Abstract
Radiotherapy plays an increasingly crucial role in the treatment of non-small cell lung cancer (NSCLC). Local tumor recurrence and tumor progression caused by intratumoral heterogeneity induced radiotherapy resistance remain the primary causes of radiotherapy failure. However, the lack of a suitable cell line model has hampered the exploration of the dynamic mechanisms of radiation resistance. We established 3 groups of equidifferent gradient dose irradiation surviving/resistant human lung cancer cell lines based on A549, H520, and H460 cells with clinical conventional fractionated radiotherapy (CFRT) (2 Gy × 20 F, 2 Gy × 30 F, and 2 Gy × 40 F). The radiosensitivity of the cells was detected by clone formation assay, EDU cell proliferation assay, neutral comet assay, and γ-H2AX immunofluorescence staining. The radiosensitivity and proliferation viability were increased in a received dose-dependent manner. Compared with parental cells, DNA double-strand breaks (DSBs) in cell lines that received higher-dose irradiation were significantly reduced. We successfully constructed equidifferent gradient dose irradiation surviving/resistant NSCLC cell lines whose radiation surviving and resistant abilities were increased in a received dose-dependent manner. This preclinical cell model could be used to dynamically observe and detect the radiation surviving/resistant biomarkers during radiotherapy stress, elucidate the mechanism of radiation resistance.
Introduction
Lung cancer is one of the most common solid tumors in the world, and non-small cell lung cancer (NSCLC) accounts for 85% of all lung cancers. Lung adenocarcinoma and lung squamous cell carcinoma are the most common subtypes of NSCLC. 1,2 The majority of lung cancer patients are diagnosed in the advanced stages, without the opportunity of surgical resection, leading to poor prognosis. Advances in lung targeted therapy, immunotherapy, and various combination treatment modes have provided new treatment directions for advanced NSCLC. In addition to radiotherapy and chemotherapy, these approaches have shown a wide and significant effect. 3,4 In the integrated management system of lung cancer, radiotherapy has always been an active and effective local treatment and has played an important role in the radical treatment of early, locally advanced lung cancer and in palliative treatment of advanced, locally advanced lung cancer. 2,5 However, radiation resistance has limited the long-term control of lung cancer, and has led to failure of local control and disease progression. 6 Therefore, elucidation of the molecular mechanism of radiotherapy resistance is required to understand its occurrence and to identify more effective treatment biomarkers to ultimately improve the lung cancer treatment response rate.
Radiotherapy kills tumors by ion beams including photon rays, electron rays, and proton rays. By causing ionization and excitation of bioactive macromolecules, and inhibiting cell energy synthesis, radioactive rays attack tumor tissues and cells, ultimately leading to tumor cell death. 7,8 The effectiveness of radiotherapy depends on the radiation sensitivity of tumor cells. Like most malignant tumors, lung cancer is composed of subclones with distinct molecular characteristics, leading to intratumoral heterogeneity. 9 Understanding the occurrence and evolution of tumor spatiotemporal heterogeneity could help to identify specific therapies for radiation resistance. 10 McAbee suggested that the biological characteristics and intratumoral heterogeneity evolution of glioma in nude mice were changed after irradiation. 11 Tumor clonal heterogeneity is considered a major factor in tumor evolution, progression, and resistance to chemoradiotherapy. 12 The regulatory mechanisms of radiation resistance have attracted increasing levels of attention. However, there is still no biomarker that can accurately indicate whether tumor cells are surviving or resistant during the course of radiotherapy. 13
CFRT for solid tumors is the most common and basic way of radiotherapy treatment. CFRT typically consists of fractionated dose (1.8∼2.0Gy/F) delivered over a period of 5-7 weeks and the total radiotherapy dose reaches 60Gy. 14 Fractionated therapy is believed to promote tumor oxygenation and facilitate the recovery of normal tissues, and hence improve the treatment response. 15,16 During fractionated radiotherapy, cancer cells would acquire radioresistance and caused treatment failure finally. To improve therapeutic outcomes, it is crucial to elucidate the underlying mechanism of intracellular treatment resistance. However, existing cancer-derived cell lines cannot pertinently reflect the heterogeneity of tumor energy changes during clinical radiotherapy. Additionally, most studies about radiation resistance mechanisms rely on research models based on parental lung cancer cell lines and radiation-resistant cell lines exposed to a certain radiation dosage. 17 -19 This discontinuous cell model ignores the effect of radiation dose differences during the establishment of a radiation-resistant cell line and does not always characterize the lung cancer surviving/resistant state. To better study the mechanism of residual recurrence after lung cancer radiotherapy, we established a series of surviving/resistant lung cancer cell lines derived from equidifferent gradient dose irradiation to explore the specific changes that occur during the stress of radiotherapy. Through multiple X-ray irradiation does and cultivation, these cell lines became a suitable model for probing the mechanism of lung cancer radiotherapy resistance.
