Abstract
The C-reactive protein is generally considered a marker of inflammation, and it is widely used in clinical practice as a minimally invasive index of any ongoing inflammatory response. Alpha-lipoic acid (ALA) supplementation can be beneficial for human health, especially in the sense of its anti-inflammatory action. The aim of this meta-analysis was to, based on the currently available highest level of evidence (prospective, randomized, double-blind, and placebo-controlled data), investigate the effect of ALA supplementation on CRP levels. Prospective, randomized, double-blind, and placebo-controlled clinical trials were extracted after a systematic search of PubMed, the Cochrane Library, the Web of Science, EMBASE, and the Scopus databases. A random effect model was used in this meta-analysis to investigate the influence of ALA on the blood CRP level. The subgroup analysis and meta-regression were used to identify the source of heterogeneity. This meta-analysis provided evidence of the positive effect of ALA on the reduction of the blood CRP level. The subgroup analysis and meta-regression results indicated that ALA can reduce the CRP level when administrated at a 600 mg dose, and not in higher or lower doses. Also, a shorter duration of study positively correlates with the reduction of CRP after ALA supplementation.
Keywords
Introduction
The C-reactive protein (CRP) is a member of the pentraxin protein family that is produced by liver cells. 1 In smaller amounts, CRP can be produced by some other cells such as vascular smooth muscle cells and macrophages, which can partly explain the possibility to use this biomarker in different fields of biomedicine. 1 The CRP is generally considered as a marker of inflammation, and it is widely used in clinical practice as a minimally invasive index of any ongoing inflammatory response. 2 Most commonly the elevated CRP levels are a consequence of some infectious disease 3 or systemic inflammatory disease. 4 A large number of diseases and conditions are accompanied by an increase in CRP values. For example, cardiovascular diseases are commonly accompanied by a rise in CRP levels. 5 Atherosclerosis, which is in part an inflammatory disease, is followed by a significant rise in blood CRP levels. 6 On the other hand, some conditions such as obesity can also be followed by a rise in CRP levels. 7 This is why obesity and some other conditions, that may simultaneously exist with inflammatory diseases, make it difficult to understand the nature of the increase in CRP values.
Alpha-lipoic acid (ALA) or thioctic acid is an organosulfur compound that can be produced by both plants and animals. 8 The ALA is also synthesized in the human body in mitochondria from octanoic acid and cysteine. 8 Both clinical and preclinical data suggest that ALA supplementation can be beneficial for human health. Most commonly the research is focused on the effect of ALA supplementation on oxidative stress, obesity, diabetes, cancer, Alzheimer’s disease, as well as many other diseases and conditions. 8 One of the highest disadvantages of ALA is its pharmacokinetic profile which restricts its potential clinical administration. The inadequate pharmacokinetic profile of ALA, which is manifested with low bioavailability and short half-life, can now be overcome with the use of innovative pharmaceutical formulations. 8
A common denominator for many diseases and conditions that are the focus of ALA research is CRP. The progression of these diseases, as well as the progression of the processes that underline these diseases, can be monitored with the CRP level. Also, therapeutic success, if the disease is inflammatory at its core, can be monitored with CRP. This is why it is crucial to determine whether ALA can affect CRP levels. Certain clinical trials were created to investigate the influence of ALA supplementation on CRP levels.9-24 Nevertheless, these clinical trials did not resolve the question of the impact of ALA on CRP levels. Some of the studies12,15,16,19,21,22 detected the reduction of CRP level after ALA use, while others9-11,13,14,17,18,20,23,24 failed to do so. Since the results of these trials were not consistent, and also the number of included participants in these trials was relatively low, there was a need to conduct a meta-analysis to resolve this problem. Previously, two meta-analyses were conducted to resolve this problem.25,26 Unfortunately, they did not include all of the relevant studies that were published up to the moment when these meta-analyses were created. Also, several new studies on this topic were published after that.
Considering the previously mentioned facts, especially focusing on potential benefits that could arise from ALA supplementation, the aim of this meta-analysis was to, based on the currently available highest level of evidence (prospective, randomized, double-blind, and placebo-controlled data), investigate the effect of ALA supplementation on CRP level.
