Abstract
Pterostilbene is a potent antioxidant and anti-inflammatory agent. However, its chemopreventive effects via anti-tyrosinase activity and inhibitory effects on melanin content have not been reported previously. Hence, this study aimed to investigate the anti-melanogenic activity of pterostilbene on UVB-irradiated B164A5 mouse melanoma cells. The effects of pterostilbene and resveratrol on cell viability were determined by MTT assay, whereas melanin content and tyrosinase assay were employed to assess melanogenesis activity. Western blot analysis was performed to determine the tyrosinase expression. Based on the MTT assay, the IC50 value of pterostilbene on UVB-irradiated B164A5 cells was 34.0 ± 3.43 μM, in comparison to resveratrol (>100 μM). Next, 5 and 10 μM pterostilbene showed a significant dose-dependent inhibition (P < .01) of tyrosinase activity in UVB-irradiated B164A5 cells at 37.14 ± 2.71% and 58.36 ± 6.8%, respectively. The findings from the tyrosinase assay also confirmed the downregulation of tyrosinase expression in UVB-irradiated B164A5 cells as measured by Western blot analysis. Finally, 10 μM pterostilbene showed a significantly decreased melanin content (P < .01) in UVB-irradiated B164A5 cells, at 27.34 ± .98 μg/mL. In conclusion, pterostilbene showed anti-melanogenic activity that was 10 times more potent than resveratrol in the UVB-irradiated B164A5 cell.
Introduction
The skin is the largest organ, comprising 16% of human body weight, and it provides various protective roles in humans. It acts as a protective barrier for physical injuries and protection from exposure to ultraviolet radiation (UV) rays. 1 Exposure to UV on the human skin can induce the accumulation of melanin. 2 Skin pigmentation is due to melanin production by melanosomes in melanocytes which acts as a protective mechanism against the DNA damaging effects of exposure to UV. Whereas keratinocytes in the skin, when stimulated by UV rays from the Sun, α-melanocyte stimulating hormone (α-MSH) production is induced and causes the upregulation of microphthalmia transcription factor (MITF). 3
Although melanin protects human skin against the harmful effects of UV rays, researchers have found that pheomelanin can cause DNA damage. 4 In addition, MITF is an essential factor in tyrosinase production, whereby tyrosinase is the most important rate-limiting enzyme in melanogenesis. 5 However, with exogenous factors such as UV rays, there is an increased generation of superoxide and hydrogen peroxide during melanogenesis. This, in turn, increases oxidative stress, which potently drives the transformation of melanocytes into melanoma. 6 Besides that, increased melanin production causes undesirable effects, primarily on cosmeceutical aspects such as melasma and darkening of the skin. 7 UVB is of particular interest in this study instead of UVA because it is more cytotoxic and genotoxic and a potent driver of photocarcinogenesis. 8
Hyperpigmentation of the skin can lead to unwanted side effects. Therefore, various compounds have been studied as anti-melanogenic agents, such as hydroquinone, arbutin, kojic acid, epigallocatechin-3-gallate, and xanthohumol. However, there are still the presence of side effects and limitations in these compounds, such as allergic reactions, irritation, cellular damage, and carcinogenesis.9-11
Resveratrol is a stilbenoid compound present in the polyphenol group. It has been discovered as a good depigmentation agent that can inhibit tyrosinase activity and reduce melanin production.
12
However, there are still limitations regarding its practical use, such as short half-life and low bioavailability. This leads to low efficacy in the absorption of resveratrol into the human body.
11
Pterostilbene is also a stilbenoid compound which is a structural analog of resveratrol. Pterostilbene can be found in Pterocarpus marsupium heartwood, grapevine, and blueberries. It has 2 methoxy groups compared to 3 hydroxyl groups in resveratrol (Figure 1). Due to the presence of methoxy groups, pterostilbene is more lipophilic than resveratrol. Hence, it has excellent membrane permeability, bioactivity, bioavailability, and a longer half-life than resveratrol.13,14 Pterostilbene has also been studied as an antioxidant agent, inflammation agent, and anti-microbial agent.15,16 However, its anti-melanogenic effects have yet to be explored. Therefore, this study aimed to determine the effects of pterostilbene as an anti-melanogenic agent on tyrosinase activity and melanin content in UVB-irradiated B164A5 mouse melanoma cells in the hope of developing a melanoma chemopreventive agent with good antioxidant properties and no side effects. Chemical structures of pterostilbene and resveratrol.
