Abstract
Although scutellarin has been extensively investigated, its effects on glioma are unclear. This study intended to reveal this regulation and the underlying mechanisms. The U251, M059K, and SF-295 cell lines were treated with gradient concentrations of scutellarin and then IC50 was calculated. SF-295 cells selected for subsequent procedures were treated with four concentrations of scutellarin. Then, proliferation, apoptosis, and cell cycle, as well as the protein and mRNA expression of significantly differentially expressed genes identified by next-generation sequencing (NGS), were examined. The curative effect of scutellarin was validated by 5-FU as the positive control. Scutellarin inhibited proliferation and induced apoptosis and G2/M cell cycle arrest in the SF-295 cell line in a dose-dependent manner. The effect of scutellarin was similar to but significantly weaker than the effect of 5-FU. The NGS results showed that genes associated with anti-apoptosis signaling pathways were significantly reduced after treatment. The Western blotting results indicated that the expressions of TP63/BIRC3/TRAF1/Bcl-2 were reduced in a dose-dependent manner, as well as the mRNA levels determined by qRT‒PCR. Our original conclusion revealed that scutellarin may inhibit glioma growth in a dose-dependent manner via the p63 signaling pathway which may provide a potential medicine for glioma chemotherapy.
Introduction
Glioma is the most common malignant tumor of the central nervous system, accounting for 80% of primary brain tumors. 1 According to its biomarkers and histological characteristics, glioma was categorized as low-grade glioma (LGG, WHO I–II) and high-grade glioma (HGG, WHO III–IV) by the World Health Organization (WHO) in 2016. Glioblastoma or GBM, which accounts for 56.6% of primary malignant brain tumors, is the most lethal type of glioma. 2 The survival of GBM patients is 3∼4 months without any treatment and 5∼6 months with positive treatment, including operation and chemo-radiotherapy. 3 In recent decades, there have been revolutionary advances in GBM treatment as a result of improvements to tumor imaging technology and micro-invasive surgery; however, the prognosis of GBM patients is still poor, as the survival after surgery is 14.2 months, 4 and the 5-year survival rate is only 5.5%. 5 The strong invasion, recurrence, and resistance to chemo-radiotherapy of GBM may be responsible for the poor prognosis. The exploration of new chemotherapy medicines that could solve chemoresistance may provide a potential approach for GBM treatment.
Traditional Chinese medicine is an excellent candidate for research on anti-cancer agents because these medicines can modulate immunity, enhance chemotherapeutic effects, inhibit undesirable reactions, and eliminate resistance to chemotherapeutics. 6 Scutellarin, a flavonoid that is the main active monomer of Scutellaria barbata and Erigeron breviscapus, 7 has been proven to have anti-inflammatory and immuno-modulatory effects. 8 At the same time, flavonoids also have anti-neoplastic effects and play anti-neoplastic roles in different cancer cell cultures through different mechanisms. 9 Scutellarin inhibits proliferation and metastasis, induces cell apoptosis and differentiation of cancer cells, and plays anti-neoplastic roles in gastric cancer, 10 colon cancer, 11 and non-small cell lung cancer (NSCLC). 12 However, the influences of scutellarin on GBM, as well as the underlying mechanisms, are still unclear. 13 In this study, we investigated the anti-tumor effect of scutellarin on GBM in in vitro cell models and revealed the possible mechanisms by combining next-generation sequencing with confirmation via Western blotting and qRT‒PCR.
Materials and Methods
Cell Culture and IC50 Calculation
The M059K, SF-295, and U251GBM cell lines were obtained from the American Type Culture Collection (Rockville, MA, USA) and cultured in complete DMEM culture medium. Scutellarin and 5-FU, which were obtained from Sigma‒Aldrich Inc., were dissolved in DMSO to make 10 mg/mL stock solutions and then diluted into gradient concentrations. Then, the three GBM cell lines were seeded in plates and treated with gradient concentrations of scutellarin.
CCK-8
Glioma cells were plated in 96-well plates (6000/well) and treated with the gradient concentrations of scutellarin mentioned above, and the cells were incubated for 48 h. Then, cell proliferation was analyzed by CCK-8 colorimetric assay (450 nm absorbance). The experiments were performed three times.