Materials and Methods
This meta-analysis was conducted in a line with the Preferred Reporting Items for Systematic Reviews and MetaAnalyses (PRISMA), 27 as well as with Cochrane Handbook guidelines. 28 The protocol of the presented meta-analysis was registered in the PROSPERO database with the identification number CRD42022321714. The PRISMA checklist is presented as a Supplementary file.
Database Search
A researcher experienced in conducting meta-analysis (M.S.) developed the search strategy. The databases searched for eligible studies were PubMed, the Web of Science, the Cochrane library, the Scopus, and the Embase database until May 2022. The query used for the literature search consisted of “(alpha-lipoic acid) OR (alpha lipoic acid) OR (thioctic acid) OR (α-lipoic acid) AND (CRP) OR (C-reactive protein).” Two researchers (Lj. Dj. and L.T.) independently carry out a literature screening by using Rayyan online tool. The identification of eligible studies was done by the previously established inclusion and exclusion criteria. Matching between these two screenings was also done by using Rayyan online tool (M.S.). For the studies that were not overlapping M.S. had the final decision. In addition, the reference list of found original and review articles was manually analyzed to increase the accuracy of the search (T.K., J.D, and D.K).
Search Strategy
The studies were included in the meta-analysis if they met the following criteria: (1) whole article published irrespective of the used language; (2) human subjects were analyzed; (3) randomized trial; (4) double-blinded trial; (5) placebo used as control; (6) minimal duration of study at least one week; (7) CRP level measured; and (8) presenting sufficient data on CRP (baseline, end of the study data or change in CRP values from baseline). The review articles, book chapters, editorials, notes, letters to the editor, abstracts published at congresses, non-human studies, retrospective studies, studies without blinding, non-randomized studies, studies without a placebo control group, studies in which CRP was not reported, and duplicate publications were excluded during the literature search process.
Data Extraction
To perform this meta-analysis, custom-made database was created. In this base, the relevant and for the selected studies important data were placed. The first part of the database consisted of data regarding study identification (first author, year of publication, country where research was conducted), the main features of the study (randomized, double-blind, and placebo control, total number of participants at the beginning and at the end of the study, total number of participants in each group at the beginning and at the end of study), explanation of ALA supplementation (daily dose, duration of treatment in weeks), study population characteristics (participants disease or condition, mean age, male to female ratio, mean systolic blood pressure–SBP, mean diastolic blood pressure–DBP, body mass index–BMI, total cholesterol, baseline low-density lipoproteins–LDL, baseline high-density lipoproteins–HDL, baseline triglycerides), results (CRP values at the beginning and at the end of the study or change from the baseline) and study quality (CCRBT (the Cochrane Collaboration Risk of Bias tool) and JADAD score.
Methodological Quality Assessment
The methodological quality of included studies was assessed with both the JADAD score 29 and the Cochrane Collaboration Risk of Bias tool (CCRBT). With JADAD score the randomization, blindness, and withdrawal of included trials were assessed. The randomization, allocation, blinding of participants and outcome, incompleteness of data, selectivity in reporting, and other biases were assessed with CCBRT.
Statistical Analysis
The primary outcome of this meta-analysis was the change in CRP levels from baseline to the end of the treatment period in response to ALA supplementation. For this meta-analysis, to achieve conservatism in the conclusion process, a random effect model was used. The final results are displayed as weighted mean difference (WMD) with 95% confidence intervals (CIs).
The inconsistency index I 2 and Chi-square test were performed to investigate the heterogeneity of results within extracted clinical trials. If the I 2 index was higher than 75%, or the Chi-square P-value was below .05, the heterogeneity was considered significant. To investigate the reason for detected heterogeneity, the meta-regression and subgroup analysis were performed. For both analyses, the covariates that could possibly influence heterogeneity were pooled out of the extracted studies. If the values for these covariates have been reported in each of the studies that entered our meta-analysis, they could be used for meta-regression or subgroup analysis. The covariates that fulfill these criteria were: dose, duration of follow-up, the total amount of administrated ALA, and age. The association of mentioned covariates values and the standardized mean difference was evaluated by performing a random effect meta-regression and subgroup analysis. In subgroup analysis for investigated variables that are continuous the median was chosen as a cutoff value. The exception to this rule was the dose where 600 mg was used as a cutoff value and three groups were created: less than 600 mg, 600 mg group, and more than 600 mg. The meta-analysis was performed by using R.
