential role in hair growth, affecting DNA replication and protein synthesis. Since the hair follicle is one of the most proliferative organs in mammals, it has focused attention on the role of EpSCs in hair growth. In studies on the effects of spermidine on hair growth, Ramot et al 36 used keratin-15 (K15) expression and K15 promoter-driven green fluorescent protein (GFP) expression as a system to enable human epithelial hair follicle (HF) stem cell function. In their studies, spermidine upregulated K15 and K19 gene expression as well as colony forming efficiency and long term proliferation of human HF EpSCs. Thus, this study provided the first evidence that spermidine has a novel role in human EpSC biology, via the hormetic upregulation of K15 and K19 expression both in vitro and in vivo. These findings are consistent with reports that ornithine decarboxylase (ODC) is expressed in the bulge region of the HF where it is colocalizes with those of K15 and K19 expression. While polyamines affect keratin composition of wool follicles, 37 it was not previously known that polyamines could upregulate the expression of human EpSC-associated keratins and to do so in an hormetic fashion.
Luteolin
Epidermal stem cells in the basal layer of the epidermis have a significant role in epidermal regeneration and have been widely used in experimental wound healing research. Luteolin-7-glucoside (L7G) has been reported to enhance wound re-epithelialization and to increase collagen deposition.
38
Follow-up research by Wan et al
39
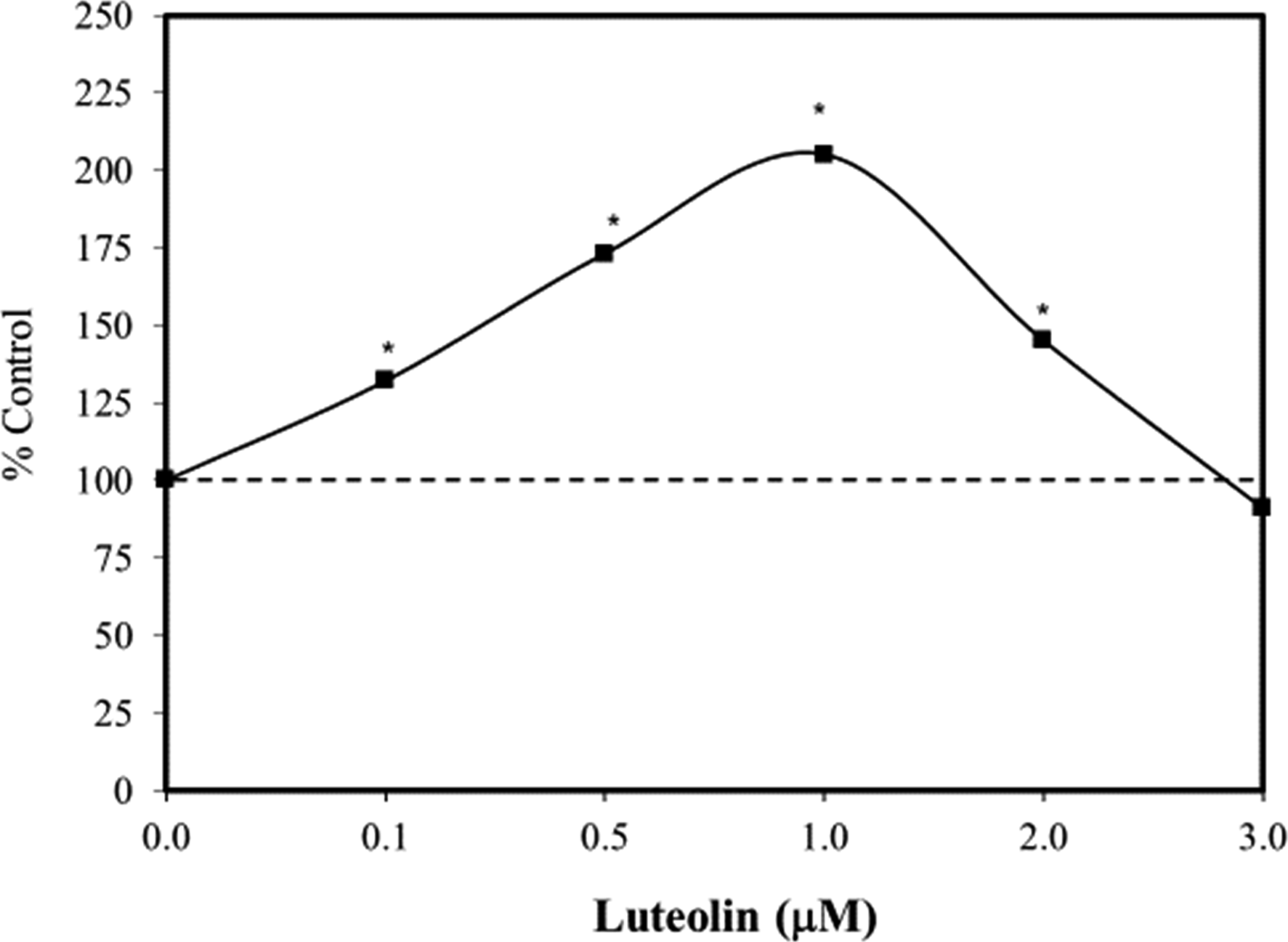
sought to clarify the effect of L7G on the proliferation of human EpSCs and underlying mechanisms. Using the MTT assay, L7G biphasically enhanced stem cell proliferation (ie, 0.1–2.0 µM) (Figure 6). Mechanistic follow-up investigations indicated that L7G induced EpSC proliferation via the upregulation of β-catenin, c-Myc, and cyclin expression at 1.0 µM, the only concentration tested for these marker endpoints. These findings suggest that L7G may have considerable therapeutic application in enhancing dermal wound healing via integrated antioxidant and anti-inflammatory mechanistic processes. The discussion of Wan et al
39
concerning the role of L7G-induced anti-inflammatory activity and its underlying wound healing properties suggested a key role of nuclear factor-erythroid-2-related factor 2 (Nrf2) activation in mediating this process.22,23,40 Effects of luteolin-7-glucoside on human skin epidermal stem cells (EpSCs) proliferation in vitro (48 hours) (MTT assay) (modified from: Wan et al.
39
).
Supporting these findings was the report of Lodhi and Singhai
41
