Abstract
Long-term exposures to low-dose radiation (LDR) may trigger several specific biological responses, including dysregulation of the immune and inflammatory systems. Here, we examined whether biodosimetry of LDR can be used to protect tissues from radiation or assess cancer risk. Mice were subjected to gamma-irradiation with repeated or single-dose LDR, and then the organ indices, peripheral hemogram, and blood biochemistry were analyzed. An antibody array was applied followed by enzyme-linked immunosorbent assay to evaluate the utility of multiple plasma proteins as biomarkers of repeated LDR in a murine model. LDR induced inapparent symptoms but slight variations in peripheral blood cell counts and alterations in blood biochemical indicator levels. Specific plasma proteins in the LDR groups were altered in response to a higher dose of irradiation at the same time points or a single-dose equivalent to the same total dose. Plasma levels of interleukin (IL)-5, IL-12p40, P-selectin, and serum amyloid A1 were associated with the LDR dose and thus may be useful as dosimetric predictors of LDR in mice. Estimating the levels of certain plasma proteins may yield promising biodosimetry parameters to accurately identify individuals exposed to LDR, facilitating risk assessment of long-term LDR exposure in individuals.
Introduction
In recent years, exposure of the general population to low-dose radiation (LDR) has increased remarkably, such as during airport security checks or medical radiographic inspection. 1 -4 Long-term exposure to LDR may trigger several specific biological responses that differ from those induced by high-dose radiation (HDR), including dysregulation of the immune and inflammatory systems 5 -8 ; downregulation of free radical scavengers and enzymes, such as superoxide dismutase and glutathione peroxidase 9 ; and DNA damage, which indicates tissue dysfunction and a risk of carcinogenesis. 10 Biodosimetry of LDR may be useful for revealing whether radiation protection is required or for predicting possible health issues, such as an increased risk of carcinogenesis. However, conventional dosimetry, including monitoring of prodromal symptoms or peripheral blood cell counts, are mostly indicated as a linear dose-response for HDR and have limited value as biomarkers for radiation damage after LDR exposure.
LDR exposure may induce diverse molecular cascades associated with metabolism and the inflammatory response. 11 -13 For example, plasma metabolic and lipidomic profile analysis showed major changes in glycerophospholipid, amino acid, and fatty acid metabolism in mice exposed to low doses of oxygen ions (16O) and protons (1H). 13,14 Additionally, 75% of radiation workers had either high monocyte chemoattractant protein-1 (MCP-1) levels or low interleukin (IL)-10 levels, and 30% exhibited dysregulated monocyte chemoattractant protein-1, tumor necrosis factor α and IL-10 activity. In contrast, only 2% of control subjects showed dysregulation of these cytokines. 12 However, several reports suggested that the cytokines may be upregulated or downregulated upon LDR exposure in mice and humans, as they are important immune regulators that respond to radiation. 5,15,16 Dysregulated expression of cytokines after LDR has not been widely investigated as a biomarker for LDR, as this may not sufficiently reflect a correlation with pathogenesis. 17
Here, we examined LDR-induced dysfunctions and abnormalities and determined plasma protein expression profiles using an antibody array in a murine model exposed to low-dose total-body irradiation. Based on the protein profiles, several proteins were selected for validation by enzyme-linked immunosorbent assay (ELISA). The present study aimed to determine whether plasma proteins are useful as biodosimetric markers.
Methods
Animals
C57BL/6 male mice (6–8-week-old) were purchased from Vital River Experimental Animal Company (Beijing, China) and housed in a controlled environment under a 12-h light/dark cycle. All animal experiments were approved by the Institutional Animal Care and Use Committee of Academy of Military Medical Sciences, China (Permit number: IACUC-DWZX-2020-700).
Radiation Exposure
Whole-body irradiation was performed using a 60Co γ-ray source (Beijing Institute of Radiation Medicine, Beijing, China). Mice were randomly divided into 5 groups and then irradiated at 0.76 Gy/min; the detailed exposures are listed in Table 1. Non-irradiated mice were used as controls. Blood samples were collected via heart puncture at 24 h after the last irradiation.
