Abstract
Acute bouts of intense exercise increase lactate concentration, which in turn stimulates brain-derived neurotrophic factor (BDNF) production. Cortisol released during intense exercise might inhibit BDNF synthesis. This study examined the acute effects of 2 protocols of strenuous exercise on serum BDNF. Seventeen physically-active healthy females (Age = 20.0 ± 0.9 yr., BMI = 23.0 ± 2.6 kg/m2) performed a strenuous cycle-ergometer graded exercise test (GXT) and a high-intensity interval training session (HIIT). Serum BDNF, serum cortisol, cortisol: BDNF ratio and blood lactate (BLa) were recorded at baseline and immediately following exercise. Although non-statistically significant, the HIIT session elicited a higher magnitude of change from baseline for BDNF (d = 0.17) and cortisol (d = 1.18) than after the GXT (d = -0.26, and d = 0.82, respectively). An interaction was found between GXT and HIIT trials and measurements on BLa levels, with higher post-exertion values after HIIT than after GXT (p < 0.0001, η2 = 0.650, 95%CI = 2.2, 5.2). The higher BLa levels did not raise circulating BDNF. The elevated cortisol levels may have overcome the effects of lactate on BDNF. However, the higher BLa induced by HIIT suggest that interval exercise modality on the long-term could be a feasible intervention to increase circulating peripheral BDNF, at least in untrained healthy women.
Introduction
Brain function is regulated by several stimuli such as pharmaceutical agents, nutritional habits and physical exercise (PE). 1 In response to PE, brain derived neurotrophic factor (BDNF) is one of the main molecules associated with the regulation of signaling pathways responsible for enhancing brain function. 2 In detail, BDNF is a neurotrophin associated with neuron growth and survival, 3 neurogenesis, axonal growth, synaptogenesis, 2,4 -6 improved learning and memory. 2,7 Evidence in rodent models have shown that BDNF is centrally synthesized, mainly at the hippocampus, 8 and studies in humans suggest a similar mechanism. 9,10 Besides to the central effects, scientific data suggest that this neurotrophin has the ability to cross the blood-brain barrier 9 -11 ; thus, the brain releases BDNF into the periphery, a response amplified during PE. 9,10 In the periphery, BDNF is involved in glucose metabolism, 12,13 and seemingly in β-oxidation. 14
In humans, there is evidence showing that peripheral circulating BDNF levels increase following chronic 15,16 and acute PE sessions 10,17 ; indeed, changes in circulating BDNF are purportedly work- and intensity-dependent. 18,19 For instance, BDNF was higher immediately after a session of high-intensity interval training (HIIT) than during a session of moderate-intensity continuous training (MICT). 18 In that study, the blood lactate (BLa) seemed to induce BDNF synthesis in the brain, thus resulting in higher peripheral BDNF levels. 18,20 HIIT is an exhaustive training modality, characterized by repeated short bouts of high-intensity exercise at near to maximal effort (i.e., ≥ 80% HRmax), interspersed with recovery periods. 21 This type of exercise is considered a time-efficient modality, resulting in similar or better metabolic adaptations (i.e., higher oxidative capacity in skeletal muscle, major glycogen storage) compared to other longer duration training protocols. 22 A single session of HIIT elicits similar metabolic demands to other continuous exhaustive exercise modalities such as a graded exercise test (GXT). 19,23 For instance, BLa levels ≥ 10 mMol/l have been reported following completion of a HIIT session in healthy women. 24,25 Additionally to BLa, stress hormones (e.g. cortisol) are also increased after exhaustive exercise protocols (HIIT and GXT). 19,23,26
A GXT is a high-lactate producing exercise protocol used to determine an individual’s functional capacity (i.e., VO2max, VO2peak) 23,27 -29 Besides, studies have shown that this exercise modality immediately increases peripheral BDNF, a response associated with synthesized lactate. 20,23,29,30 Given that GXT and HIIT exercise modalities can both considerably increase BLa concentrations, we hypothesized that these exercise protocols may increase BDNF synthesis in the brain, leading to higher peripheral BDNF levels after completion of the exercise protocols. We additionally hypothesized that BDNF changes will be proportional to circulating BLa magnitude after exhaustive exercise, and is independent from the training modality. Until now, this physiological response has not been demonstrated. Therefore, the aim of the study was to compare the acute effects of 2 exhaustive exercise modalities (GXT vs. HIIT) on serum BDNF. However, opposite to BLa, the cortisol reduces BDNF synthesis. 31 -33 Therefore, the secondary aim of this study was to assess whether 2 different exhaustive exercise modalities increase the serum cortisol levels and suppress the BDNF response to exercise.