Materials and Methods
Cell Culture
Human lung adenocarcinoma cell line A549, lung squamous cell line H520 and large cell lung cancer cell line H460 were purchased from American type culture collection (ATCC). All cell lines were cultured in RPMI-1640 containing 10% fetal bovine serum (FBS, Gibco; Thermo Fisher Scientific, Inc., MA, USA), 1% penicillin-streptomycin(Gibco; Thermo Fisher Scientific, Inc., MA, USA) and maintained at 37°C and 5% CO2 constant temperature incubator. After 20, 30 and 40 fractions (F) irradiation, all these 3 kinds of cell lines were respectively named as A549/H520/H460-20F, A549/H520/H460-30F and A549/H520/H460-40F.
Establishment of a Radiation Surviving/Resistant Cell Line Model
The medical linear accelerator (Varian, MA, USA) was used for X-ray irradiation. The irradiation conditions were: 6 MV X-rays, source skin distance was 100 cm, DT = 2 Gy, irradiation field was 10 cm × 10 cm, and the entire cell culture flask was covered with an equivalent 1.5 cm plexiglass plate for compensation. After irradiation, the cells were cultured and passaged. When cells recovered proliferative abilities, they were retreated with another CFRT (2 Gy/F). After a total of 20, 30, and 40 CFRT cycles, the radiation surviving/resistant cell line model were established. The total dose reached 40 Gy, 60 Gy, and 80 Gy, respectively, and took approximately 8-10 months.
Clone Formation Assay
A549/H520/H460-20F, −30F, and −40F cells were digested, counted, and plated in 6-well plates (Corning). After X-ray irradiation (DT = 0 Gy, 2 Gy, 4 Gy, 6 Gy, and 8 Gy) the cells were cultured for another 14 days and stained with crystal violet. A clone was defined as more than 50 cells and the colony formation rate was calculated as colony number/plating cell number. A survival curve was drawn using the “multitarget-single hitting” model: SF = 1 − (1 − exp( − k × D))^N.
EDU (5-Ethynyl-2’-deoxyuridine) Cell Proliferation Assay
After cell digestion and counting, 1 × 105 cells were incubated in a 96-well plate and cultured for 24 hours. The cell-light EDU apollo567 in vitro kit (#C10310-1, Ribobio, China) was used to label cells following the manufacturer’s instructions. After EDU labeling and incubation for 2 hours, cells were fixed with 4% paraformaldehyde and Apollo and Hoechst33342 staining were performed. Fluorescence microscopy (Olympus, Japan) was used to visualize and photograph the cells.
Neutral Comet Assay
All cell lines were cultured for 24 hours and exposed to 6 Gy, after which they continued to propagate in an incubator. At 1 hour after exposure, cells were digested to produce a 5 × 104 cell/ml single-cell suspension. Cells from the suspension were spread on agarose gels on glass slides, lysed for 3 hours, and incubated overnight in 1 × TBE. In dark conditions, electrophoresis was performed, and cells were fixed in H2O2 for 10 min. Lastly, cells were stained with propidium iodide (PI, # ST511, Beyotime, China). Comet images were photographed using a fluorescence microscope (Olympus, Japan). The comet images for each cell were independently analyzed and recorded, and at least 50 cells were analyzed per slide. The tail moment was calculated for each comet.
γ-H2AX Immunofluorescence Staining
Each cell treatment group was seeded and incubated at 37°C for 24 hours. After receiving irradiation (6 Gy X-rays), cells were cultured for another 2 hours and prepared for immunofluorescence by incubation with γ-H2AX (Phospho S139) antibody (# ab81299, Abcam, MA, USA). Firstly, cells were fixed with 4% paraformaldehyde for 20 min and then washed in Tris Buffered Saline(TBS) 3 times for 5 min each. The cell membranes were then ruptured by 0.2% Triton X-100 treatment. Cells were then incubated with the γ-H2AX (1:500) primary antibody for more than 2 hours, and washed twice with TBS for 1 min. The cells were incubated with the secondary goat anti-Rabbit IgG antibody (1:200; # ab150077, Abcam, MA, USA) for 1 hour at room temperature. Then cell nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI) for 5 min. The slides were then enclosed and observed under a fluorescence microscope (Olympus, Japan).