Results and Discussion
Evaluation of Cell Viability
Pterostilbene (Figure 2) had an IC50 value of 34.0 ± 3.43 μM. Doses below the IC50 value of pterostilbene were then chosen for the tyrosinase activity assay, melanin content assay, and Western blot, which were 2.5, 5, and 10 μM, respectively. However, the cell viability of UVB-irradiated B164A5 cells treated with pterostilbene showed hormesis at a concentration of 5 μM followed by a decrease in a dose-dependent manner. Hormesis is a process where a low dose of treatment that is damaging at higher doses induces an adaptive beneficial effect on cells. It is the cells’ adaptive response toward disruption in homeostasis.
17
Cell viability of UVB-irradiated B164A5 cells after treatment with pterostilbene, resveratrol, and menadione at concentrations ranging from 0 to 100 μM following 24 hours of treatment. The data are expressed as mean ± standard error mean based on 3 experiment replicates (n = 3). The IC50 value obtained for pterostilbene and menadione on UVB-irradiated B164A5 cells were 34.0 ± 3.43 μM and 18.0 ± 1.22 μM, respectively. No IC50 value was obtained for resveratrol on UVB-irradiated B164A5 cells.* Significant difference of cell viability percentage after treatment when compared to the negative control (P < .05) ** Significant difference of cell viability percentage after treatment when compared to the negative control (P < .01)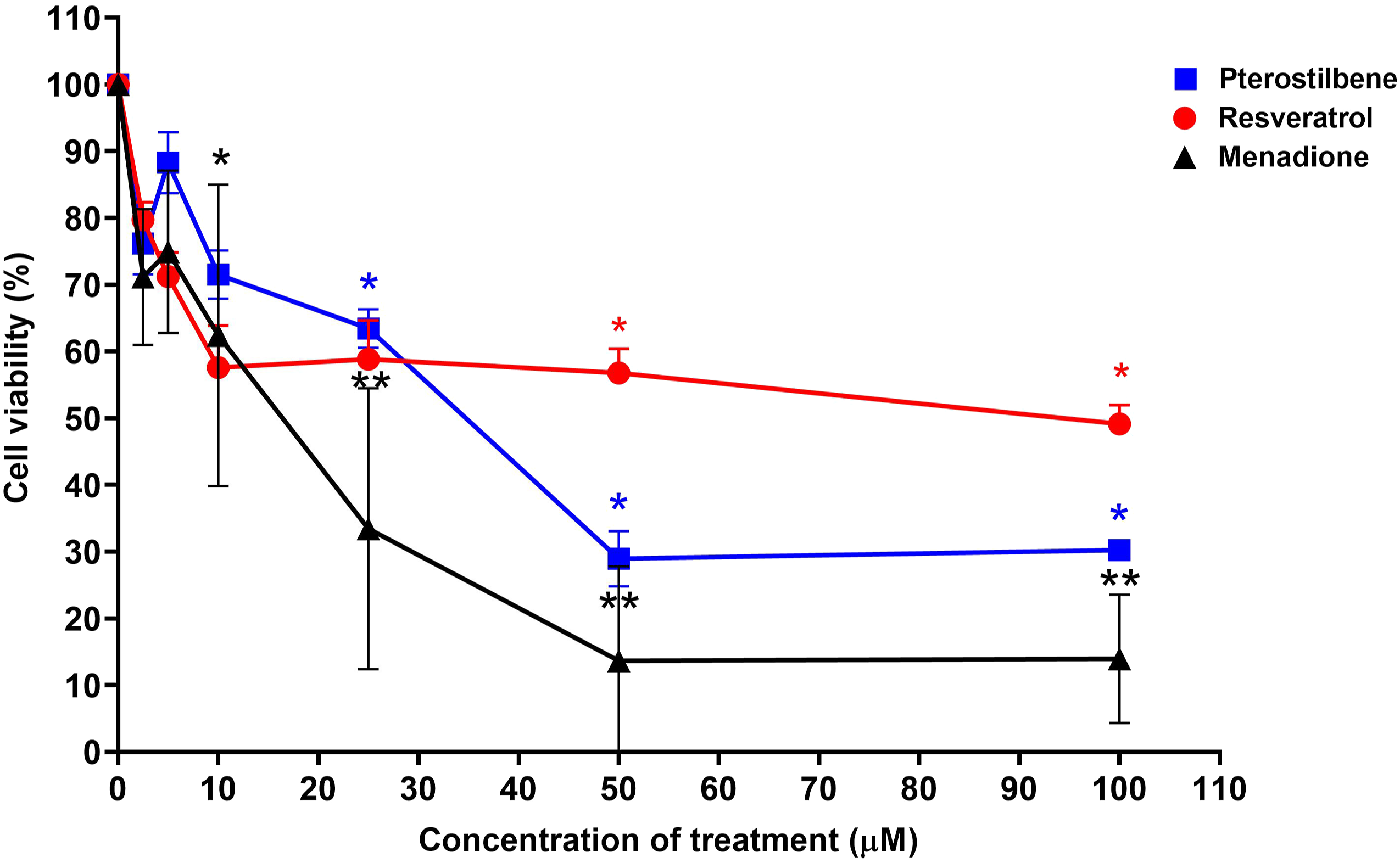
Compared to resveratrol (Figure 2), it had no IC50 value on all the tested concentrations despite a dose-dependent manner decrease. It is then posited that increased doses of resveratrol (>100 μM) would render an IC50 value. Resveratrol was chosen as the positive control for subsequent tests as it has widely been studied on its anti-melanogenesis effects.12,18 In addition, pterostilbene is a close structural analog of resveratrol, but pterostilbene has a longer half-life and good bioavailability, 19 making the effects of both compounds comparable with each other. With that, 100 μM resveratrol was chosen as the positive control dose. This is because previous research 20 has shown that treatment of B16 cells with 100 μM resveratrol is significant in reducing relative cellular melanin content and showed a potent inhibitory effect on tyrosinase activity in a cell-free investigation. A more recent study 18 also showed that treatment with 5 μM and 25 μM resveratrol reduced melanin content, but the results are not significant, compared to 100 μM resveratrol which reduced melanin production significantly.