Colony Formation
A total of 500 cells in 2 mL DMEM were plated in 6-well plates, and gradient concentrations of scutellarin were added. After 24 h of incubation, the cells were washed with PBS and stained with Giemsa staining solution. The colonies that contained more than 50 cells were counted. The experiment was repeated three times.
Cell Apoptosis
After the designated treatment, the cells were washed, harvested, and counted. A total of 5 × 105 cells were resuspended in 500 μL of binding buffer, and then 5 μL of Annexin V and 5 μL of PI were added and incubated in the dark for 15 min at room temperature, according to the manufacturer’s instructions (KGA1024, KeyGEN BioTECH, China). The apoptosis rate (%) was determined with a cytometer (CytoFLEX, Beckman Coulter, USA). The cells were also viewed under an inverse fluorescence microscope. The experiments were repeated three times.
Cell Cycle
After the designated treatment, the cells were harvested, washed with ice-cold PBS, and fixed with 70% ethanol at 4°C overnight. The ethanol was removed, and the cells were resuspended in PBS containing RNase A (50 μg/mL; KGA511, KeyGEN BioTECH, China) and then incubated with PI (50 μg/mL; KGA511, KeyGEN BioTECH, China) for 30 min in the dark prior to analysis by flow cytometry (CytoFLEX, Beckman Coulter, USA). The data were analyzed, and the percentage of cells in the G0/G1, S, or G2/M phase was calculated. DMSO-treated cells were used as untreated controls. The experiments were repeated three times.
Wound Healing Assay
In wound healing assays, cell motility was assessed by measuring the movement of cells into a wounded area. The cells were seeded in 6-well plates, and a 2.0 mm wound was generated by scraping with a 20 μl pipette tip. The rate of wound closure was monitored after 24 h, and the ratio to the width of the wound at 0 h was calculated. Each experiment was performed in triplicate.
Cell Invasion Assay
In the invasion assay, 30 μL Matrigel (BD Biosciences, Bedford, MA, USA) was added to the upper chamber of the Transwell apparatus with an 8°μm pore size membrane (Corning Incorporated, MA, USA). After the Matrigel solidified at 37°C, 1 × 105cells were added into the upper chamber; complete medium with 20% FBS was added to the lower chamber. Membranes coated with Matrigel were swabbed with a cotton swab and fixed with 100% methanol for 10 min after 24 h of incubation. The membranes with cells were incubated with crystal violet. The number of cells attached to the lower surface of the polycarbonate filter was counted. The results are expressed as the mean of triplicate experiments.
Western Blotting
Primary Antibodies Applied in This Work.

Quantitative Real-Time PCR
Primers Applied in qRT–PCR.

DNA Sequencing and Correlation Analysis
DNA sequencing was carried out via the next-generation sequencing method on the Illumina platform after library quality inspection. Pearson correlation analysis was used to obtain genes positively associated with scutellarin disposal. A heat map was generated to list the co-expressed genes and associated clinical characteristics according to scutellarin disposal. KEGG pathway enrichment analysis was conducted to determine the biological function of the related gene sets.
Statistical Analysis
Statistical analyses were performed using SPSS 22.0 statistical software (SPSS, Chicago, IL, USA). Differences between groups were analyzed, and P < .05 was considered statistically significant. Graphs were generated with Graph Prism 7 (GraphPad Software, San Diego, CA, USA).
Results
IC50 Calculation
When gradient concentrations of scutellarin were applied, the cell viability gradually decreased. Then, the IC50 values in the three cell lines were calculated; the values were 273.567 μg/mL in U251 cells, 278.557 μg/mL in M059 K cells, and 92.556 μg/mL in SF-295 cells (Figure 1A). The IC50 values in the M059K and U251 cell lines were very close to the total lethal dose, so only SF-295 cells were used in the subsequent procedures, as the IC50 value in that cell line was convenient to monitor. Scutellarin induced cell viability inhibition and clone formation inhibition. A. The result of MTT and the calculation of IC50 of scutellarin in three cell lines. B. The result of cell clone assay. C. The statistic result of cell clone assay. * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < .05; △ significant difference was detected compared to Group 3, P < .05. D. The molecular structure of scutellarin. 1. Control group; 2. A quarter LC50 group (23.14 μg/mL); 3. A half LC50 group (42.68 μg/mL); 4. LC50 group (92.56 μg/mL).