Methods
Seventeen healthy female (age = 20.0 ± 0.9 yr., body mass index [BMI] = 23.0 ± 2.6 kg/m2) undergraduate students from the Sports Faculty at the Universidad Autónoma de Baja California were recruited to voluntarily participate in the study. In accordance with the International Physical Activity Questionnaire’s (IPAQ) algorithm, and the Physical Activity Readiness Questionnaire, all participants were considered physically-active (Self-reported IPAQ short version: 4637.9 ± 2060.1 METs, minimum: 8640, maximum: 1419), free of any cardiac or respiratory problems, and capable of performing maximal or sub-maximal physical exercise. All experimental procedures were conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of the Facultad de Medicina y Psicología of Universidad Autónoma de Baja California, México, under the register number 002-2019.
The testing included 3 separate visits to the laboratory. During the first visit, participants received detailed information about all experimental procedures and were encouraged to ask any questions they might had. Then, volunteers read and signed a written informed consent. During the second and third visits, anthropometric and body composition variables were measured by bioimpedance with an Inbody™ model 770 analyzer (Seoul, Korea). For the body composition analysis, participants were instructed to refrain from eating or drinking 2-h before the test. In addition, participants were asked to abstain from performing moderate or vigorous exercise and refrain from consuming ergogenic aids 24-h before testing. Subsequently, in the second visit the female researcher performed a gender health screening of the participants (Supplemental Table 1). In addition, during the second and third visit, participants performed the GXT and the HIIT sessions, respectively (Supplemental Figure 1). All experimental procedures were performed between 11:00 and 13:00 hrs.
Immediately following the body composition assessment, a baseline 5-mL of blood was collected from an antecubital vein (pre-GXT), using a 6-mL Vacutainer tube (# 368175; Becton, Dickinson & Co., Franklin Lakes, NJ) with no additives for serum collection. Blood samples were allowed to clot at room temperature for 40-min and centrifuged at 1500 rpm for 10-min. The supernatant was collected and stored at -20°C until later analysis. Five minutes after the blood collection, resting heart rate (HRrest) (Polar FT1 HR monitor, Kempele, Finland) and systolic (SBP) and diastolic (DBP) blood pressure (Tango M2, SunTech Medical, Morrisville, NC) were recorded. In addition, resting BLa was measured from finger capillary samples (Nova Biomedical Lactate Analyzer, Waltham, MA).
After all baseline measurements were recorded, the participants performed a GXT on an electronically braked cycle-ergometer (Lode Excalibur Sport, Lode B.V., Groningen, Netherlands). Briefly, participants warmed-up during 4-min by pedaling at 20 watts (W). Then, the workload was increased to 60 W, and subsequently 20 W every 60-s until participants reached volitional exhaustion. Throughout the GXT, participants were required to maintain a pedal cadence between 70-80 rpm and remain seated on the cycle-ergometer. The trial finished when the participant was unable to maintain the cadence required or by volitional fatigue. The power (W) attained at the last completed stage was considered as the maximal aerobic power (MAP). The final stage’s immediate HR (HRpeak) and BLa were recorded, followed by recovery HR (HRR) at 60-s post-GXT. Finally, total distance (m) and work (kJ) completed during the GXT were recorded using the Lode Ergometry Manager software, version 9.4.4 (Lode B.V., Groningen, Netherlands). A 5-mL blood sample from the antecubital vein was collected and processed as described before 1-min following the GXT (post-GXT). One week after the GXT, all participants performed a HIIT session; which started with a warm-up at 40% of the previously recorded MAP, followed by a series of 30-s bouts performed at 90% MAP (high-load), and interspersed with 90-s to 120-s of active recovery at 40% MAP (low-load). The duration of HIIT was equivalent to the GXT (i.e., 12-min). HR, HRpeak, BLa, and HRR were recorded before (pre-HIIT) and immediately after (post-HIIT) the session. Session ratings of perceived exertion (RPE) were recorded using the Borg’s 6 to 20 scale immediately after finishing the last stage of the exercise protocol (last high bout in HIIT and last step in GXT). The VO2max was estimated indirectly following a methodology previously reported. 34 Each participant performed the exercise sessions the same day of the week, 1 week apart (Supplemental Figure 1).