As for menadione (Figure 2), it showed prominent cytotoxic effects toward UVB-irradiated B164A5 cells with IC50 of 18.0 ± 1.22 μM, which is in line with a previous study 21 that reported menadione had an IC50 of 14.5 μM toward B164A5 cells. Menadione has been used and studied as a cytotoxic agent for murine and human cancer cell lines.21-25 Another study 26 also showed that menadione or vitamin K3 had a substantial effect on inhibiting human melanoma cell proliferation with IC50 values of less than 10 μM. In the subsequent tests of melanin content assay and tyrosinase activity, resveratrol was chosen as the positive control because the initial goal was to suppress melanogenesis without inhibiting the growth of UVB-irradiated B16 melanoma cells. Therefore, menadione was not included in these subsequent tests because studies have shown that it is a potent cancer cell inhibitor. Hence, both pterostilbene and resveratrol displayed higher cell viability in UVB-irradiated B164A5 cells as compared to menadione. However, pterostilbene has lower cell viability in UVB-irradiated B164A5 cells when compared to resveratrol.
Although a chemopreventive agent should have lower cytotoxic effects to prevent damage toward skin cells, particularly melanocytes, to combat hyperpigmentation (a risk factor of skin cancer), this still does not undermine the potential of pterostilbene to be developed as an anti-pigmentation agent. This is because pterostilbene also has anti-cancer properties, causing apoptosis of leukemia cells and breast cancer cells. 27 In addition, pterostilbene has shown anti-metastatic effects toward B164A5 cells in the liver and a higher cytotoxic effect (19%) toward B164A5 cells cultured with hepatic sinusoidal endothelium cells in comparison to resveratrol (12%). 28 Hence, pterostilbene has apoptotic and anti-proliferative effects toward cancer cells at high concentrations. 15
Assessment of Tyrosinase Activity Inhibition
Tyrosinase is one of the most critical rate-limiting enzymes to oxidize L-DOPA into DOPAchrome, giving higher absorbance readings.