Publication Bias
The Funnel plot asymmetry evaluated by visual examination was a starting point in the investigation of potential publication bias. Additionally, trim and fill analysis was conducted to fill potentially missing studies in the funnel plot. Considering the problematic subjectivity that is present in funnel plot assessment Egger’s weighted regression tests were performed, to additionally evaluate the presence of publication bias.
Results
Search Results
The search process ended with the identification of 607 records, and 591 studies were excluded due to the unfulfillment of previously set conditions for inclusion in the meta-analysis. The remaining 16 studies were included in this meta-analysis. A more detailed explanation of the identification process of eligible studies is shown in Figure 1. Flow chart of the study selection via databases and registers in the meta-analysis.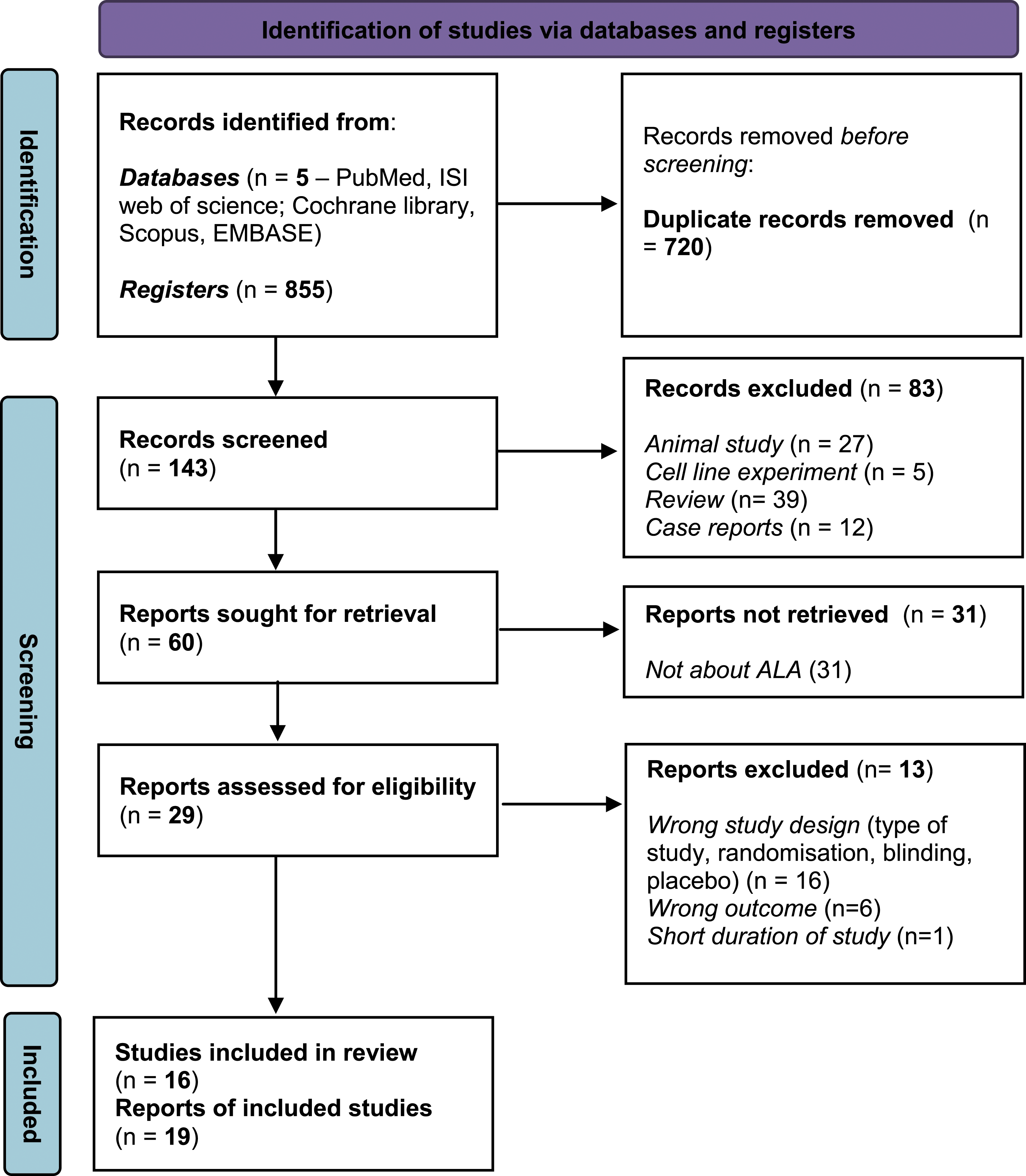
Study Characteristics
Summary of the main characteristic of included studies.