LDR Treatments.

Abbreviations: LDR, low-dose radiation; Non-IR, non-irradiated.
Antibody Arrays
Plasma was extracted from the whole blood of normal controls and mice irradiated with 0.05 Gy (×10), 0.1 Gy (×5), and 0.5 Gy (×1) via centrifugation at 1,000×g for 15 min (Heraeus Multifuge X1 R; Thermo Fisher Scientific, Waltham, MA, USA). Twenty microliters of plasma were used for the biotin label-based L-series mouse antibody array (AAM-BLG-1; Raybiotech, Atlanta, GA, USA), which was performed according to the manufacturer’s instructions.
Data Analysis
Plasma protein levels were normalized to that of the internal control and compared between the LDR and non-irradiated control groups. The intersection of target proteins between these groups was analyzed using Venny 2.1.0 (http://bioinfogp.cnb.csic.es/tools/venny/index.html). Proteins showing expression changes P < 0.5 in the 0.05 Gy (×10) group, P < 0.25 in the 0.1 Gy (×5) group, and P < 0.2 in the 0.5 Gy (×1) group were selected. A protein-protein interaction network of the common proteins was constructed using STRING (http://string-db.org) 18 and visualized in Cytoscape software (version 3.7.1, The Cytoscape Consortium, San Diego, CA, USA; http://www.cytoscape.org). 19
Body Weight and Blood Analysis
Body weight measurements and standard hematological testing of the white blood cell (WBC), red blood cell (RBC), lymphocyte (LYM), and platelet (PLT) counts were conducted at 24 h after the last irradiation. Blood was collected via heart puncture, and cell counts were determined using a hematology analyzer (Celltac E, Nihon Kohden, Tokyo, Japan). Plasma was prepared using the anticoagulant heparin lithium for blood biochemical tests, and alanine aminotransferase, aspartate aminotransferase, total proteins, albumin, globulin, albumin/globulin, total bilirubin, alkaline phosphatase, gamma-glutamyl transpeptidase, creatine kinase, glucose, urea, creatinine, cholesterol, triglyceride, calcium, phosphorus, and total bile acid levels were measured using a hematological biochemical analyzer (Cobas 6000, Roche, Basel, Switzerland) at China Agricultural University Veterinary Teaching Hospital (Beijing, China).
Elisa
Plasma samples from all groups were analyzed via sandwich ELISA to detect interleukin-5 (IL-5), IL-10, IL-12p40, matrix metalloproteinase 14 (MMP14), serum amyloid A1 (SAA1), and P-selectin (SELP) (Elabscience, Wuhan, China) levels according to the manufacturer’s instructions. In each assay, the absorbance was detected at 450 nm using a plate reader (Model 680XR; Bio-Rad, Hercules, CA, USA). Plasma protein concentrations were determined based on standard curves for each plate.
Statistical Analysis
Data are presented as the mean ± standard error of the mean (SEM). Statistical significance was assessed by one-way analysis of variance with Tukey post-test using GraphPad Prism 7.0 software (GraphPad, Inc., San Diego, CA, USA). A P-value of <0.05 was considered to indicate statistically significant results.
Results
LDR Induces Slight Peripheral Blood and Tissue Abnormality in Mice
We irradiated mice at a total dose of 0.2–0.5 Gy (×10) and 0.5 Gy (×1 or 5) and found that the body weight, spleen index, thymus index, and testicle index were not altered in all LDR mice compared to in the normal control group (Table 2). This indicates that LDR did not induce gross general changes in these tissues. Peripheral blood analysis showed decreased WBC and LYM counts, with the lowest average counts in the 0.05 Gy (×10) group (P = 0.06 for WBC count, P < 0.05 for LYM count, compared to in the normal control group), and lower RBC counts, with the lowest average count in the 0.1 Gy (×5) group. In contrast, PLT counts were increased in all LDR groups, showing significantly higher values in the 0.5 Gy groups than in the other groups [P < 0.05 for 0.5 Gy (×1), P < 0.01 for 0.1 Gy (×5) and 0.05 Gy (×10)] (Figure 1).