In the following experiments, glioma cells were divided into four groups: the control group (Group 1, 0 μg/mL), the quarter IC50 group (Group 2, 23.14 μg/mL), the half IC50 group (Group 3, 42.68 μg/mL), and the IC50 group (Group 4, 92.56 μg/mL). The dose-dependent regulatory effect of scutellarin on glioma was then examined.
Scutellarin Application Suppressed Clone Formation and Induced Apoptosis
As the clone formation assay showed, when scutellarin was applied, the clone number was significantly lower than that in the control group (Figure 1B and C, P < .05). However, the numbers decreased significantly as the concentration of scutellarin increased (Figure 1B and C, P < .05).
The cell apoptotic rate was also analyzed. When treated with scutellarin, the apoptotic rate of glioma cells increased significantly compared with that of the control group (Figure 2A and B, P < .05). In the treated groups, the apoptotic rate also increased when the scutellarin concentration increased, as there were significant differences among the groups (Figure 2A and B, P < .05). Scutellarin induced cell apoptosis and cell cycle arrest at G2/M stage. A. The result of apoptosis assay. B. The statistic result of apoptosis assay; * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < .05; △ significant difference was detected compared to Group 3, P < .05. C. The result of cell cycle assay. D. The statistic result of cell cycle assay; * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < .05; △ significant difference was detected compared to Group 3, P < .05; E. 1. Control group; 2. A quarter LC50 group (23.14 μg/mL); 3. A half LC50 group (42.68 μg/mL); 4. LC50 group (92.56 μg/mL).
Scutellarin Induced G2/M Phase Arrest in Glioma Cells
Scutellarin application significantly induced G2/M phase arrest compared with the control group (Figure 2C and D, P < .05), and the rate of G2/M phase arrest increased significantly as the concentration of scutellarin increased (Figure 2C and D, P < .05).
Scutellarin Inhibited the Migration and Invasion of Glioma Cells
In the wound healing assay, the gap in the control group was significantly narrower at 24 h than at 0 h. When scutellarin was applied, the gap was significantly wider than that in the control group. However, the gap was significantly wider in the higher dose group than in the lower dose group (Figure 3A and B, P < .05). Scutellarin inhibited migration and invasion of glioma cells. A. The result of wound healing assay. B. The statistic result of wound healing assay; * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < .05; △ significant difference was detected compared to Group 3, P < .05. C. The result of Transwell assay. D. The statistic result of Transwell assay; * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < .05; △ significant difference was detected compared to Group 3, P < .05; 1. Control group; 2. A quarter LC50 group (23.14 μg/ml); 3. A half LC50 group (42.68 μg/ml); 4. LC50 group (92.56 μg/ml).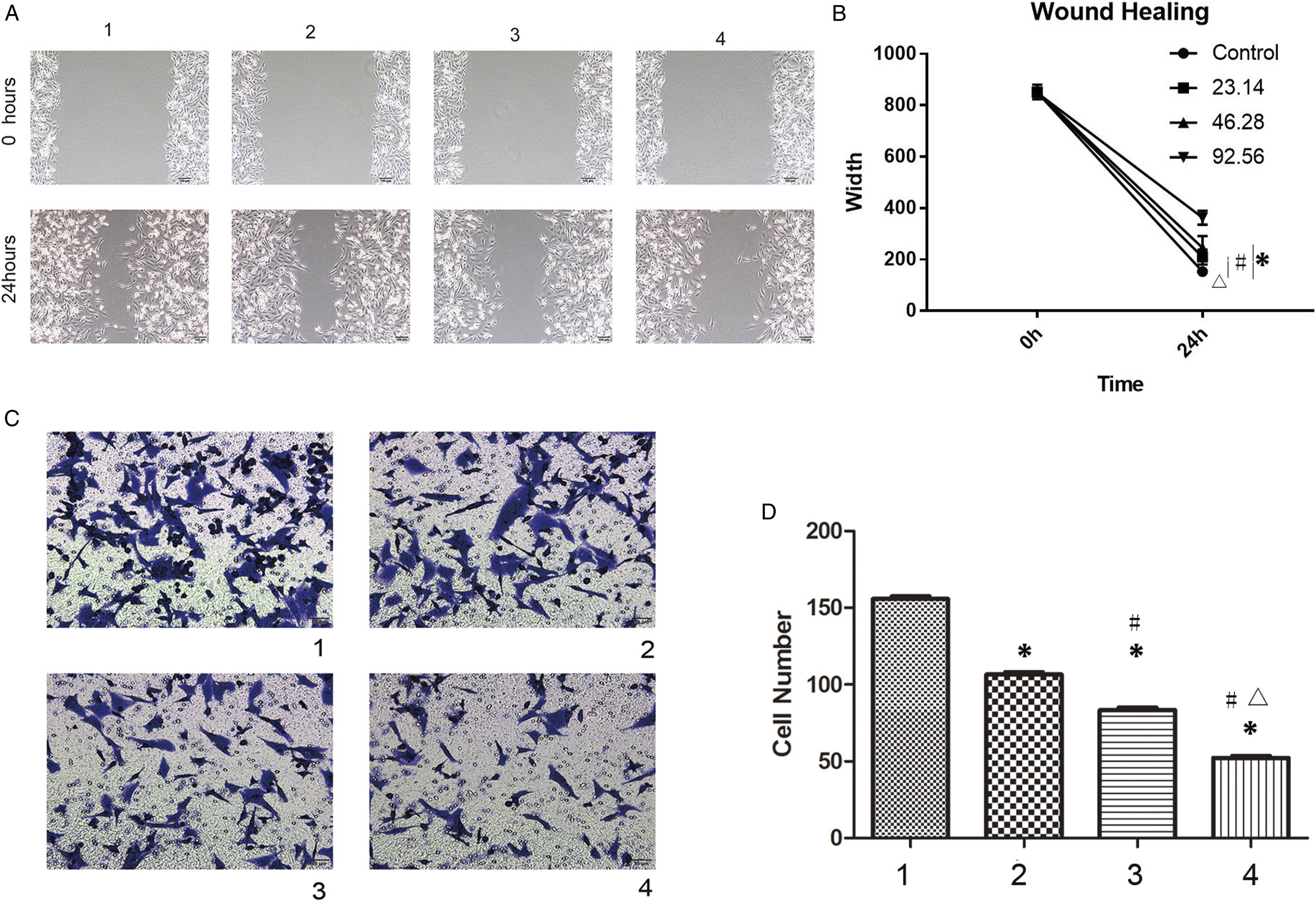
In the Transwell assay, the number of cells that crossed the membrane in the treated groups was significantly lower than that in the control group (Figure 3C and D, P < .05). The results also showed a trend of dose-dependent effects, as the cell numbers decreased as the concentration increased (Figure 3C and D, P < .05).
Validation of Scutellarin Curative Effect
In order to validate the curative effect of Scutellarin, 5-FU was applied as a positive control. The IC50 (67.907 μg/mL) of 5-FU was determined by the CCK-8 method (Supplement Material 1), and the IC50 of scutellarin mentioned above (92.56 μg/mL) was applied in this part.
The result of cell clone formation showed that the cell clone numbers of scutellarin and 5-FU group were significantly lower than the control group (Figure 4A, P < .05), and cell apoptosis result showed the apoptosis rates of scutellarin and 5-FU group were significantly higher than the control group (Figure 4B, P < .05). In addition, cell cycle was arrested in the G2/M stage both in the scutellarin and 5-FU group (Figure 4C, P < .05). Furthermore, cell migration and invasion were suppressed significantly in the scutellarin and 5-FU group, as the cell numbers transferred on to the membrane were significantly lower than the control group in Transwell experiment (the Figure 4D, P < .05) and the cell gap in 24 h of the scutellarin and 5-FU group was significantly broader than the control group in wound healing assay (Figure 4E, P < .05). However, there were significant differences between the scutellarin group and 5-FU group in all experiments (Figure 4A-E, △P < .05). The effect of Scutellarin was similar to but significant differently from the effect of 5-FU. The concentrations of IC50 were applied in test groups, Scutellarin: 92.56 μg/ml, 5-FU: 67.907 μg/ml. A. The result of cell clone assay. B. The result of cell apoptosis assay. C. The result of cell cycle assay. D. The result of Transwell assay. E. The result of wound healing assay; * significant difference was detected compared to the control group, P < .05; △significant difference was detected compared to the scutellarin group.