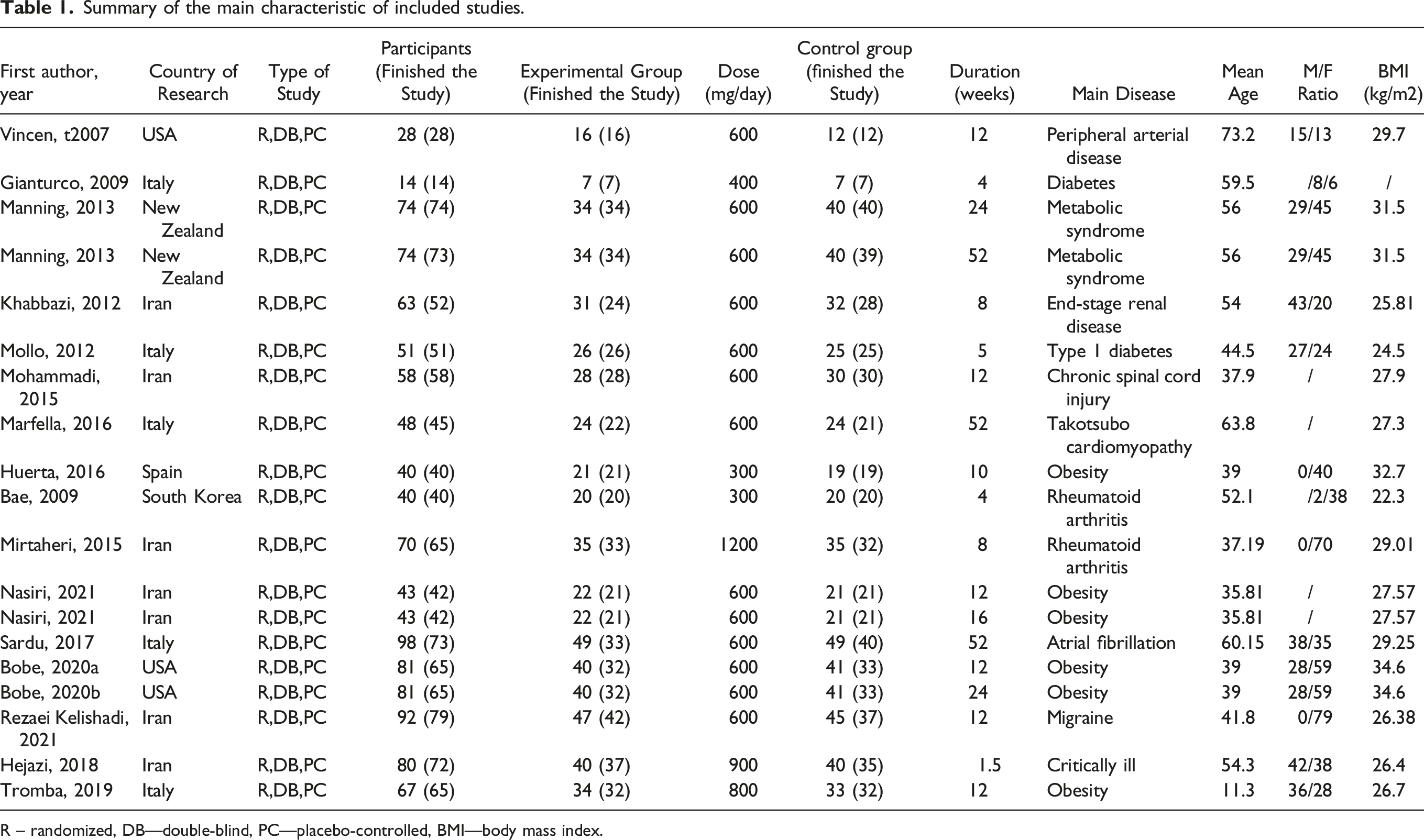
R – randomized, DB—double-blind, PC—placebo-controlled, BMI—body mass index.