Effects of γ-LDR on Body Weight and Organ Indices in Mice.a

Abbreviations: LDR, low-dose radiation; Non-IR, non-irradiated.
a Data are expressed as mean ± SEM (n = 10).
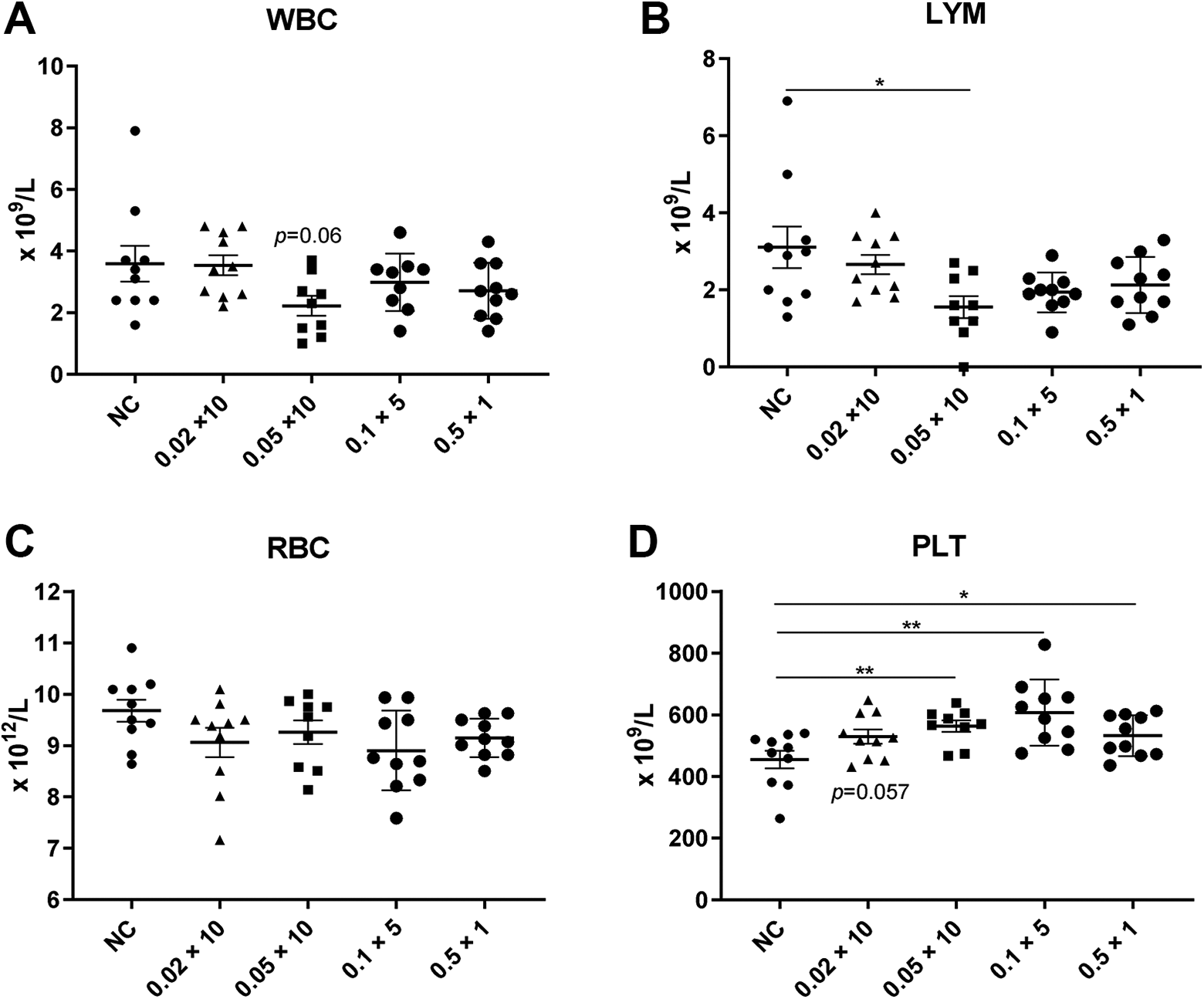
Changes in murine peripheral blood count after low-dose radiation (LDR). Peripheral blood counts of (A) white blood cells (WBC), (B) lymphocytes (LYM), (C) red blood cells (RBC), and (D) platelets (PLT) at 24 h after the last irradiation dose. Data are presented as the mean ± SEM. n = 10, *P < 0.05, **P < 0.01. NC, normal control.