Enzyme-linked immunosorbent assays (ELISA) were used to determine serum BDNF (ab99978-BDNF, Abcam, Cambridge, MA) and cortisol (3625-300a, Accu-bind, Lake Forest, CA) by following their respective manufacturer’s instructions. Absorbance was measured at 450 nm (iMark Microplate Absorbance Reader, Bio-Rad, Japan). The BDNF intra-assay coefficient of variability (CV) reported by the manufacturer was 2.8% and inter-assay CV 5.3%. The essay’s sensitivity was 2.4 µg/ml (range 15.6-1000 µg/ml). The cortisol intra-assay CV was 6.4% and inter-assay CV 7.0%. The essay’s sensitivity was 0.366 µg/ml. The cortisol: BDNF ratio (C: B) was calculated by converting cortisol concentration to the same SI unit as BDNF (from µg/dl to ng/ml) to obtain C: B. 35 The last procedure was performed to support the association between BNDF and cortisol as extensively reported before. 35 -37
All statistical analyses were conducted using IBM SPSS, version 22 (IBM Corp., Armonk, NY) with the level of significance set at p < 0.05. All data are presented as mean ± SD. Paired-samples student t-tests were used to compare mean SBD, DBP, total distance and work during exercise trials. Factorial 2 x 2 (trials: GXT, HIIT x measurements: pre, post) repeated measures ANOVA were computed for BLa, serum BDNF, serum cortisol, and C: B. Factorial 2 x 3 trials: GXT, HIIT x measurements: pre, post, 60-s recovery) repeated measures ANOVA were computed for HR. The source of explained variance on ANOVA tests was estimated by partial η2. Significant main effects or interactions were assessed using the Fisher’s Least Significant Difference post-hoc test. Effect size estimates (Cohen’s d; small ≤ 0.2, moderate = 0.5, large ≥ 0.8) were calculated for the magnitude of difference in BDNF and cortisol between pre- to post-measurements, and 95% confidence intervals (CI95%) were calculated for the mean difference estimates. Figures were created by the GraphPad Prism software (La Jolla, CA, USA).
Results
The participant characteristics are described in Table 1. Mean baseline SBP (GXT = 119.9 ± 19.9 mmHg vs. HIIT = 116.6 ± 10.5 mmHg, p = 0.403) and DBP (GXT = 73.6 ± 8.6 mmHg vs. HIIT = 74.5 ± 9.3 mmHg, p = 0.699) were similar between exercise trials. The mean distance (GXT = 4212.3 ±1262.1 m vs. HIIT = 4344.2 ± 1018.0 m, d = 0.02, p = 0.940) and total work (GXT = 63.0 ± 18.9 kJ vs. HIIT = 65.1 ± 15.2 kJ, d = 0.02, p = 0.959) were similar between trials. The RPE was higher in the HIIT (19.8 ± 0.6) than in the GXT (15.8 ± 2.3) session (p ≤ 0.0001, d = 1.98, 95%CI = 2.8, 5.4).
Descriptive Statistics of Participants (n = 17).

Abbreviations: BMI, Body mass index; MAP, Maximal aerobic power.