The p63 Signaling Pathway May be Responsible for Scutellarin-Induced Regulatory Effects
As the sequencing results showed, 383 genes were significantly differentially expressed after scutellarin treatment; 279 genes were downregulated, and 104 genes were upregulated (Figure 5A). The KEGG results analysis suggested that genes related to anti-apoptosis mechanisms were reduced after scutellarin treatment (Figure 5B). P63, which belongs to the p53 family and may play an important role in apoptosis modulation, was significantly lower in the treated group (Figure 5C). Scutellarin might modulate glioma cells via P63 signal pathway. A. The sequencing analysis indicated 279 genes downregulated while 104 genes upregulated after scutellarin treatment. B. KEGG analysis indicated signal pathway which could modulate apoptosis was downregulated. C. The transcription factor analysis showed that p63 which belongs to p53 family was downregulated after treatment. D. The result of Western blot. E. The statistic result of Western blot; * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < 0.05; △ significant difference was detected compared to Group 3, P < .05. F. The result of qRT–PCR; * significant difference was detected compared to Group 1, P < .05; # significant difference was detected compared to Group 2, P < .05; △ significant difference was detected compared to Group 3, P < .05; 1. Control group; 2. A quarter LC50 group (23.14 μg/ml); 3. A half LC50 group (42.68 μg/ml); 4. LC50 group (92.56 μg/ml).
Then, the role of the P63 signaling pathway was investigated by examining Bcl-2, TRAF-1, TP63, and BIRC3 expression via Western blot and qRT‒PCR. When scutellarin was applied, the expression of the four proteins increased in a dose-dependent manner (Figure 5D and E). The qRT‒PCR results also showed a similar trend, as the mRNA levels of Bcl-2, TRAF-1, TP63, and BIRC3 decreased when scutellarin was applied (P < .05, Figure 5F). These results demonstrated that the P63 signaling pathway may be involved in the regulatory effects of scutellarin.
Discussion
The age of GBM onset is 46.3 years old, 14 and there are no specific effective treatments. 2 According to recent literature, the morbidity rate of GBM is increasing. For example, the morbidity rate of GBM in the UK increased from 2.4/100,000 in 1995 to 5.0/100,000 in 2015, while other types of glioma showed no change. 15 The GBM morbidity in the USA will also raise in the next 30 years. 16 This phenomenon may be due to population aging and exposure to ionizing radiation in daily life. 17 The prognosis of GBM is not good, and the treatment of GBM has reached a bottleneck stage; therefore, it is urgent to explore new medicines to cure this refractory malignant tumor.
Over the past decades, natural Chinese medicinal herbs have been described as a significant source of new anti-neoplastic drug candidates. It has been proven that several herb extracts and their bioactive components contain anti-neoplastic properties on GBM cells. For example, the methanol extract of Petroselinum crispum L. showed anti-cancer effects on U87MG cells through its anti-adhesion and anti-proliferative properties, 18 and the aqueous extract of Hedyotis diffusa showed anti-proliferation effect on U87MG cells in a dose- and time-dependent manner. 19 Scutellarin, as an effective component of natural Chinese medicinal herbs, has been used to treat ischemic cardiac disease, neurological disorders, and inflammation. 20 It has been proven that scutellarin plays an important role in anti-cancer processes. Scutellarin can suppress proliferation, induce apoptosis, and arrest cell cycle progression at the G0/G1 21 stage or G2/M 10 stage. In addition, scutellarin can modulate angiogenesis, metastasis, 22 and epithelial–mesenchymal transition 23 in gastric cancer and NSCLC. In this study, we proved that scutellarin could also inhibit the proliferation, metastasis, and invasion of the SF-295 glioma cell line and induce cell cycle arrest in the G2/M phase and cell apoptosis. We also proved that the effect of scutellarin on glioma cells was dose dependent, which is in accordance with the literature. 11
Scutellarin may exert its anti-cancer effect through several signaling pathways, such as the Akt/mTOR, 23 Raf/MEK/ERK, 21 PTEN/PI3K, 11 and TGFβ-1/SMAD2/ROS pathways. 24 In this work, we analyzed the relationship between the p63 signaling pathway and the influence of scutellarin on glioma via next-generation sequencing. We found that 383 genes were differentially expressed after scutellarin treatment; 104 genes were upregulated, while 279 genes were downregulated. According to KEGG enrichment analysis, apoptosis/anti-apoptosis signal pathways were the most significant among the differentially expressed genes, and all the related genes were downregulated. TP63 and the related genes BCL2A1, BIRC3, and TRAF1, which have anti-apoptosis effects, were downregulated. Then, we examined the expression and mRNA levels of these four genes and proved that scutellarin application inhibited the mRNA and protein expression of TP63, BCL2A1, BIRC3, and TRAF1 in a dose-dependent manner.
