Abstract
Background:
MiR-664 has been demonstrated to play an important role in dermal diseases. However, the functions of miR-664 in ultraviolet B (UVB) radiation-induced keratinocytes damage remain to be elucidated.
Objective:
The present study aimed to investigate the molecular mechanisms under the UVB-induced keratinocytes damage and provide translational insights for future therapeutics and UVB protection.
Methods:
HaCaT cells were transfected with miR-664, either alone or combined with UVB irradiation. Levels of messenger RNA and protein were tested by quantitative real-time polymerase chain reaction and Western blot analyses. Cell proliferation, percentage of apoptotic cells, and expression levels of apoptosis-related factors were measured by Cell Counting Kit-8 assay, flow cytometry assay, and Western blot analysis, respectively.
Results:
We found that a significant increase in miR-664 was observed in UVB-induced HaCaT cells. Overexpressed miR-664 promoted cell vitalities and suppressed apoptosis of UVB-induced HaCaT cells. Additionally, the loss/gain of armadillo-repeat-containing protein 8 (ARMC8) rescued/blocked the effects of miR-664 on the proliferation of UVB-induced HaCaT cells.
Conclusions:
Our data demonstrate that miR-664 functions as a protective regulator in UVB-induced HaCaT cells via regulating ARMC8.
Introduction
Ultraviolet radiation (UVR) is divided into 3 spectral regions based on its biological effects. Sunlight UVR consists of 3% to 6% ultraviolet B (UVB; 315-280 nm) and 94% to 97% ultraviolet A (UVA; 400-315 nm). Negligible amounts of UVC (280-100 nm) reach the earth surface due to the filtering capacity of the ozone layer. 1 Ultraviolet radiation induces DNA damage and apoptosis in epidermal cells, and it can lead to skin aging, inflammatory responses, dysfunction of keratinocytes, and skin carcinogenesis. 2 -4 Ultraviolet B is also a major environmental factor for human nonmelanoma skin cancer. 5 However, the molecular mechanism involved in UVB-regulated cell damage remains largely unknown.
MicroRNAs (miRNAs) are a family of small noncoding RNAs that are involved in the regulation of gene expression by binding to the 3′-untranslated regions of target messenger RNAs (mRNAs). 6 -14 MicroRNAs can regulate multiple cellular processes, including UVB-mediated cell aging and cell apoptosis, through inducing RNA interference at the posttranscription level. 15 -17 According to our previous studies, miR-664 functions as an oncogene that targets multiple tumor suppressor genes, such as IRF2. 18 However, it remains unknown whether miR-664 is involved in UVB-induced cell damage.
In present study, we assessed the effect of UVB irradiation on miR-664 expression in HaCaT cells, and miR-664 was induced by UVB stimulation. MiR-664 promoted proliferation and suppressed apoptosis of HaCaT cells treated with UVB. Armadillo-repeat-containing 8 (ARMC8) is a member of the armadillo-repeat family, a protein essential for wingless signal transduction. 19 The ARMC8 overexpression enhanced UVB-stimulated cell apoptosis. Thus, these results provided a novel direction of managing UVB-mediated HaCaT cells damage.
Materials and Methods
Cell Culture and UVB Irradiation
Human benign epidermal keratinocyte cell line HaCaT (China Center for Type Culture Collection) was maintained in Dulbecco modification of Eagle medium (Invitrogen, 8118346) supplemented with 10% fetal bovine serum (Every green, 22011-8612) and 1% penicillin/streptomycin (Invitrogen, 15140163) at 37 °C in a humidified 5% CO2 atmosphere. For UVB irradiation, the culture medium was replaced with phosphate-buffered saline. After UVB irradiation, cells were cultured in fresh medium for another 24 hours.