Moreover, some tissue functions were altered after LDR. For example, creatine kinase and urea levels were gradually and significantly decreased in the 0.1 Gy (×5) and 0.5 Gy (×1) groups (P < 0.05); in contrast, cholesterol was upregulated in all groups, except for in the 0.02 Gy (×10) group (P < 0.05), in which it was significantly upregulated (Figure 2). Some other biochemical indicator levels, including those of alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, and total bile acids, were the same in all LDR groups (Figure 2, Supplemental Table 1). Taken together, these results indicate that LDR radiation induced slight abnormalities in the peripheral blood, kidneys, and lipid metabolism.

Blood biochemistry analysis in mice after low-dose radiation (LDR). (A) Alanine aminotransferase (ALT), (B) aspartate aminotransferase (AST), (C) alkaline phosphatase (ALP), (D) creatine kinase (CK), (E) urea (UREA), and (F) cholesterol (CHOL). Data are presented as the mean ± SEM. n = 5, *P < 0.05, **P < 0.01. NC, normal control.
Plasma Protein Levels Differ Between LDR and Normal Mice
To determine the plasma protein profile of LDR, the non-irradiation plasma protein profile was compared with that of plasma samples from LDR groups by antibody array. Figure 3A shows the expression of plasma proteins in the normal and 0.5 Gy total dose radiation groups with different fractionations, which revealed inconsistencies between the groups and even individuals within the same groups (Supplemental Table 2). A violin plot also showed different overall expression levels of all proteins between repeated the irradiation groups and single-dose irradiation groups (Figure 3B). Array analysis revealed eight common differentially regulated proteins in the 0.5 Gy radiation groups (Figure 3C). Activin A (Inhibin subunit β A, INHBA), C-terminal Src kinase (CSK), interferon-alpha/beta R1 (INFAR1), IL-5, IL-10, IL-12p40, and MMP14 were upregulated, whereas SELP was downregulated in the LDR groups. These proteins may also interact with each other (Figure 3D). Their expression levels are shown in Figure 3E. Some proteins, including INHBA, CSK, and IL-5, exhibited gradual upregulation in response to a decreasing number of LDR fractionations. SAA1 was downregulated (P < 0.05) in the 0.02 Gy (×10) radiation group with notable differences compared to that in the HDR group. 20,21 Taken together, although large variations in the protein expression levels within and among groups were detected, some plasma proteins were differentially altered after exposure to LDR.