Statistic Analysis
Statistic analysis were calculated by SPSS 21.0 software (Chicago, IL, USA) and GraphPad Prism v 6.01 (La Jolla, CA, USA). All data were expressed as means ± standard deviation (SD). Statistical significance was evaluated by Student’s t-test. P value< 0.05 was considered statistically significant. CASP software was used for neutral comet analysis and the comparison of TM mean between groups was performed using the Mann-Whitney u test. All experiments were repeated independently at least 3 times.
Results
Establishment of a Radiation Surviving/Resistant NSCLC Cell Model
To construct representative preclinical cell models, 3 common NSCLC cell lines, A549, H520, and H460 were used. The parental A549, H520, and H460 cells received CFRT (2Gy/F), and the cell irradiation frequency was up to 20, 30, and 40 fractions (Figure 1A). Eventually, 3 equidifferent gradient dose irradiation cell lines were harvested, and the derived cell lines were named according to radiation fractions, (Table 1). All cells were stably cultured and passaged to obtain 3 continuous equidifferent gradient dose radiation surviving and resistant cell lines (Figure 1B). The NSCLC cells were repeatedly exposed to X-rays cyclically and the cells underwent the apoptosis-survival-resistance process with accumulating radiation dose. In summary, we adopted the CFRT mode to irradiate lung cancer cells and obtained surviving/resistant cell lines with equidifferent gradient dose irradiation.
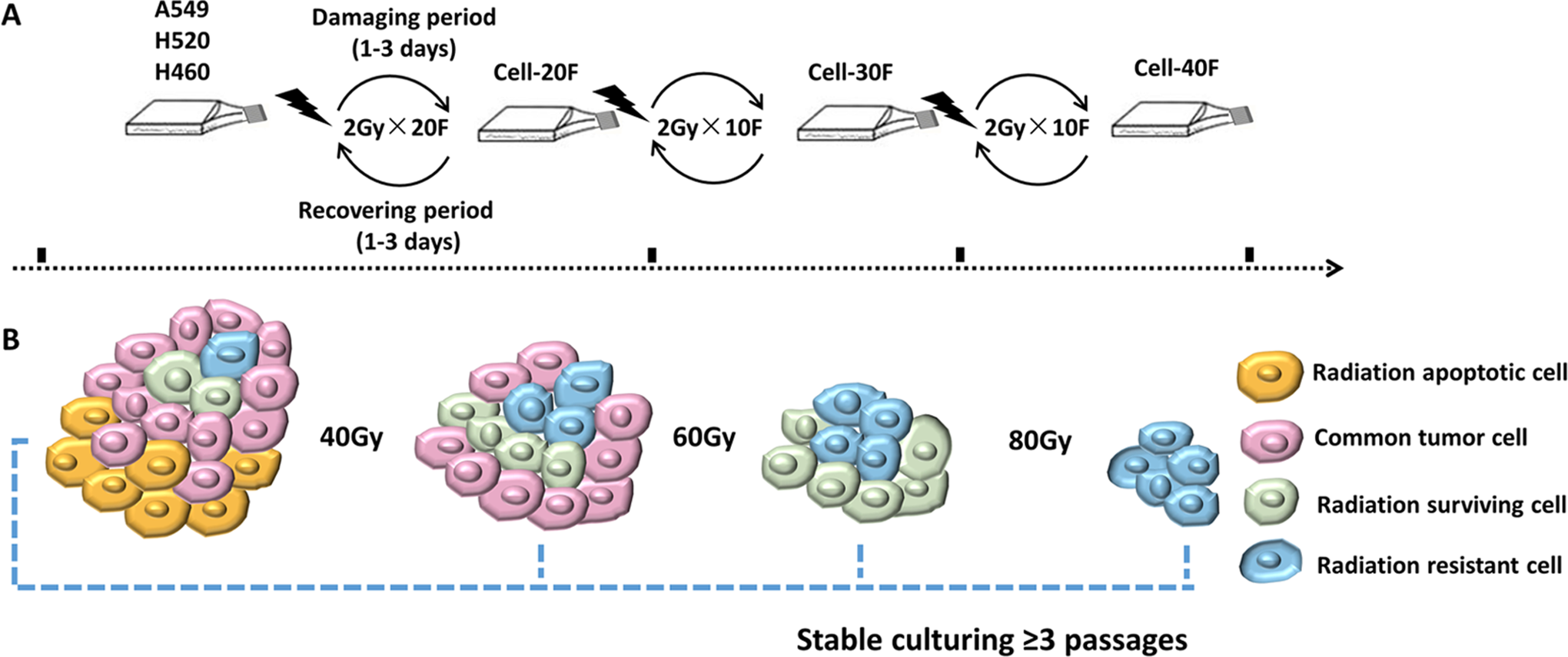
Pattern diagram for establishment of radiation surviving/resistant NSCLC cell lines. A. The conventional fractionated radiotherapy process of NSCLC cell lines. Every cell line received 2Gy/Fraction/Day X-ray irradiation. Living through damaging period, cell would reach proliferating stage, then another cycle of conventional radiotherapy was implemented. When conventional radiotherapy dosage reached 40Gy/20F © 60Gy/30F © 80Gy/40F, 3 gradient dose cell lines were stable cultured ≥3 passages and stored in liquid nitrogen. The establishing process lasted about 8-10 months. B. Model diagram for cell component changes during radiotherapy. Proliferative lung cancer cells contain heterogeneous components, where yellow spheres represent radiation apoptotic cells; pink spheres represent common tumor cells; green spheres represent radiation surviving cells and blue spheres represent radiation resistance cells. After receiving increasing radiotherapy, the resistant cell subsets gradually were screened out and occupied the main growth superiority.