Plasmids and Transfection
HaCaT cells were transiently transfected with miR-664 mimic, negative control mimic (NC mimic), miR-664 inhibitor, NC inhibitor, ARMC8 small interfering RNA (siARMC8), or negative control siRNA (siNC; RIBOBIO) using Invitrogen Lipofectamine 2000 (Invitrogen, 11668019), according to the manufacturer’s protocol.
Western Blot
Cells were lysed in sample buffer (62.5 mmol/L Tris–HCl pH 6.8, 10% glycerol, 2% sodium dodecyl sulfate [SDS]) and boiled for 5 minutes. Equal amounts of lysate (50 µg total protein) were electrophoretically separated on 10% SDS/polyacrylamide gels and transferred onto polyvinylidene fluoride membranes (Millipore, IPVH00010) followed by incubation with a 1:1000-diluted anti-ARMC8 antibody (Proteintech, 12653-1-AP), anti-Bcl-2 antibody (Proteintech, 12789-1-AP), anti-Bax antibody (Proteintech, 50599-2-Ig), anti-β-actin mouse monoclonal antibody (Santa Cruz, SC47778). Proteins were detected by incubation with horseradish peroxidase-conjugated donkey antirabbit and antimouse immunoglobulin G (1:5000).
RNA Extraction and Real-Time Quantitative Polymerase Chain Reaction
After isolation from culture, cells were treated with Trizol reagent (Ambion, 15596018), according to the manufacturer’s instructions. The RNA was quantified by Nanodrop 2000. The expression levels of ARMC8, Bcl-2, and Bax were quantified using SYBR Green Mix (CWBIO, CW2610M). The expression of mRNA was defined based on the threshold cycle (Ct), wherein Ct represents the threshold cycle for each transcript. Relative expression levels were calculated as 2 − (Ct of miRNA) − (Ct of GAPDH) after normalization with the reference to expression of GAPDH. Taqman MicroRNA Reverse Transcription Kit (Applied Biosystems) was used to make complementary DNA from the total RNA. Human miRNAs and U6 were detected by TaqMan microRNA assay (Applied Biosystems) and TaqMan Fast Advanced Master Mix (Applied Biosystems). Real-time polymerase chain reaction (RT-PCR) was performed using the Applied Biosystems Q6 Sequence Detection system.
Cell Proliferation Assay
HaCaT cells (4000 per well) cultivated on 96-well plates were transfected with siRNAs and overexpression plasmid, cell proliferation was detected after 0, 24, and 48 hours using a Cell Counting Kit (MedChemExpress, 10224196) at 450 nm as described in the manual.
Apoptosis Assay
HaCaT were seeded on a 60-mm dish and transfected with miR-664 mimic/miR-664 inhibitor or siARMC8 and cultured for 24 hours. The UVB irradiation or not and cultured for 24 hours. TransDetect Annexin V-FITC/propidium iodide (PI) cell apoptosis detection kit (TransGen Biotech) was applied according to instructions. Cell apoptosis was detected and quantified using a Guava easyCyte Flow Cytometry System (Merk Millipore) by the sum of both bottom and top right quadrants of the plots, for example, including both early and late apoptotic cells.
Statistical Analysis
Statistical tests were performed for dependent samples with an independent samples t test or 1-way analysis of variance test (SPSS version 20.0, SPSS Inc). All statistical tests incorporated 2-tailed tests and homogeneity of variance tests and were considered to reflect significant differences if *P < .05, **P < .01, or ***P < .001, details of statistical analyses including sample numbers (n) are included in the respective figure legends.
Results
MiR-664 Expression Upregulates After UVB Irradiation or MiR-664 Mimic Transfection in HaCaT Cells
To assess the potential function of miR-664 in HaCaT cells treated with UVB irradiation, we examined the expression pattern of miR-664 at different time points in HaCaT cells after irradiated with 30 mJ/cm2 UVB (0, 3, 6, 9, 12, 18, and 24 hours). The results of RT-qPCR analysis showed that the expression of miR-664 was upregulated in all time points compared with the 0 hour time point (Figure 1A). To further explore the potential role of miR-664 in HaCaT cells, the aberrant expression of miR-664 was generated by miR-664 mimic, whereas miR-664 knockdown was achieved by miR-664 inhibitor. Compared with the corresponding controls, miR-664 mimic or inhibitor could effectively increase or decrease the expression of miR-664, respectively (Figure 1B).