29
A higher absorbance corresponds to higher tyrosinase activity. The results of the tyrosinase activity inhibition by pterostilbene and resveratrol on UVB-irradiated B164A5 cells were represented as the percentage of inhibition compared to the negative control (Figure 3). Treatment of UVB-irradiated B164A5 cells with 5 and 10 μM pterostilbene showed a significant dose-dependent inhibition (P < .01) of tyrosinase activity with a percentage inhibition 37.14 ± 2.71% and 58.36 ± 6.8%, respectively, when compared to the negative control. However, there was no significant difference between the treatment of UVB-irradiated B164A5 cells with 5 and 10 μM pterostilbene relative to the positive control, 100 μM resveratrol. Regardless, when comparing 10 μM pterostilbene (58.36 ± 6.8%) that has a 10 times lesser concentration than 100 μM resveratrol (50.98 ± 6.47%), both compounds have exhibited comparable effects on tyrosinase activity inhibition in UVB-irradiated B164A5 cells. Percentage of relative tyrosinase activity inhibition in UVB-irradiated B164A5 cells after treatment with pterostilbene (Pter) and resveratrol (Resv). The data are expressed as mean ± standard error mean based on 6 experiment replicates (n = 6). Treatment with 5 and 10 μM pterostilbene showed a significant difference compared to the negative control.** Significant difference for the percentage of relative tyrosinase activity inhibition after treatment when compared to the negative control (P < .01)a Significant difference for the percentage of relative tyrosinase activity inhibition after treatment when compared to the positive control (P < .01).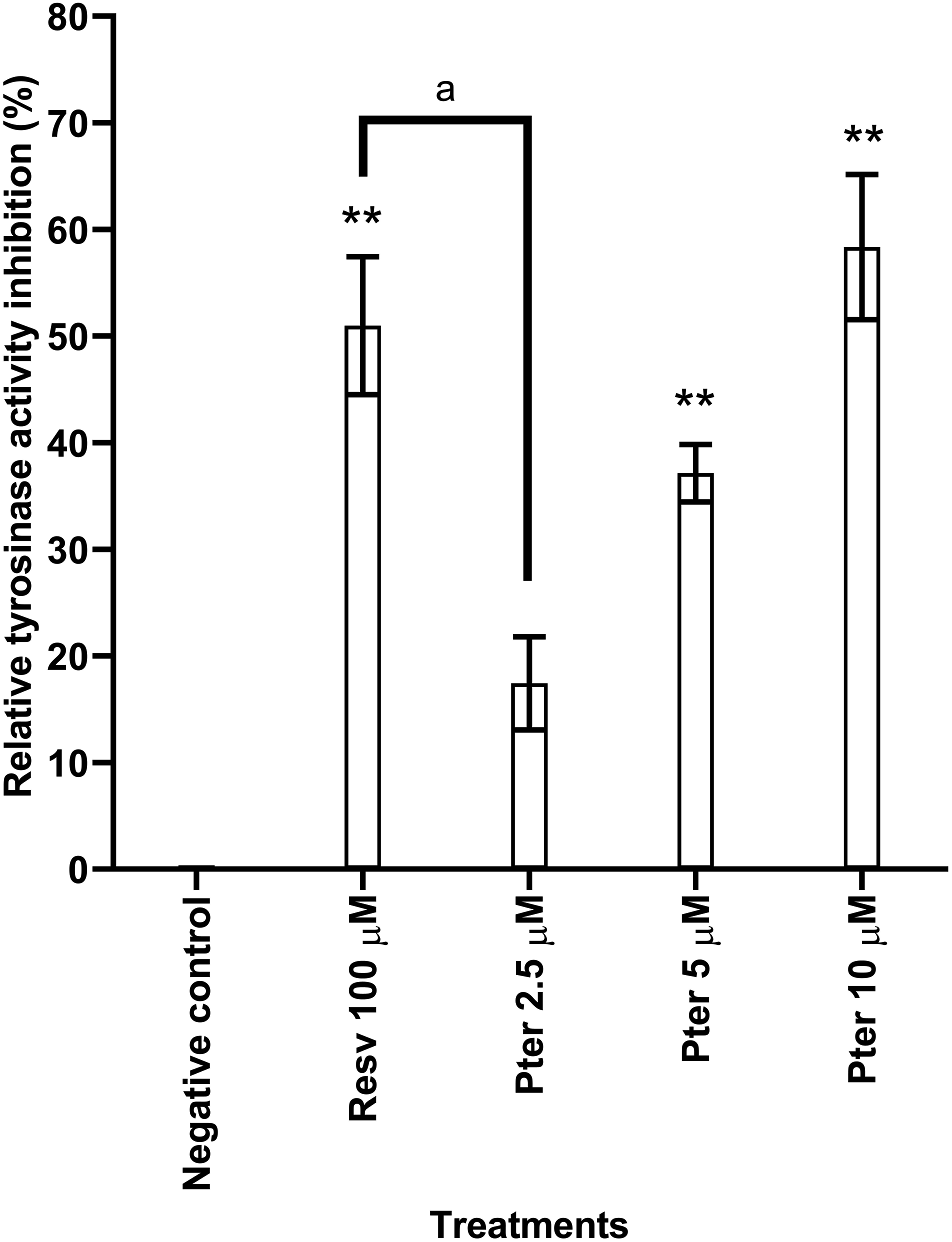
These results are supported by a study 30 that showed a significant dose-dependent decrease of the tyrosinase activity in α-MSH stimulated B164A5 cells when treated with pterostilbene. The same study 30 also reported a 58% inhibition of tyrosinase activity in B164A5 cells treated with 10 μM pterostilbene. Another study 29 has also shown that hydroxystilbene can inhibit both mushroom tyrosinase activity and B164A5 cell tyrosinase activity. As for resveratrol, it has been shown to act as a kcat inhibitor that binds and irreversibly inactivates tyrosinase, resulting in decreased melanin production. 20
