Abstract
Selenium is a fundamental trace element of the living system. Microorganisms play a crucial role in the selenium cycle, both in the environment and in life. Biogenic selenium nanoparticles have shown promising prospects for use in medicine as an antioxidant and anticancer agent. In this study, SeNPs were biosynthesized by
Introduction
Nanotechnology is an evolving technology with a broad variety of uses involving the synthesis and processing of nanomaterials (1-100 nm). Recently, there has been a great deal of focus on nanoparticle synthesis methods, including chemical, physical, and biological methods.1,2,3 There is a growing use of biological approaches for nanomaterial synthesis, involving high-yield, non-toxic, low-cost, eco-friendly, and biocompatible.2,4,5
Selenium (Se) is an important trace element for the proper and healthy life of humans, animals, bacteria, and other living systems and has an irregular distribution in the earth’s crust. 6 Selenium is well known today to play a crucial role in a variety of physiological functions in different organisms. Some of the methods used to synthesize SeNPs are physical and chemical processes. Biogenic synthesis for the preparation of SeNPs is an interesting and exciting process which is non-toxic, clean, and eco-friendly. It also has additional benefits over chemical methods, such as higher efficiency and lower costs. 7
There are many attempts to synthesize SeNPs from microorganisms such as bacteria, fungi,
8
and yeast.
9
Furthermore, few fungi can synthesize SeNPs such as
Gamma radiation is a short wave of high-energy electromagnetic radiation; it may trigger certain mutations. Tallentire 13 found that exposure of microbial cells to ionizing radiation caused a series of reactions leading to physiological changes and antimicrobial activity. These modifications are based on the absorbed dose. Thus, radiation adds additional stress to the cells, which tends to interrupt their organization.
As a result of this biochemical disruption, a wide range of changes can be observed in irradiated cells, some of which are transient and some are permanent. The specific dose of radiation is determined by changes in cell’s radiosensitivity or radioresistance. Approved by this result, the physically induced mutations of the
Low doses and low dose rates of ionizing radiation have been shown to stimulate the induction of antioxidant defense systems such as glutathione (GSH), glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase (CAT).17,18 Thus, the present investigation aims at studying the possible synthesis and characterization of SeNPs through mutating
Material and Methods
Fungal Species
A fungal organism, namely,
Preparation of Sample for Irradiation
The culture was maintained in the Czapek dox agar medium, incubated at 28°C for 7 days. The conidial suspension was prepared from the freshly raised seven-day-old culture of
Gamma Irradiation
The spore suspension which was previously prepared was exposed to different doses 10, 20, 30, 50, and 60 Gy of gamma radiation using cesium 137 as a source of gamma radiation (Gamma cell −40 Canadian, Activity 3032 Ci, Dose rate: .675 rad/second at the time of experiment at room temperature), At NCRRT, Cairo, Egypt (where, 1 Gy = 100 rad).
Biomass Preparation and Biosynthesis of SeNPs
Characterization of SeNPs
After 48 hours of synthesis, the biomasses were harvested by filtration using Whatman filter paper No. 1, followed by washing with distilled water. The red SeNPs in the pellet of collected tubes were dissolved in 10 mL of 1M Na2S and after centrifugation to remove fungal cells. The production of SeNPs in aqueous solution was monitored at the RCMB using:
UV-Visible Spectroscopy Analysis
Change in color was visually observed over a period of time. Absorption measurements were carried out using UV-visible spectrophotometer (Milton Roy Spectronic 1201) at the RCMB, Al-Azhar University.