MiR-664 was upregulated in respond to UVB radiation in HaCaT cells. A, MiR-664 expression was detected by RT-qPCR at different time points after UVB irradiation in HaCaT cells. B, The relative expression of miR-664 in HaCaT cells after transfected with miR-664 mimic/NC mimic or miR-664 inhibitor/NC inhibitor was detected by RT-qPCR. NC indicates negative control; RT-qPCR, real-time quantitative polymerase chain reaction; UVB, ultraviolet B.
MiR-664 Promotes Proliferation and Suppresses Apoptosis of UVB-Induced HaCaT Cells
To investigate the role in miR-664 in HaCaT cells treated with UVB irradiation, HaCaT cells were transfected with miR-664 mimic/NC mimic either alone or together with 30 mJ/cm2 UVB irradiation. The cell proliferation capacities were assessed by Cell Counting Kit-8 (CCK-8) assay. The cell proliferation capacity significantly increased in miR-664 mimic group (P < .01) and significantly decreased in UVB-irradiated groups compared with no irradiation groups (P < .001; Figure 2A). Next, we explored the relationship between miR-664 and the apoptosis of HaCaT cells. Apoptosis assays by Annexin V/PI double staining were performed in HaCaT cells subsequently, results revealed that the apoptosis rates in both miR-664 mimic and miR-664 mimic combined UVB irradiation groups were significantly decreased in contrast to each control group (both P < .001; Figure 2C and D).

MiR-664 promoted proliferation and suppressed apoptosis of UVB-induced HaCaT cells. A, B, The proliferation capacity of HaCaT cells transfected with miR-664 mimic/miR-664 inhibitor either alone or together with UVB irradiation was detected by CCK-8 assay. C, D, Apoptosis assay by Annexin V/PI double staining was performed in HaCaT cells transfected with miR-664 mimic/NC mimic either alone or in combination with UVB irradiation. E, F, Apoptosis assay by Annexin V/PI double staining was performed in HaCaT cells transfected with miR-664 inhibitor/NC inhibitor with UVB irradiation or not. G, Bcl-2 and Bax protein expressions were detected by Western blot after transfected with miR-664 mimic/miR-664 inhibitor either alone or together with UVB irradiation in HaCaT cells. CCK-8 indicates Cell Counting Kit-8; NC, negative control; PI, propidium iodide; UVB, ultraviolet B.
Similarly, we performed CCK-8 assay and apoptosis assay in miR-664-inhibited HaCaT cells. Results indicated that, with or without the treatment of UVB irradiation, the proliferation capacities were all significantly decreased in miR-664 inhibition groups (both P < .01), and the proliferation capacities were significantly decreased in UVB-irradiated groups compared with no irradiation groups (P < .001; Figure 2B). The apoptosis rates in both miR-664 inhibition and miR-664 inhibition combined UVB irradiation groups were significantly increased (both P < .01; Figure 2E and F).
Western blot analysis was performed afterward, by which the expression of apoptosis-related markers (Bcl-2 and Bax) were detected. The results suggested that the expression of Bcl-2 was upregulated in the case of miR-664 overexpression and downregulated in the case of miR-664 inhibition or UVB irradiation, the expression alteration of Bax was just the reverse (Figure 2G). Together, these results indicate that miR-664 promotes proliferation and suppresses apoptosis of HaCaT cells treated with UVB irradiation.