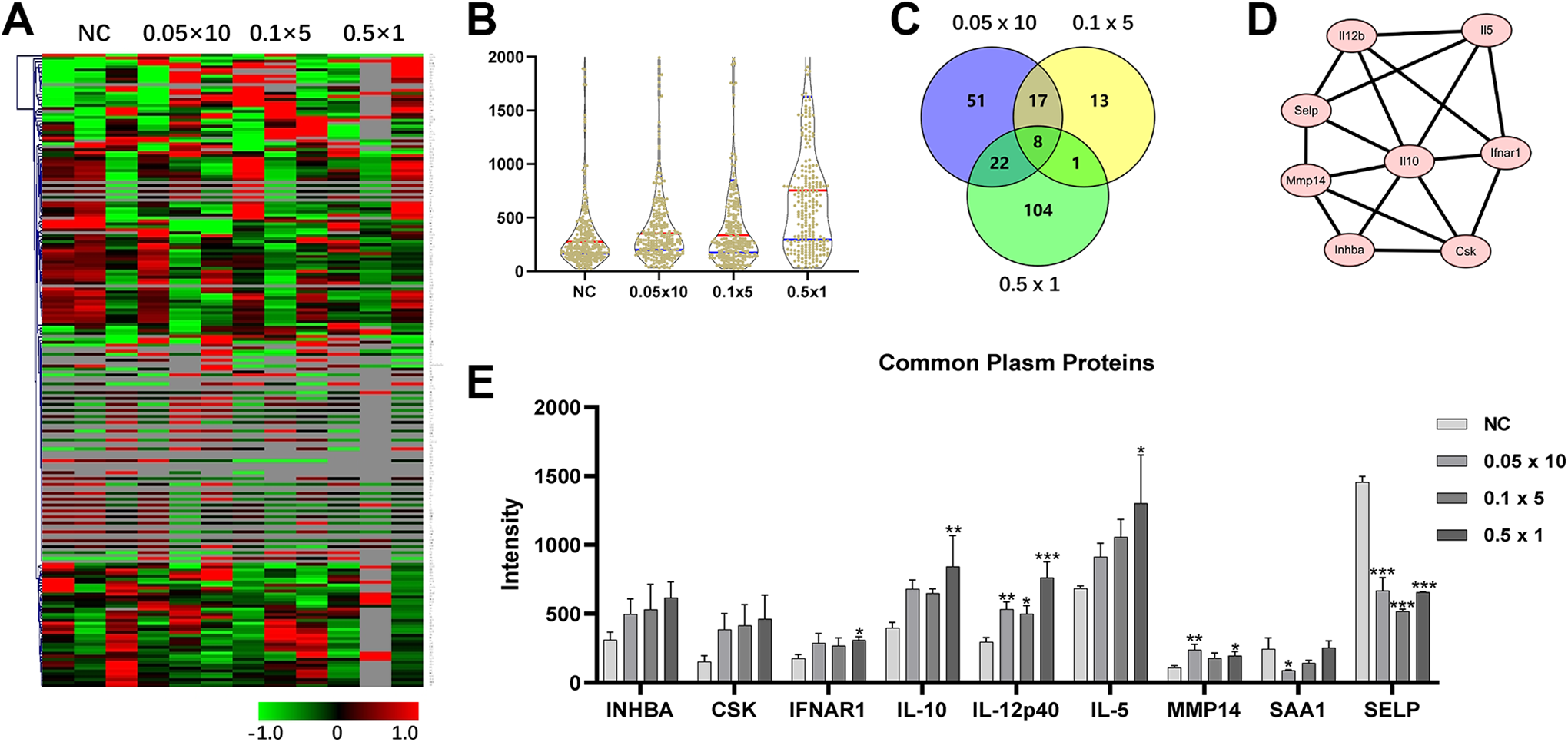
Comparison of plasma protein levels before and after low-dose irradiation (LDR) using an antibody array. (A) Heatmap of plasma protein expression between non-radiation control and low-dose radiation groups is shown. The heatmap was drawn using MultiExperiment Viewer (4.9.0). (B) Violin plot shows Avery expression levels of total analyzed proteins in each group. (C) Venny analysis indicated that the common plasma proteins were differentially altered between pre-irradiation and post-irradiation samples in all LDR groups. (D) Protein-protein interaction network of differentially altered proteins from the common set of LDR groups is shown. (E) Expression of selected plasma proteins from the antibody array in each LDR group. Data are expressed as the mean ± SEM. n = 3, *P < 0.05, **P < 0.01, ***P < 0.001. NC, normal control.
Plasma Protein Levels Were Altered in Response to Reduced Time Fractions at the Same Dose
We confirmed the plasma protein levels in the different time fractions for the same total dose radiation [0.05 Gy (×10), 0.1 Gy (×5), and 0.5 Gy (×1)] and found that plasma IL-5, IL-10, and IL-12p40 levels gradually increased, whereas those of SELP gradually and significantly decreased in response to fewer fractionations (Figure 4A-D). The level of plasma MMP14 was gradually, but not significantly, upregulated (Figure 4E). The SAA1 level was unaffected by repetitive LDR but was lower than that in the control group (P < 0.01) (Figure 4F). These results suggest that repeated exposures induced fewer alterations in plasma protein levels compared to single-dose or fewer fractionations at the same total dose.