It has been proven that powerful anti-apoptosis characteristics are the distinguishing features of cancer cells and are responsible for malignant progression as well as chemotherapy resistance. 25 Bcl-2, which is encoded by BCL2A1, belongs to the Bcl-2 family and can modulate apoptosis. It participates in many important biological processes, such as embryonic development and tumorigenesis, by regulating apoptosis via reaction with BAX/BID/PUMA.26,27 However, inhibitors targeting Bcl-2 have been explored and tested in several malignant tumors at present. BIRC3, which is also known as cellular inhibitor of apoptosis 2 (cIAP2), is a protein that belongs to the IAP family that is upregulated in human cancers and can regulate cell apoptosis and survival.28,29 BIRC3 can promote ubiquitinoylation by binding to TRAF1/2 to inhibit apoptosis, 30 and it induces chemotherapy resistance and causes poor prognosis. 29 However, TRAF1, which belongs to the TRAF family, can react with IAP to accept TNF-mediated anti-apoptosis signals. Overactivation of TRAF1 can induce the proliferation of cancer cells and induce chemotherapy resistance.11,31-33 In this study, when scutellarin was applied, the mRNA and protein expression of Bcl-2, BIRC3, and TRAF1 were downregulated. These phenomena may be responsible for inhibiting proliferation and inducing apoptosis in glioma cells.
As an important member of the p53 superfamily, TP63 can also modulate cell apoptosis and cell cycle arrest, which are vital for tumorigenesis. 34 Different subtypes of TP63 are formed via selective gene splicing, which are named Tap63 subtype and ΔNp63 subtype. The Tap63 subtype exhibits pro-apoptotic activity mediated by death receptors (CD 95, TNF, and TRAIL) and the mitochondrial pathway (BAX and PUMA). In contrast, ΔNp63 can act as an anti-apoptosis factor by competitively binding to the TP63 promoter and forming an inactive quaternary structure. 35 However, malignant tumor progression and chemotherapy resistance induced by the upregulation of TP63 have been observed in breast cancer, 36 cervical cancer, 37 and lung cancer. 38 In our work, TP63 expression and mRNA levels were inhibited by scutellarin treatment, and this inhibition was more obvious with increasing concentration. The results of TP63/TRAF1/BIRC3/Bcl-2 inhibition indicated that scutellarin may modulate glioma proliferation and metastasis/invasion via the p63 signaling pathway. However, the regulatory interactions between the four genes TP63, BCL2A1, BIRC3, and TRAF1, as well as whether scutellarin can also inhibit the proliferation and invasion of glioma stem cells (GCSs), remain unclear. Further studies will be conducted to resolve the remaining questions.
Our study proved that scutellarin could inhibit glioma growth by a dose-dependent manner and could be an alternative but not main medicine for glioma therapy as the effect of scutellarin was very close to but significantly weaker than conventional chemotherapy drug 5-FU at IC50. However, the influence of scutellarin on drug resistance glioma was still unclear, and the underlying mechanisms need further investigation. The curative effect of scutellarin may be enhanced by RNAi etc., with the biotechnology development, and scutellarin is expected to become a highly effective medicine for glioma chemotherapy.
Conclusion
Our study proved that scutellarin could inhibit proliferation, metastasis, and invasion while inducing apoptosis and cell cycle arrest by modulating the p63 pathway in a dose-dependent manner. Scutellarin may be a potential medicine for glioma chemotherapy.
Supplemental Material
Supplemental Material - Scutellarin Inhibits Glioblastoma Growth in a Dose-dependent Manner by Suppressing the p63 Signaling Pathway
Supplemental Material for Scutellarin Inhibits Glioblastoma Growth in a Dose-dependent Manner by Suppressing the p63 Signaling Pathway by Yongjie Chen and Wenlan Li in Dose-Response.
Footnotes
Acknowledgments
The authors would also thank Dr. Yuwen Song for providing technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by The Fundamental Research Funds for the Provincial University, No. 31041210047.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