that explored wound healing actions of luteolin in streptozotocin (STZ)-exposed Wistar albino rats demonstrating its anti-oxidant role, although the study was limited due to the use of only two treatment concentrations. The authors extracted luteolin from dried leaves of the plant
The antioxidant components (SOD, CAT, GSH), protein content of the tissue, and hydroxyproline were also increased in 0.5% luteolin treated group compared to control group. Similar results were reported by Ozay et al 42 where Wistar diabetic and non-diabetic rat models treated with luteolin 0.5% and 1% ointment showed statistically significant increased granulation tissue thickness, re-epithelialization, angiogenesis, and wound contraction. These findings suggest that luteolin has potential as a wound healing promoter and deserves further exploration in clinical studies.
Quercetin
Epidermal stem cells located in the basal layer of the skin epidermis have an important role in wound healing. Such EpSCs became activated to proliferate with the newly formed cells migrating and then differentiating to affect a regenerated and repaired epidermis. During this repair process the Wnt/β-catenin signaling pathway becomes activated, enhancing wound healing. Quercetin, a plant-derived polyphenol, has been widely reported to enhance the healing of skin lesions, including excision wounds, pressure ulcer lesions, and diabetic wounds.43-45 The quercetin treatment has been found to speed the healing of dermal wounds, enhancing re-epithelialization and down-regulating of fibrotic responses due to skin damage.44,46 During cell culture studies quercetin enhances β-catenin expression in keratinocytes
47
and increases keratinocyte proliferation and migration.48,49 These findings led Wang et al
50
to question whether quercetin would enhance EpSC proliferation. In their follow-up study, Wang et al
50
(2020) showed that quercetin enhanced human EpSC proliferation in vitro using BrdU incorporation over 24–72 hours. It is interesting to note that the stimulatory response pattern changed significantly over the 3 days. Day 1, there was a biphasic concentration dose response with a marked inhibitory response at the highest concentration (5 µM) (Figure 7). However, the inhibitory concentration became stimulatory on days 2 and 3. Using the optimal stimulatory concentration of days 2 and 3 (1 µM) the quercetin induced PCNA, epidermal thickness in cultured human skin, and increases in β1-inegrin and a6-integrin expressing cells. In a similar fashion, the quercetin (1 µM) enhanced the expression of β-catenin, c-Myc, and cyclic A2 and E1. Effects of quercetin on human epidermal stem cell proliferation in vitro (BrdU) (modified from: Wang et al.