Protein Expression of Tyrosinase Using Western Blot
Figure 4 shows the β-actin and tyrosinase protein band from the Western blot analysis using UVB-irradiated B164A5 cell lysate treated with pterostilbene and resveratrol. It was observed that after treatment with 10 μM pterostilbene, the expression of tyrosinase protein in UVB-irradiated B164A5 cells was reduced significantly. Protein bands of β-actin and tyrosinase from Western blot analysis using UVB-irradiated B164A5 cell lysate treated with pterostilbene (Pter) and resveratrol (Resv). The expression of tyrosinase protein was inhibited in UVB-irradiated B164A5 cells treated with 100 μM resveratrol and 10 μM pterostilbene for 24 hours. β-actin was used as a housekeeping protein.
Besides that, resveratrol also inhibits tyrosinase activity by reducing the protein expressions of tyrosinase (as observed in this study) alongside TRP-1, TRP-2, and MITF. 11 Pterostilbene could also exhibit a similar mechanism of action just like resveratrol, but further studies are needed to validate the pathways and components involved in melanogenesis.
Assessment of Melanin Content
Melanin content of UVB-irradiated B164A5 cells treated with pterostilbene showed a significant reduction (P < .01) at 10 μM (27.34 ± .98 μg/mL) compared to the negative control (43.81 ± 3.7 μg/mL). As for the positive control, 100 μM resveratrol significantly reduced melanin content (25.54 ± 3.04 μg/mL) than the negative control. The change in melanin content of UVB-irradiated B164A5 cells with regard to their respective treatments are illustrated in Figure 5. When comparing 10 μM pterostilbene (27.34 ± .98 μg/mL) that has a 10 times lesser concentration to 100 μM resveratrol (25.54 ± 3.04 μg/mL), both compounds have exhibited comparable effects on reducing melanin production in UVB-irradiated B164A5 cells. Melanin content of UVB-irradiated B164A5 cells after treatment with pterostilbene (Pter) and resveratrol (Resv). The data are expressed as mean ± standard error mean based on 6 experiment replicates (n = 6). Treatment with 5 and 10 μM pterostilbene showed a significant difference compared to the negative control.** Significant difference of melanin content after treatment compared to the negative control (P < .01).