Expression of ARMC8 Is Downregulated by UVB Irradiation or MiR-664 Mimic Transfection in HaCaT Cells
Then, we explored whether the downstream target genes of miR-664 play a function in HaCaT cells treated with UVB irradiation. ARMC8, which is one of the predicted target genes of miR-664, was chosen for further experiments. We examined the expression pattern of ARMC8 at different time points in HaCaT cells after irradiated with 30 mJ/cm2 UVB by RT-qPCR and Western blot. The expression of ARMC8 in both assays was downregulated in HaCaT cells after treatment with UVB irradiation (Figure 3A and B). Subsequently, we detected the ARMC8 expression of HaCaT cells in different miR-664 status (overexpression or inhibition) with 30 mJ/cm2 UVB irradiation or not. Obviously, the expression of ARMC8 was negatively correlated with the expression of miR-664 (Figure 3C and D).

Expression of ARMC8 was downregulated by UVB irradiation or miR-664 mimic transfection in HaCaT cells. A, The relative mRNA expression of ARMC8 was detected by RT-qPCR at different time points after UVB irradiation in HaCaT cells. B, The protein expression of ARMC8 was detected by Western blot at different time points after UVB irradiation in HaCaT cells. C, The relative mRNA expression of ARMC8 was detected by RT-qPCR after transfected with miR-664 mimic/NC mimic or miR-664 inhibitor/NC inhibitor in HaCaT cells. D, ARMC8 protein expression was detected by Western blot after transfected with miR-664 mimic/miR-664 inhibitor either alone or together with UVB irradiation in HaCaT cells. ARMC8 indicates armadillo-repeat-containing protein 8; mRNA, messenger RNA; NC, negative control; RT-qPCR, real-time quantitative polymerase chain reaction; UVB, ultraviolet B.
Armadillo-Repeat-Containing Protein 8 Knockdown Inhibits UVB-Induced HaCaT Cells Apoptosis
To further explore the potential role of ARMC8 in HaCaT cells, we knocked down ARMC8 by transfecting siRNA oligos into HaCaT cells. The reduction of ARMC8 mRNA and protein levels was achieved in 2 different siARMC8-treated cells (Figure 4A and B). We assessed the apoptosis rates of HaCaT cells transfected with siARMC8/siNC either alone or together with 30 mJ/cm2 UVB irradiation by Annexin V/PI double staining. Results revealed that knockdown of ARMC8 alleviated the apoptosis of HaCaT cells but not to a significant extent (P > .05). However, in combination with UVB irradiation, the apoptosis of ARMC8 knockdown HaCaT cells alleviated significantly (P < .05; Figure 4C and D). The proliferation capacities of ARMC8-silenced HaCaT cells with 30 mJ/cm2 UVB irradiation or not were assessed. Results suggested that UVB irradiation significantly inhibited the proliferation capacity of HaCaT cells, and knockdown of ARMC8 did not influence the proliferation capacity significantly (P < .001; Figure 4E).

ARMC8 knockdown inhibited UVB-induced HaCaT cells apoptosis. A, The relative mRNA expression of ARMC8 was detected by RT-qPCR after transfected with siARMC8/siNC in HaCaT cells. B, ARMC8 protein expression was detected by Western blot after transfected with siARMC8/siNC in HaCaT cells. C, D, Apoptosis assay by Annexin V/PI double staining was performed in HaCaT cells after transfected with siARMC8/siNC either alone or in combination with UVB irradiation. E, The proliferation capacity of HaCaT cells transfected with siARMC8/siNC either alone or together with UVB irradiation were detected by CCK-8 assay. F, ARMC8, Bcl-2, and Bax protein expressions were detected by Western blot after transfected with siARMC8/siNC either alone or together with UVB irradiation in HaCaT cells. ARMC8 indicates armadillo-repeat-containing protein 8; CCK-8, Cell Counting Kit-8; mRNA, messenger RNA; NC, negative control; PI, propidium iodide; RT-qPCR, real-time quantitative polymerase chain reaction; siARMC8, ARMC8 small interfering RNA; siNC, negative control siRNA; UVB, ultraviolet B.