An interaction was found between GXT and HIIT trials and measurements on mean BLa (p ≤ 0.0001, η2 = 0.650). Two participants were considered outlier and were excluded from the analysis for BLa. The BLa increased from pre- to post-exertion in the GXT (Pre = 1.8 ± 0.9 vs. Post = 12.6 ± 3.1 mMol/l, p < 0.0001, 95%CI = 10.9, 14.3) and HIIT (Pre = 1.8 ± 1.0 vs. Post = 16.3 ± 2.8 mMol/l, p < 0.0001, 95%CI = 14.7, 17.8) conditions. Post-exertion BLa values were considerably higher in the HIIT compared to the GXT condition (p < 0.0001, 95%CI = 2.2, 5.2, Figure 1A). The magnitude of the change was large in both HIIT (d = 6.43) and GXT (d = 4.17) sessions.
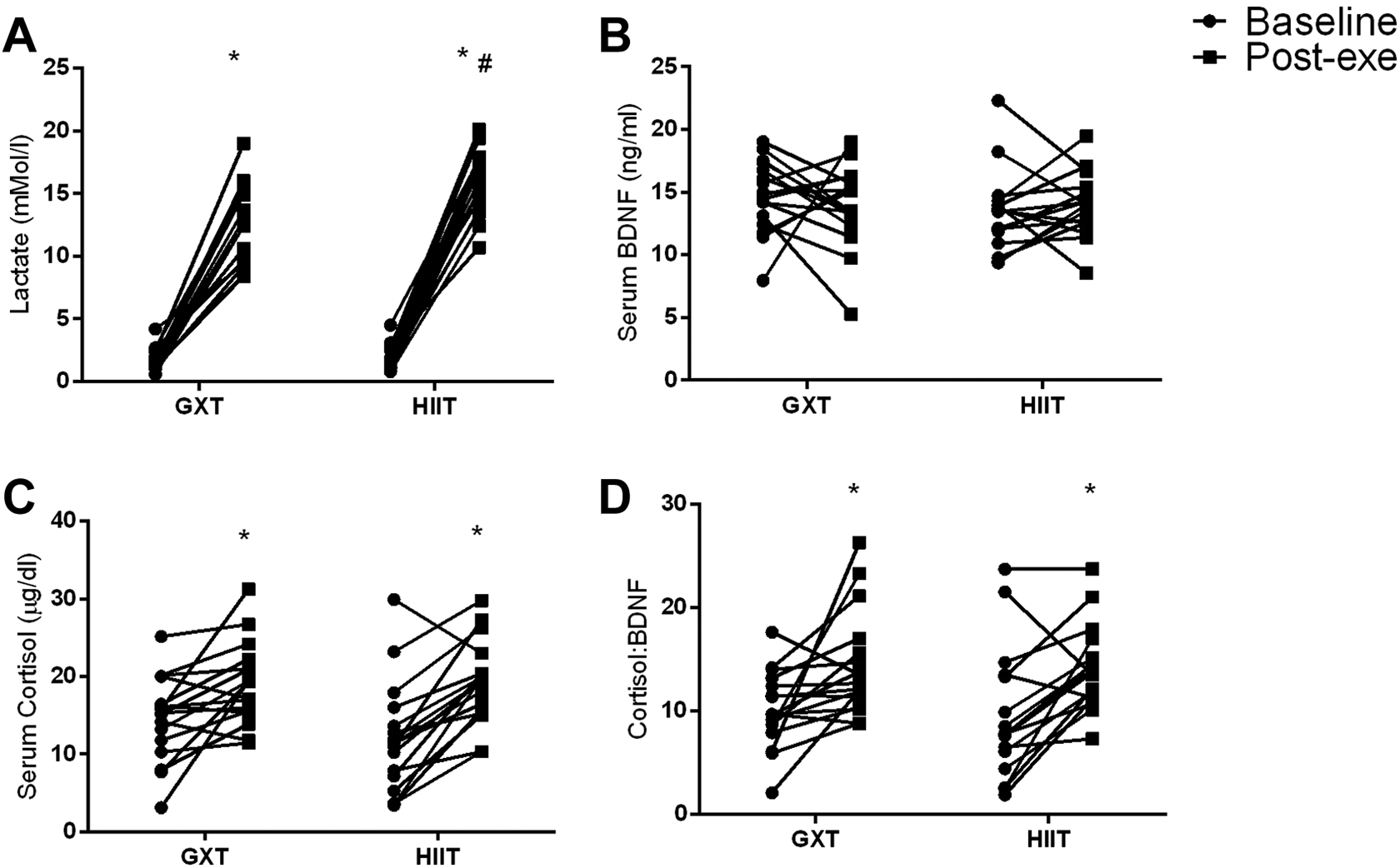
Linear dotplots depicting the response of individual data values for (
No interaction was found between GXT and HIIT trials and measurements on mean serum BDNF concentrations (p = 0.388, η2 = 0.047). The magnitude of the change was small in both, the HIIT (d = 0.17) and the GXT (d = -0.26) sessions. No interaction was found between GXT and HIIT trials and measurements on serum cortisol concentrations (p = 0.139, η2 = 0.132). Regardless of the trial performed, serum cortisol increased following exertion (Pre = 13.2 ± 1.2 vs. Post = 19.1 ± 0.9 µg/dl, p ≤ 0.0001, η2 = 0.715, 95%CI = 3.9, 7.9) (Figure 1C). The magnitude of the change was large in both, the HIIT (d = 1.18) and the GXT (d = 0.82) sessions.
No interaction was found between GXT and HIIT trials and measurements on mean C: B (p = 0.813, η2 = 0.004). Regardless of the trial performed, C: B increased following exertion (Pre = 9.8 ± 0.9 vs. Post = 14.2 ± 0.8, p ≤ 0.0001, η2 = 0.572, 95%CI = 2.4, 6.5) (Figure 1D). The magnitude of the change was large in the HIIT session (d = 0.89) and small in the GXT session (d = -0.09) (see Supplemental Table 2 for mean and SD data of BDNF, cortisol and C: B values).