Plasma protein levels in mice in response to low-dose radiation (LDR), determined by ELISA. A, Interleukin (IL)-5, (B) IL-12p40, (C) P-selectin (SELP), (D) IL-10, (E) matrix metalloproteinase 14 (MMP14), (F) serum amyloid A (SAA1). Data are expressed as the mean ± SEM; n = 6-8 mice for each group. *P < 0.05, **P < 0.05, ***P < 0.05, ****P < 0.0001. NC, normal control.
Certain Plasma Proteins May Serve as Dosimetric Markers of LDR
To identify possible biodosimetric markers of LDR, selected plasma proteins from the 0, 0.2, and 0.5 Gy groups with ×10 exposure were validated by sandwich ELISA. As shown in Figure 5, several proteins, including IL-5 and IL-12p40, were significantly and gradually upregulated, whereas SAA1 and SELP were downregulated in response to 0-0.5 Gy with ×10 exposure. However, IL-10 and MMP14 were gradually upregulated but only IL-10 was significantly upregulated in the 0.5 Gy group (P < 0.05) (Figure 5). Taken together, these results indicate that some plasma proteins, including IL-5, IL-12p40, SELP, and SAA1, are potential biodosimetric markers in mice after long exposure to LDR.

Plasma protein expression levels in mice were altered in response to a fraction of low-dose radiation (LDR). (A) Interleukin (IL)-5, (B) IL-12p40, (C) P-selectin (SELP), (D) IL-10, (E) matrix metalloproteinase 14 (MMP14), and (F) serum amyloid A (SAA1). Data are expressed as the mean ± SEM; n = 6-8 mice for each group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. NC, normal control.
Discussion
The present study focused on the association between plasma proteins and abnormality caused by LDR and revealed that multiple proteins were altered in response to the number of fractions and dose of LDR. We identified several plasma proteins that can serve as potential predictors of LDR. Multiple studies have suggested that LDR triggers organ- or tissue-specific responses in humans and mice, including dysregulation of the immune system, 6,12 kidneys, 22 and other organs. Some reports indicated that the levels of proteins/cytokines, 5,7 metabolites, 13,14 and nucleic acids 23 in the plasma and tissues are altered after LDR, which may be useful biodosimetric markers for LDR.
We found that the body weights and organ indices of mice were not altered by LDR. Although WBC, RBC, and most LYM [except 0.05 Gy (×10)] counts exhibited a decreasing trend, these counts were not significantly changed in the LDR groups. In contrast, the PLT count was increased after LDR, suggesting that LDR slightly influences hematopoiesis, particularly thrombopoiesis. Furthermore, the low WBC and LYM counts and high PLT counts in the 0.05 Gy (×10) group indicated that multiple or long-term LDR induces more serious abnormalities than single or short-term radiation even at the same dosage. Although the indicators of liver function were unchanged, some indicators in the kidneys, muscles, and metabolism were slightly altered after LDR. These changes were still within the normal range, indicating that longer exposure to a varied dose of LDR induces greater alterations and dysregulation in these tissues or biological processes. These results are consistent with those showing that LDR can modify the progression of chronic renal failure, 22 alter lipid metabolism, 13,24 or induce other damage. 9 Collectively, these findings suggest that repeated LDR caused mild tissue abnormalities in mice, although no alteration were detected at the body or tissue levels. Further experimental studies are required to determine the details and molecular mechanisms of these abnormalities.