Nomenclature of 3 Radiation Surviving/Resistant NSCLC Cell Lines.

1Adenocarcinoma.
2Squamous carcinoma.
3Large cell carcinoma.
The Survival Fraction of Irradiation Resistant Cells Exceeded That of Irradiation Surviving Cells in NSCLC
We constructed 3 groups of surviving/resistant cell lines with equidifferent gradient dose irradiation. To evaluate the radiosensitivity of these cell lines, parental A549, H520, and H460 cells and their derived surviving/resistant cell lines were exposed to gradually increased doses of 0 Gy, 2 Gy, 4 Gy, 6 Gy, and 8 Gy. Survival curves of A549/H520/H460, A549/H520/H460-20F, A549/H520/H460-30F, and A549/H520/H460-40F were obtained (Figure 2). Compared with A549-30F, H520-30F, and H460-30F, the A549-40F, H520-40F, and H460-40F cell lines showed significant radiation resistance when exposed to 2 Gy, 4 Gy, 6 Gy, and 8 Gy (p < 0.05) and A549/H520/H460-20F and A549/H520/H460-30F had moderate radiosensitivity. For all 3 different dosage cell lines, we observed a gradual dose-dependent increase of radiation resistance when compared to the parental cell lines.

Radiosensitivity of established radiation surviving/resistant cell lines and parental cells. A. B. C. Representative photographs and corresponding growth curves in colony formation assay. X-rays irradiation was enforced on radiation surviving/resistant cell model and parental cells, with dosage = 2 Gy, 4 Gy, 6 Gy and 8 Gy, respectively. The colony formation rate was calculated. Three independent experiments were conducted; The bar graphs showed the means ± SD (n = 3). *** P < 0.001, ** P < 0.01, *P < 0.05. vs A549-30F © H520-30F © H460-30F.
Irradiation Resistant Cells Had an Apparent Proliferation Ability in NSCLC
We speculated that radiation tolerant cell lines had increased proliferative abilities. Cell proliferative potential can be detected by EDU cell proliferation assay. We observed that, compared with A549 (0.2451 ± 0.3806), the proliferation ability of A549-20F (0.3240 ± 0.3448), A549-30F (0.3454 ± 0.3869), and A549-40F (0.4849 ± 0.2222) were significantly increased (Figure 3A). Similar results were observed for the H520 and H460 surviving/resistant cell lines (Figure 3B-C). Consistent with the survival curve data, these results indicated that proliferative ability positively correlated with radiation dosage in these cells.