Next, we detected ARMC8 and apoptosis-related protein (Bcl-2, Bax) levels by Western blot in HaCaT cells transfected with siARMC8/siNC with 30 mJ/cm2 UVB irradiation or not. There is no obvious alteration in the expressions of apoptosis-related proteins (Figure 4F). In summary, ARMC8 knockdown inhibits apoptosis of HaCaT cells treated with UVB irradiation to a certain extent.
ARMC8 Overexpression Inhibits Proliferation and Accelerates Apoptosis of UVB-Induced HaCaT Cells
Aberrant expression of ARMC8 was performed by expression vector (pCMV-ARMC8) transfection. The validation of ARMC8 overexpression at mRNA and protein levels was performed in plasmid-transfected cells, as well as the NC cells (pCMV-NC transfected; Figure 5A and B). The cell apoptosis rate significantly increased after ARMC8 overexpression both in the UVB irradiated or nonirradiated groups (both P < .01; Figure 5C and D). The ARMC8 overexpression was found to inhibit the proliferation capacity of HaCaT cells with UVB irradiation or not (P < .001; Figure 5E). Results of Western blot showed that ARMC8 overexpression decreased the protein level of Bcl-2 and increased the protein level of Bax under circumstances of UVB irradiation and no UVB irradiation (Figure 5F). Taken together, ARMC8 overexpression inhibits proliferation and accelerates apoptosis of HaCaT cells treated with UVB irradiation.
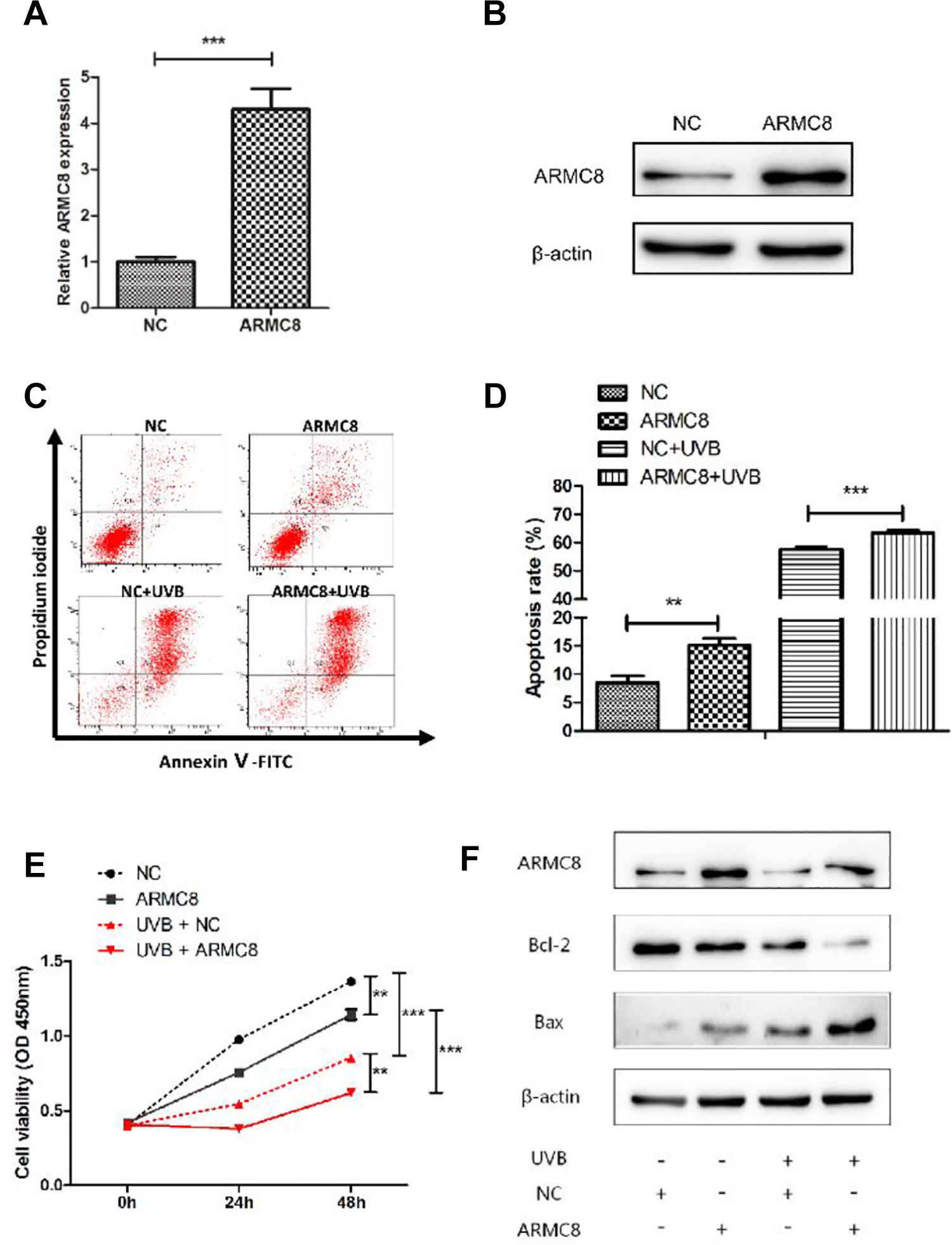
ARMC8 overexpression inhibited proliferation and accelerated apoptosis of UVB-induced HaCaT cells. A, The relative mRNA expression of ARMC8 was detected by RT-qPCR after transfected with pCMV-ARMC8/pCMV-NC in HaCaT cells. B, ARMC8 protein expression was detected by Western blot after transfected with pCMV-ARMC8/pCMV-NC in HaCaT cells. C, D, Apoptosis assay by Annexin V/PI double staining was performed in HaCaT cells transfected with pCMV-ARMC8/pCMV-NC with UVB irradiation or not. E, The proliferation capacity