50
).
When β-catenin expression was inhibited, it led to the suppression of quercetin induced upregulation of β-catenin, c-Myc, and cyclin A2 and E1. When c-Myc was inhibited, it suppressed the upregulation of c-Myc and cyclin A2 by quercetin. Further, when the estrogen receptor antagonist ICI 187,780 was administered, it prevented quercetin induced cell proliferation and upregulation of β-catenin, c-Myc, and cyclin A2. These findings indicate that quercetin enhanced EpSC proliferation via the estrogen receptor (ER)-induced activation of the β-catenin/c-Myc/cyclin A2 signaling pathway and the ER independent cyclic E1 upregulation of cyclin E1.
Epidermal Stem Cell Marker Enhancement
An important area of skin biology involves finding practical means to sustain and enhance the stem cell properties of human epidermal keratinocytes. Aging affects both the epidermal and dermal skin components, along with progressive homeostasis instability, as is typically seen with a distributed balance between proliferation and differentiation of the epidermis. 51 EpSCs provide the constant renewal of the epidermis throughout the entire lifespan. Given their role in sustaining the viability of the skin, the EpSCs are protected in a dermatologically structured “niche” which is anchored to the dermal epidermal junction (DEJ) via several key proteins (ie, integrin β1, and α6, which bind to Type II collagen and laminin). In differentiation processes these protein binding interactions with the basement membrane become altered, permitting the migration of the differentiated cells. Attempts to sustain the stemness of EpSCs has employed a variety of experimental strategies, but typically including enhancing multiple stem cell biomarkers along with functional performance assays, such as those involving cell viability, cell proliferation, and capacity to resist various types of oxidative stress. Studies showing the capacity to enhance EpSC stemness within the context of an hormetic dose response framework include selenium, 52 kaempferol, 53 and abalone. 54
c-Myc—Regulating Epidermal Stem Cell Proliferation and Differentiation
A large population of patients with squamous cell carcinoma who have been treated for a prolonged period with immunosuppression strategies display v-Myc myelocytomatosis viral oncogene homologues (Myc) amplification.
55
However, Myc occurs typically at low levels in epidermal regions such as the basal layer of the interfollicular epidermis (IFE) and in the bulb of growth hair follicles (HF) where cell proliferation typically occurs.
55
In general, EpSCs divide/proliferate infrequently.
56
When Myc is activated in the epidermis, it stimulates cell proliferation. The activity of Myc in the epidermis is negatively regulated, keeping EpSCs in a relative state of quiescence by two proteins (Blimp1-in sebaceous gland cells and Lrig1 in IFE). While Myc activation is characteristically associated with cell proliferation, it can also stimulate differentiation.
57
Since the cells that are expanded following Myc activation tend to be those regulated by Blimp and Lrig1, it has been hypothesized that Myc-induced differentiation may be an evolutionarily based and highly conserved processes to prevent uncontrolled cell proliferation, therefore, preventing tumor formation by the EpSC compartment.56,58 Berta et al
59
hypothesized that a low Myc activation enhances cell proliferation and may promote tumor formation. However, at higher levels of activation Myc triggers the ARF/p53 tumor suppression pathway, which reduces cell proliferation while enhancing cell differentiation. The epidermal terminal differentiation affects a loss of cell proliferation capacity, a process common to all stem cell types. These observations led Berta et al
59
to hypothesize that the epidermis may respond in a “dose-dependent” manner to different levels of Myc activation, with cell proliferation found at low activation with differentiation being selected at higher Myc activities.