The Methodological Quality of Included Trials
The methodological quality of included trials ranged from low quality to high-quality trials with JADAD scores in a range of 2 to 5. The median score of included trials was 5. In total 9 trials had a JADAD score of 5, four trials had a score of 4, one trial had a score of 3, and two trials had a score of 2. Other aspects of methodological quality are presented in the Supplementary material.
Effect of Alpha-lipoic Acid on C-Reactive Protein
The CRP level was determined in all of the extracted trials. The baseline and end of the treatment period CRP level or the change in CRP level between these two periods were pooled after which the meta-analysis was conducted. The obtained result demonstrates significant reduction in CRP level after ALA supplementation (16 studies, 19 study arms, 1040 subjects, WMD: −3.95, 95% CI: −7.09 to −.81, SEM: 1.6, P = .014) (Figure 2). Nevertheless, significant heterogeneity was registered in terms of ALA administration effects on CRP (I
2
= 99.97%, P < .0001). Meta-analysis of the effect of ALA on CRP.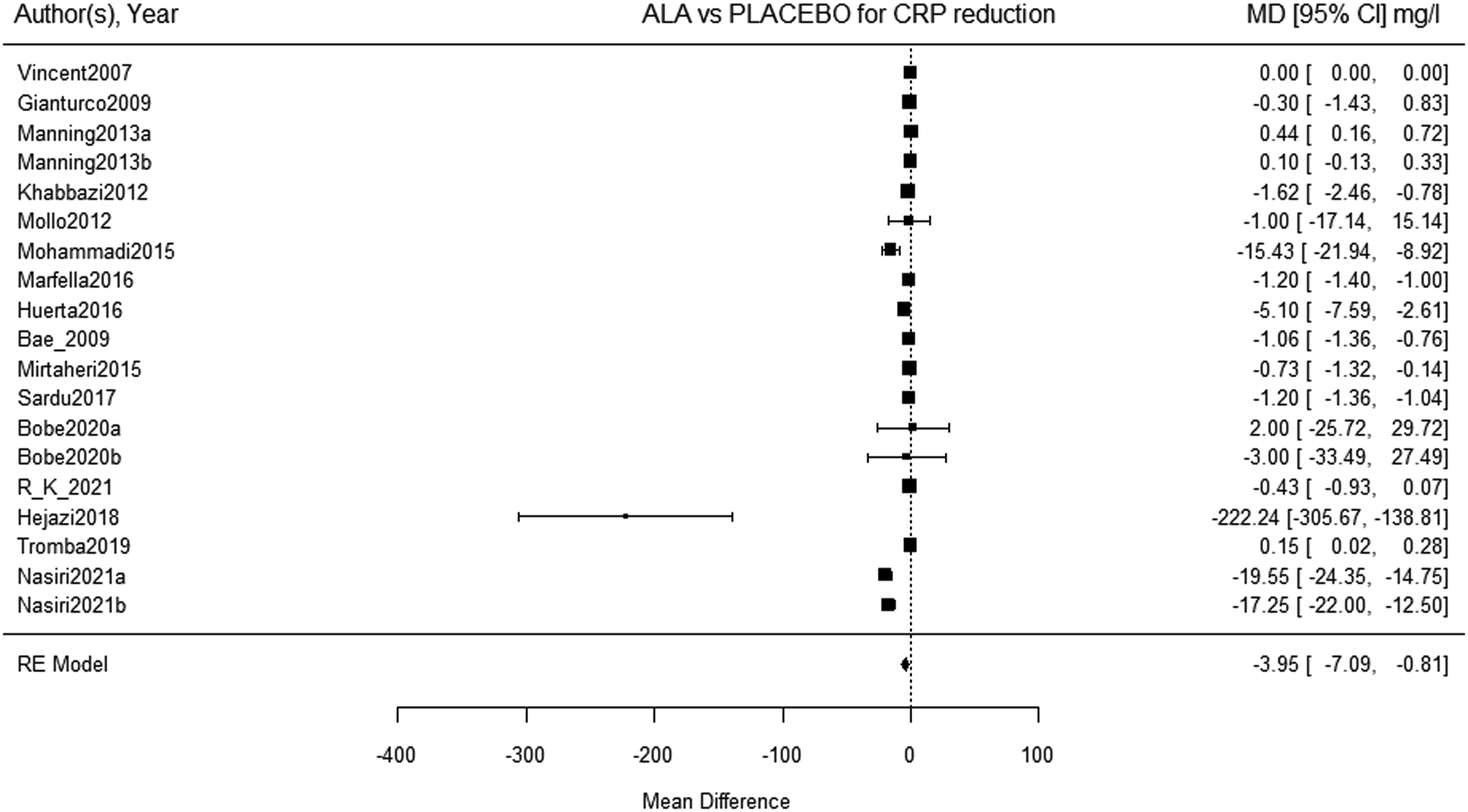
Meta-Regression and Subgroup Analysis
The results of the meta-regression analysis.