Transmission Electron Microscopy (TEM)
For TEM analysis, a drop of the solution was placed on the carbon-coated copper grids and dried by allowing water to evaporate at room temperature. Electron micrographs were obtained using a JEOL JEM-1010 transmission electron microscope at 70 kV. 20
Energy Dispersive Analysis of X-ray (EDX)
The presence of elemental selenium was confirmed through EDX. The EDX microanalysis was carried out by the X-ray micro-analyzer (Oxford 6587 INCA) attached to the JEOL JSM-5500 LV scanning electron microscope at 20 kV at the RCMB, Al-Azhar University. The EDX spectrum recorded in the spot profile mode from one of the densely populated silver nanoparticles’ region on the surface of the film. 6
Electron Microscopy
For TEM preparation, the samples were fixed in 3% glutaraldehyde in .1 M sodium cacodylate buffer (pH 7.0) for 2 hours at room temperature, rinsed in the same buffer, and post-fixed in 1% osmium tetroxide for 2 hours at room temperature. The samples were dehydrated in an ethanol series ranging from 10% to 90% for 15 minutes in each alcohol dilution and finally with absolute ethanol for 30 minutes. Samples were infiltrated with epoxy resin and acetone through a graded series till finally in pure resin. Ultrathin sections were collected on copper grids. Sections were then double-stained in uranyl acetate followed by lead citrate. Stained sections were observed with a JEOL JEM 1010 transmission electron microscope at 70 kV at the RCMB, Al-Azhar University.21,22
Determination of Reduced Glutathione (GSH)
Harvested fungus is grinded by mortar in phosphate buffer saline 1:10 and then centrifuged. GSH was assayed in the resulted solution based upon the development of a relatively stable yellow color when 5,5′-dithiobis-(2-nitrobenzoic acid) is added to sulphdril compounds according to Beutler et al. 23
Results and Discussion
Effect of Gamma-Irradiated Penicillium citrinum on the Biosynthesis of SeNPs
The fungal cells were exposed to different gamma rays by doses 10, 20, 30, 40, 50, and 60 Gy. The fungal cells were exposed to radiation doses from 10 to 30 Gy, and formed reddish cells were observed after the growth of the irradiated cells with 10 mM sodium hydrogen selenite; the reddish color decreased with increase in radiation from 40 to 60 Gy. However, there is no change in the color of un-irradiated cells after growth with 10 mM sodium hydrogen selenite (Figure 1). From the above results, it was observed that the exposure of Mycosynthesis of SeNPs after exposure to different doses of gamma radiation. (A) Un-irradiated fungal pellets after growth with 10 mM of sodium hydrogen selenite. (B) 10 Gy. (C) 20 Gy. (D) 30 Gy. (E) 40 Gy. (F) 50 Gy. (G) 60 Gy.
Measurement of Elemental Selenium Produced by Fungal Reduction of Selenite Using Na2S Solution
A red-brown liquid without any turbidity resulting from the addition of Na2S to the red SeNPs was subject to a different characterization analysis for selenium nanoparticles. Intracellular SeNPs can be determined by Na2S, as the alkalinity of the 1-M Na2S solution can dissolve cell membranes and allow for the evaluation of intracellular SeNPs deposits as well as SeNPs bound to extracellular protein. 28
UV-Visible Spectroscopy Analysis
From the results in Figure 2, it appeared that the absorption spectra of nano-Se after exposure of UV-visible absorption spectrum obtained for selenium nanoparticles synthesized by 
Microscopic Characterization by TEM