55
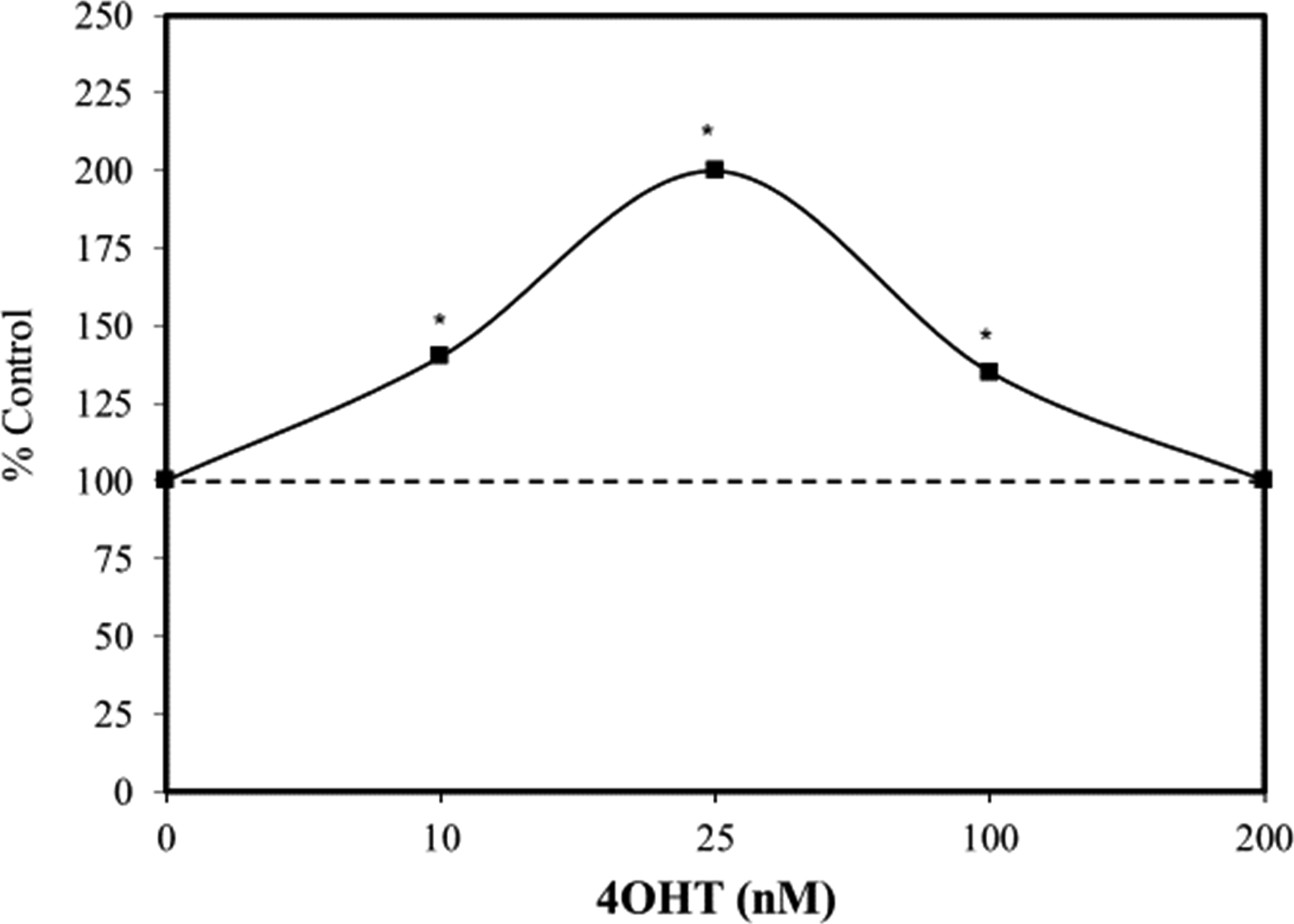
Supportive experiments using 4OHT (4-hydroxy-tamoxifen) induced differential activation of Myc in a transgenic mouse model (Figure 8). These researchers observed low levels of Myc activation enhanced proliferation which are differentiated into sebaceous glands and IFE at higher Myc activity levels. It was concluded that Myc-induced differentiation blocks uncontrolled proliferation and neoplasia formation and acts as a mechanism to prevent tumor induction. This finding that Myc in human EpSC can dose-dependently and inversely regulate the occurrence of cell proliferation and differentiation was conceptually challenged, when initially proposed
57
but has been widely confirmed.55,59,60 Of interest is that the transition from cell proliferation to cell differentiation occurs within a dose response framework. The transition from cell proliferation occurs when it reaches the hormetic maxima of 30-60% greater than the control which then shifts to differentiation which likewise has its own hormetic maxima.