No interaction was found between GXT and HIIT trials and measurements on mean HR values (p = 0.227, η2 = 0.088). Regardless of the trial performed, HR values increased from baseline following exertion. Post-hoc analysis showed that HR baseline values were lower than values recorded immediately following exertion (Pre = 79.4 ± 2.4 vs. immediately post = 190.5 ± 1.6 bpm, p ≤ 0.0001, 95%CI = 105.9, 116.3) and after 60-s during recovery (60-s recovery = 165.3 ± 2.0 bpm, p ≤ 0.0001, 95%CI = 80.6, 91.2). Recovery HR values were lower than immediately post-exertion values (p ≤ 0.0001, 95%CI = 20.3, 30.1).
Discussion
The current study was designed to compare the acute effects of 2 exhaustive exercise protocols (i.e., GXT vs HIIT) on serum BDNF in healthy physically-active women. The research hypothesis was that both exhaustive exercise modalities would increase BLa and elicit a raise in circulating BDNF. Our data indicate that acute exhaustive exercise did not change significantly circulating BDNF following training. Additionally, higher serum cortisol levels were found following both exhaustive exercises. The increase in cortisol concentration could explain the lack of effect of exercise on circulating BDNF. Finally, the current study found that HIIT greatly increases BLa more than GXT, this was an unexpected finding, since work and distance performed during HIIT and GXT were similar. The speed revolution was higher in HIIT (high bouts); therefore, it is possible that the muscle contraction rate was greater in HIIT compared to GXT; enhancing the metabolic process in interval training. This condition could partially explain the difference in BLa between protocols. Further studies are necessary to elucidate the differences of BLa response between GXT and HIIT. It is worth mentioning that this biochemical marker did not affect circulating BDNF immediately following the exercise, a response that does not support our hypothesis.