The data obtained from transmission electron-micrograph of biosynthesized SeNPs by TEM micrograph of the biosynthesized selenium nanoparticles after 
These results indicate that the prepared particle size gets smaller and the distribution of particle size is enhanced with irradiation dose, increasing until 30 Gy, and the diameter of particles’ size increased with increasing dose from 40 to 60 Gy. The present finding is consistent with the findings of past studies which concluded that the diameter of nano-Se after exposure to 2 Gy decreased to 25.8 ± 5.2 nm, 4 Gy decreased to 22.4 ± 5.8 nm, and 6 Gy decreased to 19.82 ± 4.7 nm. These findings suggest that small doses of irradiation do not destroy the selenium nanoparticle structures but may induce higher selenium ion concentrations.
29
The AuNPs were biosynthesized by
Energy Dispersive X-ray (EDX) Analysis
Energy dispersive x-ray (EDX) spectroscopy showed typical absorption for SeKα peaks approximately at 11.45 keV for all samples after exposure of
The present finding is consistent with the findings of past studies which concluded that the typical absorption of SeLα and SeKα peaks by electron-dense selenium nanoparticles was approximately 1.37 keV and 11.22 keV, respectively, that indicates bacterial uptake and transformation of selenite into intracellular SeNPs.8,16,25,38 The presence of SeKβ nanoparticles that had a peak at 12.49 keV is also recorded by Dhanjal and Cameotra
25
(see Figure 4). Histogram showing the percent of the biosynthesized SeNPs and EDX graphs of the biosynthesized SeNPs after 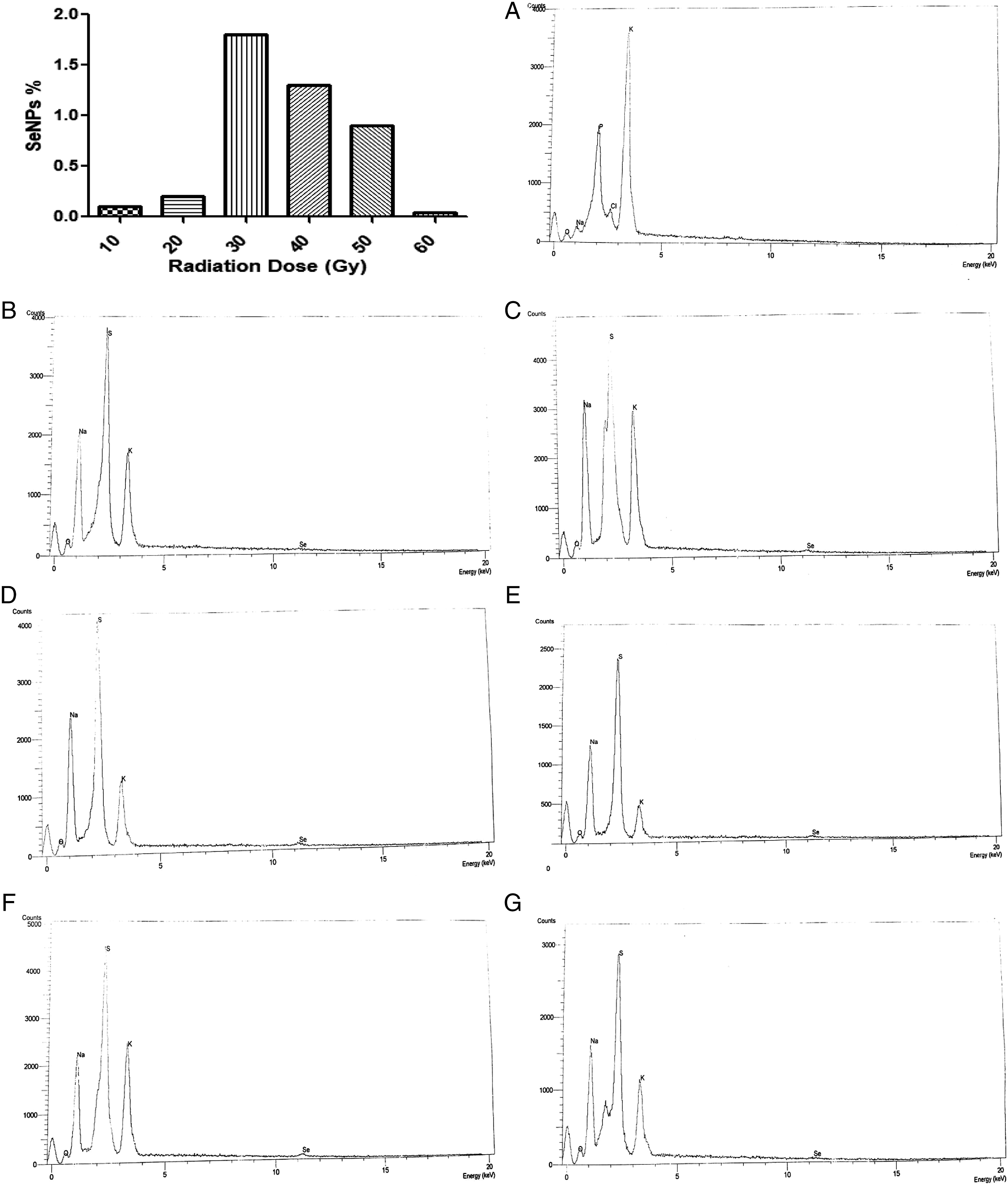
Electron Microscopic Observations
TEM was used to determine the location of SeNPs produced by Transmission electron micrographs of elemental selenium nanoparticles produced by the biomass of 
Reduced Glutathione Content (GSH)
GSH increased by increasing the radiation dose until 30 Gy but decreased again with the higher doses as shown in Figure 6. Selenite may be reduced to SeNPs by reaction with protein/peptide reactive thiol groups (activated at the plasma membrane), suggested as a general microbial detoxification reaction to oxyanions. Among these protein/peptides is the glutathione (GSH)/glutathione reductase (GR) system which is responsible for the formation of SeNPs. These proteins/peptides may act as oxido-reductase enzymes or as protons antitransporters.
42
Glutathione (GSH or reduced glutathione) is a gamma-glutamyl-cysteinylglycine tripeptide and the major intracellular antioxidant in several organisms. GSH effectively protects the cells against a variety of free radicals including reactive oxygen species. It has 2 forms, the reduced form of glutathione (GSH) and the oxidized form (GSSG), where sulfhydryl bonds bind 2 GSH moieties. Glutathione peroxidase (GPx) and glutathione-s-transferase (GST) conduct the detoxification reactions effectively using GSH, transforming it to GSSG. Glutathione reductase (GR) runs the recovery pathway by transforming GSSG to GSH with the loss of NADPH and restoring the cellular GSH reservoir. Therefore, GSH and GSH-dependent enzymes are essential to preserve the body’s normal redox balance and assist in cell survival under stress conditions. GSH and dependent enzymes give living cells a survival advantage against radiation.
43
Reduced glutathione content after adding 10 mM of sodium hydrogen selenite to irradiated fungus exposed to different doses of gamma radiation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