These results correlate well with the results obtained in the tyrosinase activity assay and protein expression, as tyrosinase is a rate-limiting enzyme in melanin production. 31 The inhibition of tyrosinase activity will inhibit the conversion of tyrosine into DOPA and DOPA into DOPAquinone. DOPAquinone is the substrate for the subsequent production of melanin. Hence, melanin production will be inhibited or reduced. 3 As a whole, this study shows that pterostilbene reduced the melanogenesis activity in UVB-irradiated B164A5 cells by inhibiting the rate-limiting enzyme tyrosinase. Further supporting these findings is a study on Arthrophytum scoparium, a type of plant rich in phenolic compounds, which caused a reduction in melanin content associated with the downregulation of tyrosinase protein expression. 32
Conclusion
Pterostilbene inhibited tyrosinase activity and reduced the melanin content of UVB-irradiated B164A5 cells in a dose-dependent manner. Western blotting also showed that pterostilbene inhibited the melanogenesis process by inhibiting the tyrosinase activity and downregulating the protein expression of tyrosinase. Further research on the potential of pterostilbene as a chemopreventive agent is required to understand the full spectrum of its beneficial and cell viability effects in normal human melanocytes.
Materials and Methods
Chemicals
Pterostilbene and resveratrol were purchased from Friendemann Schmidt (Germany). Dulbecco’s Modified Eagle Medium (DMEM) with L-glutamine and HEPES, fetal bovine serum (FBS), Pen-Strep (100 U/ml penicillin and 100 μg/mL streptomycin), trypan blue .4%, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) salt, trypsin-EDTA (.25% trypsin, 1 mM EDTA.4Na), Triton X-100, 1%, phenylmethylsulfonyl fluoride (PMSF), Coomasie Blue G250, phosphoric acid, L-3,4-dihydroxyphenylalanine (L-DOPA), sodium hydroxide, menadione, phosphate-buffered saline (PBS), DMSO, ethanol, and methanol were purchased from Sigma Aldrich (USA).
Stock Preparation of Pterostilbene and Resveratrol
10 mM pterostilbene stock solution was prepared by solubilizing 5.126 mg pterostilbene powder into 2 mL 100% DMSO. 10 mM resveratrol stock solution was prepared by solubilizing 4.57 mg resveratrol into 2 mL 100% DMSO. The stock solution was mixed using a vortex for 5 to10 minutes then filtered using a .45 μm syringe filter. The stock solution was stored at −20°C until further use. When required, the stock is diluted in media to achieve the desired concentration in μM, considering that DMSO needs to be less than 1% to avoid cytotoxicity of treatment.
Preparation of Cell Culture
The B164A5 mouse melanoma cell line was purchased from the European Collection of Authenticated Cell Cultures (ECACC). The adherent cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) enriched with 10% fetal bovine serum (FBS) and 1% Pen-Strep (100 U/ml penicillin and 100 μg/mL streptomycin) at the Biocompatibility and Toxicology Laboratory, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia. The cells were incubated in a humidified atmosphere at 37°C in 5% CO2 until 70–80% confluency is achieved within 2–3 days and were subjected to sub-culture.
Cell Seeding and In Vitro Treatment
B164A5 mouse melanoma cells were seeded at a 1 × 105 cells/ml concentration and incubated for 24 hours to allow cell attachment to culture plates. The cells were then exposed to UVB irradiation at 30 mJ/cm2 before treatment to enhance the melanogenesis activity in the cells. B164A5 cells were then treated with pterostilbene or resveratrol (positive control) for 24 hours following the experimental design in this study.
Evaluation of Cell Viability
Cell viability of UVB-irradiated B164A5 cells treated with pterostilbene and resveratrol were measured to obtain the IC50 value for dose optimization purposes. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay
7
was used to assess the viability of B164A5 cells treated with different concentrations of treatment compounds. Menadione and resveratrol were used as positive controls for the MTT assay, while the negative control was the cells without treatment. The assay was performed in a 96-well plate, and the seeded cells were incubated for 24 hours before being irradiated with UVB at 30 mJ/cm2. The cells were treated with pterostilbene, resveratrol, and menadione at concentrations ranging from 2.5 to 100 μM. After 24 hours of treatment, 20 μL MTT (5 mg/mL) solution was added to each well and incubated for 4 hours. Following 4 hours of incubation, DMSO was added to each well to dissolve the formazan crystals, and the plates were further incubated for another 15 minutes. Then, the plate was shaken for 5 minutes, and the absorbance was read using a microplate reader (Bio-Rad, USA) at 570 nm. The cell viability was calculated using the following formula
7
The doses of pterostilbene and resveratrol below the half-maximal inhibitory concentration (IC50) were selected for further tests.