Subgroup analysis of ALA administration and its effect on CRP level.

ALA–Alpha-lipoic acid; CI–Confidence interval; SEM–Standard error of measurement.
Publication Bias
The investigation of publication bias was initially done by visual examination of funnel plot asymmetry (Figure 3). The visual examination of the funnel plot indicates the presence of publication bias. The presence of publication bias was confirmed with Egger’s weighted regression test (P < .0001). Trim and fill analysis shows that the estimated number of missing studies on the right side is zero. Publication bias evaluated by the Funnel plot asymmetry.
Discussion
The results of this meta-analysis, which are based on randomized, double-blind, and placebo-controlled clinical trials, indicate that ALA has a significant effect on inflammation, caused by various pathological processes, which is manifested by a significant decrease in CRP level. The heterogeneity measured in this meta-analysis was significant. The meta-regression analysis indicated that the duration of ALA supplementation has a pivotal role in the success of ALA-induced CRP reduction. The results of subgroup analysis indicate that 12 weeks or shorter duration of ALA supplementation is associated with a significant reduction in CRP level. This indicates a short-term and transient effect of ALA on CRP levels. Also, in studies in which ALA was used in a 600 mg dose the CRP reduction was statistically significant. In studies in which ALA was administrated in higher or lower doses, there has been no significant reduction in CRP levels indicating the possible U-shape curve of ALA action. Additionally, if the total concentration of ALA administrated during the supplementation period didn't exceed 50400 mg, a significant reduction in CRP level could be recorded.
The influence that ALA has on different pathological conditions was widely investigated. A critical number of clinical trials, regarding ALA use, was reached and several meta-analyses on different topics were conducted. The conducted meta-analysis revealed the influence that ALA has on lipid profile, male infertility, weight loss, glycemic control, and renal function in patients with diabetic nephropathy.30-33
Previously, two meta-analyses were performed to determine whether ALA supplementation has an effect on the reduction of CRP levels. Both of these meta-analyses were created around the same time in the past.25,26 Also, both of them included only clinical studies that were published up to the year 2017. In the first meta-analysis, conducted by Saboori et al, 25 11 studies were included to address the question of the ALA effect on CRP levels. The studies that were included in the first meta-analysis were randomized and placebo controlled. Also, in this meta-analysis, the authors included 2 studies that were not double-blinded.34,35 The second meta-analysis conducted by Haghighatdoost and Hariri 26 included a total of 11 studies, with 14 study arms. It is important to point out that some of the included studies in the second meta-analysis have not used a placebo as a comparator. 33 It could also be noted that some of the included studies used ALA in combination with other supplements or drugs. 33 It is interesting that after comparing these two meta-analyses it could be seen that although performed in the same period only 6 studies overlapped in both meta-analyses. Nevertheless, the result of both meta-analyses indicates that ALA supplementation decreases CRP levels. Considering previously listed facts regarding the two meta-analyses, also the fact that the most quality clinical trials are randomized, double-blind, and placebo-controlled trials, as well as a fact that several studies with ambiguous results have been published on this topic since 2017, we decided to create a new meta-analysis to have a final clue on the raised question. One of the two previously conducted meta-analyses indicated that the dose of ALA is not a source of heterogeneity and that it has not any significant influence on the CRP level. 32 Additionally, they postulated that the dose of 400 mg/day was sufficient enough to lower the CRP level in a patient with an elevated level of this inflammatory biomarker. 32 The results of our meta-analysis show a completely different situation with a dose marked as a definite source of heterogeneity and a dose of 400 mg as insufficient for the reduction of CRP level. The explanation of these differences lies in the fact that several studies were published on this topic since 2017 giving a new perspective on the investigated question, and in fact that we only included randomized, double-blind, and placebo-controlled trials.