As important regulators of the immune system, certain cytokines in the plasma are highly induced upon irradiation. 20,21 Comparison of the protein profiles between normal control and LDR mice revealed that most plasma proteins were altered after LDR with inconsistent expression, even between individuals within the same group, indicating significant individual differences. Among these proteins, some were selected based on their consistent expression and showed consistent upregulation or downregulation after LDR. Protein-protein interaction analysis further indicated that these LDR-induced proteins are functionally related. We confirmed that the expression of plasma IL-5, IL-10, IL-12p40, and SELP was altered following single-dose irradiation compared to after repeated irradiation at the same total dose. Moreover, the expression of plasma IL-5, IL-12p40, SELP, and SAA1 was altered in response to LDR dose. Some of these proteins, including IL-55 and IL-10, 5,7,12 have been reported to be upregulated during LDR, and IL-12p40 25 and SAA1 21 are altered after exposure to radiation and correlated with radiation damage, whereas SELP 26 and MMP14 27,28 are involved in radiation-induced pathological processes. These results suggest that the proteins play roles in regulating LDR-induced tissue damage and can serve as dosimetric indicators of LDR.
Some serum proteins were reported to be altered in response to all doses of radiation; for examples, IL-5 and IL-10 levels were consistently elevated in splenocytes according to RT-PCR analysis after 0.01–2 Gy exposure on day 1 5 and in the blood according to ELISA after 3-12 Gy exposure 29 on days 1-4 (IL-5) or days 4-7 (IL-10) post-irradiation, which are suitable for dosimetry of HDR. Here, we identified serum IL-5 and IL-10 as biomarkers for LDR. SAA1 was previously reported to be upregulated in response to >2 Gy HDR, but was invariant 30,31 or irregularly changed to approximately 1-2 Gy at 24–48 h after irradiation. 32 However, our data show that SAA1 was significantly downregulated at a radiation dose as low as 0.2-0.5 Gy. Therefore, SAA1 can be used as a biomarker of both HDR and LDR with bilateral regulation and indicates the complexity of regulation between LDR and HDR. The underlying mechanism and boundary of LDR-induced downregulation and HDR-induced upregulation require further investigation.
Based on the antibody array data, high variation within and among groups was observed, which may have decreased the number of candidate biomarker proteins for LDR. For instance, some known LDR-related cytokines, including IL-6 5,7 and tumor necrosis factor α, 7,12 were not identified in the antibody array, perhaps because of insufficient samples along with high inter-individual variation in protein levels resulting from hyper-radiosensitivity. 20 This also indicates that plasma proteins other than those identified in this study are potential biomarkers.
Conclusion
Our data preliminarily indicate that LDR induces protein dysregulation in the blood and tissues, and certain plasma proteins can be considered as indicators of LDR in mice. Although some plasma proteins may have been missed because of inter-individual variations, 33 our results provide insight into biomarkers of LDR or LDR-induced abnormality or damage.
Supplemental Material
Supplemental Material, sj-pdf-1-dos-10.1177_15593258211016257 - Plasma Proteins As Biodosimetric Markers of Low-Dose Radiation in Mice
Supplemental Material, sj-pdf-1-dos-10.1177_15593258211016257 for Plasma Proteins As Biodosimetric Markers of Low-Dose Radiation in Mice by Changhui Ge, Yanjie Liang, Yaocang Zhang, Fei Su, Lu Chen, Feiyue Ma, Kexin Ding, Jie Zhu, Xiaofei Zheng and Hanjiang Fu in Dose-Response
Supplemental Material
Supplemental Material, sj-pdf-2-dos-10.1177_15593258211016257 - Plasma Proteins As Biodosimetric Markers of Low-Dose Radiation in Mice
Supplemental Material, sj-pdf-2-dos-10.1177_15593258211016257 for Plasma Proteins As Biodosimetric Markers of Low-Dose Radiation in Mice by Changhui Ge, Yanjie Liang, Yaocang Zhang, Fei Su, Lu Chen, Feiyue Ma, Kexin Ding, Jie Zhu, Xiaofei Zheng and Hanjiang Fu in Dose-Response
Footnotes
Author Contributions
Changhui Ge and Yanjie Liang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Key Scientific Research Projects (Grant number BWS18J008) and the Chinese National Natural Science Foundation projects (Grant numbers 91540202 and 81773038).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