of HaCaT cells transfected with pCMV-ARMC8/pCMV-NC either alone or together with UVB irradiation was detected by CCK-8 assay. F, ARMC8, Bcl-2, and Bax protein expressions were detected by Western blot after transfected with pCMV-ARMC8/pCMV-NC either alone or together with UVB irradiation in HaCaT cells. ARMC8 indicates armadillo-repeat-containing protein 8; CCK-8, Cell Counting Kit-8; mRNA, messenger RNA; PI, propidium iodide; RT-qPCR, real-time quantitative polymerase chain reaction; UVB, ultraviolet B.
ARMC8 Knockdown Can Reverse the Effects of MiR-664 Inhibition on UVB-Induced HaCaT Cells
To understand whether ARMC8 could reverse the effects of miR-664 on UVB-induced HaCaT cell proliferation and apoptosis, we knocked down miR-664 and ARMC8 simultaneously. As depicted in Figure 6A, when compared with the NC inhibitor + siNC group, cell proliferation capacity was markedly inhibited in the miR-664 inhibitor + siNC group, and cell proliferation capacity in the miR-664 inhibitor + siARMC8 group was increased significantly compared with the miR-664 inhibitor + siNC group (both P < .001; Figure 6A). In the UVB-induced HaCaT cells, ARMC8 knockdown could also alleviate the proliferation repression caused by miR-664 inhibitor (P < .01; Figure 6B). Likewise, we assessed the apoptosis rate of all 8 groups, and results indicated that ARMC8 knockdown reduced the apoptosis rate in both UVB-induced HaCaT cells (both P < .05) and no UVB irradiation HaCaT cells (both P < .01). Finally, we performed Western blot analysis to determine the protein levels of ARMC8 and apoptosis-related proteins (Bcl-2, Bax). Results revealed that the protein level of Bcl-2 increased by ARMC8 knockdown, and the level of Bax was just the reverse whether with UVB irradiation or not (Figure 6E). In conclusion, ARMC8 knockdown can reverse the effects of miR-664 inhibitor on UVB-induced HaCaT cell proliferation repression and apoptosis.