61
The integration of the regulation of stem cell biology within an hormetic strategy provides an evolutionary explanation for cancer mediated regulation. It suggests an hormetic strategy directs the Myc-mediated proliferation/differentiation regulatory framework that affects tumor promotion incidence. Effects of 4-hydroxy-tamoxifen (4OHT) on c-Myc expression in transgenic mice (modified from: Berta et al.
59
).
Discussion
The present findings complement and extend a recent extensive effort to discover, document, and assess the occurrence and biological/biomedical significance of hormetic dose responses in stem cell biology. Hormetic dose responses have been commonly reported for adipose derived stem cells (ADSCs), 1 apical papilla stem cells (APSCs), 7 bone marrow stem cells (BMSCs), 62 dental pulp stem cells (DPSCs), 4 embryonic stem cells (ESCs), 3 neuronal stem cells (NSCs), 2 and periodontal ligament stem cells (PDLSCs). 5 Based on the substantial occurrence of hormetic dose responses affecting cell viability, cell survival, cell proliferation, cell differentiation, and a capacity to enhance resilience to inflammatory conditions, a broad spectrum of stem cell types use adaptive strategies that conform to the quantitative features of the hormetic dose response, displaying a substantial degree of generality. Despite these widespread occurrences of hormesis within the stem cell literature these findings are typically unrecognized as a general dose response concept with clinical implications. The recent spate of papers on the role of hormesis in stem cell functioning is likely to enhance the recognition of the significance of hormetic-biphasic dose responses and their role in stem cell biology.
Hormetic dose responses were reported in over 100 highly diverse chemical and physical agents with some common dietary supplements, such as resveratrol and EGCG, showing such biphasic dose responses in multiple stem cell types.1-9 For example, resveratrol has been shown to induce hormetic effects in six stem cell types (eg, adipose, 1 bone, 62 embryonic, 3 endothelial, 8 muscle, 6 neural 2 ). These responses conform to the quantitative features of the hormetic dose response, findings that extend the generality of the hormesis concept. 15 The present findings show that a broad spectrum of chemical classes induce hormetic response in EpSCs. Despite the occurrence of hormetic dose responses by EpSCs, the extent to which this has been reported is modest compared to other more studied stem cells (eg, adipose, neural, embryonic, dental). The quantitative features of the dose responses seen with EpSCs also match closely with a vast hormetic literature63,64 as well as those reported for other types of stem cells. That is, the amplitude of the EpSC hormetic responses is 150% (median) with the median stimulation width being 10 fold. The median number of concentration/doses is 5.
Therapeutic Considerations
While little consideration was given to the potential therapeutic implication of the occurrence of hormetic biphasic responses in EpSCs in translational science, there has not been the case with the potential applications of NO on dermal wound healing. The EpSC findings have become integrated within a broader research domain concerning the effects of NO on the wound healing process, including how it affects keratinocyte and fibroblast activation, cell proliferation and cell migration. 34 Of further therapeutic interest were studies addressing age-related changes in EpSCs. In such cases, research efforts have explored how to enhance the “stemness” of the EpSCs, and as an intervention to slow the occurrence of aging. 52 Of potential considerable fundamental significance has been the research of Watt and colleagues concerning the effects of Myc activation on EpSC proliferation and differentiation. 59 These findings show a type of regulatory control over the extent of cell proliferation, not only limiting it to a maximum of about 30–60% greater than control values, but then transitioning the cells into terminal differentiation. Watt recognized this as a fail-safe protective mechanism that blocks possible tumor development/progression. The present analysis shows that the fail-safe tumor promotion mechanism is a manifestation of hormetic processes. 65 Finally, the area of EpSC and hormesis research is quite limited relative to other types of stem cells. Thus, there is considerable need for more research activity in this area. The general patterns of the literature direction of stem cells and hormesis have been consistent and would be helpful for future research directions with respect to EpSCs and hormetic hypotheses. This would provide a detailed and coherent research guide for future EpSC and hormesis research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: EJC acknowledges longtime support from the US Air Force (AFOSR FA9550-19-1-0413) and ExxonMobil Foundation (S18200000000256). The U.S. Government is authorized to reproduce and distribute for governmental purposes notwithstanding any copyright notation thereon. The views and conclusions contained herein are those of the author and should not be interpreted as necessarily representing policies or endorsement, either expressed or implied. Sponsors had no involvement in study design, collection, analysis, interpretation, writing and decision to and where to submit for publication consideration.