Tyrosinase Activity Assay
Tyrosinase activity assay was carried out using the method described by Lee et al. 33 B164A5 cells were seeded into 6-well plates and incubated for 24 hours at 37°C in 5% CO2. The cells were then irradiated with UVB at 30 mJ/cm2 and treated with 2.5, 5, and 10 μM pterostilbene for 24 hours, respectively. 100 μM resveratrol was used as the positive control, while the untreated cells were used as the negative control. B164A5 cells were lysed with .2 mM PMSF and 1% Triton X-100 in 67 mM sodium phosphate buffer. The supernatant was then collected and stored in Eppendorf tubes. This was followed by the Bradford protein assay for protein quantification, whereby absorbance was measured at 595 nm. To assess tyrosinase activity, 100 μL supernatant containing 40 μg protein was mixed with 100 μL 12.5 mM L-DOPA and incubated for 30 minutes in 37°C. The absorbance was then measured at 475 nm using a microplate reader.
Western Blot Analysis
Western blotting was carried out using the method described by Burnette. 34 B164A5 cells were seeded into 6-well plates, incubated for 24 hours, and treated as described in the tyrosinase activity assay. Replicated cells of the same dose were pooled and lysed in a 1X radioimmunoprecipitation assay (RIPA) buffer. The supernatants were then collected and mixed with Laemmli buffer to form loading samples. The loading samples containing 20 μg protein were then loaded and separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The separated proteins were then blotted onto a nitrocellulose membrane. The membrane was then blocked with 5% skimmed milk and washed using Tris-buffered saline with Tween 20 solution. After that, the blots were incubated with primary antibody: mouse anti-tyrosinase antibody (St John’s Laboratory, United Kingdom) and secondary antibody: horseradish peroxidase-conjugated secondary antibody (Cell Signaling Technology, USA). The bound antibodies were then detected by using an enhanced chemiluminescence kit.
Melanin Content Assay
The method was carried out as described by Yoon et al 30 with some modifications. B164A5 cells were seeded in 6-well plates and subjected to 24 hours of incubation to allow for cell attachment. The cells were then irradiated with UVB at 30 mJ/cm2 and treated with 2.5, 5, and 10 μM pterostilbene for 24 hours, respectively. 100 μM resveratrol was used as the positive control, while the untreated cells were used as the negative control. B164A5 cells were lysed with .2 mM PMSF and 1% Triton X-100 in 67 mM sodium phosphate buffer. The pellets were collected to measure the melanin content in the cells. Then, 1 N of sodium hydroxide was added to the pellet to extract the melanin and incubated for 30 minutes at 37°C. The absorbance was then measured at 405 nm using a microplate reader. Melanin content was calculated by using the synthetic melanin standard curve (Supplementary Figure 1).
Statistical Analysis
All data were expressed as mean ± standard error mean (SEM). Statistical analysis was performed using IBM Statistical Package for Social Sciences (SPSS) v23 by employing one-way ANOVA for comparison between means. The data were considered to be statistically significant when P < .05.
Supplemental Material
sj-pdf-1-dos-10.1177_15593258211047651 – Supplemental Material for Pterostilbene Inhibits the Melanogenesis Activity in UVB-Irradiated B164A5 Cells
Supplemental Material, sj-pdf-1-dos-10.1177_15593258211047651 for Pterostilbene Inhibits the Melanogenesis Activity in UVB-Irradiated B164A5 Cells by Dayang Fredalina Basri, Leong Chen Lew, Raveena Vaidheswary Muralitharan, Tava Shelan Nagapan and Ahmad Rohi Ghazali in Dose-Response
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universiti Kebangsaan Malaysia (DIP-2018-034).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