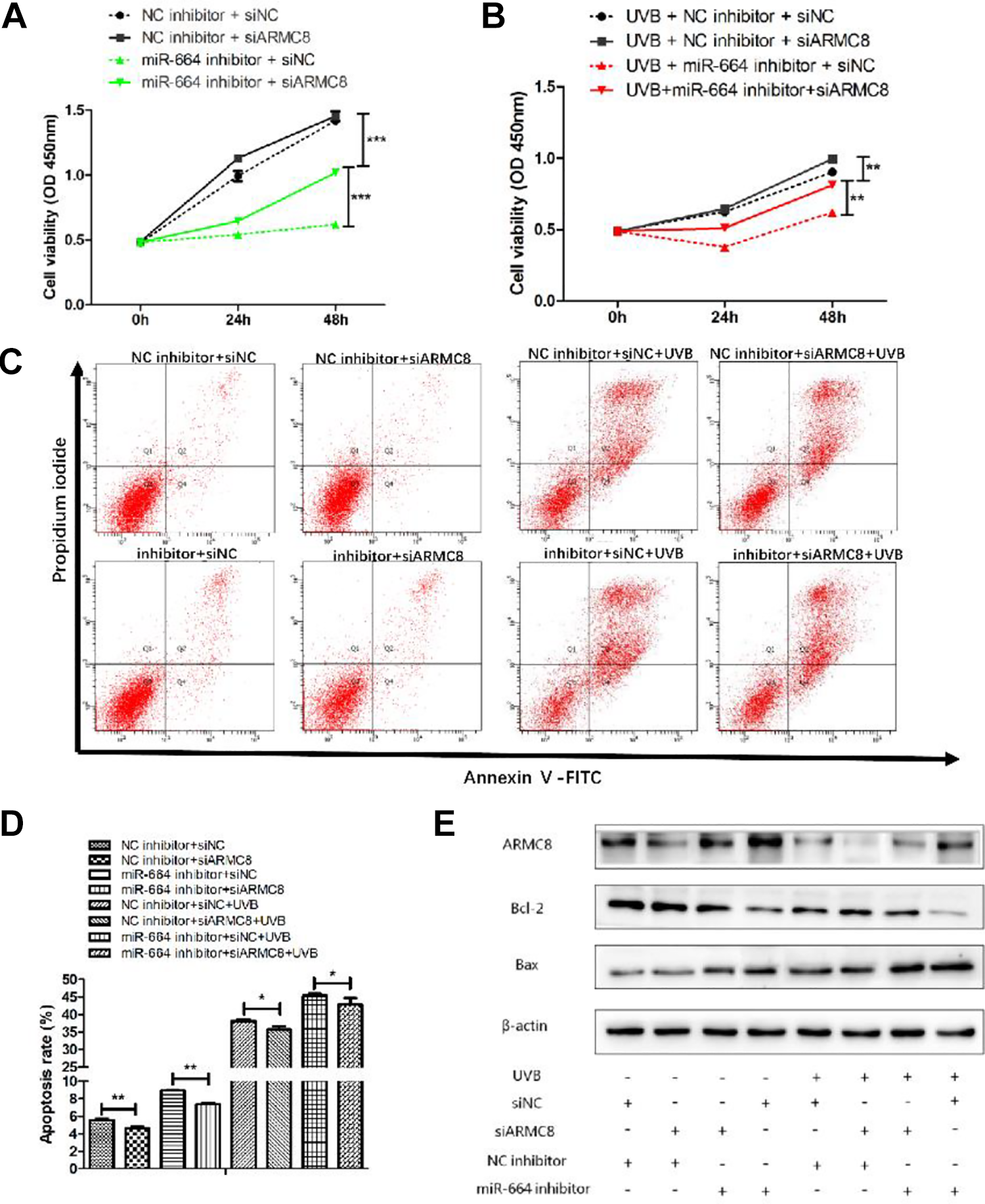
ARMC8 knockdown could reverse the effects of miR-664 inhibition on UVB-induced HaCaT cells. A, B, The proliferation capacity was detected by CCK-8 assay after transfected with siARMC8/siNC and miR-664 inhibitor/NC inhibitor either alone or together with UVB irradiation in HaCaT cells. C, D, Apoptosis assay by Annexin V/PI double staining was performed in HaCaT cells transfected with siARMC8/siNC and miR-664 inhibitor/NC inhibitor with UVB irradiation or not. E, ARMC8, Bcl-2, and Bax protein expressions were detected by Western blot transfected with siARMC8/siNC and miR-664 inhibitor/NC inhibitor either alone or together with UVB irradiation in HaCaT cells. ARMC8 indicates armadillo-repeat-containing protein 8; CCK-8, Cell Counting Kit-8; NC, negative control; PI, propidium iodide; UVB, ultraviolet B.