Representative EDU+ cell staining in radiation surviving/resistant cell models and parental cells. A. B. C. EDU+ cells fluorescence expression in A549/H520/H460, A549/H520/H460-20F, A549/H520/H460-30F and A549/H520/H460-40F. The EDU+ cells were marked with red fluorescence. The cell nucleus were stained with DAPI presented with blue fluorescence (magnification, ×200) .Quantity of EDU+ cells in every cell line were calculated in corresponding column diagram. Three independent experiments were conducted; The bar graphs showed the means ± SD (n = 3). *** P <0.001, ** P < 0.01, *P < 0.05. vs A549, H520, H460.
Radiation Damage in Irradiation Resistant Cells Was Decreased in NSCLC
Exposure of tumor cells to radiation induces DNA double-strand breaks (DSB). The residual DNA of a DSB can be observed as the “tail” of a comet. Therefore, the comet assay is often used as an indicator of cell radiosensitivity. We examined radiation-induced DNA damage using the neutral comet assay (Figure 4A-C). The degree of damage degree in A549-20F (p < 0.0001), A549-30F (p < 0.0001), and A549-40F (p < 0.0001) cells was significantly reduced when compared with parental A549 cells. Similar results were also observed for H520- and H460-derived cell lines (p < 0.0001).

Neutral comet assay revealed the distribution of radiation damage. A. B. C. Representative photographs and corresponding scatter diagrams in A549/H520/H460, A549/H520/H460-20F, A549/H520/H460-30F and A549/H520/H460-40F. The “comet” cells were stained with PI and presented with red fluorescence (magnification, ×400) .Quantity of tail moment in every cell was calculated in corresponding scatter diagrams. At least 50 cells on each slide were recorded. *** P < 0.001, ** P < 0.01, *P < 0.05. vs A549, H520, H460.