It is known that BDNF is a protein sensitive to exhaustive acute exercises. 18,38 Others have suggested that BLa can be a key molecule responsible for inducing BDNF synthesis in the brain. 20,39 In the current study, the BLa increased significantly after both exercise interventions; in fact, higher BLa levels were observed following HIIT. However, there were similar BDNF concentrations following both exercise interventions. These data do not support our hypothesis that peripheral lactate produced following acute strenuous exercise affects circulating BDNF; others have reported similar findings. 19 The apparent lack of BDNF secretion or synthesis can be partially explained by the higher serum cortisol following both strenuous exercise modalities. 31 This phenomena has been reported before, where BDNF increased immediately after the GXT while cortisol levels did not change following an acute ramp exercise. 23 A possible physiological mechanism involved a downregulation of BDNF expression. 32,33 Similarly, chronic studies showed the same physiological response between cortisol and BDNF. For instance, Barari et al. reported that 4 weeks of endurance and circuit resistance exercise increased BDNF levels in young sedentary adults but did not modify cortisol levels. 40 Likewise, 8 weeks of aerobic exercise increased circulating BDNF levels but did not modify peripheral cortisol concentrations. 41 The C: B response reported in this study, could indirectly indicate the increase of cortisol produced in the hypothalamic-pituitary-adrenal axis is an inhibitor of central and peripheral BDNF synthesis. 31
On the other hand, we do not disregard a possible minimal influence of peripheral BLa in BDNF synthesis and expression during acute strenuous exercise. It has been demonstrated in rodents that during exhaustive exercise, the lactate produced in the brain (mainly astrocytes) is more relevant for brain function than the peripheral lactate. 42 Perhaps, during acute exhaustive exercise, the peripheral lactate is used predominantly for ATP synthesis in the brain rather than the regulation of genes associated with cognition as BDNF is. 43
Finally, we do not discard a role of functional capacity (i.e., VO2max and physical activity levels) on the BDNF response. It has been demonstrated that these variables are inversely related to circulating BDNF levels. 44 -46 The participants in the current study were physically active, thus, this condition could explain the lack of BDNF response following both strenuous exercise modalities. Additionally, the menstrual cycle was monitored in the participants (Supplemental Table 1), showing a variation between the follicular and luteal phase in the subjects. In regard to the previous data, we expect that steroid levels (i.e., estrogen) in the participants were heterogeneous. Since estrogen helps regulate BDNF synthesis and activity, 47 -49 we therefore do not dispute that the lack of BDNF response during both exercises were a consequence of the variability of estrogen levels in our participants.
Conclusion
The current study demonstrated that acute HIIT induces higher BLa changes than GXT. However, the increased BLa levels did not raise the circulating BDNF. The elevated cortisol levels observed following strenuous exercise could overcome the lactate effects on BDNF, at least in untrained healthy women. However, we do not discard that the higher BLa induced by HIIT, could be a metabolically feasible intervention to increase BDNF’s synthesis in the brain and its circulation in the periphery in a healthy population on the long term. 39
Supplemental Material
Supplemental Material, sj-pdf-1-dos-10.1177_1559325820970818 - Acute Systemic Response Of BDNF, Lactate and Cortisol to Strenuous Exercise Modalities in Healthy Untrained Women
Supplemental Material, sj-pdf-1-dos-10.1177_1559325820970818 for Acute Systemic Response Of BDNF, Lactate and Cortisol to Strenuous Exercise Modalities in Healthy Untrained Women by Patricia C. García-Suárez, Iván Rentería, José Moncada-Jiménez, Andrew C. Fry and Alberto Jiménez-Maldonado in Dose-Response
Supplemental Material
Supplemental Material, sj-tif-1-dos-10.1177_1559325820970818 - Acute Systemic Response Of BDNF, Lactate and Cortisol to Strenuous Exercise Modalities in Healthy Untrained Women
Supplemental Material, sj-tif-1-dos-10.1177_1559325820970818 for Acute Systemic Response Of BDNF, Lactate and Cortisol to Strenuous Exercise Modalities in Healthy Untrained Women by Patricia C. García-Suárez, Iván Rentería, José Moncada-Jiménez, Andrew C. Fry and Alberto Jiménez-Maldonado in Dose-Response
Footnotes
Acknowledgments
We thank the participants for their honest effort and commitment to the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current project was partially supported by the “20va Convocatoria Interna de Apoyo a Proyectos de Investigación de la Universidad Autónoma de Baja California, México” (Register Number 431/2/C/39/20) to Iván Rentería.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