Discussion
Recent studies have shown that UVB can regulate miRNAs expression, which is involved in cell proliferation, differentiation, apoptosis, autophagy, carcinogenesis, and DNA damage response. 20 -24 It indicates that miRNAs play a vital role in UVB-mediated cell responses.
Some studies have revealed that UVB radiation could induce the alteration of miRNAs expression in HaCaT cells. MicroR-1246 promotes UVB-induced apoptosis by downregulating RTKN2 expression. 25 MicroR-23a regulates DNA damage repair and apoptosis in UVB-irradiated HaCaT cells. 26 UVB irradiation induces the upregulation of miR-141 expression, and miR-141 expression increases apoptosis in HaCaT cells. 27 MicroR-145 alleviates interleukin-6-induced increase in sensitivity to UVB irradiation by downregulating MyD88 in HaCaT cells. 28 Ultraviolet B irradiation inhibits iASPP (Inhibitor of Apoptosis Stimulating Protein of p53) expression through inducing miR-340 expression, thereby promoting RPE cell apoptosis and suppressing cell proliferation. 29 In our present study, we found that the expression of miR-664 upregulated in UVB-radiated HaCaT cells. And miR-664 promoted cell proliferation and suppressed cell apoptosis in HaCaT cells after UVB irradiation. Thus, we speculated that miR-664 could protect against UVB radiation-induced cell damage.
Armadillo-repeat-containing protein 8 belongs to the family of armadillo-repeat-containing proteins, which have been found to be involved in diverse cellular functions including cell–cell contacts and intracellular signaling. 30 Herein, we assessed the possible effect of ARMC8 in UVB-induced damage on HaCaT cells. After UVB irradiation, ARMC8 mRNA and protein levels were significantly reduced, suggesting the potential role of ARMC8 in UVB-mediated HaCaT cell damage. In addition, ARMC8 knockdown partially reversed the effect of UVB irradiation and miR-664 inhibition on HaCaT cell proliferation and apoptosis, as well as apoptosis-related proteins. These results suggest that silence of ARMC8 could efficiently reverse UVB-mediated HaCaT cell proliferation and cell apoptosis.
Conclusion
Our data confirm that UVB-induced HaCaT cell proliferation repression and apoptosis are regulated by miR-664/ARMC8 axis. Overexpression of miR-664 protects UVB-induced HaCaT cell against proliferation inhibition and apoptosis via downregulating ARMC8. Hence, miR-664/ARMC8 would play a potential role in clinical treatment of UVB-mediated skin cell damage.
Footnotes
Authors’ Note
C.Z. and X.X. contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship, and/or publication this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the grant from the National Natural Science Foundation of China (nos. 31971167, 81673105, and 81472922; ![]() ) and the Natural Science Foundation of Guangdong Province (no. 2020A151501664).
) and the Natural Science Foundation of Guangdong Province (no. 2020A151501664).