Expression of γ-H2AX Foci Was Decreased in NSCLC Radiation Surviving/Resistant Cells
Tumor cells damaged by ionizing radiation, leading to DNA DSB, also cause the formation of H2AX (S139) phosphorylation foci. We examined γ-H2AX expression using immunofluorescence staining. In 3 radiation surviving/resistant lung cancer cell groups, the formation of γ-H2AX foci was reduced (Figure 5A-C). A549/H520/H460-30F (p < 0.0001) and A549/H520/H460-40F (p < 0.0001) cell lines had significantly fewer γ-H2AX foci than did every parental cell line. However, for A549/H520/H460 cell lines, only A549-20F (p = 0.0147) and H460-20F (p = 0.0139) had significantly fewer γ-H2AX foci than did their parental cell lines. Taken together, these results indicated that the accumulation of γ-H2AX foci reduced significantly in radiation surviving/resistant lung cancer cells with increasing radiation dose. These results indicated that with the increasing of dose exposure, the radiation damage was reduced, suggesting that radiation resistance was gradually increasing.
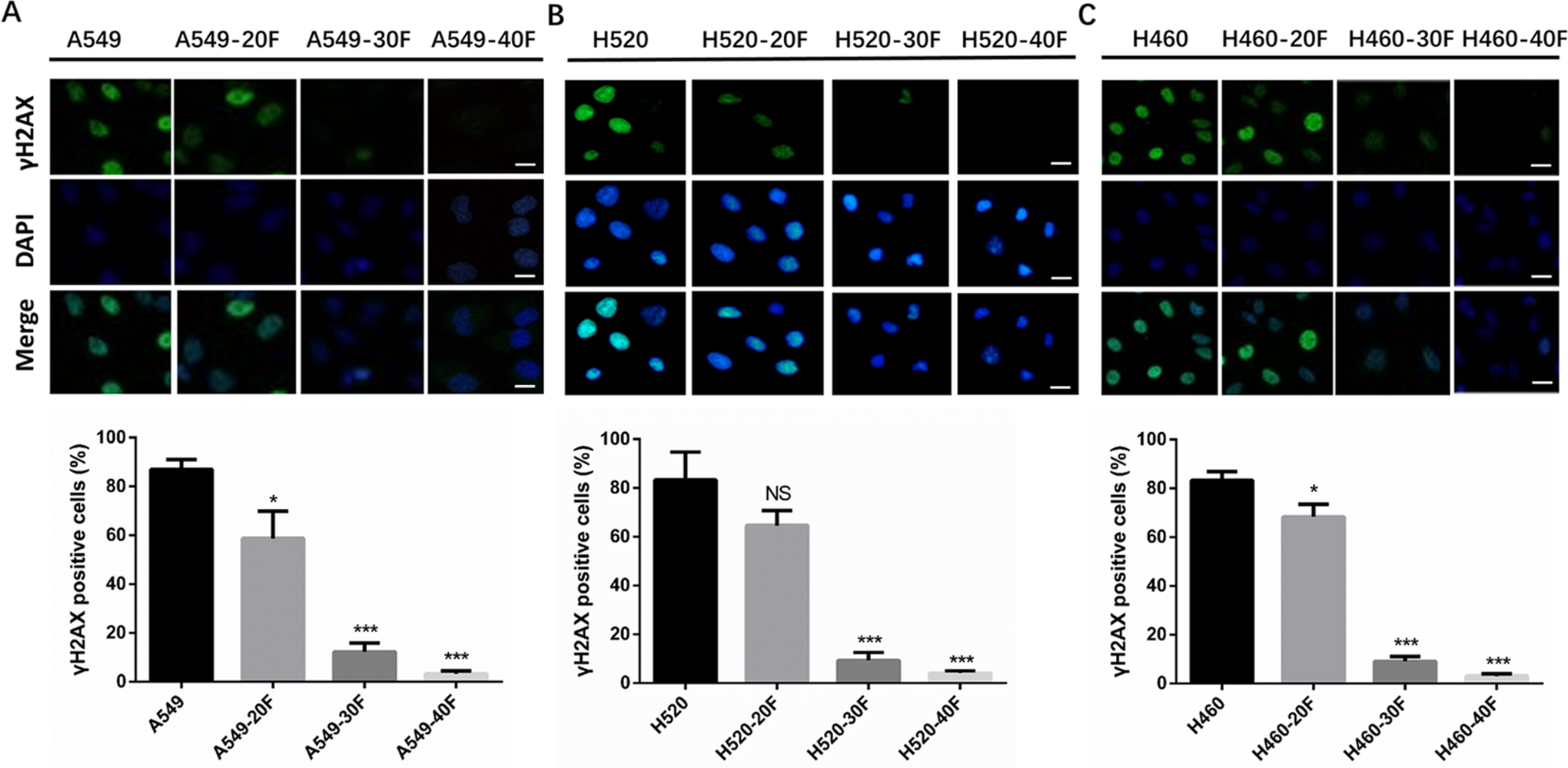
γ-H2AX immunofluorescence in radiation surviving/resistant NSCLC cell models. A. B. C. Representative photographs and corresponding column diagram in A549/H520/H460, A549/H520/H460-20F, A549/H520/H460-30F and A549/H520/H460-40F. The γ-H2AX expression was marked with red fluorescence. The cell nucleus was stained with DAPI presented with blue fluorescence (magnification, ×400) .Quantity of γ-H2AX expression was calculated in corresponding column diagram. The bar graphs showed the means ± SD (n = 3). ***P < 0.001, ** P < 0.01, *P < 0.05. vs A549, H520, H460.