The meta-regression analysis performed in the present meta-analysis revealed that only study duration is inversely associated with ALA supplementation effect on CRP levels suggesting a significant duration-dependent effect of ALA on CRP levels. Even more, our subgroup analysis showed that 12 weeks (median) or shorter ALA supplementation significantly reduce CRP levels. On the other hand, results from a previously conducted meta-analysis by Saboori et al 32 showed that ALA supplementation caused significant CRP reduction when trial duration was more than 8 weeks (median), while subgroup analysis from Haghighatdoost and Hariri 33 study showed no significant CRP reduction when study duration was 9 weeks or longer (median). The observed differences between ours and other published meta-analyses regarding the duration-dependent effect of ALA on CRP levels should be attributed to the difference in the studies involved in the analysis. Although there is no doubt that ALA exhibits a time-dependent effect on CRP levels, the question of the adequate therapeutic length of ALA supplementation to reduce CRP still remains open.
The result that was obtained with our meta-analysis, which indicates that CRP levels can be reduced after ALA supplementation in a dose and duration-dependent manner was not a surprise, since next to the clinical study's ambiguous results there have been certain indications in animal studies that associated ALA supplementation and the reduction of CRP. 36 Still, the understanding of the exact role that ALA has in CRP level control remains incomprehensible, especially if one takes into consideration that in a certain situation in which the CRP level is decreased ALA has, paradoxically, the potential to increase it. 37
The antioxidative and anti-inflammatory roles are one of the best established ALA functions in the human body. The regulation of inflammation by ALA is probably mediated via nuclear factor κB (NF-κB), while oxidative stress is regulated through the Nuclear erythroid 2-related factor (Nrf2) signaling pathway.38 The CRP is capable of binding to the CD32/CD64 receptor which activates the NF-κB signal cascade and induces the inflammation process.38 Probably ALA, by reducing the CRP levels, prevents overactivation of NF-κB signal cascade and reduces the inflammation. The exact molecular link between ALA and the reduction of CRP is not familiar and needs to be established.
The influence of ALA on CRP level, which has been proven in our meta-analysis, needs to be interpreted beyond the pattern where ALA influences the inflammation process and as a consequence of that, the reduction in CRP level is noted. The CRP is not just a simple follower of the inflammatory process. This is important because there has been an increasing rise in evidence suggesting that CRP is a pathological factor responsible for the development variety of diseases (cardiovascular diseases, kidney diseases, diabetes, etc.).38 Observed through these data, the results of our meta-analysis may put forward ALA as a treatment option for numerous diseases.
Conclusion
The present meta-analysis provided evidence of the positive effect of ALA supplementation on the reduction of the blood CRP level. However, considering subgroup analysis and meta-regression results we can strongly suggest that ALA can reduce the CRP level when administrated at 600 mg dose, and not in higher or lower doses, and when the overall concentration of ALA administrated does not exceed 50400 mg. Additionally, a shorter duration of study positively correlates with the reduction of CRP after ALA supplementation.
Supplemental Material
Supplemental Material - The Effect of α-lipoic Acid on C-Reactive Protein Level: A Meta-analysis of Randomized, Double-Blind, and Placebo-Controlled Studies
Supplemental Material for The Effect of α-lipoic Acid on C-Reactive Protein Level: A Meta-analysis of Randomized, Double-Blind, and Placebo-Controlled Studies by Ljiljana Đukić, Lazar Trajković, Tamara Knežević, Jelena Dimitrijević, Danijela Krstić, and Marko Stojanović in Dose-Response
Supplemental Material
Supplemental Material - The Effect of α-lipoic Acid on C-Reactive Protein Level: A Meta-analysis of Randomized, Double-Blind, and Placebo-Controlled Studies
Supplemental Material for The Effect of α-lipoic Acid on C-Reactive Protein Level: A Meta-analysis of Randomized, Double-Blind, and Placebo-Controlled Studies by Ljiljana Đukić, Lazar Trajković, Tamara Knežević, Jelena Dimitrijević, Danijela Krstić, and Marko Stojanović in Dose-Response
Footnotes
Acknowledgments
Only the authors were responsible for the study design, data collection and analysis, publishing decision making, and the final preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was Supported by the Ministry of Education, Science and Technological Development Republic of Serbia, with Grant 451-03-9/2021-14/200110.
Authors' Note
All analyses were based on previously published studies; thus, no ethical approval and patient consent are required.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