Discussion
In the comprehensive treatment of NSCLC, radiotherapy has become increasingly prominent. New radiation therapy techniques, such as stereotactic body radiation therapy, is more frequently being used as an effective radical treatment for small solitary lung cancer. 20 In particular, some randomized, prospective data showed that stereotactic body radiation therapy or stereotactic ablative radiotherapy could significantly improve survival benefits in patients with oligometastatic NSCLC. 21 -23 Additionally, the combination of radiotherapy and immune checkpoint inhibitors shows a synergistic anti-tumor response in clinical and preclinical studies. 24 -26 Although radiotherapy has an obvious curative effect, the incidence of locoregional relapse after concurrent chemoradiotherapy remains high at 24-50%. 27,28
The generation of tumor cell radioresistance is a complex process involving multiple genes, multiple factors and multiple mechanisms. However, the specific mechanism of radiotherapy resistance to lung cancer cells is still unclear. Previous studies have demonstrated that the radioresistance of cancer was attributed to many biological mechanisms. One of the reasons is the existence of intrinsic cancer stem cells (CSCs), which represent a small, but radioresistant cell subpopulation that exists in heterogeneous tumors. Glioblastoma cancer-stem like cells (GSCs) have self-renewal, genomic stability and tumor initiating capacity, which display stubborn resistance to radiotherapy. 29 Moreover, cancer cells can undergo epithelial-mesenchymal transition (EMT) and then cause radioresistance. 30 When radioresistant NSCLC cells are exposed to ionizing radiation, CB11 induced cell death by inhibiting EMT. 31 There are also researches report that tumor microenvironment (TME) plays a vital role in determining the responses of radiotherapy. 32 Lee etc. found that TGF-β and hypoxia/reoxygenation promote radioresistance of lung cancer cells and tumor progression through ROS-mediated activation of Nrf2 and EGFR. 33 And many inflammatory factors, chemokines and related signaling pathways play an important role in radiation resistance in lung cancer cells. 34 More and more studies present clonal heterogeneity within tumors are considered to be one of the main factors of radiotherapy resistance too. After irradiation, cells within the tumor react differently, with radio-sensitive and radio-resistant components existing. We speculated that radiation resistance caused by intratumoral heterogeneity led to tumor recurrence and progression. 35 To evaluate radiotherapy efficacy, Zhou reported that non-invasive MRI could monitor the physiological changes caused by radiotherapy. 36 However, there remained no convincing preclinical model to dynamically monitor the occurrence of radiation resistance. The previous established cell lines model only consisted of parental cells and a single derived radiation-resistant cell line, 18 -20 and could not fully recapitulate changes in biomolecular properties during radiotherapy. Recently, a series of head and neck cancer isogenic cell lines with different radiosensitivities has been established. Using next-generation sequencing, scientists described exactly how the molecular pathways change during the process of radiotherapy resistance. 37 Similar cell models are necessary for studying radiation resistance in NSCLC. Our team has established the radiation surviving/resistant cell lines based on equidifferent gradient CFRT, focusing on a series of cell lines with different radiosensitivity. Using these cell models, the relationship between changes in radiation dosage and intratumoral heterogeneity could be better clarified.
We constructed 3 groups of equidifferent gradient dose lung cancer cell lines and detected their radiosensitivity. Analysis of their survival curves showed that A549/H520/H460-30F and A549/H520/H460-40F were closer to the radiotherapy resistance state, and that A549/H520/H460-20F was closer to the radiotherapy sensitivity state. Our equidifferent gradient dose cell lines preferentially simulated the cell sensitivity–survival–resistance stage during radiotherapy. Our data indicate that increasing radiation dose results in significantly increased radiation resistance. Exposure to ionizing radiation contributes to the generation of more radiation-resistant cells, including those expressing cancer stem cell markers. 38 By means of various omics detection, such as next-generation sequencing, our preclinical experimental models could be used to increase our understanding of the dynamic changes in the continuous transition from radiotherapy sensitivity to resistance, to explore heterogeneity in the radiotherapy process, and to evaluate the dynamic subcloning evolution of different states over time.
However, the regulation of multicellular organisms is complicated. In many cases, clinical characteristics of NSCLC, including tumor stage and volume, tumor microenvironment characteristics, and intrinsic cellular characteristics might influence tumor therapeutic response. As a result, enhanced DNA repair, regulation of cell cycle progression, and scavenging reactive oxygen species would promote the radiotherapy effect. 39 Interaction between the tumor microenvironment and radiotherapy should also be considered. The relationship between intratumoral heterogeneity and radiation resistance mechanism needs to be further explored using in vivo experiments. Here, we have described the establishment of radiation surviving/resistant lung cancer cells based on equidifferent gradient dose irradiation, which practically simulates the process of clinical CFRT. This provides a good preclinical model for exploring the mechanism of radiotherapy resistance and NSCLC recurrence in the course of radiotherapy.
Conclusions
We constructed a serial of suitable cell lines to dynamically monitoring changes during radiotherapy. These equidifferent gradient dose irradiation surviving/resistant NSCLC cell lines acquired dose-dependent radio-resistant ability. With these preclinical cell models, we could dynamically research the radiation surviving/resistant biomarkers, elucidate the mechanism of radiation resistance.
Footnotes
Author Contribution
Lijuan Wang and Xiaoxia Zhu are authors contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [No. 81972853] and [81572279], Clinical Research Startup Program of Southern Medical University by High-level University Construction Funding of Guangdong Provincial Department of Education [LC2019ZD009].
